Abstract
A simple, economical and mild solution chemistry method was used to synthesize diverse nickel oxide (NiO) nanostructures employing methionine as a growth-directing agent. The as-synthesized NiO nanostructures were observed to possess a unique skein-shape morphology with uniform spherical distribution. The NiO nanoskein (NiO NSk) formation was extensively studied using X-ray diffraction (XRD), X-ray photoelectron microscopy (XPS), scanning electron microscopy (SEM) and Fourier transform infrared (FTIR) techniques, respectively. The unique NiO NSks exhibited better electrocatalytic activity towards glucose oxidation in alkaline media, enabling the development of a highly sensitive non-enzymatic glucose sensor. The observed analytical properties included high sensitivity (1915 μA mM−1 cm−2), wide linear range (0.1–5.0 mM), low detection limit (0.7 μM), higher stability and reproducibility. Moreover, the sensor is selective in the presence of interfering species such as ascorbic acid (AA), uric acid (UA) and dopamine (DP) during the non-enzymatic glucose sensing. The worthy-of-notice electrocatalytic activity and economical feasible preparation of NiO NSk-shaped electroactive material for direct glucose-sensing applications make the present study of high interest for the fabrication of low-cost devices. A NiO NSk-based glucose sensor has also been employed for glucose determination in human serum with adequate results, suggesting high potential for the routine monitoring of glucose from biotechnology, clinical and food industry samples.

Graphical representation of NiO NSks formation
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The study of metal oxides has attracted the attention of material scientists due to their optical, electrical, magnetic and catalytic properties, which make them technologically beneficial [1]. Currently, considerable interest in nanocrystalline oxide materials exists owing to their unusual properties. Decreasing particle size and fabricating unique morphologies result in some remarkable electronic phenomena [2]. For example, Li et al. reported the fabrication of NiO hollow nanospheres and its electrocatalytic potential towards amperometric sensing [3]. Similarly, Umar et al. prepared polyhedral nanocages of MgO, which were determined to be an effective electrode material in bio-sensing of glucose [4], and very recently, Chen et al. and Yi et al. prepared novel Co3O4/PbO2 core–shell nanorod arrays and nickel oxide–carbon nanotube hybrid nanobelts, respectively, and examined their electrochemical capability towards enzyme-free glucose determination [5, 6]. At present, various metal oxides like NiO [7, 8], Co3O4 [9], RuO2 [10], and MnO2 [11] are being heavily investigated due to their small size, high surface-to-volume ratio, better crystalline quality and Debye length relative to their dimensions. Among these metal oxides, NiO nanostructures particularly are believed to possess electrocatalytic response besides the simplicity offered in their growth procedures [12–14]. Interestingly, most properties of metal oxides including electrochemical behaviour are largely influenced by their morphological features [15]. Thus, obtaining an attractive morphology with efficient properties besides their application in sensing technology is an important task to explore. Due to such consideration, a great deal of interest is growing towards the employment of various bio-compatible templates to obtain more morphologically unique and special nanostructures contemporary to conventional structures of metal oxides. In addition to the growth-directing capability, bio-templates enable controlled synthesis, high reliability and consistence in synthesis, as a consequence of which the development of a robust and stable sensor system can be assured. Thus, fabrication routes for such morphologically unique-shaped nanostructures which are efficient in improving the sensitivity and selectivity of sensors, particularly bio sensors, need to be extensively explored [16].
As the determination and monitoring of glucose is gaining high interest regardless of its practice in routine analysis, today’s scientific research has thus very much focused its attention towards portable, convenient and cost-effective glucose sensors [17, 18]. Especially in the case of diabetic patients, the regular, fast and precise monitoring of glucose concentration is essential in order to ensure effective treatment and prevention of sudden diabetic shock during the critical conditions (blood glucose level higher or lower than the physiological value (4.4–6.6 mM)) [19]. Hence, there is an increasing need for the development of miniaturized, economic, handy sensitive, precise and fast glucose sensors/biosensors [20]. In this regard, various techniques have been proposed and applied for the quantification of glucose including infrared spectroscopy, Raman spectroscopy, photoacoustic spectroscopy [21–23], surface plasmon resonance biosensor [24], capacitive determination [25], electrochemiluminescence [26] and colorimetric biosensor [27]. However, the electrochemical technique is considered to be more useful with advantages like easy miniaturization of devices [28, 29].
There are two kinds of electrochemical glucose sensors, glucose oxidase enzyme-based biosensors and enzyme-free glucose sensors [30]. The enzyme-based glucose biosensors are highly selective, sensitive, quick and reproducible. However, such sensors, besides being expensive, are accompanied by disadvantages like chemical and thermal instabilities which reduce their analytical usage [31]. In order to overcome these disadvantages, a lot of work has been devoted for the direct measurement of glucose concentrations by developing enzyme-free glucose sensors. Such sensors have been fabricated using various metals including Pt [8], Au [32], Cu [33] and bimetallic-like Pt–Pb [34], Pt–Te [35], etc., but the high cost, weak selectivity and instability of such sensors make them an unsuitable candidate from the perspective of practical usage. Such drawbacks of enzyme-free glucose sensors based on metals have been removed by the advancement in the field of nanotechnology and different electrode nanomaterials including well-ordered Pt nanotubes [36], Pt nanoparticles [37], Pt–Ru nanoparticles [38], Ni-Pd nanoparticles [39], nanoporous Pt–Pb [40], Pt–Pb nanowires [41] and three-dimensional Au films which have been successfully used to modify the electrodes. The aforementioned reports clearly indicate the significance and requirement for the investigation of cost-effective materials for the purpose of sensing.
Although several attempts have been published regarding the fabrication of NiO nanostructures [42], there still remains much interest in designing simpler, inexpensive, consistent and more versatile fabrication routes to produce unique NiO nanostructures and to study their structure-dependent electrochemical properties. Keeping this in mind, the present study is focused on the synthesis of new and novel nanostructures of NiO, obtained via assistance of methionine (an amino acid) used as a bio-template. The prepared skein-shaped morphological features of NiO demonstrated extremely high potential for stimulating electronic communication during electrocatalytic oxidation of glucose, which facilitated the development of a sensitive, selective, robust, economic and stable non-enzymatic glucose sensor. The reliability of the developed sensor for routine glucose analysis was evaluated by precise determination of glucose from human blood sera.
Experimental
Chemicals and reagents
Analytical-grade nickel nitrate hexahydrate (Ni(NO3)2·6H2O), methionine (HO2CCHCH2CH2SCH3), 33 % ammonia (NH3), d-glucose (C6H12O6), dopamine (C8H11NO2), ascorbic acid (C6H8O6), uric acid (C5H4N4O3) and sodium hydroxide (NaOH) were purchased from Sigma-Aldrich. Nafion, 1.0 %, was prepared in isopropanol (Merck), while other stock solutions were prepared using Milli-Q water.
Apparatus and electrochemical measurements
Material characterization was carried out using scanning electron microscopy (SEM) model JSM 6380 of Jeol, Japan. X-ray diffraction (XRD) pattern was recorded by model D-8 of Bruker using CuKα radiation (λ = 1.54050 Å) and (2θ) scale range of 30 to 80°. The Fourier transform infrared (FTIR) study was performed by model Nicolet 5700 of Thermo, after incorporating the air-dried sample in a solid-state KBr disc. The X-ray photoelectron microscopy (XPS) experiment was performed by an ESCA200 spectrometer in ultrahigh vacuum using a base pressure of 10−10 mbar. The measurement chamber was associated with a monochromatic Al (Kα) X-ray source using photons with frequency (hυ = 1486.6 eV). The electrochemical and amperometric measurements were performed using Bipotentiostate model 760, USA.
Synthesis of skein-shaped NiO nanostructures
The methionine-assisted nickel oxide nanostructures were obtained by using the low-temperature aqueous chemical growth method. In the synthetic procedure, 0.1 M nickel nitrate (Ni(NO3)2·6H2O) and 1 g of methionine (HO2CCHCH2CH2SCH3) were homogenized in 100 ml of deionized water followed by adding 05 ml of 33 % (NH3) solution. The beaker containing the growth solution was tightly sealed with aluminium foil, and the growth solution was kept in a preheated electric oven at 95 °C for 4–6 h. At the completion of growth, Ni(OH)2 nanomaterial was collected, washed to remove exterior impurities and dried at room temperature. Complete conversion of hydroxide to the pure oxide phase of NiO was thus achieved after annealing the precursor material at 450 °C for 2–3 h in an electrical furnace.
Fabrication and electrochemical assessment of the NiO NSk-modified glassy carbon electrode
Prior to the surface modification, a glassy carbon electrode (GCE, diameter 3 mm) was polished with 1 and 0.05 μm alumina paste followed by washing with deionized water and ethanol, respectively. Then, 05 μl of a well-sonicated suspension of as-synthesized NiO nanostructures in ethanol (5 mg ml−1) was drop casted over the surface of mirror polished GCE and dried under ambient air. The entrapment of NiO NSks was fully assured by casting a similar volume of 1 % Nafion solution onto the modified layer. The fabricated electrode was denoted as GCE/NiO NSks/Nafion. The modified electrode was further used as working electrode in the electrochemical cell system in combination with silver–silver chloride wire (Ag/AgCl) used as reference and platinum (Pt) as a counter electrode. The entire glucose-sensing study was carried out using cyclic voltammetry (CV) as a primary mode with the following optimized conditions: 0 to 0.7 V initial potential range, 0.05 V s−1 scan rate, 5 s quiet time, 1 × 10−6 (A/V) sensitivity and 9 ml of 0.1 M NaOH used as an electrolytic solution.
Glucose sensing in human blood sera
The real practical application of GCE/NiO NSks/Nafion was carried by analysing real blood samples, collected from few personally known individuals. The blood serum was obtained via high-speed centrifugation followed by proper treatment and adequate dilution before analysis. A similar sensing procedure was adopted as described above and the obtained results were found satisfactory when compared with those achieved with the commercially available glucose meter.
Results and discussion
Structural characterization of NiO skein-shaped nanostructures
Figure 1 represents the XRD diffractogram obtained for NiO NSks. The diffraction patterns located at 43.16, 62.77 and 75.30 are consistent with pure-phase NiO (JCPDS card no. 71-1179) and could be attributed to (200), (220) and (311) crystal planes of a face-centred cubic (FCC). The obtained intensity suggests that the prepared nanostructures are highly crystalline in nature [43].
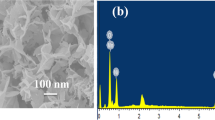
XPS was applied to reveal the surface chemical composition of NiO NSks. As shown in Fig. 2(b), the two peaks at 873.0 and 885.9 eV, with a spin orbit splitting gap of 12.9 eV, are attributed to Ni 2p1/2 and Ni 2p3/2. The bonding energy for O 1s in Fig. 2(c) is 529.8 eV, attributed to the lattice oxygen of NiO. The absence of any other peaks within the full-scale spectrum shown in Fig. 2(a) clearly indicates the surface composition of pure Ni and O. The obtained results are in good agreement with previous reports on NiO nanostructures [44, 45].
Figure 3 shows the results obtained from the SEM imagining of as-synthesized NiO nanostructures. The SEM observation revealed spherical skein morphology with uniform size and high density distribution. Figure 3a–d represents low- and high-resolution images, where hollow cavities within each skein can be visualized. It can be depicted that the formation of such nanostructures takes place via interconnected nanorods during the nucleation process. The observed morphology based on its unique cavitation and size homogeneity and greater surface area offered by connecting tentacles could be responsible to produce themselves as a potential candidate in the enzyme-free sensing of glucose by declaring the high electrocatalytic properties. The average diameter for present nanoskeins was measured to be within 200–300 nm in size. Without the methionine used as template, the obtained NiO NSks would have uncontrollably grown to giant structures and tended to agglomerate as shown in Fig. S1. In addition, they were also found to deceive presently observed electrocatalytic behaviour. This led to the conclusion that the methionine in this case plays an extremely crucial role in simultaneous control of the growth and morphology of synthesized NiO nanostructures. The proposed mechanism can be suggested in this way: the methionine molecule resulted in the formation of a spherical micelle around nickel hydroxide nanostructures and also provided a large number of nucleation sites for the growth of nickel hydroxide nanocrystals. The use of methionine resulted in a subsequent increase in the conglomeration of the nickel hydroxide nanostructures which led to the formation of the nanoskein-like structure of NiO.
Figure 4 represents the FTIR spectrum of standard methionine, precursor material (Ni(OH)2) before and after annealing, respectively. The comparison of spectra in Fig. 4(a, b) clearly depicts an interaction between functional moieties of methionine and prepared Ni(OH)2. The shift observed in the vibrational frequencies for CO stretching from 2094 to 2120 cm−1 and NH band from 3160 to 3180 cm−1 in Fig. 4(b) indicates metal ligand interactions [46]. In addition, the FTIR band observed at 2580 cm−1 resulting from standard methionine was observed to shift to a slightly higher wavelength, i.e. 2590 cm−1, which suggested the existence of intermolecular hydrogen bonding between the protonated amine and the carboxylate groups of methionine during metal ligand interactions [47]. The FTIR band residing at 500 cm−1 is attributed to metal-oxide vibrational stretching. The FTIR spectrum recorded after annealing the precursor material is provided in Fig. 4(c). Since no bands associated with that of methionine were detected, this assures complete removal of bio-template from NiO NSks. However, a sharp band residing near 500 cm−1 attributed to NiO was observed which further confirms the formation of pure NiO nanomaterial. Based on the collected information, a reasonable scheme for the formation of the NiO nanostructures is presented in the following schematic diagram (Scheme 1).
The enzyme-free detection of glucose using novel nanostructures of NiO
To explore sensing properties of novel nanostructures of NiO, cyclic voltammetry was selected as a powerful tool for studying electrochemical behaviours. Figure 5 depicts the CV response of bare and GCE/NiO NSks/Nafion both in the absence and presence of 0.1 mM glucose recorded at a scan rate of 0.05 V s−1. It can be clearly seen that bare GCE has failed to exhibit any prominent response either in the absence or presence of glucose molecules. In contrast to this, GCE/NiO NSks/Nafion clearly exhibits a beautiful pair of oxidation and reduction peaks corresponding to Ni (II)/(III) [48]. In addition, the peak current (Ip) response recorded for GCE/NiO NSks/Nafion in glucose solution is very much higher than the CV response obtained for GCE/NiO NSks/Nafion in blank solution. The observed phenomena reflect the extreme potential of NiO NSks towards oxidation of glucose. The measured responses of the proposed sensor device are in good agreement with the other reported NiO-based sensors [16, 49, 50]. The description for the sensing mechanism of the developed enzyme-free glucose sensor by using the novel nanostructures of NiO can be said as first Ni2+ is electrochemically oxidized to Ni3+ in alkaline solution. The unique morphological features allow enhanced generation of an electron which oxidizes glucose to gluconic acid and reduces Ni3+ to Ni2+ ions and results in a large anodic peak Ip increase [48]. Thus, it is safe to say that typical structural characteristics along with a large surface area of NiO nanoskein are responsible for observed high sensitivity and reactivity of the developed glucose sensor compared to those presented in Table 1.
To achieve the best possible Ip responses during glucose determination, optimization of the most crucial parameters was carried out using GCE/NiO NSks/Nafion in 0.1 mM glucose solution. Figure 6a presents the CV responses for scan rate, recorded with respect to incremental scan rate in the range from 0.05 to 1.0 V s−1. Successive enhancement in Ip response can be observed with the electrochemical process believed to be diffusion controlled as interpreted from the inset of Fig. 6a showing linear proportionality between Ip and the square root of the scan rate.
CV measurement of a GCE/NiO NSks/Nafion at various scan rates from 0.05 to 1.0 mV s−1 in 0.1 M NaOH with 0.1 mM glucose with inset linear graph between Ip response and square root of scan rate, b variation of Ip with electrolytic volume in range from 1.0 to 10.0 ml, c variation NiO NSks casting volume in range from 05 to 20 μl at 0.52 V
The Ip responses of GCE/NiO NSks/Nafion under various electrolytic volumes ranging from 1.0 to 10 ml were also recorded. Figure 6b indicates that the maximum Ip responses can be achieved with 9 ml of NaOH during glucose determination. However, other than the 9-ml decline in Ip response, and shift in potential towards lower value were found eminent.
The Ip responses for NiO NSk casting volume in the range from 5 to 15 μl for GCE/NiO NSks/Nafion were further studied. The corresponding CV measurements are presented in Fig. 6c. As expected, successive increment in Ip was noted with increasing casting volume. However, 5 μl was selected as an optimum volume as it was found to exhibit most stable Ip responses, unlike other volumes (>5 μl) which showed instability caused by erosion of casted material from the electrode surface.
The analytical data for the developed non-enzymatic sensor was obtained via plotting a linear calibration graph between the Ip response of GCE/NiO NSks/Nafion and glucose concentration in the range between 0.1 and 5.0 mM as shown in Fig. 7a. Although the sensor works very well outside the selected calibration range, but to ensure the working potential of the developed sensor in the physiological glucose range and for appropriate data presentation, such range was considered best suitable. The analytically important characteristics of the developed sensor were obtained through linear regression analysis of Ip vs. glucose concentration at the fixed potential of 0.52 V as shown in the inset of Fig. 7a. The developed equation Y = (134 ± 2)x (μM) + 175 ± 6 (R = 0.997) was used to calculate the sensitivity of the developed sensor by dividing the obtained slope with the electrode surface area (0.07 cm−1). The sensitivity was calculated to be 1915 μA mM−1 cm−2 with limit of detection (LOD) and limit of quantification (LOQ) determined to be 0.7 (S/N = 3) and 4.3 μM, respectively. In addition, subsequent amperometric measurements, presented in Fig. 7b, were recorded at the fixed potential of +0.52 V (vs. Ag/AgCl) with increasing glucose concentration in the range from 0.1 to 4.5 μM. It can be clearly seen that sensor response achieves >90 % of steady current in less than 9 s.
The obtained analytical characteristics of the developed sensor were compared with other NiO-based non-enzymatic sensors as shown in Table 1. It is clear that the developed non-enzymatic glucose sensor based on NiO NSks is associated with acceptable advantages compared to the reported enzyme-free glucose sensors. Thus, besides the development of a sensitive and stable non-enzymatic glucose sensor, the easy, consistent, environment friendly and low-cost synthesis protocol makes NiO NSks a potential candidate for sensitive electrochemical glucose sensing compared to the recently reported sensors.
Specificity, reproducibility and stability of GCE/NiO NSks/Nafion
The specificity of the GCE/NiO NSks/Nafion was evaluated by recording its response in the presence of interfering species like dopamine (DA), ascorbic acid (AA) and uric acid (UA), which presented in the physiological fluids are considered to produce most interferences during non-enzymatic glucose oxidation [36]. CV responses were obtained at the normal physiological levels of DA (0.1 mM), UA (0.1 mM), AA (0.1 mM) in the presence of 0.1 mM glucose and 0.1 M NaOH solution at 0.52 V. It can be seen in Fig. 8a that the developed sensor possesses high anti-interference capability suggesting higher selectivity during glucose sensing. The reproducibility of the sensor was evaluated by preparing ten GCE/NiO NSks/Nafion under the same fabrication conditions and recording Ip responses for the electrode with 1 mM glucose solution in 0.1 M NaOH solution at 0.48 V as shown in Fig. 8b. The relative standard deviation (RSD) was 1.5 %, suggesting a highly reproducible nature of the developed sensor. Similarly, the stability of the developed sensor was examined by measuring the Ip response of GCE/NiO NSks/Nafion during 3 weeks of its storage in ambient air conditions. A very small decrease in Ip response (2.5 %) was observed compared to the initial response, which reflected the long-term stability of the sensor.
CV measurements recorded for a specificity of GCE/NiO NSks/Nafion in the presence of common electroactive interferents, b GCE/NiO NSks/Nafion reproducibility with inset showing Ip responses for 10 similarly modified electrodes, c Ip response measured for GCE/NiO NSks/Nafion up to 6 weeks for the same electrode stored in ambient air environment
Poisoning effect of halides
Halide poisoning is an important issue when developing a non-enzymatic glucose sensor [51, 52]. A large number of studies have addressed strong halide poisoning for noble metal electrodes such as gold (Au) [53, 54] and platinum (Pt) [55]. Thus, the effect of halides particularly chloride (Cl−1) and bromide (Br−1) was studied for poisoning during non-enzymatic glucose sensing. Figure S2 shows the corresponding CV measurements recorded in the presence of Cl−1 and Br− ions within the concentration range of 0.1–0.3 M. It can be seen that no significant changes have occurred on the glucose oxidation peak in the presence of both halides. The observed strong tolerance of NiO NSks towards halides may be attributed to the lower susceptibility of metal oxides towards halide poisoning compared to their metal counterparts as suggested by several other studies [56, 57].
Practical applications
To investigate GCE/NiO NSks/Nafion for practical applications, it was subjected to glucose analysis in human blood serum samples. The separated blood serum was two fold diluted before analysis and linear equation mentioned above was used for concentration evaluation. The obtained results, presented in Table 2, were further confirmed authentic by cross analysing the same samples with a commercial glucometer.
Conclusion
In summary, we have developed a sensitive, selective, performance-durable and cost-effective non-enzymatic glucose sensor based on unique skein-shaped nanostructures of NiO. The NiO NSk-based sensor demonstrated acceptable sensitivity (1915 μA mM−1 cm−2), low detection limit (0.7 μM) and high stability besides a wide linear working range (up to 5 mM). The high performance can be ascribed to the unique morphological features of NiO which were obtained with the assistance of methionine used as a growth-directing agent. All the obtained data suggested GCE/NiO NSks/Nafion as a promising candidate in the development of a non-enzymatic glucose sensor. Furthermore, this work also demonstrated a promising route to fabricate unique skein-shaped NiO nanostructures with controlled shape and size.
References
Heli H, Sattarahmady N, Vais RD, Karimian K (2014) Nickel hydroxide nanopetals: one-pot green synthesis, characterization and application for the electrocatalytic oxidation and sensitive detection of montelukast. Sensor Actuat B: Chem 196:631–639
Combustible solid precursors to nanocrystalline oxide materials. In: Chemistry of nanocrystalline oxide materials. pp 9-41. doi:10.1142/9789812793157_0002
Li C, Liu Y, Li L, Du Z, Xu S, Zhang M, Yin X, Wang T (2008) A novel amperometric biosensor based on NiO hollow nanospheres for biosensing glucose. Talanta 77(1):455–459
Umar A, Rahman MM, Hahn Y-B (2009) MgO polyhedral nanocages and nanocrystals based glucose biosensor. Electrochem Commun 11(7):1353–1357
Chen T, Li X, Qiu C, Zhu W, Ma H, Chen S, Meng O (2014) Electrochemical sensing of glucose by carbon cloth-supported Co3O4/PbO2 core-shell nanorod arrays. Biosens Bioelectron 53:200–206
Yi W, Yang D, Chen H, Liu P, Tan J, Li H (2014) A highly sensitive nonenzymatic glucose sensor based on nickel oxide–carbon nanotube hybrid nanobelts. J Solid State Electrochem 18(4):899–908
Xu J, Gao L, Cao J, Wang W, Chen Z (2011) Electrochemical capacitance of nickel oxide nanotubes synthesized in anodic aluminum oxide templates. J Solid State Electrochem 15(9):2005–2011
Vassilyev YB, Khazova OA, Nikolaeva NN (1985) Kinetics and mechanism of glucose electrooxidation on different electrode-catalysts: part I. Adsorption and oxidation on platinum. J Electroanal Chem Interfacial Electrochem 196(1):105–125
Ding Y, Wang Y, Su L, Bellagamba M, Zhang H, Lei Y (2010) Electrospun Co3O4 nanofibers for sensitive and selective glucose detection. Biosens Bioelectron 26(2):542–548
Lyons MEG, Fitzgerald CA, Smyth MR (1994) Glucose oxidation at ruthenium dioxide based electrodes. Anly 119(5):855–861
Chen J, Zhang W-D, Ye J-S (2008) Nonenzymatic electrochemical glucose sensor based on MnO2/MWNTs nanocomposite. Electrochem Commun 10(9):1268–1271
Chekin F, Tahermansouri H, Besharat M (2014) Nickel oxide nanoparticles prepared by gelatin and their application toward the oxygen evolution reaction. J Solid State Electrochem 18(3):747–753
Elumalai P, Zosel J, Guth U, Miura N (2009) NO2 sensing properties of YSZ-based sensor using NiO and Cr-doped NiO sensing electrodes at high temperature. Ionics 15(4):405–411
Wu Z, Jiang L, Zhu Y, Xu C, Ye Y, Wang X (2012) Synthesis of mesoporous NiO nanosheet and its application on mercury (II) sensor. J Solid State Electrochem 16(10):3171–3177
Wang D, Wang Q, Wang T (2013) Controlled synthesis of porous nickel oxide nanostructures and their electrochemical capacitive behaviors. Ionics 19(3):559–570
Ding Y, Liu Y, Parisi J, Zhang L, Lei Y (2011) A novel NiO–Au hybrid nanobelts based sensor for sensitive and selective glucose detection. Biosens Bioelectron 28(1):393–398
Park H, Seo Yoon H, Patil U, Anoop R, Lee J, Lim J, Lee W, Chan Jun S (2014) Radio frequency based label-free detection of glucose. Biosens Bioelectron 54:141–145
Meng L, Jin J, Yang G, Lu T, Zhang H, Cai C (2009) Nonenzymatic electrochemical detection of glucose based on palladium–single-walled carbon nanotube hybrid nanostructures. Anal Chem 81(17):7271–7280
Wang J (2007) Electrochemical glucose biosensors. Chem Rev 108(2):814–825
Sljukic B, Banks CE, Salter C, Crossley A, Compton RG (2006) Electrochemically polymerised composites of multi-walled carbon nanotubes and poly(vinylferrocene) and their use as modified electrodes: application to glucose sensing. Anly 131(5):670–677
Song C, Pehrsson PE, Zhao W (2006) Optical enzymatic detection of glucose based on hydrogen peroxide-sensitive HiPco carbon nanotubes. J Mater Res 21(11):2817–2823
Barone PW, Parker RS, Strano MS (2005) In Vivo fluorescence detection of glucose using a single-walled carbon nanotube optical sensor: design, fluorophore properties, advantages, and disadvantages. Anal Chem 77(23):7556–7562
Spanner G, Nießner R (1996) New concept for the non-invasive determination of physiological glucose concentrations using modulated laser diodes. Fresenius J Anal Chem 354(3):306–310
Shen XW, Huang CZ, Li YF (2007) Localized surface plasmon resonance sensing detection of glucose in the serum samples of diabetes sufferers based on the redox reaction of chlorauric acid. Talanta 72(4):1432–1437
Cheng Z, Wang E, Yang X (2001) Capacitive detection of glucose using molecularly imprinted polymers. Biosens Bioelectron 16(3):179–185
Forster RJ, Hogan CF (2000) Electrochemiluminescent metallopolymer coatings: combined light and current detection in flow injection analysis. Anal Chem 72(22):5576–5582
Morikawa MA, Kimizuka N, Yoshihara M, Endo T (2002) New colorimetric detection of glucose by means of electron-accepting indicators: ligand substitution of [Fe(acac)3-n(phen)n]n + complexes triggered by electron transfer from glucose oxidase. Chemistry 8(24):5580–5584
Liu X, Li Y, Liu X, Zeng X, Kong B, Luo S, Wei W (2012) Simple sensor for simultaneous determination of dihydroxybenzene isomers. J Solid State Electrochem 16(3):883–889
Wu C, Wang X, Ju B, Bai Y, Jiang L, Wu H, Zhao Q, Gao J, Wang X, Yi L (2013) Supercapacitive behaviors of the nitrogen-enriched activated mesocarbon microbead in aqueous electrolytes. J Solid State Electrochem 17(6):1693–1700
Luo L, Li F, Zhu L, Ding Y, Zhang Z, Deng D, Lu B (2013) Nonenzymatic glucose sensor based on nickel(II)oxide/ordered mesoporous carbon modified glassy carbon electrode. Colloid Surface B 102:307–311
Katakis I, Domínguez E (1995) Characterization and stabilization of enzyme biosensors. TrAC Trends Anal Chem 14(7):310–319
Adzic RR, Hsiao MW, Yeager EB (1989) Electrochemical oxidation of glucose on single crystal gold surfaces. J Electroanal Chem IntElectrochem 260(2):475–485
Nagy L, Nagy G, Hajós P (2001) Copper electrode based amperometric detector cell for sugar and organic acid measurements. Sensor Actuat B: Chem 76(1–3):494–499
Sun Y, Buck H, Mallouk TE (2001) Combinatorial discovery of alloy electrocatalysts for amperometric glucose sensors. Anal Chem 73(7):1599–1604
Guascito MR, Chirizzi D, Malitesta C, Siciliano M, Siciliano T, Tepore A (2012) Amperometric non-enzymatic bimetallic glucose sensor based on platinum tellurium microtubes modified electrode. Electrochem Commun 22:45–48
Yuan JH, Wang K, Xia XH (2005) Highly ordered platinum-nanotubule arrays for amperometric glucose sensing. Adv Funct Mater 15(5):803–809
Kang X, Mai Z, Zou X, Cai P, Mo J (2008) Glucose biosensors based on platinum nanoparticles-deposited carbon nanotubes in sol-gel chitosan/silica hybrid. Talanta 74(4):879–886
Li L-H, Zhang W-D, Ye J-S (2008) Electrocatalytic oxidation of glucose at carbon nanotubes supported PtRu nanoparticles and its detection. Electroanalysis 20(20):2212–2216
Miao F, Tao B, Sun L, Liu T, You J, Wang L, Chu PK (2009) Amperometric glucose sensor based on 3D ordered nickel–palladium nanomaterial supported by silicon MCP array. Sensor Actuat B: Chem 141(1):338–342
Wang J, Thomas DF, Chen A (2008) Nonenzymatic electrochemical glucose sensor based on nanoporous PtPb networks. Anal Chem 80(4):997–1004
Bai Y, Sun Y, Sun C (2008) Pt-Pb nanowire array electrode for enzyme-free glucose detection. Biosens Bioelectron 24(4):579–585
Nathan T, Aziz A, Noor AF, Prabaharan SRS (2008) Nanostructured NiO for electrochemical capacitors: synthesis and electrochemical properties. J Solid State Electrochem 12(7–8):1003–1009
Liu S, Yu B, Zhang T (2013) A novel non-enzymatic glucose sensor based on NiO hollow spheres. Electrochim Acta 102:104–107
Chigane M, Ishikawa M (1998) XRD and XPS characterization of electrochromic nickel oxide thin films prepared by electrolysis-chemical deposition. J Chem Soc Faraday Trans 94(24):3665–3670
Grosvenor AP, Biesinger MC, Smart RSC, McIntyre NS (2006) New interpretations of XPS spectra of nickel metal and oxides. Surf Sci 600(9):1771–1779
Kalwar NH, Sirajuddin SSTH, Abro MI, Tagar ZA, Hassan SS, Junejo Y, Khattak MI (2011) Synthesis of l-methionine stabilized nickel nanowires and their application for catalytic oxidative transfer hydrogenation of isopropanol. Appl Catal, A 400(1–2):215–220
Krchova T, Kotek J, Jirak D, Havlickova J, Cisarova I, Hermann P (2013) Lanthanide(iii) complexes of aminoethyl-DO3A as PARACEST contrast agents based on decoordination of the weakly bound amino group. Dalton Trans 42(44):15735–15747
Li X, Hu A, Jiang J, Ding R, Liu J, Huang X (2011) Preparation of nickel oxide and carbon nanosheet array and its application in glucose sensing. J Solid State Chem 184(10):2738–2743
Lu W, Qin X, Asiri AM, Al-Youbi AO, Sun X (2013) Facile synthesis of novel Ni(ii)-based metal-organic coordination polymer nanoparticle/reduced graphene oxide nanocomposites and their application for highly sensitive and selective nonenzymatic glucose sensing. Anly 138(2):429–433
Lu W, Qin X, Asiri AM, Al-Youbi AO, Sun X (2013) Ni foam: a novel three-dimensional porous sensing platform for sensitive and selective nonenzymatic glucose detection. Anly 138(2):417–420
Jeong H, Kim J (2012) Electrochemical oxidation of glucose at nanoporous black gold surfaces in the presence of high concentration of chloride ions and application to amperometric detection. Electrochim Acta 80:383–389
Pasta M, La Mantia F, Cui Y (2010) A new approach to glucose sensing at gold electrodes. Electrochem Commun 12(10):1407–1410
Seo B, Kim J (2010) Electrooxidation of glucose at nanoporous gold surfaces: structure dependent electrocatalysis and its application to amperometric detection. Electroanalysis 22(9):939–945
Hsiao MW, Adzic RR, Yeager EB (1992) The effects of adsorbed anions on the oxidation of D-glucose on gold single crystal electrodes. Electrochim Acta 37(2):357–363
Kamath VN, Lal H (1970) Halide adsorption and the anodic oxidation of chemisorbed methanol on platinum. J Electroanal Chem Interfacial Electrochem 24(1):125–135
Luo S, Su F, Liu C, Li J, Liu R, Xiao Y, Li Y, Liu X, Cai Q (2011) A new method for fabricating a CuO/TiO2 nanotube arrays electrode and its application as a sensitive nonenzymatic glucose sensor. Talanta 86:157–163
Liu Y, Teng H, Hou H, You T (2009) Nonenzymatic glucose sensor based on renewable electrospun Ni nanoparticle-loaded carbon nanofiber paste electrode. Biosens Bioelectron 24(11):3329–3334
Shamsipur M, Najafi M, Hosseini M-RM (2010) Highly improved electrooxidation of glucose at a nickel(II) oxide/multi-walled carbon nanotube modified glassy carbon electrode. Bioelectrochemistry 77(2):120–124
Lv W, Jin F-M, Guo Q, Yang Q-H, Kang F (2012) DNA-dispersed graphene/NiO hybrid materials for highly sensitive non-enzymatic glucose sensor. Electrochim Acta 73:129–135
Zhong Y, Li Y, Li S, Feng S, Zhang Y (2014) Nonenzymatic hydrogen peroxide biosensor based on four different morphologies of cuprous oxide nanocrystals. RSC Adv 4(76):40638–40642
Acknowledgments
We acknowledge the Higher Education Commission, Islamabad, Pakistan, for provision of financial assistance during this research and the National Centre of Excellence in Analytical Chemistry, Jamshoro, for their facilities.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 442 kb)
Rights and permissions
About this article
Cite this article
Soomro, R.A., Ibupoto, Z.H., Sirajuddin et al. Controlled synthesis and electrochemical application of skein-shaped NiO nanostructures. J Solid State Electrochem 19, 913–922 (2015). https://doi.org/10.1007/s10008-014-2700-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2700-z