Abstract
In contrast to mesophiles, in which levels of trehalose and phosphatidic acids (PA) increased only under heat shock (HS), in thermophiles trehalose and PA were predominant under optimal growth conditions. To study the role of trehalose protection in the adaptation of thermophiles to various stressors, the composition of osmolytes and membrane lipids in the thermophilic fungus Rhizomucor miehei was studied under cold (CS), osmotic (OS) and oxidative (OxS) shocks. CS resulted in no accumulation of glycerol in the mycelium, while the amount of trehalose decreased. The main lipid changes were the increase in the PA proportion with simultaneous decrease of sterols (St), the increase of the unsaturation degree of polar lipids and the decrease of the ergosterol proportion in total St. OS did not cause changes in the lipid composition, but led to the decrease of ergosterol proportion too. Despite the low ability of Mucorales to produce polyols, increase in the level of arabitol and glycerol was observed under OS. OxS led to the decrease of trehalose level and had no effect on the lipid composition. Thus, our results show the similarity (OS) and the difference (CS and OxS) between adaptation mechanisms of thermophiles and mesophiles.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Extremophiles are divided into groups according to optimal growth conditions, such as temperature (psychrophiles, thermophiles and hyperthermophiles), pH (acidophiles and alkaliphiles), pressure (barophiles), ionic force (halophiles) (Weber et al. 2007; Rampelotto 2013). Thermophilia of fungi, unlike bacteria, is significantly lower in temperature; these eukaryotes are unable to grow at temperatures above 62 °C probably due to the inability to form functional and thermostable membranes of intracellular organelles (Tansey and Brock 1972). However, the ability of eukaryotes to withstand adverse pH and salinity levels or desiccation conditions is not different from that of prokaryotes (Brown and Fritz 2019).
Life in extreme conditions is possible due to the adaptation mechanisms to one or several adverse factors, formed in the process of evolution. The study of these mechanisms in thermophilic fungi, as well as the study of adaptation mechanisms to stress factors in mesophiles are interconnected. There is a hypothesis that thermophiles are using the heat shock adaptation mechanisms intrinsic to mesophilic fungi (Feofilova and Tereshina 1999). Experimental data confirming this hypothesis were obtained: large amount of trehalose is accumulated in the cytosol, and the proportion of phosphatidic acids in the membrane lipids composition increases significantly both in mesophilic fungi under the action of HS (Tereshina et al. 2010, 2011), as well as in thermophiles under optimal conditions (Ianutsevich et al. 2016b). However, unlike mesophiles, thermophiles did not acquire heat resistance after HS treatment, which was accompanied by a sharp drop of trehalose level, while the proportion of PA in the composition of membrane lipids increased.
High level of trehalose in the mycelium of thermophilic fungi under optimal growth conditions suggests that they may use trehalose protection to adapt to various stressors, but no such information was found in the literature. Trehalose and polyols are the main fungal osmolytes, which are not only compatible solutes, but also perform cytoprotective functions (Yancey 2005; Yancey and Siebenaller 2015).
Trehalose, unlike polyols, is a unique cytoprotector that protects not only cell macromolecules, but also membranes under stress conditions and has antioxidant and regulatory functions (Patel and Williamson 2016; Rubio-Texeira et al. 2016; Glatz et al. 2016). Previously expressed hypothesis about trehalose being a universal protector under any stress conditions (Crowe 2007) has not been experimentally confirmed. Study of the response of Aspergillus niger to heat osmotic, oxidative and cold shocks showed that the trehalose level increased significantly only after HS, whereas in response to cold and osmotic shock glycerol level increased, while oxidative shock had no effect on the composition of carbohydrates (Ianutsevich et al. 2016a). Osmolytes production in response to osmotic and cold stress is regulated through the HOG-pathway (Jennings 1985; Hayashi and Maeda 2006; Duran et al. 2010). Different polyols are involved in protection against osmotic shock in fungi–glycerol in halophilic yeast (Kogej et al. 2007) and A. niger (Ianutsevich et al. 2016a), arabitol in halotolerant fungus Fusarium sp. (Smolyanyuk et al. 2013).
Proper functional state of the membrane is necessary for stress resistance. It has been demonstrated, that changes in the composition of membrane lipids and their unsaturation degree are important for adaptation. In response to thermal, cold, osmotic and oxidative shocks in A. niger, the same pattern was observed: the proportion of non-bilayer phosphatidic acids in the composition of membrane lipids increased significantly, in contrast to the Hazel hypothesis on increasing proportion of bilayer lipids under stress impact (Hazel 1995). The liquid-mosaic model of the membrane structure proposed in 1972 (Singer and Nicolson 1972) was significantly improved (Nicolson 2014). New data on the signal functions of membranes were obtained (Ernst et al. 2016), the heterogeneous structure of the membrane has been experimentally proven. Non-bilayer lipids drew more attention, due to their participation in the formation of membrane microdomains such as rafts, caveolae, eisosomes, detergent-resistant parts of membranes (Vigh et al. 2005; Douglas and Konopka 2014; Carquin et al. 2016). The role of membrane lipids in the perception of external signals, the regulation of enzyme activity and gene expression, the intracellular transport of proteins, endo- and exocytosis, virulence was demonstrated (Kooijman et al. 2003; McMahon and Gallop 2005; Rella et al. 2016; Welte and Gould 2017).
Thermophilic fungi are well adapted to life at elevated temperatures, however, there is no information on the mechanisms of adaptation of thermophiles to other stress factors. Is trehalose protection in thermophiles sufficient for adaptation to osmotic, cold, oxidative shocks, or is the production of other osmolytes required? Therefore, Mucorales thermophilic fungus Rhizomucor miehei, unable to synthesize large amounts of polyols, is an interesting object to study (Jennings 1985).
The aim of this work was to study the composition of membrane lipids and osmolytes of thermophilic fungus R. miehei under cold, oxydative and osmotic shocks.
Materials and methods
Strain, media and cultivation
The study was carried out with the thermophilic fungus Rhizomucor miehei (Cooney & R. Emerson 1964) Schipper 1978 VKM F-1365 (Lichtheimiaceae, Mucoromycetes, Zygomycota), which was grown on wort agar slants at 41–43 ºC for 5 days. Liquid medium was inoculated with the spore suspension to the final concentration of 5 × 105–106 spores/ml. The fungus was grown in submerged culture in 250 ml flasks with 50 ml of Goodwin medium (Garton et al. 1951) on a KE-12-250T electromagnetic shaker (150 rpm) at the optimum temperature of 41–43 °C. Cold shock conditions were created by transferring the submerged fungal culture, grown under optimal conditions for 24 h ( trophophase stage), to 20 °C for 3 and 6 h, while maintaining the same aeration conditions. Oxidative shock conditions were created by adding hydrogen peroxide to the trophophase submerged culture to the concentrations of 10, 30 and 50 mM and continuing the cultivation for 3 h. To create osmotic shock sodium chloride was added to the trophophase submerged culture up to the concentration of 0.125, 0.25 and 0.5 M, and continuing the cultivation for another 3 and 6 h. Fungal biomass was filtered through caprone, washed with distilled water, stored at – 21 °C. Dry biomass was determined gravimetrically.
Lipids and osmolytes analysis
Lipids and carbohydrates analyses were performed as described earlier (Ianutsevich et al. 2016b). Briefly, lipids were extracted by the Nichols method (Nichols 1963) with phospholipase-deactivating isopropanol, separated by two-dimensional (polar lipids) or one-dimensional (neutral lipids) thin-layer chromatography (TLC) and quantified using standard compounds with densitometry method (DENS software). To study the composition of fatty acids the polar lipid fraction was isolated using one-dimensional TLC in neutral lipid system. The polar lipid spots remaining at the start were scraped out, eluted with a mixture of chloroform: methanol (1:1), then extract was evaporated and methanolysis was carried out using 2.5% H2SO4 in methanol solution for 2 h at 85 °C. To determine the composition of fatty acids, their methyl esters were analyzed by GLC. Soluble cytosol carbohydrates were extracted with boiling water, proteins and charged compounds were removed, trimethylsilyl derivatives of sugars were obtained and analyzed by GLC, using an internal standard.
Sterols and triterpenoids fraction were isolated from the lipid extract by saponification. Approximately 10 mg of lipids was suspended in 10 ml of 6% (w/v) KOH in methanol solution and left at 85 °C for 2 h. Complete removal of acylglycerols was checked by TLC. The composition of the sterol fraction was analyzed by GC–MS. Silyl derivatives were obtained to analyze individual components of the mixture. Analytical samples were dissolved at 20 °C in N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA) silylating reagent, and fully derivatized within 30 min at 70 °C with occasional stirring, the mixture was cooled and the residue was dissolved in hexane. Derivatives were analyzed using the Agilent Technologies equipment consisting of a gas chromatograph 7890 (column HP-5, 50 m × 320 µm × 1.05 µm) and mass-selective detector 5975C with quadrupole mass analyzer. Chromatography temperature program: at 40 °C—isotherm 2 min, followed by programmed heating up to 250 °C at a speed of 5 °C/min, at 250 °C—isotherm 15 min, followed by programmed heating up to 320 °C at a speed of 25 °C/min, at 320 °C—isotherm for 30 min. The injector had the split ratio of 1:50. Injector temperature was 250 °C. Interface temperature was 280 °C. Carrier gas-helium; flow rate 1 ml/min. The chromatogram presented is the total ion current chromatogram. MS detection conditions: ionizing electron energy of 70 eV; registration of mass spectra in positive ions in the range from 20 to 450 (m/z), scan speed of 2.5 scans/s. Software—ChemStation version E02.00. Identification of the component composition (qualitative analysis) was performed using NIST database, as well as the corresponding values of retention times of the reference compounds.
Statistical analysis
For each variant experiments were performed three times (n = 3). The differences among the means were compared using one-way ANOVA (P ≤ 0.05). Post hoc Tukey HSD test was used for pairwise comparison between control and shock variants. Statistical analysis was carried out using MS Office Excel. In the figures and tables an asterisk (*) represents a significant difference (P ≤ 0.05), each data point is a mean value ± SE (n = 3).
Results
Sugars and polyols composition under different shocks
The micromycete R. miehei is a true thermophile, since it has optimum growth temperature at 41–43 °C and does not grow at 20 °C (Yanutsevich et al. 2014). Previously, we have shown that trehalose plays an important role in thermophilia. The amount of this disaccharide at all growth stages in submerged culture of R. miehei varied from 8 to 10% of dry weight (Ianutsevich et al. 2016b). Present research was dedicated to studying the mechanisms of adaptation to various stress factors of R. miehei, having powerful trehalose protection.
Cytosol carbohydrates and polyols of R. miehei under optimal conditions (control variant) were presented mainly by trehalose, a small amount of glucose (2–3% of the total sugars) and traces of arabitol and glycerol (Fig. 1). Under the cold shock conditions (20 °C) for 3 h no significant changes were observed (Fig. 1). Increasing the duration of CS up to 6 h resulted in a three-fold drop in the amount of trehalose, with no effect on other carbohydrates or polyols.
Composition of the main soluble cytosol carbohydrates of R. miehei mycelium under cold shock. C—Control, CS—Cold shock, 3 and 6 h, respectively. The fungus was grown in submerged culture at optimal conditions (41–43 °C) for 24 h (trophophase), then it was transferred to 20 °C and cultivation was continued for 3 and 6 h. Soluble cytosol carbohydrates were extracted four times with boiling water, proteins and charged compounds were removed, trimethylsilyl derivatives were obtained and analyzed by GLC. After 6 h of cold shock amount of trehalose decreased threefold
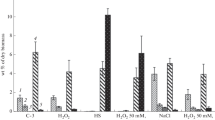
Exposure to 10 mM H2O2 for 3 h did not lead to any significant changes in the composition of carbohydrates (Fig. 2). The increase of the H2O2 concentration up to 30 and 50 mM was accompanied by a sharp decrease in the amount of trehalose by 2 and 8 times, respectively, while the amount of glucose did not change.
Composition of the main soluble cytosol carbohydrates of R. miehei mycelium under oxidative shock. C-3—Control variant after 3 h. The fungus was grown in submerged culture at optimal conditions (41–43 °C) for 24 h (trophophase), then H2O2 was added to the concentration of 10, 30 and 50 mM and cultivation was continued for 3 h. Soluble cytosol carbohydrates were extracted four times with boiling water, proteins and charged compounds were removed, trimethylsilyl derivatives were obtained and analyzed by GLC. After 3 h of oxidative shock amount of trehalose decreased significantly
The osmotic impact of 0.125 M NaCl for 3 h did not lead to any changes in the carbohydrates composition (Fig. 3). Higher NaCl concentration (0.25 M) resulted in an increase of arabitol level, the proportion of polyols rising up to 30%. Further increase of the salt concentration to 0.5 M led to a fivefold drop of the trehalose level, which resulted in the increase of polyols proportion up to 45%. To ensure that the polyols level reached its maximum, the duration of OS was prolonged to 6 h. No significant changes were observed. The only noticeable effect was the increase of the polyols proportion up to 40%, due to the increase of the arabitol and glycerol levels in 0.25 M NaCl variant.
Composition of the main soluble cytosol carbohydrates of R. miehei mycelium under osmotic shock. C—Control, OS—Osmotic shock, 3 and 6 h, respectively. The fungus was grown in submerged culture at optimal conditions (41–43 °C) for 24 h (trophophase), then NaCl was added to the concentration of 0.125, 0.250 and 0.500 M and cultivation was continued for 3 and 6 h. Soluble cytosol carbohydrates were extracted four times with boiling water, proteins and charged compounds were removed, trimethylsilyl derivatives were obtained and analyzed by GLC. After 3–6 h of osmotic shock polyols levels increased (glycerol and arabitol). Higher concentration of NaCl (0.500 M) resulted in the significant decrease of trehalose amount
Membrane lipids under different shocks
To study how changes in the membrane lipids composition contribute to the adaptation to various stressors, quantitative analysis of lipids was performed. Predominant membrane lipids of the thermophilic fungus during growth under optimum conditions were PA, PE and St (Fig. 4). With time, the proportion of PA remained consistently high, and proportions of sterols and X1 lipid increased, while levels of PE, PC and CL decreased. Unidentified polar lipid X1 had properties of glycerophospholipids (was saponified), but it also contained a glycoside group, demonstrating positive reaction with alpha-naphthol. The proportion of sphingolipids also increased with time but did not exceed 5%.
Membrane lipids composition of R. miehei grown in submerged culture at optimal conditions for 12, 18, 36 and 42 h. Polar lipids were extracted using the Nichols method (first with phospholipase deactivating isopropanol, then with the mixture of isopropanol and chloroform), separated by two-dimensional TLC and quantified using standard compounds (phosphatidylcholine, glycoceramide and ergosterol) followed by densitometry method (DENS software). With time, an increase in the proportion of St and X1 was detected, while PE, PC and CL decreased. The relative amount of PA remained consistently high
After 3 and 6 h of CS, similar changes were observed in the membrane lipids of R. miehei—a significant increase in the proportion of phosphatidic acids with simultaneous decrease of sterols level (Fig. 5).
Membrane lipids composition of R. miehei under cold shock. C—Control, CS—Cold shock, 3 and 6 h, respectively. The fungus, grown in submerged culture at optimal conditions (41–43 °C) for 24 h (trophophase), was transferred to 20 °C and cultivation continued for 3 and 6 h. Polar lipids were extracted using the Nichols method (first with phospholipase deactivating isopropanol, then with the mixture of isopropanol and chloroform), separated by two-dimensional TLC and quantified using standard compounds (phosphatidylcholine, glycoceramide and ergosterol) followed by densitometry method (DENS software). Cold shock led to the increase in the PA and decrease in St proportions
The osmotic effect of 0.125 and 0.25 M NaCl for 3 h did not result in any noticeable changes in the membrane lipids composition, while higher NaCl concentration (0.5 M) led to the slight increase in the proportion of sterols (Fig. 6).
Membrane lipids composition of R. miehei under osmotic shock. C-3—Control variant after 3 h. The fungus was grown in submerged culture at optimal conditions (41–43 °C) during 24 h (trophophase), then NaCl was added to the concentration of 0.125, 0.250 and 0.500 M and cultivation was continued for 3 h. Polar lipids were extracted using the Nichols method (first with phospholipase deactivating isopropanol, then with the mixture of isopropanol and chloroform), separated by two-dimensional TLC and quantified using standard compounds (phosphatidylcholine, glycoceramide and ergosterol) followed by densitometry method (DENS software). Osmotic shock did not cause significant changes in the composition of membrane lipids
Oxidative shock (10 mM H2O2) caused no changes in the composition of membrane lipids, whereas higher concentrations of hydrogen peroxide (30 and 50 mM) led to the slight increase in the proportions of PA and sterols, while PE and PC decreased (Fig. 7).
Membrane lipids composition of R. miehei under oxidative shock. C-3—Control variant after 3 h. The fungus was grown in submerged culture at optimal conditions (41–43 °C) during 24 h (trophophase), then H2O2 was added to the concentrations of 10, 30 and 50 mM and cultivation was continued for 3 h. Polar lipids were extracted using the Nichols method (first with phospholipase deactivating isopropanol, then with the mixture of isopropanol and chloroform), separated by two-dimensional TLC and quantified using standard compounds (phosphatidylcholine, glycoceramide and ergosterol) followed by densitometry method (DENS software). Oxidative shock led to the increase in PA and St and decrease in PE and PC proportions
Sterols and triterpenoids fraction of the R. miehei mycelium was represented by 7 compounds, ergosterol being predominant (up to 70% of the total sterols) (Fig. 8 Table 1). Characteristic feature was the presence of two acetylated sterols—ergosta-5,7,22-trien-3β-yl acetate and ergosta-7,22-dien-3β-yl acetate. The qualitative composition did not change under the influence of the studied stressors, while significant changes in the ratio of were observed. Cold shock had the most pronounced effect, leading to the 1.5-fold decrease of the ergosterol proportion, while proportions of ergosta-7,22-dien-3β-ol and ergosta-3,5,7,9-tetraene increased three and two times, respectively (Table 1). Oxidative shock had no significant impact on the sterols and triterpenoids composition, whereas osmotic shock led to the increase in the proportion of ergosta-7,22-dien-3β-ol with simultaneous decrease of ergosterol.
Chromatograms of fungal sterols and triterpenoids under osmotic, oxidative and cold shocks. a Control variant. To create osmotic shock (b), NaCl was added to the submerged culture at the trophophase stage (24 h) to a concentration of 0.500 M and cultivation continued for 3 h; to create oxidative shock (c) H2O2 was added to a concentration of 50 mM and incubated for 3 h; to create cold shock (d), the submerged culture was transferred to 20 °C and cultivation continued for 6 h. Lipids were extracted using Nichols method (first with phospholipase deactivating isopropanol, then with the mixture of isopropanol and chloroform). Sterols and triterpenoids fraction were isolated from the lipid extract by saponification, using 6% (w/v) KOH in methanol solution at 85 °C for 2 h. The composition of the sterol and triterpenoids fraction was analyzed by GC–MS. Silyl derivatives were obtained to analyze individual components of the mixture. Identification of the component composition (qualitative analysis) was performed using NIST database, as well as the corresponding values of retention times of the reference compounds. The fungal sterols and triterpenoids were represented by 7 components, ergosterol being predominant. 1: ergosta-5,7,9(11),22-tetraene, 2: ergosta-3,5,7,9-tetraene, 3: ergosta-5,7,9(11),22-tetraen-3β-ol, 4: ergosta-5,7,22-trien-3β-ol, 5: ergosta-7,22-dien-3β-ol, 6: ergosta-5,7,22-trien-3β-ylacetate, 7: ergosta-7,22-dien-3β-ylacetate (refer to Table 1)
Fatty acids of the phospholipids under different stressors
To evaluate how changes of the unsaturation degree of membrane lipids contribute to the adaptation, the fraction of polar lipids was isolated and their fatty acids composition was analyzed. Main fatty acids of the R. miehei membrane phospholipids were palmitic (C16:0), stearic (C18:0), oleic (C18:1n9c), linoleic (C18:2n6c) and α-linolenic (C18:3n3). Amounts of C12:0, C14:0, C14:1, 15:0, C16:1, C17:0, C17:1 fatty acids were minor. It was demonstrated that only exposure to cold shock for 6 h led to the increase in the unsaturation degree of the polar lipids, whereas osmotic and oxidative shocks did not cause any significant changes in fatty acid composition (Table 2).
Discussion
The obtained results provide the opportunity to compare adaptation mechanisms of thermophilic and mesophilic fungi. In mesophilic fungi under optimal conditions trehalose was the minor component, but its level increased rapidly in response to HS (Thevelein 1996; Vigh et al. 2005; Tereshina et al. 2011). Previously it was shown that thermophilic fungus R. miehei has high level of trehalose at all stages of growth under optimal conditions, but HS resulted in sharp drop of this disaccharide level (Yanutsevich et al. 2014). In mesophilic fungi, in contrast to thermophiles, the significant increase in the proportion of non-bilayer phosphatidic acids in the composition of membrane lipids was observed under HS conditions (Tereshina et al. 2011). In R. miehei, as well as in previously studied thermophilic fungi Rhizomucor tauricus and Myceliophthora thermophila (Ianutsevich et al. 2016b) a high proportion of PA was registered at all growth stages, and under the influence of HS its proportion further increased, as in mesophiles.
During osmotic shock, sensors, localized in the plasma membrane, induce the high osmolarity glycerol (HOG) pathway, mediated by mitogen-activated protein kinase (MAPK cascade) (Duran et al. 2010). Thus, halophilic yeasts accumulate glycerol in the cytosol (Kogej et al. 2007), and halotolerant filamentous fungus Fusarium sp.—arabitol (Smolyanyuk et al. 2013). Moreover, it was suggested that trehalose might be a universal protector under different types of stress (Elbein et al. 2003; Crowe 2007; Iturriaga et al. 2009). Indeed, in the pathogenic yeasts Candida albicans and Cryptococcus neoformans the level of trehalose was increased 50-fold in response to OxS, OS and HS (Nwaka and Holzer Nwaka and Holzer 1998). But in A. niger trehalose participated only in the adaptation to HS, but not OS, OxS and CS (Ianutsevich et al. 2016a). In the present study it was demonstrated, that despite the high level of trehalose in the mycelium of R. miehei, OS led to further polyols accumulation, such as arabitol and glycerol (Fig. 3). Moreover, increasing concentrations of NaCl up to 0.5 M led to the significant reduction of the trehalose level. It should be emphasized that thermophilic fungus R. miehei belongs to Mucorales, which usually do not have polyols in the composition of soluble cytosol carbohydrates, in contrast to the asco- and basidiomycetes (Jennings 1985). In the membrane lipids composition of mesophilic fungus A. niger proportion of PA increased significantly under OS conditions, while proportions of the PE and PC decreased, and unsaturation degree of the main phospholipids increased (Ianutsevich et al. 2016a), while in thermophilic R. miehei we did not observe significant changes in the composition of membrane lipids (Fig. 6) and their unsaturation degree (Table 2). Therefore, in this study we demonstrated that polyols are necessary for adaptation to OS in thermophilic fungus, similar to the OS response of mesophilic fungi (Ianutsevich et al. 2016a). However, unlike mesophilic fungi, there were no significant changes in the membrane lipid composition of thermophilic R. miehei under OS conditions.
Response of the studied thermophile to cold shock is different from mesophilic fungi. For mesophiles (Jennings 1985; Hayashi and Maeda 2006; Ianutsevich et al. 2016a) the activation of the HOG pathway and the accumulation of glycerol, similar to OS response, was shown in response to CS. A significant decrease in the level of trehalose was observed for the thermophilic fungus, while there was no noticeable increase in the amount of glycerol or other polyols (Fig. 1), which indicates either the sufficiency of the trehalose level for protection against CS or the inhibition of glycerol synthesis in thermophilic fungus under cold shock conditions. Similar changes in the composition of membrane lipids under CS were observed in thermophilic R. miehei and previously studied mesophilic A. niger (Ianutsevich et al. 2016a)–increase in the proportion of PA (Fig. 5) and the unsaturation degree of membrane phospholipids (Table 2). The difference between the cold shock responses of mesophilic and thermophilic fungi is in the osmolyte composition changes–glycerol was not involved in adaptation to CS in thermophilic R. miehei.
In mycelial fungi response to oxidative shock created by hydrogen peroxide includes a two- to threefold increase in catalase activity (Angelova et al. 2005). As shown previously, the oxidative shock created by hydrogen peroxide did not affect the composition of osmolytes in A. niger, but reduced their amounts (Ianutsevich et al. 2016a), and in thermophilic R. miehei oxidative shock reduced the level of trehalose significantly (Fig. 7). Oxidative shock led to similar changes in the composition of membrane lipids in both thermopile as mesophile, the proportion of PA increased and PC decreased, while the unsaturation degree of phospholipids did not change significantly (Fig. 7 Table 2). Thus thermophilic and mesophilic fungi have similar responses to OxS.
It is worth noting, that all of the studied stressors caused a significant drop in trehalose level in thermophilic fungus, and only osmotic shock led to the production of polyols–glycerol and arabitol. These data show that a high initial level of trehalose might be sufficient for adaptation to oxidative and cold, but not osmotic shocks.
PA can fulfil both the regulatory and structural functions (Balogh et al. 2013; Csoboz et al. 2013; Glatz et al. 2016). Non-bilayer lipids are a crucial part of membranes and are of key importance for vesicular transport and membrane remodeling, for membranes fusion and fission (Putta et al. 2016). Characteristic feature of the membrane lipids composition of the thermophilic fungus under optimal growth conditions was a high proportion of non-bilayer phospholipids—PA and PE, accounting for about 50% of the total (Fig. 4). Considering that the proportion of sterols is about 20%, the proportion of the bilayer lipids accounts for no more than 30%, indicating the unusual structure of the membranes of this thermophilic micromycete. An interesting take at the role of non-bilayer lipids, necessary for the integral membrane proteins functioning, was given after describing a new cubic phase—an ordered three-dimensional water–lipid system in which lipids are organized into a strongly curved, interlaced bilayer network (Landau and Rosenbusch 1996). Moreover, in R. miehei proportion of unidentified phospholipid X1, containing glycoside group, reached 10–18% of the total, but the proportion of sphingolipids did not exceed 5%. In response to different shocks, the proportion of PA was not reduced significantly, while under CS and OxS conditions it even increased. These data indicate the importance of PA not just for thermophilia, but also for the adaptation to other various stressors.
Sterols are polyfunctional compounds involved in the processes of growth and reproduction, biofilm formation, virulence (Rella et al. 2016; Rodrigues 2018). Sterols affect the viscosity of the membranes and are involved in the formation of rafts together with sphingolipids (Alvarez et al. 2007). The sterols and triterpenoids fraction in R. miehei is presented mainly by ergosterol (up to 70% of the total) and four minor sterols. However, the composition of the fraction differs from that of other Zygomycetes. Six pathogenic Mucorales have up to 14 components in the composition of sterols (Müller et al. 2018), while only 2 of them can be found in R. miehei—ergosterol and ergosta-7,22-dien-3β-ol. The studied stressors did not affect the qualitative composition of sterols, however, under CS and OS conditions a significant decrease in the proportion of ergosterol was observed, while more saturated ergosta-7,22-dien-3β-ol increased. Previously, an increase in the proportions of dihydroergosterols with a decrease in ergosterol was observed during adaptation to elevated cultivation temperatures in basidiomycete Favolaschia manipularis (Senik et al. 2019), which, together with our data, confirms the participation of sterols in adaptive stress response. The fungi are able to produce (Huang et al. 2012; Li et al. 2013) and transform (Parra et al. 2009) diverse triterpenoids. In addition, some fungi, such as phytopathogenic Phytophthora cactorum, unable to epoxidize squalene, can accumulate polycyclic isopentenoids produced by the plant, which, as the authors suggest, stimulate growth of the fungus (Nes et al. 1982). We have found no information about the presence of the ergosta-5,7,9(11),22-tetraene and ergosta-3,5,7,9-tetraene in fungi and their possible functions, but in basidiomycete Suillus luteus other triterpenoids without hydroxyl group (ergosta- 3,5,7,9(11),22-pentaene, ergosta- 2,5,7,9(11),14,22-hexaene) were found (Nieto and Ávila 2008).
Thus, obtained data showed the importance of polyols for the adaptation of thermophiles to OS, despite the high content of trehalose in the mycelium. All studied stress impacts, as well as previously described HS (Yanutsevich et al. 2014), resulted in a decrease in the trehalose amount in the fungal mycelium, indicating greater sensitivity of the trehalose synthesis to stressors. High proportion of PA was observed under optimal growth conditions and did not decrease under the influence of various stress factors, even increasing as a result of CS and OS, which shows the importance of these non-bilayer phospholipids not only for thermophilia but also for stress adaptation. In addition, a major change in the composition of sterols under CS and OS conditions was observed: a significant decrease in the proportion of ergosterol and increase in the proportion of ergosta-7,22-dien-3β-ol. Taken together, these data show the active participation of osmolytes and membrane lipids in the adaptation to stress in thermophiles.
Abbreviations
- CL:
-
Cardiolipins
- CS:
-
Cold shock
- HS:
-
Heat shock
- LPE:
-
Lysophosphatidylethanolamines
- OS:
-
Osmotic shock
- OxS:
-
Oxidative shock
- PA:
-
Phosphatidic acids
- PC:
-
Phosphatidylcholines,
- PE:
-
Phosphatidylethanolamines
- PI:
-
Phosphatidylinositols
- PS:
-
Phosphatidylserines
- SL:
-
Sphingolipids
- St:
-
Sterols
- X1:
-
Unidentified lipid
References
Alvarez FJ, Douglas LM, Konopka JB (2007) Sterol-rich plasma membrane domains in fungi. Eukaryot Cell 6:755–763. https://doi.org/10.1128/EC.00008-07
Angelova M, Pashova SB, Spasova BK, Vassilev S, Slokoska LS (2005) Oxidative stress response of filamentous fungi induced by hydrogen peroxide and paraquat. Mycol Res 109:150–158. https://doi.org/10.1017/S0953756204001352
Balogh G, Péter M, Glatz A, Gombos I, Török Z, Horváth I, Harwood JL, Vigh L (2013) Key role of lipids in heat stress management. FEBS Lett 587:1970–1980. https://doi.org/10.1016/j.febslet.2013.05.016
Brown SR, Fritz SC (2019) Eukaryotic organisms of continental hydrothermal systems. Extremophiles 23:367–376. https://doi.org/10.1007/s00792-019-01101-y
Carquin M, D’Auria L, Pollet H, Bongarzone ER, Tyteca D (2016) Recent progress on lipid lateral heterogeneity in plasma membranes: from rafts to submicrometric domains. Prog Lipid Res 62:1–24. https://doi.org/10.1016/j.plipres.2015.12.004
Crowe JH (2007) Trehalose as a “chemical chaperone”: fact and fantasy. Adv Exp Med Biol 594:143–158. https://doi.org/10.1007/978-0-387-39975-1_13
Csoboz B, Balogh GE, Kusz E, Gombos I, Peter M, Crul T, Gungor B, Haracska L, Bogdanovics G, Torok Z, Horvath I, Vigh L (2013) Membrane fluidity matters: hyperthermia from the aspects of lipids and membranes. Int J Hyperth 29:491–499. https://doi.org/10.3109/02656736.2013.808765
Douglas LM, Konopka JB (2014) Fungal membrane organization: the eisosome concept. Annu Rev Microbiol. https://doi.org/10.1146/annurev-micro-091313-103507
Duran R, Cary JW, Calvo AM (2010) Role of the osmotic stress regulatory pathway in morphogenesis and secondary metabolism in filamentous fungi. Toxins (Basel) 2:367–381. https://doi.org/10.3390/toxins2040367
Elbein AD, Pan YT, Pastuszak I, Carroll D (2003) New insights on trehalose: a multifunctional molecule. Glycobiology 13:17R–27R. https://doi.org/10.1093/glycob/cwg047
Ernst R, Ejsing CS, Antonny B (2016) Homeoviscous adaptation and the regulation of membrane lipids. J Mol Biol 428:4776–4791. https://doi.org/10.1016/j.jmb.2016.08.013
Feofilova EP, Tereshina VM (1999) Thermophilicity of mycelial fungi in the context of biochemical adaptation to thermal stress (review). Appl Biochem Microbiol 35:486–494
Garton GA, Goodwin TW, Lijinsky W (1951) Studies in carotenogenesis. 1. General conditions governing β-carotene synthesis by the fungus Phycomyces blakesleeanus Burgeff. Biochem J 48:154–163. https://doi.org/10.1042/bj0480154
Glatz A, Pilbat A, Németh GL, Vince-Kontár K, Jósvay K, Hunya Á, Udvardy A, Gombos I, Péter M, Balogh G, Horváth I, Vígh L, Török Z (2016) Involvement of small heat shock proteins, trehalose, and lipids in the thermal stress management in Schizosaccharomyces pombe. Cell Stress Chaperon 21:327–338. https://doi.org/10.1007/s12192-015-0662-4
Hayashi M, Maeda T (2006) Activation of the HOG pathway upon cold stress in Saccharomyces cerevisiae. J Biochem 139:797–803. https://doi.org/10.1093/jb/mvj089
Hazel JR (1995) Thermal adaptation in biological membranes: is homeoviscous adaptation the explanation? Annu Rev Physiol 57:19–42. https://doi.org/10.1146/annurev.ph.57.030195.000315
Huang L, Li J, Ye H, Li C, Wang H, Liu B, Zhang Y (2012) Molecular characterization of the pentacyclic triterpenoid biosynthetic pathway in Catharanthus roseus. Planta 236:1571–1581. https://doi.org/10.1007/s00425-012-1712-0
Ianutsevich EA, Danilova OA, Groza NV, Tereshina VM (2016a) Membrane lipids and cytosol carbohydrates in Aspergillus niger under osmotic, oxidative, and cold impact. Microbiology (Russian Fed) 85:302–310. https://doi.org/10.1134/S0026261716030152
Ianutsevich EA, Kotlova ER, Danilova OA, Tereshina VM, Groza NV (2016b) Heat shock response of thermophilic fungi: membrane lipids and soluble carbohydrates under elevated temperatures. Microbiology 162:989–999. https://doi.org/10.1099/mic.0.000279
Iturriaga G, Suárez R, Nova-Franco B (2009) Trehalose metabolism: from osmoprotection to signaling. Int J Mol Sci 10:3793–3810. https://doi.org/10.3390/ijms10093793
Jennings DH (1985) Polyol metabolism in fungi. Adv Microb Physiol. 25:149–193. https://doi.org/10.1016/S0065-2911(08)60292-1
Kogej T, Stein M, Volkmann M, Gorbushina AA, Galinski EA, Gunde-Cimerman N (2007) Osmotic adaptation of the halophilic fungus Hortaea werneckii: role of osmolytes and melanization. Microbiology 153:4261–4273. https://doi.org/10.1099/mic.0.2007/010751-0
Kooijman EE, Chupin V, de Kruijff B, Burger KNJ (2003) Modulation of membrane curvature by phosphatidic acid and lysophosphatidic acid. Traffic 4:162–174. https://doi.org/10.1034/j.1600-0854.2003.00086.x
Landau EM, Rosenbusch JP (1996) Lipidic cubic phases: a novel concept for the crystallization of membrane proteins. Proc Natl Acad Sci 93:14532–14535. https://doi.org/10.1073/pnas.93.25.14532
Li Y-X, Himaya S, Kim S-K (2013) Triterpenoids of marine origin as anti-cancer agents. Molecules 18:7886–7909. https://doi.org/10.3390/molecules18077886
McMahon HT, Gallop JL (2005) Membrane curvature and mechanisms of dynamic cell membrane remodelling. Nature 438:590–596. https://doi.org/10.1038/nature04396
Müller C, Neugebauer T, Zill P, Lass-Flörl C, Bracher F, Binder U (2018) Sterol composition of clinically relevant mucorales and changes resulting from posaconazole treatment. Molecules 23:1218. https://doi.org/10.3390/molecules23051218
Nes WD, Saunders GA, Heftmann E (1982) Role of steroids and triterpenoids in the growth and reproduction of Phytophthora cactorum. Lipids 17:178–183. https://doi.org/10.1007/BF02535100
Nichols BW (1963) Separation of the lipids of photosynthetic tissues: Improvements in analysis by thin-layer chromatography. Biochim Biophys Acta Spec Sect Lipids Relat Subj 70:417–422. https://doi.org/10.1016/0926-6542(63)90060-X
Nicolson GL (2014) The fluid-mosaic model of membrane structure: still relevant to understanding the structure, function and dynamics of biological membranes after more than 40 years. Biochim Biophys Acta Biomembr 1838:1451–1466. https://doi.org/10.1016/j.bbamem.2013.10.019
Nieto I, Ávila I (2008) Determination of fatty acids and triterpenoid compounds from the fruiting body of Suillus luteus. Rev Colomb Química 37:297–304
Nwaka S, Holzer H (1998) Molecular biology of trehalose and the trehalases in the yeast Saccharomyces cerevisiae. Prog Nucleic Acid Res Mol Biol 58:197–237. https://doi.org/10.1016/s0079-6603(08)60037-9
Parra A, Rivas F, Garcia-Granados A, Martinez A (2009) Microbial transformation of triterpenoids. Mini Rev Org Chem 6:307–320. https://doi.org/10.2174/157019309789371569
Patel TK, Williamson JD (2016) Mannitol in plants, fungi, and plant–fungal interactions. Trends Plant Sci 21:486–497. https://doi.org/10.1016/j.tplants.2016.01.006
Putta P, Rankenberg J, Korver RA, van Wijk R, Munnik T, Testerink C, Kooijman EE (2016) Phosphatidic acid binding proteins display differential binding as a function of membrane curvature stress and chemical properties. Biochim Biophys Acta Biomembr 1858:2709–2716. https://doi.org/10.1016/j.bbamem.2016.07.014
Rampelotto P (2013) Extremophiles and extreme environments. Life 3:482–485. https://doi.org/10.3390/life3030482
Rella A, Farnoud AM, Del Poeta M (2016) Plasma membrane lipids and their role in fungal virulence. Prog Lipid Res 61:63–72. https://doi.org/10.1016/j.plipres.2015.11.003
Rodrigues ML (2018) The multifunctional fungal ergosterol. MBio 9:1–5. https://doi.org/10.1128/mBio.01755-18
Rubio-Texeira M, Van Zeebroeck G, Thevelein JM (2016) 10 trehalose metabolism: enzymatic pathways and physiological functions. In: Hoffmeister D (ed) Biochemistry and molecular biology. Springer, Cham, pp 191–277. https://doi.org/10.1007/978-3-319-27790-5_10
Senik SV, Psurtseva NV, Shavarda AL, Kotlova ER (2019) Role of lipids in the thermal plasticity of basidial fungus Favolaschia manipularis. Can J Microbiol 65:870–879. https://doi.org/10.1139/cjm-2019-0284
Singer SJ, Nicolson GL (1972) The fluid mosaic model of the structure of cell membranes. Science 175:720–731. https://doi.org/10.1126/science.175.4023.720
Smolyanyuk EV, Bilanenko EN, Tereshina VM, Kachalkin AV, Kamzolkina OV (2013) Effect of sodium chloride concentration in the medium on the composition of the membrane lipids and carbohydrates in the cytosol of the fungus Fusarium sp. Microbiology 82:600–608. https://doi.org/10.1134/S0026261713050111
Tansey MR, Brock TD (1972) The upper temperature limit for eukaryotic organisms. Proc Natl Acad Sci USA 69:2426–2428. https://doi.org/10.1073/pnas.69.9.2426
Tereshina VM, Memorskaya AS, Kotlova ER, Feofilova EP (2010) Membrane lipid and cytosol carbohydrate composition in Aspergillus niger under heat shock. Microbiology 79:40–46. https://doi.org/10.1134/S0026261710010066
Tereshina VM, Memorskaya AS, Kotlova ER (2011) The effect of different heat influences on composition of membrane lipids and cytosol carbohydrates in mycelial fungi. Microbiology 80:455–460. https://doi.org/10.1134/S0026261711040199
Thevelein JM (1996) Regulation of trehalose metabolism and its relevance to cell growth and function. In: Brambl R, Marzluf GA (eds) Biochemistry and molecular biology. Springer, Berlin, pp 395–420. https://doi.org/10.1007/978-3-662-10367-8_19
Vigh L, Escribá PV, Sonnleitner A, Sonnleitner M, Piotto S, Maresca B, Horváth I, Harwood JL (2005) The significance of lipid composition for membrane activity: new concepts and ways of assessing function. Prog Lipid Res 44:303–344. https://doi.org/10.1016/j.plipres.2005.08.001
Weber AP, Horst RJ, Barbier GG, Oesterhelt C (2007) Metabolism and metabolomics of eukaryotes living under extreme conditions. Int Rev Cytol 256:1–34. https://doi.org/10.1016/S0074-7696(07)56001-8
Welte MA, Gould AP (2017) Lipid droplet functions beyond energy storage. Biochim Biophys Acta Mol Cell Biol Lipids 1862:1260–1272. https://doi.org/10.1016/j.bbalip.2017.07.006
Yancey PH (2005) Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J Exp Biol 208:2819–2830. https://doi.org/10.1242/jeb.01730
Yancey PH, Siebenaller JF (2015) Co-evolution of proteins and solutions: protein adaptation versus cytoprotective micromolecules and their roles in marine organisms. J Exp Biol 218:1880–1896. https://doi.org/10.1242/jeb.114355
Yanutsevich EA, Memorskaya AS, Groza NV, Kochkina GA, Tereshina VM (2014) Heat shock response in the thermophilic fungus Rhizomucor miehei. Microbiology 83:498–504. https://doi.org/10.1134/S0026261714050282
Acknowledgements
The study was partially supported by the Russian Foundation for Basic Research (Project 18-04-00488).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Albers.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ianutsevich, E.A., Danilova, O.A., Kurilov, D.V. et al. Osmolytes and membrane lipids in adaptive response of thermophilic fungus Rhizomucor miehei to cold, osmotic and oxidative shocks. Extremophiles 24, 391–401 (2020). https://doi.org/10.1007/s00792-020-01163-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-020-01163-3