Abstract
The deleterious effects of ionizing radiation are a major concern of the modern world. In the last decades, outstanding interest has been given to developing new therapeutic tools designed for protection against the toxic effects of ionizing radiation. Deinococcus spp. are among the most radioresistant organisms on Earth, being able to survive extreme doses of radiation, 1000-fold higher than most vertebrates. The molecular mechanisms underlying DNA repair and biomolecular protection, which are responsible for the remarkable radioresistance of Deinococcus bacteria, have been a debatable subject for the last 60 years. This paper is focused on the most recent findings regarding the molecular background of radioresistance and on Deinococcus bacteria response to oxidative stress. Novel proteins and genes involved in the highly regulated DNA repair processes, and enzymatic and non- enzymatic antioxidant systems are presented. In addition, a recently proposed mechanism that may contribute to oxidative damage protection in Deinococcus bacteria is discussed. A better understanding of these molecular mechanisms may draw future perspectives for counteracting radiation-related toxicity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As only few radioprotectors are currently available for therapy, in the last decades there has been an outstanding interest in developing new agents to decrease the side effects of exposure to ionizing radiation in high-risk individuals. These could be accidentally or professionally exposed to ionizing radiation and include: cleanup crews involved in nuclear mishaps or attacks, astronauts, international flight crews and pilots, nuclear power plant personnel, and some medical professionals (e.g., radiologists).
Furthermore, ionizing radiation used in cancer therapy injures normal tissues surrounding the tumor, which represents a cause for treatment toxicity and a limiting factor that ultimately dictates the dose, volume and technique of radiation. There is an urgent need for developing efficient radioprotectors, which could be administered before radiation exposure for protecting normal cells. The advantages of efficient radioprotective agents would be enormous, since they would allow a higher and more efficient dose to be used and decrease the side effects of radiotherapy. Mitigators are radioprotective agents that are given after exposure to counteract the toxic effects of radiation and to increase survival after a nuclear hazard.
Resistance to ionizing radiation damage varies from cell to cell, and from organism to organism. Physiological factors able to predict the ability of a particular cell to survive radiation-induced damage are yet controversial. It appears that the more complex is an organism, the more is it sensitive to radiation-induced damage. Vertebrates show greater sensitivity to the effects of DNA-damaging agents, such as ionizing radiation, UV light and desiccation. While humans cannot survive a whole-body exposure of 10 Gy (Gray; the SI unit of absorbed radiation dose) and 200 Gy is lethal for most bacteria, Deinococcus radiodurans can survive acute exposures of 15,000 Gy (Daly et al. 1994) and chronic exposures of 60 Gy/h (Venkateswaran et al. 2000). Ionizing radiation, UV light and desiccation generate reactive oxygen species (ROS), which have extremely harmful effects on all macromolecules in a cell: proteins, DNA, RNA and lipids (Du and Gebicki 2004; Daly et al. 2007; Bosshard et al. 2010; Krisko and Radman 2010).
The first species of the Deinococcaceae family to be discovered, D. radiodurans, has been identified in 1956 during a study that aimed to test if gamma radiation could be used as a method of sterilization (Anderson et al. 1956). Deinococcus radiodurans (DR) was found in a can of meat that had been exposed to a dose of radiation that presumably killed all known organisms. Later, it was found that DR could survive doses of radiation of up to 15,000 Gy. This extreme radiation resistance seems to be an evolutionary adaptation to dehydration, since no such highly radioactive natural environment has yet been discovered on Earth (Mattimore and Battista 1996). Genome sequences have been published for six members of the Deinococcus genus: D. radiodurans (White et al. 1999), D. geothermalis (White et al. 1999; Makarova et al. 2007; de Groot et al. 2009), D. proteolyticus (Copeland et al. 2012), D. wulumuqiensis (Xu et al. 2013), D. xibeiensis (Hu et al. 2013) and D. phoenicis (Stepanov et al. 2014).
This paper aims to focus on the molecular background of resistance to ionizing radiation in Deinococcus species and on the influence of oxidative damage on cells.
Mechanisms underlying radioresistance
Ionizing radiation generates wide-ranging molecular damage in a cell, affecting DNA, RNA, proteins and lipids. Hence, to elucidate the mechanism that allows some organisms to survive exposures to high doses of radiation, a large number of factors are to be taken into account. The main mechanisms proposed suggest that radioresistant organisms:
-
(a)
have unique DNA repair mechanisms, far more effective than those of other organisms;
-
(b)
use enzymatic and nonenzymatic antioxidant systems;
-
(c)
possess a highly efficient cellular cleansing system, which exports damaged nucleotides out of the cell.
Proteins involved in genome preservation and DNA repair mechanisms in Deinococcus bacteria
Extreme exposures to desiccation and ionizing radiation cause DNA single-strand breaks and double-strand breaks (DSBs) (Mattimore and Battista 1996), resulting in hundreds of short DNA fragments (Battista et al. 1999). It has been observed that Deinococcus bacteria accumulate approximately the same amount of genomic damage as radiosensitive bacteria (Daly 2009). Typically, 100 Gy generates approximately 1 DSB/haploid genome (Ghosal et al. 2005).
Notably, each cell of D. radiodurans bacteria has at least two genome copies, a feature which is mandatory for DSB repair by recombination. However, contrary to what would be expected, a higher number of genome copies do not enhance radioresistance in D. radiodurans (Harsojo et al. Harsojo and Matsuyama 1981).
Deinococcus DNA repair system is similar to that of the radiosensitive E.coli and Schewanella oneidensis (Daly 2009; Ghosal et al. 2005) and what is even more intriguing is that the DSB repair system in D. radiodurans resembles that of yeast cells (Symington et al. 2014).
Basically, DSBs repair in D. radiodurans is mainly achieved by extended synthesis-dependent strand annealing (ESDSA), which is as a two-step process. The first stage involves a Pol I-dependent mechanism necessary for fragment reassembly. Essentially, a helicase (UvrD), a 5′-3′ exonuclease (RecJ), RecA and two DNA polymerases, Pol I and Pol III, are required for this step. The second process is RecA dependent and results in mature, circular chromosomes (Zahradka et al. 2006). However, there is evidence that a large number of DSBs are repaired prior to ESDSA through single-strand annealing, a process which generates relatively large DNA fragments, which probably serve afterward as substrates for ESDSA (Daly and Minton 1996).
The RecA protein of D. radiodurans (DrRecA) has an essential role in DNA repair after extreme irradiation. The loss of RecA function entails a dramatic decrease in radiation resistance (Earl et al. 2002). Briefly, DrRecA binds to double-stranded DNA (dsDNA) and, in the presence of ATP, forms long helical filaments. DrRecA forms filaments on dsDNA that nucleate much faster than E. RecA (EcRecA), but extend slower (Hsu et al. 2011). These filaments exhibit interconvertible active and inactive states. Addition of single-stranded DNA (ssDNA) activates ATP hydrolysis of the DrRecA–dsDNA filament (Ngo et al. 2013). The inactive state of RecA may provide a new point of regulation and further studies are needed to uncover its regulatory mechanisms (Fig. 1).
The RecA-dependent double-strand break repair mechanism in D. radiodurans. In normal conditions, double-stranded genomic DNA is bound by DrRecA in an inactive state. Following exposure to ionizing radiation, double-strand breakage of genomic DNA may occur. The helicase UvrD is thought to unwind the DNA duplex, along with other DNA helicases; following DNA unwinding, RecJ digests the exposed 5′-DNA, creating 3′-overhangs. Single-stranded proteins (SSBs) wrap around the 3′-overhang. The presence of ssDNA in the cell activates the ATPase activity of DrRecA. RecA transfer from dsDNA to ssDNA is mediated by the RecFOR pathway (process not shown in this figure). The RecA-mediated strand invasion of a homologous duplex forms a migrating D-loop. DNA synthesis process is catalyzed by Pol I and Pol III. The newly synthesized single strands, which progressively detach from the D-loop, anneal with complementary strands. The resulting DNA fragments are joined into circular chromosomes by RecA-dependent crossovers. [Modified from ref. Ngo et al. 2013 with permission from The American Society for Biochemistry and Molecular Biology, Inc. and based on data from ref. Slade and Radman (2011)]
Proteins structurally and functionally similar to DrRecA are found in virtually all organisms, whether eukaryotic, prokaryotic or archaeal (Seitz et al. 1998). Most likely, evolutionary adaptation to different environmental stresses inflicted mutations on recA gene. In D. radiodurans, RecA protein displays a much different approach to DNA repair, because it is needed for genome restoration after prolonged periods of desiccation (Mattimore and Battista 1996), whereas RecA homologs in eukaryotes are only necessary for the repair of replication errors.
Comparative genomic studies show that D. geothermalis RecA (DgeRecA) protein has 87.6 % identity and 95.9 % similarity with DrRecA protein; Deinococcus murrayi RecA (DmuRecA) protein has 86.9 % identity and 96.9 % similarity with DrRecA protein. All three enzymes, DrRecA, DgeRecA and DmuRecA show more than 50 % identity to the EcRecA protein (Gutman et al. 1994; Wanarska et al. 2011). In vitro studies show that, unlike EcRecA, DrRecA has a preference toward binding dsDNA when placed in a reaction mixture containing both dsDNA and ssDNA (Kim 2006). In contrast to DrRecA’s binding preference, DgeRecA and DmuRecA bind more readily to ssDNA when both ssDNA and dsDNA are present in the same reaction mixture. DgeRecA and DmuRecA proteins are able to hydrolyze ATP and dATP in the presence of ssDNA and in the DNA strand exchange conditions (Wanarska et al. 2011). These findings suggest that RecA-mediated DNA repair mechanisms may differ from one Deinococcus species to another.
Piechura et al. demonstrated that RecA mutations affecting the Asp residue at positions 276 (D276), D276A and D276N increase radiation resistance of E. coli mutant strains up to 3 kGy (Piechura et al. 2015). Further studies should be performed to determine if mutations at equivalent sites on RAD51, the eukaryotic homolog of RecA, have a similar effect on radiation resistance.
RecFOR pathway includes RecF, RecO and RecR. DrRecR is a recombination mediator protein (RMP) with a dimeric architecture in solution. RecR binds RecO, forming a complex with a four-to-two stoichiometry. RecO binding strongly stimulates the formation of RecR tetramers. The crystal structure of the RecOR complex reveals a RecR tetrameric ring with one RecO monomer on either side, which can switch from an “open” conformation to a “closed” one. This hetero-hexameric complex interacts with a single-stranded DNA-binding protein (SSB)-coated ssDNA substrate. Subsequent to ssDNA binding within the RecR ring, the RecOR complex is locked in the “open” conformation (Radzimanowski et al. 2013).
DrRecQ possesses an unusual architecture of the helicase catalytic core domain, essential for the ATPase and DNA-unwinding abilities of RecQ proteins (Wang et al. 2013). Moreover, the DNA-binding motif of DrRecQ displays interdomain flexibility, which may be involved in regulating DNA recognition (Chen et al. 2014).
RecX is considered to be involved in the switch between stress response and normal metabolism in D. radiodurans, inhibiting the expression of some proteins involved in the DNA repair process (RecA, SSB, PprA) and enhancing the expression of some metabolism-related proteins (Sheng et al. 2009).
DdrB (DNA damage response B) is an SSB, unique to Deinococcus species. In D. radiodurans, proteomic analysis showed that, among DNA repair proteins, DdrB is the second most abundant radiation-induced protein. This result indicates vigorous protection conferred to ssDNA fragments as the basic defense strategy against DNA damage of D. radiodurans (Basu and Apte 2012). DdrB from D. geothermalis was found to contain an unusual ssDNA- binding fold that is structurally and topologically different from that of all other SSBs characterized to date. DdrB is able to promote strand annealing of complementary ssDNA fragments (Sugiman-Marangos and Junop 2010). Therefore, it has been inferred that DdrB may play an important role in single-strand annealing DNA repair process (Bouthier de la Tour et al. 2011), by analogy with the eukaryotic Rad52 protein (Singleton et al. 2002). Single molecule measurements indicate that SSBs can act as a sliding platform that shifts on DNA strands via a “sliding-with-bulge” mechanism and recruits the interacting proteins in DNA recombination and repair processes (Xu et al. 2010; Zhou et al. 2011). DdrB in D. geothermalis and DdrB in D. radiodurans have amino acid sequences that are 72 % identical, indicating that these two proteins probably display similar DNA-binding characteristics (Norais et al. 2009; Sugiman-Marangos & Junop 2010). Furthermore, the crystal structure of DdrB in complex with ssDNA and the pentameric structure of DdrB was found to assemble into a higher-order oligomeric structure that wraps around ssDNA (Sugiman-Marangos et al. 2013). Furthermore, DdrB has been reported to inhibit RecJ exonuclease activity and protect oligonucleotides (Jiao et al. 2012), shielding ssDNA produced during repair processes. Sugiman-Marangos et al. propose a mechanism of single-strand annealing in which DdrB adopts a two-ring forming oligomeric structure, similar to the eukaryotic protein Rad52 (Sugiman-Marangos et al. 2013). Recently, a novel protein encoded by the dr1245 gene was identified, which may have chaperon activity toward DdrB and possibly other substrates. Studies showed that a Δdr1245 strain is impaired in growth, but radioresistance is not affected (Norais et al. 2013).
PprI (or IrrE) protein is specific to bacteria belonging to Deinococcaceae and Thermaceae families. It is a regulatory protein that stimulates expression of recA and other DNA repair genes (Hua et al. 2003), as well as genes involved in various metabolic pathways and enhances catalase activity (Lu et al. 2009, 2012), acting like a general switch. PprI has proteolytic activity and in the presence of Mn2+ ions, PprI cleaves DdrO, hence suppressing its function. The cleavage site seems to be the same in D. radiodurans and D. deserti. It is thought that PprI and DdrO mediate a novel and unique DNA damage response pathway, which differs markedly from the LexA-SOS response system in E.coli (Wang et al. 2015a, b).
In D. radiodurans, DNA repair proteins, protein kinases and phosphoproteins form a multiprotein complex (Kota and Misra 2008). Following exposure to ionizing radiation, protein kinases seem to be up-regulated, while phosphatases and phosphodiesterases tend to be down-regulated (Kamble et al. 2009).
Ferric uptake regulator (Fur) controls the expression of genes involved in the uptake of some divalent ions and regulates the intracellular redox status. Fur family proteins generally act as repressors in the response to Fe(II), Mn(II), Zn(II) and Ni(II), referred to as Fur, Mur, Zur and Nur, respectively (Ahn et al. 2006; Diaz-Mireles et al. 2004; Ul Hussain Shah et al. 2014; Fuangthong& Helman Fuangthong and Helmann 2003). Two Fur homologs have been described in D. radiodurans: PerR-like protein and dr0865, a Mur regulator protein.
DR0171, a helix–turn–helix (HTH)-containing DNA-binding protein, plays the role of a regulator of the transcriptional response to radiation damage in D. radiodurans (Lu et al. 2011). It may mediate the switch from DNA degradation to synthesis and repair (Udupa et al. 1994). This switch is an important step in DNA repair mechanisms, still incompletely characterized. An intriguing fact is that this protein seems to be Deinococcus specific and it may stand as an example of adaptation to DNA-damaging agents (Makarova et al. 2001).
Other recently characterized genes and proteins involved in DNA repair and oxidative stress response in Deinococcus radiodurans are included in Table 1.
Potential role of small RNAs in radiation resistance
Small non-coding RNAs (sRNAs) regulate DNA repair gene expression (Gottesman 2005). The small size of sRNA genes (400 bp) would allow them to remain largely undamaged after exposure to extreme levels of ionizing radiation, compared to protein-encoding genes (1000–2000 bp). The functional sRNAs transcribed from DNA fragments after irradiation may contribute to genome reconstruction. Eight sRNAs showed differential expression after acute ionizing radiation (15 kGy) in D. radiodurans and seven of these sRNAs were also confirmed in D. geothermalis. Potential sRNA binding targets were predicted to be mRNAs that encode proteins contributing to radiation survival, such as RecA, RuvA and RadA. Other predicted mRNAs are involved in stress response mechanisms or global gene regulation, for instance the transcriptional regulator DR0074 belonging to the TetR family, DR0097 protein belonging to the ferredoxin-NADP+ reductase family and the MerR family transcriptional regulator (Tsai et al. 2015). Luan et al. also reported the up-regulation of sRNA expression in response to high doses of radiation, mainly including tRNAs, which may modulate the translation of RNA to protein. In addition, many putative antisense-RNAs may play important roles in various processes involved in radiation resistance, e.g., glycolysis pathway, homologous recombination and manganese transport throughout the cells (Luan et al. 2014).
Dual effects of ROS on cells
ROS include free radicals: superoxide radicals (O ·−2 ), hydroxyl radicals (HO·) and hydrogen peroxide (H2O2), with the last oxygen species being highly reactive, although it does not contain unpaired electrons in the molecular orbital.
Ionizing radiation generates ROS, which, depending on the context, can act both as oxidants and reductants. ROS can damage cell structures and promote genomic instability and DNA mutations (Hunt et al. 1998). Radiation directly causes only 20 % of DNA damage, while the remaining 80 % is indirectly caused by HO· (Ghosal et al. 2005).
In bacteria, manganese (Mn) and iron (Fe) ions significantly influence the cellular redox status, as represented in Fig. 2.
Mn and Fe cellular redox cycling in response to ionizing radiation. (1) Radiation-triggered water radiolysis generates HO· (hydroxyl radical), H+ (proton) and \({\text{e}}_{\text{aq}}^{-}\) (hydrated electron); (2) HO· radicals may react with each other and form H2O2, which, due to its uncharged molecular structure, can further diffuse throughout the cell membranes; (3) Fenton reaction: Fe2+ + H2O2 → Fe3+ + HO· + HO− (hydroxide anion); (4) Haber–Weiss reaction: 2 Fe3+ + H2O2 → 2 Fe(II) + O2 + 2 H+; (5) Mn2+ oxidation: Mn2+ + O ·−2 + 2 H+ → Mn3+ + H2O2; (6) Mn3+ reduction: 2 Mn3+ + H2O2 → 2 Mn2+ + O2 + 2 H+; (7) 2H2O2 → 2H2O + O2 (reaction catalyzed by the catalase enzyme); (8) dissolved O2 generates O ∙−2 , through a reaction with \({\text{e}}_{\text{aq}}^{ - }\); (9) the resulting O ∙−2 can reform H2O2 if protons are available in the intracellular medium. Modified from Daly et al. 2007 with permission from the publisher
O ∙−2 and H2O2 do not react directly with DNA (Imlay 2008), but with proteins that contain exposed iron–sulfur (Fe–S), haem groups, cysteine residues or cation-binding sites, where an iron-catalyzed oxidation reaction occurs (Culotta and Daly 2013). Due to the fact that O ·−2 is negatively charged, it has greater damaging potential than H2O2 for Fe–S clusters.
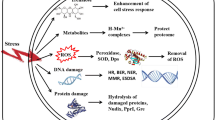
Although cellular macromolecules cannot be protected from the direct damaging effects of UV or ionizing radiation, antioxidants can increase the survival of cells exposed to such types of radiation. Antioxidants decrease ROS‑mediated toxicity, secondary to the radiation direct effects (Fig. 3).
A better understanding of the interactions between ROS and antioxidants from Deinococcus species may have far-reaching implications in increasing the efficiency of radiotherapy and in protecting normal tissue from radiation-induced damage.
Antioxidant systems in Deinococcus bacteria
DNA reassembly in Deinococcus bacteria requires a multitude of DNA repair enzymes. This is why survival of these bacteria most probably depends on their ability to maintain structural and functional integrity of proteins involved in DNA repair, DNA synthesis and diverse metabolic pathways. Hence, resistance to oxidative stress is thought to result from the work of very efficient systems that protect proteins from oxidative damage and allows Deinococcus bacteria to survive high doses of radiation.
Over the last decades, scientists have gathered proof that the amount of protein damage is better correlated with the survival of bacteria after irradiation than the amount of DNA damage (Daly 2009). More likely, stress-resistance capacity of Deinococcus bacteria has not been improved by acquiring unique DNA repair mechanisms, but by compiling diverse ROS-scavenging systems from exogenous sources (Makarova et al. 2007; Daly 2012; Krisko and Radman 2010).
To explain the radiation resistance of these bacteria, some studies focused on the antioxidant enzymes in Deinococcus-Thermus phylum, such as catalases and peroxidases.
The katE (dr1998) gene encodes the most abundant catalase in D. radiodurans, important for H2O2 scavenging (Markillie et al. 1999). The expression of katE is co-regulated by ROS-responding proteins PerR and OxyR. Thus, in normal conditions, when cells do not require excess catalase, its expression is repressed by DrPerR and, when Deinococcus cells are exposed to oxidative stress, OxyR promotes the expression of katE (Liu et al. 2014a, b). DtxR (DR2539) and DR0865 are also suggested to regulate KatE activity (Chen et al. 2010). This mechanism may partially explain D. radiodurans’ ability to adapt to fluctuant environment stresses (Liu et al. 2014a, b).
The superoxide dismutase (SOD) gene may be strongly induced in the early stages of irradiation, considered as an early defense strategy against oxidative stress damage caused by ROS (Luan et al. 2014).
It has been proved that the genome of the radiosensitive bacterium Shewanella oneidensis encodes similar types of catalases and peroxidases as D. radiodurans; however, they differ widely in resistance to oxidative stress and IR (D10 = 0.07 kGy for S. oneidensis. D10 = dose required for a 90 % inactivation of viable CFU). Therefore, other systems must explain the radioresistance of these bacteria. Non-enzymatic antioxidants may complement AOE, playing a role in oxidative stress resistance, as ROS scavengers. Non-enzymatic small-molecule antioxidants in D. radiodurans include pyrroloquinoline quinone (Misra et al. 2004; Rajpurohit et al. 2013b), carotenoids and low molecular weight manganese complexes.
Structures and roles of carotenoids in some species of Deinococcus bacteria
Carotenoids are natural pigments found in photosynthetic and non-photosynthetic organisms. While in the first category of bacteria and plants they play a role in photoprotection against the damaging effects of free radicals created by light and oxygen, in non-phototrophic bacteria carotenoids play a role in cellular protection as ROS scavengers. Furthermore, it is well established that carotenoids protect and stabilize membranes of thermophilic bacteria against photooxidation and extremely high temperatures (Tian and Hua 2010).
Carotenoids are reported to be present in most extremophiles discovered on Earth and a lot of work has been done to clarify their contribution to the mechanisms that allow extreme bacteria to survive exposure to different stress types. It has been noticed that carotenoids in Deinococcus bacteria have unique structures and there have been attempts made to understand the complete scheme of structure–activity correlations (Tian and Hua 2010).
Deinococcus radiodurans synthesizes deinoxanthin (Fig. 3), a unique ketocarotenoid (Lemee et al. 1997; Lysenko et al. 2011). There is evidence that the intracellular level of protein oxidation following treatment with H2O2 in a carotenogenesis-deficient mutant was approximately 25 % higher than that in the wild type (Tian et al. 2009). Deinoxanthin shows higher antioxidant activity than lycopene and ß-carotene (Tian et al. 2007). These findings prove that carotenoids in D. radiodurans inhibit oxidative damage to proteins, but how carotenoids interact with proteins or other macromolecules in vivo remains an open question.
Orange carotenoid protein (OCP) is a photoactive protein discovered in cyanobacteria, containing a keto-carotenoid as photoresponsive chromophore (Kirilovsky and Kerfeld 2013). The carotenoid–protein interaction in OCP is explained by the presence of hydrogen bonds between the carbonyl group of the carotenoid (hydroxyechinenone) and specific amino acids from the C-terminal domain (Wilson et al. 2010). Notably, there are some structural similarities between hydroxyechinenone and deinoxanthin, e.g., the carbonyl group in C-4 position. This analogy could imply that a similar protein might exist in Deinococcus bacteria, although none has yet been identified.
Notably, deinoxanthin was demonstrated to act as a prooxidant at high doses, inducing ROS production in cancer cells (Choi et al. 2014) and algae (Li et al. 2015). An extract from D. radiodurans containing deinoxanthin induced apoptotic effects in HepG2 (human liver carcinoma cell line), PC-3 (human prostate cancer cell line) and HT-29 (human colorectal adenocarcinoma cell line) cells (Choi et al. 2014). The apparent conflict between prooxidant and antioxidant activities of deinoxanthin needs further investigation and a better understanding of the antioxidant activity of carotenoids in Deinococcus bacteria should elucidate this contradiction.
Recently, there has been an increased interest in elucidating the biosynthetic pathways of carotenoids in D. radiodurans. CruF, CrtD and CrtO enzymes have been experimentally confirmed (Sun et al. 2009). However, the enzyme that catalyzes the hydroxylation of C-2 of the ß-ring has not been identified and this carotenoid 2-hydroxylase is presumed to be an entirely new enzyme type (Tian and Hua 2010).
Carotenoid biosynthetic enzymes could potentially be used in biotechnology for creating genetically engineered strains and genetically modified economic crops with increased resistance to DNA damage. Moreover, carotenoid β-ring hydroxylase and ketolase are promiscuous candidates for the synthesis of functional xanthophylls (Misawa 2011). Geranylgeranyl diphosphate synthase and other enzymes involved in carotenoid biosynthetic pathway can be used as molecular targets in genetic engineering strategies for increasing deinoxanthin production in Deinococcus microorganisms (Liu et al. 2014a).
The effects of carotenoids from Deinococcus bacteria on the immune function have not yet been evaluated. Therefore, this might be a subject of future studies, since there is evidence that carotenoids can influence immune function through their ability to regulate membrane fluidity and gap-junctional communication (Chew and Park 2014). Astaxanthin, a carotenoid with a similar chemical structure to deinoxanthin, augments T-dependent antigen-specific humoral immune responses (Jyonouchi et al. 1995).
A recent study brings a new perspective regarding the mechanism of action of carotenoids, stating that their oxidative breakdown products or their metabolites might be the actual bioactive agents. Thus, oxygen-rich, ß carotene–oxygen copolymers might be responsible for the antioxidant properties of these compounds. The same study also states that these copolymers appear to be common to most, if not all carotenoid compounds (Johnston et al. 2014).
Role of Mn(II) species. Mn/Fe homeostasis
Metal ions are important enzyme cofactors for many proteins involved in DNA synthesis and repair, ROS scavenging and electron transport (Ghosal et al. 2005). Therefore, they play an essential role in regulating intracellular redox cycling, in resistance to oxidative stress and ionizing radiation.
The intracellular Mn(II)/Fe(II) ratio in radiation-sensitive bacteria ranges from 0.0001 to 0.007, while this ratio varied from 0.033 to 0.523 in Deinococcus species. These ratios were 70- to 300-fold higher in Deinococcus species as compared to those in radiation-sensitive bacteria (Daly et al. 2004).
Whereas Fe2+ levels in radioresistant and radiosensitive bacteria are approximately the same, Mn2+ concentrations in D. radiodurans are up to 30 mM, 15–150 times higher than in radiosensitive bacteria. An excess of Mn2+ is usually toxic to a normal cell. Although the precise mechanism of Mn2+ cell toxicity is not well understood, it is thought to be associated with Mn2+ interaction with other cations, such as Fe2+, Zn2+ and Cu2+ (Banh et al. Banh et al. 2013). With cooperation from ROS, these cations can bind to proteins and can convert side-chain amine groups into carbonyls. Increased protein carbonyl content is used as a biomarker of oxidative stress (Dalle-Donne et al. 2006) and has been associated with several diseases, such as aging (Stadtman 2006), diabetes (Pandey et al. 2010), cancer (Ma et al. 2013) and cardiovascular disease (Barsotti et al. 2011). Therefore, Deinococccus’ “proteome shields” may serve as a model for future treatment strategies of the above-mentioned pathologies.
In Deinococcus bacteria, the unusual Mn2+ accumulation has been strongly correlated with ionizing radiation resistance (Daly et al. 2004, 2007; Daly 2009). Daly et al. (2010) demonstrated in vitro that there is a link between radiation resistance, manganese accumulation and protein protection in radioresistant microorganisms. They demonstrated that ultrafiltered, protein-free preparations of D. radiodurans cell extracts, containing Mn2+, inorganic phosphate, peptides, amino acids, nucleosides and bases have the ability to prevent in vitro ionizing radiation-induced protein carbonylation purified from E. coli and preserve the restriction endonuclease BamH1 activity up to 12.5 kGy. The ultrafiltrate prevented protein oxidation, but did not significantly protect DNA at high doses of ionizing radiation (Daly et al. 2010). This finding is in agreement with previous studies which highlighted the antioxidant activity of Mn2+ complexes. For example, it was demonstrated that amino acids and peptides form complexes with Mn2+, which scavenge ROS (Berlett et al. 1990), and Mn2+ and orthophosphate form complexes which can remove superoxide anion (O ·2 ˉ) (Barnese et al. 2008).
Furthermore, human Jurkat T cells survived γ-rays exposure (16 Gy) after treatment with DR ultrafiltrate. This finding proves that metabolic interventions in animal cells could bring about new perspectives to anti-aging and radiotherapies, reducing the damage to non-cancerous cells and the side effects of radiotherapy (Daly et al. 2010).
In D. radiodurans, Mn/Fe homeostasis seems to be tightly controlled by a network of regulatory genes, which cooperate with antioxidant systems under extreme conditions and contribute to cell survival. Two types of Mn transporter systems have been identified: the proton (H+)-dependent (MntH) transporter and the ATP-binding cassette (ABC) transporters (Sun et al. 2012).
DR2539 protein is a negative regulator of Mn-dependent transporter genes (dr2283) and katE (dr1998), and a positive regulator of Fe-dependent transporter genes (dr1219 and drb0125) (Chen et al. 2010). DR2539 binds specifically to the promoter of the MntH gene (Sun et al. 2012).
PerR-like protein acts as a negative transcription regulator of katE and dps (DNA-binding proteins from starved cells), which protect DNA from oxidative damage and play a role in maintaining metal homeostasis (Liu et al. 2014b). Most probably, the following four proteins: PerR, OxyR, DR2539 (transcriptional regulators of the diphtheria toxin repressor family; Chen et al. 2010) and DR0865 (Fur homolog) regulate the oxidative stress response of D. radiodurans, but further research is needed to describe the interactions between them. Moreover, comparative genomics studies showed that the drperR genes had an above 50 % sequence identity with genes from D. radiodurans, D. gobiensis, D. geothermalis and D. proteolyticus (Liu et al. 2014b). A dr0865 mutant had a decreased survival rate after exposure to various stress factors, such as manganese stress, hydrogen peroxide and gamma and UV irradiation than the wild-type R1 strain. Dr0865 gene regulates the manganese efflux pump gene mntE and Mn ABC transporter genes, and plays an important role in preserving the homeostasis of intracellular Mn2+ (Ul Hussain Shah et al. 2014).
In D. radiodurans, the main energetic source is represented by the amino acids and peptides resulting from proteolysis. Ionizing radiation triggers proteolytic activity, which increases intracellular concentration of amino acids and peptides resulting from degradation of proteins damaged under oxidative stress conditions (Liu et al. 2003). These metabolic products bind Mn2+ and form low molecular weight (LMW)—Mn2+ complexes, which will subsequently assist the recovery process of Deinococcus cells.
Some progress has recently been made in consolidating the theory that LMW–Mn2+ complexes play a significant role in radiation resistance. Using advanced paramagnetic resonance techniques, Sharma et al. (2013) demonstrated in vivo that Mn2+ ions exist in D. radiodurans predominantly as LMW complexes with orthophosphate and nitrogenous metabolites (nucleosides and free bases released as DNA damage products).
SODs have been shown to have potential use in providing differential protection for normal cells or tissues against the side effects induced by radiation-induced damage and anticancer therapies (Lefaix et al. 1996; Delanian et al. 1994; Carpenter et al. 2005; Epperly et al. 2004). Thus, a few classes of low molecular weight SOD mimetics have been synthesized and characterized: Mn(III) salens, Mn(III) meso-porphyrins, and Cu(II) diisopropylsalicylate (CuDIPS) and nonmetal-based nitroxide free radicals (Cabello et al. 2007; Salvemini and Cuzzocrea 2003). Hence, the finding of LMW–Mn2+ complex in Deinococcus bacteria, with a higher antioxidant activity than that of SOD, could bring potential benefit for the protection of normal cells surrounding the irradiated area in cancer therapy. LMW manganese complexes were also shown to scavenge superoxide ions in other bacterial species: Bacillus subtilis, Neisseria gonorrhoeae, E. coli, Staphylococcus aureus and the yeast Saccharomyces cerevisiae (Lisher and Giedroc 2013).
Furthermore, based on the composition of extracts from D. radiodurans, Gaidamakova et al. (2012) reconstituted a radioprotective complex containing Mn2+ and a decapeptide (DP; H-Asp-Glu-His-Gly-Thr-Ala-Val-Met-Leu- Lys-OH) in the orthophosphate (Pi) buffer. The Mn2+ complex preserved antigenic structures in aqueous preparations of bacteriophage lambda, Venezuelan equine encephalitis virus and Staphylococcus aureus during irradiation (25–40 kGy) and stimulated B and T cell-dependent protection against methicillin-resistant S. aureus (MRSA) in mice. These results might bring new perspectives to the industry of vaccines produced by irradiation, since the major drawback of this method is the destruction of the immunogenic epitopes needed to mount protective immune responses.
Therefore, an increased Mn/Fe ratio has a twofold benefit for the cell. LMW–Mn complexes act as efficient scavengers of H2O2 and O ·2 ˉ and decrease HO· production by the Fenton reaction. Moreover, Mn2+ plays a role in chromosomal condensation by neutralizing the electrostatic repulsion between phosphate groups in D. radiodurans chromosomes (Daly et al. 2004).
Extracellular dGMP increases Deinococcus radiodurans resistance to oxidative damage
Following irradiation, damaged bases and nucleotides resulting from digestion of DNA free ends are exported from Deinococcus cells and further processed by DRB0067, an extracellular nuclease, associated with the outer membrane (Mitchel 1978; Li et al. 2013). The extracellular nuclease in D. radiodurans seems to be secreted by a pathway that is similar to the Sec pathway in E. coli (Wang et al. 2015a, b). ssDNA fragments are released in the extracellular medium following exposures to doses of ionizing radiation greater than 5 kGy; they are degraded to deoxyribonucleosides 5′- monophosphate (dNMPs) by the same extracellular nuclease and these dNMPs reenter the cell once DNA synthesis has started (Li et al. 2013). Notably, DRB0067 is the only extracellular nuclease found in Deinococcus bacteria (Li et al. 2013).
Mainly, the extracellular nuclease serves two purposes in D. radiodurans, namely:
-
1.
processes extracellular DNA fragments, protecting the bacteria from mutagenesis and
-
2.
creates free dNMPs, which reenter the cell and help in the genome reconstruction process (Li et al. 2013).
Extracellular deoxyriboguanosine monophosphate (dGMP) plays an important role in the Deinococcal defense against oxidation-induced damage. Deinococcus cells that had dGMPs in the extracellular medium showed a 57-fold higher resistance to H2O2 treatment than cells that were grown in a medium lacking dGMPs. The presence of extracellular dGMPs increases the expression of drb0067 and other genes associated with resistance to oxidative damage, including genes involved in the regulation of manganese/iron homeostasis (Li et al. 2013; Fig. 4).
Extracellular dGMP (deoxyriboguanosine monophosphate) effects on Deinococcus cells (based on data from ref. Li et al. 2013)
It is possible that extracellular dGMP, after its reincorporation into cells, could be converted to cyclic guanosine monophosphate (cGMP), which could act as a second messenger, protecting cells from oxidative damage, as demonstrated in eukaryotic cells (Curatola et al. 2011; Li et al. 2013).
Conclusions
Current knowledge suggests that effective management of oxidative stress caused by γ radiation in Deinococcus bacteria and the mechanisms conferring radiation resistance result from the cross-talk of various molecular pathways. Further studies should focus on understanding the nature of the effective interactions between gene expression and metabolic regulatory pathways, instead of addressing only a single level of regulation. Also, the synergy of the DNA repair system in Deinococcus bacteria is not completely understood and may reveal important data about radiation resistance.
Taking into account that the genome of D. radiodurans was sequenced in 1999 (White et al. 1999), missing data might be due to the limitations of sequencing techniques. Therefore, next-generation sequencing techniques could be essential for revealing new genes involved in DNA repair mechanisms, ROS response pathways, carotenoid biosynthesis, etc. Moreover, correlations between genomic and proteomic research could unravel exciting findings regarding adaptive and evolutionary mechanisms and cellular response to oxidative stress. More work needs to be done to identify the secondary messengers and signaling enzymes, as well as their contributions to Deinococccus’ extreme resistance to ionizing radiation.
Further studies should bring new insight into cancer therapy and radioprotection as molecular mechanisms behind radioresistance are uncovered. The cellular redox status after irradiation may have major implications in explaining the contribution of metabolism to the phenotypic changes associated with radiation-induced DNA and protein damage, as well as suggesting new molecular targets for anticancer therapy.
References
Ahn BE, Cha J, Lee EJ, Han AR, Thompson CJ, Roe JH (2006) Nur, a nickel responsive regulator of the Fur family, regulates superoxide dismutases and nickel transport in Streptomyces coelicolor. Mol Microbiol 59:1848–1858
Anderson A, Nordan H, Cain R, Parrish G, Duggan D (1956) Studies on a radioresistant micrococcus. I. Isolation, morphology, cultural characteristics, and resistance to gamma radiation. Food Technol 10:575–578
Appukuttan D, Seo HS, Jeong S, Im S, Joe M, Song D, Choi J, Lim S (2015) Expression and Mutational Analysis of DinB-Like Protein DR0053 in Deinococcus radiodurans. PLoS ONE 10(2):e0118275. doi:10.1371/journal.pone.0118275
Banh A, Chavez V, Doi J, Nguyen A, Hernandez S et al (2013) Manganese (Mn) oxidation increases intracellular Mn in Pseudomonas putida GB-1. PLoS ONE 8:e77835. doi:10.1371/journal.pone.0077835
Barnese K, Gralla EB, Cabelli DE, Valentine JS (2008) Manganous phosphate acts as a superoxide dismutase. J Am Chem Soc 130(14):4604–4606
Barsotti A, Fabbi P, Fedele M, Garibaldi S, Balbi M, Bezante GP, Risso D, Indiveri F, Ghigliotti G, Brunelli C (2011) Role of advanced oxidation protein products and Thiol ratio in patients with acute coronary syndromes. Clin Biochem 44(8–9):605–611
Basu B, Apte SK (2012) Gamma radiation-induced proteome of Deinococcus radiodurans primarily targets DNA repair and oxidative stress alleviation. Mol Cell Proteomics 11(M111):011734
Battista JR, Earl AM, Park MJ (1999) Why is Deinococcus radiodurans so resistant to ionizing radiation? Trends Microbiol 7:362–365
Berlett BS, Chock PB, Yim MB, Stadtman ER (1990) Manganese(II) catalyzes the bicarbonate-dependent oxidation of amino acids by hydrogen peroxide and the amino acid-facilitated dismutation of hydrogen peroxide. Proc Natl Acad Sci USA 87(1):389–393
Bosshard F, Riedel K, Schneider T, Geiser C, Bucheli M, Egli T (2010) Protein oxidation and aggregation in UVA-irradiated Escherichia coli cells as signs of accelerated cellular senescence. Environ Microbiol 12:2931–2945
Bouthier de la Tour C, Boisnard S, Norais C, Toueille M, Bentchikou E, Vannier F, Cox MM, Sommer S, Servant P (2011) The deinococcal DdrB protein is involved in an early step of DNA double strand break repair and in plasmid transformation through its single-strand annealing activity. DNA Repair (Amst) 10:1223–1231
Cabello CM, Bair WB 3rd, Wondrak GT (2007) Experimental therapeutics: targeting the redox Achilles heel of cancer. Curr Opin Investig Drugs 8:1022–1037
Carpenter M, Epperly MW, Agarwal A, Nie S, Hricisak L, Niu Y, Greenberger JS (2005) Inhalation delivery of manganese superoxide dismutase-plasmid/liposomes protects the murine lung from irradiation damage. Gene Ther 12:685–693
Chen H, Wu R, Xu G, Fang X, Qiu X, Guo H, Tian B, Hua Y (2010) DR2539 is a novel DtxR-like regulator of Mn/Fe ion homeostasis and antioxidant enzyme in Deinococcus radiodurans. Biochem Biophys Res Commun 396:413–418
Chen SC, Huang CH, Yang CS, Way TD, Chang MC, Chen Y (2014) Crystal structure of Deinococcus radiodurans RecQ helicase catalytic core domain: the interdomain flexibility. BioMed Res Int. doi:10.1155/2014/342725
Chew BP, Park JS (2014) Carotenoid action on the immune response. J Nutr 134(1):257S–261S
Choi YJ, Hur JM, Lim S, Jo M, Kim DH, Choi JI (2014) Induction of apoptosis by deinoxanthin in human cancer cells. Anticancer Res 34(4):1829–1835
Copeland A et al (2012) Complete genome sequence of the orange-red pigmented, radioresistant Deinococcus proteolyticus type strain (MRP(T)). Stand Genomic Sci 6(2):240–250
Cox MM, Keck JL, Battista JR (2010) Rising from the Ashes: DNA Repair in Deinococcus radiodurans. PLoS Genet 6(1):e1000815. doi:10.1371/journal.pgen.1000815
Culotta VC, Daly MJ (2013) Manganese complexes: diverse metabolic routes to oxidative stress resistance in prokaryotes and yeast. Antioxid Redox Signal 19(9):933–944
Curatola AM, Xu J, Hendricks-Munoz KD (2011) Cyclic GMP protects endothelial progenitors from oxidative stress. Angiogenesis 14:267–279
Dalle-Donne I, Aldini G, Carini M, Colombo R, Rossi R, Milzani A (2006) Protein carbonylation, cellular dysfunction, and disease progression. J Cell Mol Med 10:389–406
Daly MJ (2009) A new perspective on radiation resistance based on Deinococcus radiodurans. Nat Rev Microbiol 7:237–245
Daly MJ (2012) Death by protein damage in irradiated cells. DNA Repair (Amst) 11(1):12–21
Daly MJ, Minton KW (1996) An alternative pathway of recombination of chromosomal fragments precedes recA-dependent recombination in the radioresistant bacterium Deinococcus radiodurans. J Bacteriol 178:4461–4471
Daly MJ, Ouyang L, Fuchs P, Minton KW (1994) In vivo damage and recA-dependent repair of plasmid and chromosomal DNA in the radiation- resistant bacterium Deinococcus radiodurans. J Bacteriol 176:3508–3517
Daly MJ, Gaidamakova EK, Matrosova VY, Vasilenko A, Zhai M et al (2004) Accumulation of Mn(II) in Deinococcus radiodurans facilitates gamma-radiation resistance. Science 306:1025–1028. doi:10.1126/science.1103185
Daly MJ, Gaidamakova EK, Matrosova VY, Vasilenko A, Zhai M et al (2007) Protein Oxidation Implicated as the Primary Determinant of Bacterial Radioresistance. PLoS Biol 5(4):e92. doi:10.1371/journal.pbio.0050092
Daly MJ, Gaidamakova EK, Matrosova VY, Kiang JG, Fukumoto R, Lee DY, Wehr NB, Viteri GA, Berlett BS, Levine RL (2010) Small-molecule antioxidant proteome-shields in Deinococcus radiodurans. PLoS One 5(9):e12570. doi:10.1371/journal.pone.0012570
Das AD, Misra HS (2012) DR2417, a hypothetical protein characterized as a novel β-CASP family nuclease in radiation resistant bacterium, Deinococcus radiodurans. Biochim Biophys Acta 7:1052–1061
de Groot A, Dulermo R, Ortet P, Blanchard L, Guérin P et al (2009) Alliance of Proteomics and Genomics to Unravel the Specificities of Sahara Bacterium Deinococcus deserti. PLoS Genet 5(3):e1000434. doi:10.1371/journal.pgen.1000434
Delanian S, Baillet F, Huart J, Lefaix JL, Maulard C, Housset M (1994) Successful treatment of radiation-induced fibrosis using liposomal Cu/Zn superoxide dismutase: clinical trial. Radiother Oncol 32:12–20
Diaz-Mireles E, Wexler M, Sawers G, Bellini D, Todd JD, Johnston AW (2004) The Fur-like protein Mur of Rhizobium leguminosarum is a Mn(2 +)-responsive transcriptional regulator. Microbiology 150:1447–1456
Du J, Gebicki JM (2004) Proteins are major initial cell targets of hydroxyl free radicals. Int J Biochem Cell Biol 36:2334–2343
Earl AM, Mohundro MM, Mian IS, Battista JR (2002) The IrrE protein of Deinococcus radiodurans R1 is a novel regulator of recA expression. J Bacteriol 184:6216–6224
Epperly MW, Carpenter M, Agarwal A, Mitra P, Nie S, Greenberger JS (2004) Intraoral manganese superoxide dismutase-plasmid/liposome (MnSOD-PL) radioprotective gene therapy decreases ionizing irradiation-induced murine mucosal cell cycling and apoptosis. In Vivo 18:401–410
Fuangthong M, Helmann JD (2003) Recognition of DNA by three ferric uptake regulator (Fur) homologs in Bacillus subtilis. J Bacteriol 185:6348–6357
Gaidamakova EK, Myles IA, McDaniel DP, Fowler CJ, Valdez PA, Naik S, Gayen M, Gupta P, Sharma A, Glass PJ, Maheshwari RK, Datta SK, Daly MJ (2012) Preserving immunogenicity of lethally irradiated viral and bacterial vaccine epitopes using a radio- protective Mn2+-Peptide complex from Deinococcus. Cell Host Microbe 12(1):117–124
Ghosal D, Omelchenko MV, Gaidamakova EK, Matrosova VY, Vasilenko A, Venkateswaran A, Zhai M, Kostandarithes HM, Brim H, Makarova KS, Wackett LP, Fredrickson JK, Daly MJ (2005) How radiation kills cells: survival of Deinococcus radiodurans and Shewanella oneidensis under oxidative stress. FEMS Microbiol Rev 29:361–375
Gottesman S (2005) Micros for microbes: non-coding regulatory RNAs in bacteria. Trends Genet 21:399–404
Gutman PD, Carroll JD, Masters CI, Minton KW (1994) Sequencing, targeted mutagenesis and expression of a recA gene required for the extreme radioresistance of Deinococcus radiodurans. Gene 141:31–37
Harsojo Kitayama S, Matsuyama A (1981) Genome multiplicity and radiation resistance in Micrococcus radiodurans. J Biochem 90:877–880
Hsu HF, Ngo KV, Chitteni-Pattu S, Cox MM, Li HW (2011) Investigating Deinococcus radiodurans RecA protein filament formation on double-stranded DNA by a real-time single-molecule approach. Biochemistry 50:8270–8280
Hu Y, Xu X, Song P, Jiang L, Zhang Z, Huang H (2013) Draft Genome Sequence of Deinococcus xibeiensis R13, a New Carotenoid-Producing Strain. Genome Announc. doi:10.1128/genomeA.00987-13
Hua YJ, Narumi I, Gao GJ, Tian B, Satoh K, Kitayama S et al (2003) PprI: a general switch responsible for extreme radioresistance of Deinococcus radiodurans. Biochem Bioph Res Commun 306:354–360
Hunt CR, Sim JE, Sullivan SJ, Featherstone T, Golden W, Von Kapp-Herr C, Hock RA, Gomez RA, Parsian AJ, Spitz DR (1998) Genomic instability and catalase gene amplifi cation induced by chronic exposure to oxidative stress. Cancer Res 58(17):3986–3992
Imlay JA (2008) Cellular defenses against superoxide and hydrogen peroxide. Annu Rev Biochem 77:755–776
Jiao J, Wang L, Xia W, Li M, Sun H, Xu G, Tian B, Hua Y (2012) Function and biochemical characterization of RecJ in Deinococcus radiodurans. DNA Repair (Amst) 11:349–356
Johnston JB, Nickerson JG, Daroszewski J, Mogg TJ, Burton GW (2014) Biologically Active Polymers from spontaneous carotenoid oxidation: a new frontier in carotenoid activity. PLoS ONE. doi:10.1371/journal.pone.0111346
Jyonouchi H, Sun S, Tomita Y, Gross MD (1995) Astaxanthin, a carotenoid without vitamin A activity, augments antibody responses in cultures including T-helper cell clones and suboptimal doses of antigen. J Nutr 125(10):2483–2492
Kamble VA, Misra HS (2010) The SbcCD complex of Deinococcus radiodurans contributes to radioresistance and DNA strand break repair in vivo and exhibits Mre11–Rad50 type activity in vitro. DNA Repair 9:488–494
Kamble VA, Rajpurohit YS, Srivastava AK, Misra HS (2009) Increased synthesis of signaling molecules coincides with reversible inhibition of nucleolytic activity during postirradiation recovery of Deinococcus radiodurans. FEMS Microbiol Lett 303:18–25
Kim JI (2006) Analysis of double stranded DNA-dependent activities of Deinococcus radiodurans RecA protein. J. Microbiol 44:508–514
Kirilovsky D, Kerfeld CA (2013) The Orange Carotenoid Protein: a blue-green light photoactive protein. Photochem Photobiol Sci 12(7):1135–1143
Kota S, Misra HS (2008) Identification of a DNA processing complex from Deinococcus radiodurans. Biochem Cell Biol 86:448–458
Krisko A, Radman M (2010) Protein damage and death by radiation in Escherichia coli and Deinococcus radiodurans. Proc Natl Acad Sci USA 107:14373–14377
Lefaix JL, Delanian S, Leplat JJ, Tricaud Y, Martin M, Nimrod A, Baillet F, Daburon F (1996) Successful treatment of radiation-induced fibrosis using Cu/Zn-SOD and Mn-SOD: an experimental study. Int J Radiat Oncol Biol Phys 35:305–312
Lemee L, Peuchant E, Clerc M, Brunner M, Pfander H (1997) Deinoxanthin: a new carotenoid isolated from Deinococcus radiodurans. Tetrahedron 53:919–926
Li M, Sun H, Feng Q, Lu H, Zhao Y, Zhang H, Xu X, Jiao J, Wang L, Hua Y (2013) Extracellular dGMP Enhances Deinococcus radiodurans Tolerance to Oxidative Stress. PLoS ONE 8(1):e54420. doi:10.1371/journal.pone.0054420
Li Y, Zhu H, Lei X, Zhang H, Guan C, Chen Z, Zheng W, Xu H, Tian Y, Yu Z, Zheng T (2015) The first evidence of deinoxanthin from Deinococcus sp. Y35 with strong algicidal effect on the toxic dinoflagellate Alexandrium tamarense. J Hazard Mater 290:87–95
Lisher JP, Giedroc DP (2013) Manganese acquisition and homeostasis at the host-pathogen interface. Front Cell Infect Microbiol 3:91. doi:10.3389/fcimb.2013.00091
Liu Y, Zhou J, Omelchenko MV, Beliaev AS, Venkateswaran A, Stair J, Wu L, Thompson DK, Xu D, Rogozin IB, Gaidamakova EK, Zhai M, Makarova KS, Koonin EV, Daly MJ (2003) Transcriptome dynamics of Deinococcus radiodurans recovering from ionizing radiation. Proc Natl Acad Sci USA 100:4191–4196
Liu C, Sun Z, Shen S, Lin L, Li T, Tian B, Hua Y (2014a) Identification and characterization of the geranylgeranyl diphosphate synthase in Deinococcus radiodurans. Lett Appl Microbiol 58(3):219–224
Liu C, Wang L, Li T, Lin L, Dai S, Tian B, Hua Y (2014b) A PerR-like protein involved in response to oxidative stress in the extreme bacterium Deinococcus radiodurans. Biochem Biophys Res Commun 450(1):575–580
Lu H, Gao G, Xu G, Fan L, Yin L, Shen B, Hua Y (2009) Deinococcus radiodurans PprI switches on DNA damage response and cellular survival networks after radiation damage. Mol Cell Proteomics 8(3):481–494
Lu H, Xia W, Chen H, Yin L, Zhao X, Xu G, Hua Y (2011) Characterization of the role of DR0171 in transcriptional response to radiation in the extremely radioresistant bacterium Deinococcus radiodurans. Arch Microbiol 193(10):741–750
Lu H, Chen H, Xu G, Shah AM, Hua Y (2012) DNA binding is essential for PprI function in response to radiation damage in Deinococcus radiodurans. DNA Repair (Amst) 77(2):139–145
Luan H, Meng N, Fu J, Chen X, Xu X et al (2014) Genome-Wide Transcriptome and Antioxidant Analyses on Gamma-Irradiated Phases of Deinococcus radiodurans R1. PLoS ONE 9(1):e85649. doi:10.1371/journal.pone.0085649
Lysenko VS, Chistyakov VA, Zimakov DV, Soier VG, Sazykina MA, Sazykina LMI, Sazykin IS, Krasnov VP (2011) Separation and Mass Spectrometry Identification of Carotenoid Complex from Radioresistant Bacteria Deinococcus radiodurans. J Anal Chem 66(13):49–52
Ma Y, Zhang L, Rong S et al (2013) Relation between gastric cancer and protein oxidation, DNA damage, and lipid peroxidation. Oxid Med Cell Longev 2013:543760. doi:10.1155/2013/543760
Makarova KS, Aravind L, Wolf YI, Tatusov RL, Minton K, Koonin EV, Daly MJ (2001) Genome of the extremely radiation resistant bacterium Deinococcus radiodurans viewed from the perspective of comparative genomics. Microbiol Mol Biol Rev 65:44–79
Makarova KS, Omelchenko MV, Gaidamakova EK, Matrosova VY, Vasilenko A et al (2007) Deinococcus geothermalis: the Pool of Extreme Radiation Resistance Genes Shrinks. PLoS ONE 2(9):e955. doi:10.1371/journal.pone.0000955
Markillie LM, Varnum SM, Hradecky P, Wong KK (1999) Targeted mutagenesis by duplication insertion in the radioresistant bacterium Deinococcus radiodurans: radiation sensitivities of catalase (katA) and superoxide dismutase (sodA) mutants. J Bacteriol 181:666–669
Masters CI, Moseley BE, Minton KW (1991) AP endonuclease and uracil DNA glycosylase activities in Deinococcus radiodurans. Mutat Res 254:263–272
Mattimore V, Battista JR (1996) Radioresistance of Deinococcus radiodurans: functions necessary to survive ionizing radiation are also necessary to survive prolonged desiccation. J Bacteriol 178:633–637
Misawa N (2011) Carotenoid β-ring hydroxylase and ketolase from marine bacteria—promiscuous enzymes for synthesizing functional xanthophylls. Mar Drugs 9(5):757–771
Misra HS, Khairnar NP, Barik A, Indira Priyadarsini K, Mohan H, Apte SK (2004) Pyrroloquinoline-quinone: a reactive oxygen species scavenger in bacteria. FEBS Lett 578(1–2):26–30
Mitchel RE (1978) The radiation-releasable cell wall nuclease of Micrococcus radiodurans. Purification and properties of the native enzyme. Biochim Biophys Acta 524:362–372
Ngo KV, Molzberger ET, Chitteni-Pattu S, Cox MM (2013) Regulation of Deinococcus radiodurans RecA protein function via modulation of active and inactive nucleoprotein filament states. J Biol Chem 288(29):21351–21366. doi:10.1074/jbc.M113.459230
Norais CA, Chitteni-Pattu S, Wood EA, Inman RB, Cox MM (2009) DdrB protein, an alternative Deinococcus radiodurans SSB induced by ionizing radiation. J Biol Chem 284:21402–21411
Norais C, Servant P, Bouthier-de-la-Tour C, Coureux P-D, Ithurbide S et al (2013) The Deinococcus radiodurans DR1245 Protein, a DdrB Partner Homologous to YbjN Proteins and Reminiscent of Type III Secretion System Chaperones. PLoS ONE 8(2):e56558. doi:10.1371/journal.pone.0056558
Onodera T, Satoh K, Ohta T, Narumi I (2013) Deinococcus radiodurans YgjD and YeaZ are involved in the repair of DNA cross-links. Extremophiles 77(1):171–179
Pandey KB, Mishra N, Rizvi SI (2010) Protein oxidation biomarkers in plasma of type 2 diabetic patients. Clin Biochem 43(4–5):508–511
Piechura JR, Tseng TL, Hsu HF, Byrne RT, Windgassen TA, Chitteni-Pattu S, Battista JR, Li HW, Cox MM (2015) Biochemical characterization of RecA variants that contribute to extreme resistance to ionizing radiation. DNA Repair (Amst) 26:30–43. doi:10.1016/j.dnarep.2014.12.001
Radzimanowski J, Dehez F, Round A, Bidon-Chanal A, McSweeney S, Timmins J (2013) An ‘open’ structure of the RecOR complex supports ssDNA binding within the core of the complex. Nucleic Acids Res 41(16):7972–7986
Rajpurohit YS, Misra HS (2013) Structure-function study of deinococcal serine/threonine protein kinase implicates its kinase activity and DNA repair protein phosphorylation roles in radioresistance of Deinococcus radiodurans. Int J Biochem Cell Biol 45(11):2541–2552
Rajpurohit YS, Desai SS, Misra HS (2013) Pyrroloquinoline quinone and a quinoprotein kinase support γ-radiation resistance in Deinococcus radiodurans and regulate gene expression. J Basic Microbiol 53(6):518–531
Salvemini D, Cuzzocrea S (2003) Therapeutic potential of superoxide dismutase mimetics as therapeutic agents in critical care medicine. Crit Care Med 31:S29–S38
Seitz EM, Brockman JP, Sandler SJ, Clark AJ, Kowalczykowski SC (1998) RadA protein is an archaeal RecA protein homolog that catalyzes DNA strand exchange. Genes Dev 12(9):1248–1253
Sheng D, Jao J, Li M, Xu P, Zhang J (2009) RecX is involved in the switch between DNA damage response and normal metabolism in D. radiodurans. J Biochem 146(3):337–342
Singleton MR, Wentzell LM, Liu Y, West SC, Wigley DB (2002) Structure of the single-strand annealing domain of human RAD52 protein. Proc Natl Acad Sci USA 99:13492–13497
Slade D, Radman M (2011) Oxidative Stress Resistance in Deinococcus radiodurans. Microbiol Mol Biol Rev 75(1):133–191
Stadtman ER (2006) Protein oxidation and aging. Free Radic Res 40(12):1250–1258
Stepanov VG, Vaishampayan P, Venkateswaran K, Fox GE (2014) Draft Genome Sequence of Deinococcus phoenicis, a Novel Strain Isolated during the Phoenix Lander Spacecraft assembly. Genome Announc. doi:10.1128/genomeA.00301-14
Sugiman-Marangos S, Junop MS (2010) The structure of DdrB from Deinococcus: a new fold for single-stranded DNA binding proteins. Nucleic Acids Res 55(10):3432–3440
Sugiman-Marangos SN, Peel JK, Weiss YM, Ghirlando R, Junop MS (2013) Crystal structure of the DdrB/ssDNA complex from Deinococcus radiodurans reveals a DNA binding surface involving higher-order oligomeric states. Nucleic Acids Res 41(21):9934–9944
Sun Z, Shen S, Tian B, Wang H, Xu Z, Wang L, Hua Y (2009) Functional analysis of γ-carotene ketolase involved in the carotenoid biosynthesis of Deinococcus radiodurans. FEMS Microbiol Lett 301:21–27
Sun H, Li M, Xu G, Chen H, Jiao J, Tian B, Wang L, Hua Y (2012) Regulation of MntH by a dual Mn(II)- and Fe(II)-dependent transcriptional repressor (DR2539) in Deinococcus radiodurans. PLoS ONE 7:e35057
Symington LS, Rothstein R, Lisby M (2014) Mechanisms and regulation of mitotic recombination in Saccharomyces cerevisiae. Genetics 198(3):795–835
Tian B, Hua Y (2010) Carotenoid biosynthesis in extremophilic Deinococcus-Thermus bacteria. Trends Microbiol 18(11):512–520
Tian B, Xu Z, Sun Z, Lin J, Hua Y (2007) Evaluation of the antioxidant effects of carotenoids from Deinococcus radiodurans through targeted mutagenesis, chemiluminescence, and DNA damage analyses. Biochim Biophys Acta 1770:902–911
Tian B, Sun Z, Shen S, Wang H, Jiao J, Wang L, Hu Y, Hua Y (2009) Effects of carotenoids from Deinococcus radiodurans on protein oxidation. Lett Appl Microbiol 49:689–694
Tsai CH, Liao R, Chou B, Contreras LM (2015) Transcriptional analysis of Deinococcus radiodurans reveals novel small RNAs that are differentially expressed under ionizing radiation. Appl Environ Microbiol 81(5):1754–1764. doi:10.1128/AEM.03709-14
Udupa KS, O’Cain PA, Mattimore V, Battista JR (1994) Novel ionizing radiation-sensitive mutants of Deinococcus radiodurans. J Bacteriol 176:7439–7446
Ul Hussain Shah AM, Zhao Y, Wang Y, Yan G, Zhang Q et al (2014) A Mur Regulator Protein in the Extremophilic Bacterium Deinococcus radiodurans. PLoS ONE 9(9):e106341. doi:10.1371/journal.pone.0106341
Venkateswaran A, McFarlan SC, Ghosal D, Minton KW, Vasilenko A, Makarova K, Wackett LP, Daly MJ (2000) Physiologic determinants of radiation resistance in Deinococcus radiodurans. Appl Environ Microbiol 66:2620–2626
Wanarska M, Krawczyk B, Hildebrandt P, Kur J (2011) RecA proteins from Deinococcus geothermalis and Deinococcus murrayi—cloning, purification and biochemical characterisation. BMC Mol Bio 12:17. doi:10.1186/1471-2199-12-17
Wang W, Hou H, Du Q, Zhang W, Liu G, Shtykova EV, Xu J, Liu P, Dong Y (2013) Solution small angle X-ray scattering (SAXS) studies of RecQ from Deinococcus radiodurans and its complexes with junction DNA substrates. J Biol Chem 288(45):32414–32423
Wang L, Tan H, Cheng K, Li M, Xu X, Wang J, Hua Y (2015a) Sec Pathway Influences the Growth of Deinococcus radiodurans. Curr Microbiol 70(5):651–656
Wang Y, Xu Q, Lu H, Lin L, Wang L, Xu H, Cui X, Zhang H, Li T, Hua Y (2015b) Protease Activity of PprI facilitates DNA damage response: Mn(2 +)-dependence and substrate sequence-specificity of the proteolytic reaction. PLoS ONE 10(3):e0122071. doi:10.1371/journal.pone.0122071
White O et al (1999) Genome sequence of the radioresistant bacterium Deinococcus radiodurans R1. Science 286:1571–1577
Wilson A, Kinney JN, Zwart PH, Punginelli C, D’Haene S, Perreau F, Klein MG, Kirilovsky D, Kerfeld CA (2010) Structural determinants underlying photoprotection in the photoactive orange carotenoid protein of cyanobacteria. J Biol Chem 285(24):18364–18375
Xu G, Lu H, Wang L, Chen H, Xu Z, Hu Y, Tian B, Hua Y (2010) DdrB stimulates single-stranded DNA annealing and facilitates RecA-independent DNA repair in Deinococcus radiodurans. DNA Repair 9(7):805–812
Xu X, Jiang L, Zhang Z, Shi Y, Huang H (2013) Genome Sequence of a Gamma- and UV-Ray-Resistant Strain, Deinococcus wulumuqiensis R12. Genome Announc. doi:10.1128/genomeA.00206-13
Zahradka K, Slade D, Bailone A et al (2006) Reassembly of shattered chromosomes in Deinococcus radiodurans. Nature 443(7111):569–573
Zhang H, Xu Q, Lu M, Xu X, Wang Y, Wang L, Zhao Y, Hua Y (2014) Structural and functional studies of MutS2 from Deinococcus radiodurans. DNA Repair 21:111–119
Zhou R, Kozlov AG, Roy R, Zhang J, Korolev S, Lohman TM, Ha T (2011) SSB functions as a sliding platform that migrates on DNA via reptation. Cell 146(2):222–232
Acknowledgments
This work received financial support through the project entitled “CERO—Career profile: Romanian Researcher”, Grant Number POSDRU/159/1.5/S/135760, cofinanced by the European Social Fund for Sectorial Operational Programme Human Resources Development 2007–2013. We are grateful to Dr. Gina Manda from the Radiobiology Laboratory of “Victor Babes” National Institute of Pathology, Bucharest, Romania, for her valuable suggestions and for revising the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Albers.
Rights and permissions
About this article
Cite this article
Munteanu, A.C., Uivarosi, V. & Andries, A. Recent progress in understanding the molecular mechanisms of radioresistance in Deinococcus bacteria. Extremophiles 19, 707–719 (2015). https://doi.org/10.1007/s00792-015-0759-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-015-0759-9