Abstract.
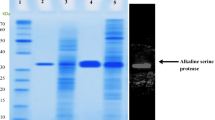
A gene, tayI, encoding a novel subtilisin-like protease, designated thermicin, from the extremely thermophilic bacterium Thermoanaerobacter yonseiensis KB-1 (DSM 13777) was cloned by using a sequence tag containing the consensus sequence of proteases. The gene consisted of 1,239 nucleotides, and the deduced amino acid sequence indicated that it is a preproenzyme with a 311-residue mature protein composed of canonical catalytic residues (Asp29, His64, and Ser252). Thermicin was overproduced in E. coli as a fusion protein with a histidine tag and purified by nickel nitrilotriacetic acid affinity chromatography. Thermicin from E. coli showed maximum proteolytic activity at 92.5°C and pH 9.0, and its half-life was 30 h at 80°C. In order to determine cleavage specificity, thermicin was incubated with insulin β chain, and the resulting peptides were analyzed by matrix assisted laser desorption/ionization-time of flight mass spectrometry. The carboxyl group side of the Val12, Leu15,17, Gly23, and Pro28 residues was cleaved. Thermicin is well known to hydrolyze Gly- and Pro-rich collagens. The K m and k cat /K m values of thermicin for the hydrolysis of the synthetic substrate L-Gly-Pro- p-nitroaniline were 54.16 µM and 142.05 (105 s–1 M–1), respectively, at 92.5°C and pH 9.0. Amino acid sequence comparison and phylogenetic analysis indicated that this enzyme belongs to a new subgroup with respect to its molecular evolution, when compared with previously characterized subtilisins. This result indicates that thermicin is a novel enzyme different from other thermostable proteases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Jang, H., Kim, B., Pyun, Y. et al. A novel subtilisin-like serine protease from Thermoanaerobacter yonseiensis KB-1: its cloning, expression, and biochemical properties. Extremophiles 6, 233–243 (2002). https://doi.org/10.1007/s00792-001-0248-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00792-001-0248-1