Abstract
Objective
There is a controversy over the influence of obesity on the periodontal treatment outcome in patients with chronic periodontitis (CP). The aim of the present systematic review was to evaluate the efficacy of non-surgical periodontal therapy (NSPT) in the management of CP among obese and non-obese patients.
Materials and methods
The addressed focused question was “What is the efficacy, of NSPT with respect to clinical, radiographic, biochemical, microbiological, and patient-centered outcomes in obese as compared to non-obese chronic periodontitis patients?” Databases were searched from 1977 up to and including December 2014 using relevant key indexing terms. Unpublished data, experimental studies, letters to the editor, review articles, case reports, and commentaries were excluded. Meta-analysis of three studies was performed.
Results
Five clinical studies were included. The total number of patients ranged between 30 and 260 individuals. The mean age of patients was between 42.5 and 48.8 years. In three studies, the clinical periodontal parameters (plaque index (PI), gingival bleeding index (GBI), periodontal pocket depth (PPD), and clinical attachment loss (CAL)) in obese and non-obese patients following NSPT was comparable. Meta-analysis of PPD and CAL among obese and non-obese subjects showed comparable outcomes (PPD P = 0.91, I 2 67.36 %; CAL P = 0.87, I 2 77.16 %). However, in three studies, NSPT resulted in a significantly better clinical periodontal outcome among non-obese subjects than obese subjects. The difference in the levels of serum pro-inflammatory cytokine levels (IL-1β, IL-6, TNF-α, IFN-γ, leptin, adiponectin, and CRP) among obese and non-obese patients following treatment for CP was inconsistent.
Conclusion
It remains unclear whether NSPT has a significantly higher impact on the clinical periodontal outcomes in obese patients than in non-obese patients with chronic periodontitis, given that the number of selected studies was relatively low and the reported findings were inconsistent.
Clinical relevance
Although the effect of obesity on the outcome of NSPT still remains unclear, nevertheless clinicians are prompted to manage obesity prior to and during periodontal treatment
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-surgical periodontal therapy (NSPT) remains the gold standard in the management of chronic periodontitis (CP). NSPT involves mechanical removal of bacterial biofilm and deposits with scaling and root planing (SRP), creating a local environment and microbiota harmonious with the periodontal health. NSPT results in the replacement of periodontal inflammatory tissue with highly perfused and collagen-rich connective tissues [1, 2]. This causes gingival tissue to shrink in the apical direction, towards the root surface [3]. Efficacy of NSPT in the management of periodontitis is established, and clinical trials have shown reduction of inflammation, pocket depth reduction, and clinical attachment gains following NSPT [4–7].
Obesity is defined as the accumulation of superfluous amounts of fat in the body, to a degree that may debilitate health [8, 9]. According to WHO criteria [8], individuals with body mass index (BMI) over 30 kg/m2 are categorized as obese. Around 300 million individuals are estimated to be obese globally [10]. Obesity is linked to increased morbidity and is culpable to major risk factor for different diseases including diabetes mellitus, hypertension, bone problems, and even cancers [11, 12]. Obese subjects show elevated levels of serum inflammatory biomarkers (secreted from adipocytes), which modulate immunologic, metabolic, and inflammatory responses [13–15]. Conversely, serum cytokine levels tend to decrease with weight loss [16]. Raised levels of pro-inflammatory cytokine levels result in a hyper-inflammatory state leading to increased susceptibility to infections and poor wound healing [14, 17–19]. Increased BMI and body fat have also been linked to periodontitis; however, the exact pathophysiological mechanism for this association is still unknown [20, 21]. The association between obesity and periodontal disease was first reported in obese Zucker rats by Perlstein and Bissada [22]. Recent systematic review has demonstrated the association between obesity and periodontal disease [23]. Laboratory-based investigations have suggested the increased levels of pro-inflammatory cytokines (such as interleukin (IL) 1, IL 6, IL 1 beta (β), tumor necrosis factor (TNF)) in obese patients to be the probable cause of increased periodontal destruction [24, 25]. Furthermore, increased cytokine levels are also associated with insulin resistance, leading to diabetes and increased risk of periodontal damage [26].
Studies [25, 27] have assessed the efficacy of NSPT in the management of chronic periodontitis among obese and non-obese patients. For instance, Zuza et al. [25] compared 52 patients with and without obesity for the treatment response of NSPT in the management of CP. The results showed comparable periodontal conditions among obese and non-obese subjects at 12 weeks follow-up [25]. Dias Goncalves et al. [27], however, comparing 48 obese and non-obese patients for periodontal treatment outcome, reported significantly higher periodontal pocket depths and clinical attachment loss in obese individuals as compared to non-obese subjects at 24 weeks follow-up. In this regard, it seems that there is a controversy over the influence of obesity on the periodontal treatment outcome in CP patients when compared to non-obese controls. In a systematic review by Suvan et al. [28] aimed at investigating the association between obesity and clinical periodontal outcomes, they concluded that there is insufficient clinical evidence with reference to the development of appropriate clinical protocols for the management of obese patients having chronic periodontitis. Therefore, the aim of the present study was to systematically review the efficacy of NSPT in the management of CP among obese and non-obese subjects.
Materials and methods
Focused question
Based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [29], a specific question was constructed according to the PICO (Participants, Interventions, Control, Outcomes) principle [30]. The addressed focused question was “What is the efficacy, of NSPT with respect to clinical, radiographic, biochemical, microbiological and patient-centered outcomes in obese as compared to non-obese chronic periodontitis patients?”
-
(P) Participants—It was essential for participants to be obese and have chronic periodontitis
-
Types of interventions—The intervention of interest was non-surgical periodontal therapy (NSPT).
-
(C) Control intervention—Non-surgical periodontal therapy (NSPT) in non-obese patients with chronic periodontitis
-
(O) Outcome measures
-
Primary—Changes in clinical attachment loss (CAL) and periodontal pocket depth (PPD) from the baseline measurement to follow-up.
-
Secondary—(1) Plaque index (PI), (2) gingival bleeding index (GBI), (3) bleeding on probing (BOP), (4) serum levels of pro-inflammatory cytokines, (5) bone loss, (6) bacterial counts, (7) pain and discomfort, and (8) esthetics were secondary outcomes.
Search protocol
In order to identify studies relevant to the PICO question, the MEDLINE database, the EMBASE database, the Cochrane Central Register of Controlled Trials (CENTRAL), Scopus, Web of Knowledge, and Google Scholar databases were electronically searched for available data. Databases were searched from 1977 up to and including December 2014 using different combinations of the following key words:
Filters: humans
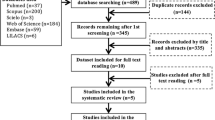
(“Periodontitis” [MeSH terms] OR “Periodontal Diseases” [MeSH terms]) AND (“Obesity” [MeSH terms] OR “Obese” OR “Body Mass Index”) AND (“Root planing” [MeSH terms] OR “Periodontal treatment” OR “Periodontal therapy”) OR (Adipokines [MeSH] OR Cytokines [MeSH]) OR “Bone loss” OR “periodontopathogens” OR “Patient-centered outcomes”. Titles and abstracts of studies identified using the above-mentioned protocol were screened by two authors (ZA and FV). Hand searching of the following journals was performed: Journal of Clinical Periodontology, Journal of Periodontology, Journal of Periodontal Research, Clinical Oral Investigation, Journal of Dental Research, Obesity Research & Clinical Practice, Disease Markers, and Saudi Medical Journal. Full texts of studies identified to be relevant by title and abstract were read by authors (ZA and FV) and evaluated in accordance with the following eligibility criteria: clinical studies, obese patient with chronic periodontitis (without systemic disease) receiving NSPT, and presence of control group. Letters to the Editor, historic reviews, commentaries, experimental studies, and case reports were excluded. Relevant original and review articles were also hand searched. This was done to identify any studies that could have remained unidentified in the previous step and checked for disagreement via discussion among the authors. A summary of the literature search strategy according to the PRISMA guidelines is shown in Fig. 1. The information from the accepted studies was tabulated according to the (1) study design, (2) subject demographics, (3) study groups, (4) mean BMI, (5) follow-up period, and (6) main outcome. Kappa scores (Cohen’s ĸ coefficient) were employed during full-text assessment to ensure eligibility and level of agreement between the reviewers [31, 32].
In order to answer the focused question, meta-analyses were conducted separately for each of the two outcomes: CAL and PPD. The mean differences between the reduction in PPD and gain in CAL in obese and non-obese patients were estimated as the effect size measures. Heterogeneity among the included studies for each outcome was assessed using Q-statistics and I 2 statistic. Meta-analyses of only three studies [27, 34, 35] were performed which reported overall mean values of clinical periodontal parameters. The remaining two studies [25, 33] reported overall site percentages for PPD and CAL. Therefore, due to non-uniformity in reported date, these studies were not incorporated in the meta-analyses. The authors were unable to perform meta-analyses with regard to biochemical outcomes (cytokine levels) because of considerable variation in the types and number of cytokines investigated in the included studies (Table 4). All above statistical analyses were carried out using MedCalc Software v 15.8.
Quality assessment
Two authors (ZA and FV) independently assessed the methodological quality of the included studies according to a grading system developed by the Swedish Council on Technology Assessment in Health Care [36]. The following criteria were used for assessing the methodological quality of the studies included in the present review:
-
Grade A (High)—A randomized controlled trial or prospective study, composed of a well-defined control group; defined diagnosis and end points; diagnostic reliability tests and reproducibility tests described; and blinded outcome measurements (all criteria should be met).
-
Grade B (Moderate)—A cohort study or retrospective study, composed of a defined control group; defined diagnosis and end points; and diagnostic reliability tests and reproducibility tests described (all criteria should be met; otherwise, grade C).
-
Grade C (Low)—One or more of the following settings are encountered: poorly defined patient material, unclear diagnosis and end points, and large attrition of the samples.
Level of evidence
For each study included in the present review, level of evidence was judged in accordance with the following scale [37]:
-
Strong Scientific Support (Evidence Grade 1)—Conclusion is based on at least two studies with level A evidence. Studies with opposite conclusions may lower the evidence grade.
-
Moderately Strong Support (Evidence Grade 2)—Conclusion is based on one study with strong evidence (A) and at least two with moderately strong evidence (B). Studies with opposite conclusions may lower the evidence grade.
-
Limited Scientific Support (Evidence Grade 3)—Conclusion is based on at least two studies with moderately strong evidence (B). If studies contradicting the conclusion exist, the scientific basis is judged as contradictory or insufficient.
-
Inconclusive Scientific Support (Evidence Grade 4)—If studies fulfilling the evidence criteria are lacking, the scientific basis for conclusion is considered insufficient.
Results
General characteristics of the studies
From the 36 articles assessed (full-text) for eligibility, 31 studies were excluded (Appendix 1). In total, five studies [25, 27, 33–35] were included and processed for data extraction (Fig. 1). All studies, [25, 27, 33–35] included were non-randomized controlled trials. The study by Suvan et al. [33] was secondary analysis of primary clinical studies [38–41]. The other studies [25, 27, 33–35] were performed at outpatient clinics of universities. The total number of patients ranged between 40 [34] and 260 [33] individuals. All studies [25, 27, 33–35] reported the mean age of study participants, which was between 42.5 years [35] and 48.8 years [27] (age range 27 to 77). All studies [25, 27, 33–35] reported the number of female participants, which ranged from 22 [27] to 137 [33] individuals (Table 1).
In the studies by Suvan et al. [33] and Altay et al. [35], 87 and 11 smokers were included, respectively. The kappa value for inter-reviewer agreement was 0.86.
In all the studies, NSPT was performed via full-mouth non-surgical scaling and root planning (SRP) with standard oral hygiene instructions. Four studies, [25, 27, 33, 35] reported subjects receiving full-mouth mechanical debridement with hand curettes together with ultrasonic instruments. One study [34] did not report the type of instrumentation used for non-surgical periodontal therapy. In addition to SRP, Altay and colleagues [35] applied full-mouth disinfection protocol as described by Quirynen et al. [42].
Obesity defining parameters of the included studies
All studies [25, 27, 33–35] followed WHO classification of obesity in adults. In the study by Dias Gonçalves et al. [27], individuals with body mass indexes (BMI) of ≥30–40 and ≥20–29.9 kg/m2 were categorized as obese and non-obese, respectively. Two studies [25, 35] used waist circumference (WC) to assess abdominal adiposity. This was divided into two categories, normal and high, using the cutoff point of >102 cm for males and >88 cm for females based on Adult Treatment Panel III guidelines [43]. In addition, Zuza et al. [25] and Dias Gonçalves et al. [27] used waist-hip ratio (WHR) of ≥0.8 for females and ≥0.9 for males as their case definitions for obesity [44]. Body fat (BF) categorized as ≥33 % for women and ≥25 % for men was also used by Zuza et al. [25] (Table 2).
Clinical periodontal inflammatory parameters
In three [25, 27, 34] and two studies [25, 27], full-mouth percentage of PI and GBI for obese and non-obese patients was reported. However, in the study by Altay et al. [35], full-mouth PI index and GBI index scores were reported. Four studies [25, 27, 34, 35] reported BOP and PPD in obese and non-obese subjects at follow-up, respectively. Mean CAL for obese and non-obese subjects was reported by four studies [25, 27, 34, 35], which ranged from 2.5 to 4.4 mm and 3.9 to 2.2 mm, respectively. In the study by Zuza et al. [25], PPD and CAL were reported as the percentage of sites with ≤3, 4–6, and ≥7 mm. In the study by Suvan et al. [33], mean PI, GBI, and PPD (percentage of >4 mm pocket depth) of obese and non-obese ranged from 68 to 24 % and 60 to 18 % for PI, 70 to 25 % and 60 to 18 % for GBI, and 50 to 20 % and 47 to 15 % for PPD, respectively, at follow-up. These results are summarized in Table 3. In one study [35], a total of seven teeth in five patients were extracted in test group, whereas a total of five teeth in four patients were extracted in control group.
All studies [25, 27, 33–35] reported significant reduction of clinical periodontal inflammatory parameters in both obese (test) and non-obese (control) subjects following NSPT. In three studies [25, 34, 35], the clinical periodontal condition in both test and control groups at follow-up was comparable. In the study by Suvan et al. [33], clinical periodontal condition among non-obese subjects was significantly better than obese subjects at follow-up. Dias Goncalves et al. [27] reported PPD and CAL to be significantly better among non-obese than obese subjects after NSPT at 6 months follow-up.
Meta-analyses were performed for only three studies [27, 34, 35] reporting means and standard deviations (Figs. 2 and 3). For all analyses, heterogeneity was found to be statistically significant; therefore, random-effects model were employed. For the effects of periodontal treatment on clinical parameters in obese and non-obese patients, no differences could be observed for both the parameters (PPD and CAL) among obese and non-obese subjects (PPD P = 0.91, I 2 67.36 %; CAL P = 0.87, I 2 77.16 %).
Serum levels of inflammatory cytokines among the included studies
Four studies [25, 27, 34, 35] assessed and reported the serum inflammatory cytokine levels among obese and non-obese subjects receiving NSPT, using the enzyme-linked immunosorbent assay (ELISA). The cytokines reported were IL 1β, IL-6, TNF-α, interferon-gamma (IFN-γ), leptin, adiponectin, and C-reactive protein (CRP). Zuza et al. [25] reported concentrations of IL1β, IL-6, TNF-α, and IFN-γ in obese and non-obese subjects. TNF-α and IL-6 was significantly higher in obese than non-obese subjects at follow-up [25]. Dias Gonçalves et al. [27] reported serum levels of adiponectin and leptin in obese and non-obese subjects. Serum levels of adiponectin and leptin in both obese and non-obese subjects were comparable to baseline at follow-up [27].
In a study by Al-Zahrani and AlGhamdi [34], serum CRP levels among obese and non-obese subjects who received NSPT for the management of chronic periodontitis was reported. They showed a significant but comparable reduction in CRP levels in both obese and non-obese subjects at follow-up [27]. In the study by Altay et al. [35], serum levels of TNF-α, IL-6, CRP, and leptin, among obese and non-obese subjects, were reported. At follow-up, TNF-α, IL-6, and leptin showed significant reduction in obese subjects; however, only IL-6 levels significantly reduced in non-obese subjects [35] (Table 4).
Quality assessment
On the basis of the methodological quality [36], all studies [25, 27, 33–35] were graded as “moderate.” Moderate grading was mainly based on non-randomized assignment of patients and lack of blinded assessment of outcome measures in the included studies (Table 1). In the study by Suvan et al. [33], blinded assessment of outcome measures was performed.
Discussion
To our knowledge from indexed literature, this is the first study that systematically reviewed the efficacy of NSPT in the management of CP among obese and non-obese patients. Interestingly, in 40 % of studies [27, 33], periodontal outcome was significantly poor in obese subjects as compared to non-obese subjects at follow-up. Multiple mechanisms are suggested for the association of obesity and periodontitis in the literature. It is known that a pro-inflammatory state is produced in obesity due to the increased serum levels of pro-inflammatory biomarkers (such as IL 1β, IL-6, TNF-α, IFN-γ, CRP) [45]. Moreover, altered immune response because of compromised functional state of immune cells in obese state has been reported [18]. These systemic factors could be responsible for the significantly inferior periodontal outcome following NSPT in obese subjects as compared to non-obese at follow-up.
By contrast, 60 % of the included studies (nearly 33 % of the total included population) [25, 34, 35] showed comparable improvements in the clinical periodontal outcomes (such as BOP, PPD, CAL) among obese and non-obese subjects at follow-up after NSPT. In addition, outcomes of the meta-analysis [27, 34, 35] showed comparable PPD and CAL among obese and non-obese subjects at follow-up. One factor that could have influenced the outcome of NSPT in obese and non-obese subjects could be gender type. It is pertinent to mention that in all studies [25, 34, 35], showing similar periodontal outcomes among obese and non-obese subjects at follow-up, the percentage of females in the obese group (77 to 100 %) was significantly higher as compared to non-obese (0 to 30.5 %). In addition, female subjects reported in the studies [25, 27, 33–35] were in fertile age (43.7–48 years) and did not receive any medication that could interfere with menstrual cycle. It is reported that the female ovarian hormones released during the menstrual cycle may exaggerate pre-existing inflammation in the periodontium [46, 47]. Another, important factor that could have influenced the outcomes in the included studies [25, 27, 33–35] was the number of extracted teeth in the obese individuals during the follow-up period. For instance in the study by Altay et al. [35], seven teeth were extracted among the obese subjects during the follow-up period. However, four studies [25, 27, 33, 34] failed to report the number of missing and extracted teeth during follow-up. A reduction in the pocket depths assessed due to extractions could have influenced the overall difference in PPD and CAL among obese and non-obese subjects. Furthermore, obesity is known to have a negative impact on the inflammatory, immunologic, and wound healing responses in the human body. [11, 13, 15] Therefore, it is possible that if there were standardization of gender distribution and adequate reporting and consideration of lost and extracted teeth among the obese and non-obese patients in these studies, periodontal outcome following NSPT would differ among obese and non-obese subjects.
In the present systematic review, four studies [25, 27, 34, 35] compared serum cytokine levels following NSPT in obese and non-obese subjects. A common finding among these studies was the higher level of serum cytokines in the obese as compared to controls at baseline and follow-up. An explanation in this regard may lie in the ability of adipose tissues to act as an endocrine gland, releasing higher levels of pro-inflammatory cytokines [48]. In addition, the pro-inflammatory state also results in increased monocytes and macrophages (produce cytokines for immune regulation) causing further localized elevation in inflammatory cytokine levels [49]. Moreover, Lundin et al. [50] evaluated cytokine levels in the GCF including TNF-α in obese patients; they demonstrated a positive correlation between BMI (40 kg/m2) and cytokine levels, i.e., increased cytokine levels with increasing BMI.
All studies [25, 27, 33–35] that fulfilled our eligibility criteria showed significant improvement in clinical periodontal parameters following NSPT in both obese and non-obese individuals. However, among the studies [25, 27, 34, 35] included in the present review, the impact of NSPT on the levels of serum cytokines in obese subjects was variable. It is likely that other confounding factors such as habitual tobacco smoking [51, 52] could have influenced the overall therapeutic effect of NSPT in obese patients. Therefore, it is possible that the smoking habit of subjects included in the study by Altay et al. [35] could have sustained the periodontal inflammation preventing the reduction in serum CRP levels in obese subjects. Furthermore, it appears that issues related to methodology of studies have confounded their outcomes. It is therefore suggested that further controlled trials with strict inclusion and exclusion criteria and longer follow-up periods should be performed to ascertain the outcome of NSPT in obese chronic periodontitis patients.
From a clinical point of view, although the effect of obesity on the outcome of NSPT still remains unclear, nevertheless, clinicians are prompted to manage obesity prior to and during periodontal treatment.
Conclusion
It remains unclear whether NSPT has a significantly higher impact on the clinical periodontal outcomes in obese patients than in non-obese patients with chronic periodontitis, given that the number of selected studies was relatively low and the reported findings were inconsistent.
References
Tagge DL, O’Leary TJ, El-Kafrawy AH (1975) The clinical and histological response of periodontal pockets to root planing and oral hygiene. J Periodontol 46:527–533
Biagini G, Checchi L, Miccoli MC, Vasi V, Castaldini C (1988) Root curettage and gingival repair in periodontitis. J Periodontol 59:124–129
Caton JG, Zander HA (1979) The attachment between tooth and gingival tissues after periodic root planing and soft tissue curettage. J Periodontol 50:462–466
Smart GJ, Wilson M, Davies EH, Kieser JB (1990) The assessment of ultrasonic root surface debridement by determination of residual endotoxin levels. J Clin Periodontol 17:174–178
Heitz-Mayfield LJA, Trombelli L, Heitz F, Needleman I, Moles D (2002) A systematic review of the effect of surgical debridement vs. non-surgical debridement for the treatment of chronic periodontitis. J Clin Periodontol 29:92–102
Drisko CH (2001) Nonsurgical periodontal therapy. Periodontol 2000(25):77–88
Cobb CM (1996) Non-surgical pocket therapy: mechanical. Ann Periodontol 1:443–490
World Health Organization (2000) Obesity: preventing and managing the global epidemic 894: World Health Organization
Kopelman P, Jebb S, Butland B (2007) Executive summary: foresight ‘tackling obesities: future choices’ project. Obes Rev 8:vi–ix
Field AE, Coakley EH, Must A, Spadano JL, Laird N, Dietz WH, Rimm E, Colditz GA (2001) Impact of overweight on the risk of developing common chronic diseases during a 10-year period. Arch Int Med 161:1581–1586
Rasouli N, Kern PA (2008) Adipocytokines and the metabolic complications of obesity. J Clin Endocrinol Metab 93:s64–s73
Kim S, Popkin BM (2006) Commentary: understanding the epidemiology of overweight and obesity—a real global public health concern. Int J Epidemiol 35:60–67
Coppack SW (2001) Pro-inflammatory cytokines and adipose tissue. Proc Nutr Soc 60:349–356
Ziccardi P, Nappo F, Giugliano G, Esposito K, Marfella R, Cioffi M, D’Andrea F, Molinari AM, Giugliano D (2002) Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 105:804–809
Falagas ME, Kompoti M (2006) Obesity and infection. Lancet Infect Dis 6:438–446
Ouchi N, Parker JL, Lugus JJ, Walsh K (2011) Adipokines in inflammation and metabolic disease. Nat Rev Immunol 11:85–97
Saxlin T, Ylöstalo P, Suominen-Taipale L, Männistö S, Knuuttila M (2011) Association between periodontal infection and obesity: results of the Health 2000 Survey. J Clin Periodontol 38:236–242
Maury E, Brichard SM (2010) Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol Cell Endocrinol 314:1–16
Wilson JA, Clark JJ (2003) Obesity: impediment to wound healing. Crit Care Nurs Q 26:119–132
Muñoz-Torres FJ, Jimenez MC, Rivas-Tumanyan S, Joshipura KJ (2014) Associations between measures of central adiposity and periodontitis among older adults. Comm Dent Oral Epidemiol 42:170–177
Dalla Vecchia CF, Susin C, Rösing CK, Oppermann RV, Albandar JM (2005) Overweight and obesity as risk indicators for periodontitis in adults. J Periodontol 76:1721–1728
Perlstein MI, Bissada NF (1977) Influence of obesity and hypertension on the severity of periodontitis in rats. Oral Surg Oral Med Oral Pathol 43:707–719
Moura-Grec PGD, Marsicano JA, Carvalho CAPD, Sales-Peres SHDC (2014) Obesity and periodontitis: systematic review and meta-analysis. Cien Saude Colet 19:1763–1772
Kose O, Canakci V, Canakci CF, Yildirim A, Kermen E, Arabaci T, Gungor A (2015) The effects of obesity on local and circulating levels of tumor necrosis factor-α and interleukin-6 in patients with chronic periodontitis. J Periodontol Imp Dent 7:7–14
Zuza EP, Barroso EM, Carrareto ALV, Pires JR, Carlos IZ, Theodoro LH, Toledo BEC (2011) The role of obesity as a modifying factor in patients undergoing non-surgical periodontal therapy. J Periodontol 82:676–682
Genco RJ, Grossi SG, Ho A, Nishimura F, Murayama Y (2005) A proposed model linking inflammation to obesity, diabetes, and periodontal infections. J Periodontol 76:2075–2084
Dias Goncalves TE, Feres M, Zimmermann GS, Faveri M, Figueiredo LC, Braga PG, Duarte PM (2014) Effects of scaling and root planing on clinical response and serum levels of adipocytokines in obese patients with chronic periodontitis. J Periodontol 86:53–61
Suvan J, D’Aiuto F, Moles DR, Petrie A, Donos N (2011) Association between overweight/obesity and periodontitis in adults. A systematic review. Obes Rev 12:e381–e404
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Int Med 151:264–269
Boudin F, Nie JY, Bartlett JC, Grad R, Pluye P, Dawes M (2010) Combining classifiers for robust PICO element detection. BMC Med Inform Decis Mak 10:29
Roberts C (2008) Modelling patterns of agreement for nominal scales. Stat Med 27:810–830
Chang CH (2014) Cohens’s kappa for capturing discrimination. Int Health 6:125–129
Suvan J, Petrie A, Moles DR, Nibali L, Patel K, Darbar U, Donos N, Tonetti M, D’Aiuto F (2013) Body mass index as a predictive factor of periodontal therapy outcomes. J Dent Res:0022034513511084
Al-Zahrani MS, AlGhamdi HS (2012) Effect of periodontal treatment on serum C-reactive protein level in obese and normal-weight women affected with chronic periodontitis. Saudi Med J 33:309–314
Altay U, Gürgan CA, Ağbaht K (2013) Changes in inflammatory and metabolic parameters after periodontal treatment in patients with and without obesity. J Periodontol 84:13–23
Bondemark L, Holm AK, Hansen K, Axelsson S, Mohlin B, Brattstrom V, Paulin G, Pietila T (2007) Long-term stability of orthodontic treatment and patient satisfaction: a systematic review. Angle Orthod 77:181–191
Mohlin B, Axelsson S, Paulin G, Pietilä T, Bondemark L, Brattström V, Hansen K, Holm AK (2007) TMD in relation to malocclusion and orthodontic treatment: a systematic review. Angle Orthod 77:542–548
D’Aiuto F, Nibali L, Parkar M, Suvan J, Tonetti MS (2005) Short-term effects of intensive periodontal therapy on serum inflammatory markers and cholesterol. J Dent Res 84:269–273
D’Aiuto F, Parkar M, Andreou G, Suvan J, Brett PM, Ready D, Tonetti MS (2004) Periodontitis and systemic inflammation: control of the local infection is associated with a reduction in serum inflammatory markers. J Dent Res 83:156–160
D’Aiuto F, Parkar M, Nibali L, Suvan J, Lessem J, Tonetti MS (2006) Periodontal infections cause changes in traditional and novel cardiovascular risk factors: results from a randomized controlled clinical trial. Am Heart J 151:977–984
Tonetti MS, D’Aiuto F, Nibali L, Donald A, Storry C, Parkar M, Suvan J, Hingorani AD, Vallance P, Deanfield J (2007) Treatment of periodontitis and endothelial function. N Engl J Med 356:911–920
Quirynen M, Bollen CML, Vandekerckhove BNA, Dekeyser C, Papaioannou W, Eyssen H (1995) Full-vs. partial-mouth disinfection in the treatment of periodontal infections: short-term clinical and microbiological observations. J Dent Res 74:1459–1467
Panel NCEPNE (2002) Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106:3143
Saito T, Shimazaki Y, Koga T, Tsuzuki M, Ohshima A (2001) Relationship between upper body obesity and periodontitis. J Dent Res 80:1631–1636
Akman PT, Fentoglu O, Yilmaz G, Arpak N (2012) Serum plasminogen activator inhibitor-1 and tumor necrosis factor-α levels in obesity and periodontal disease. J Periodontol 83:1057–1062
Shourie V, Dwarakanath CD, Prashanth GV, Alampalli RV, Padmanabhan S, Bali S (2011) The effect of menstrual cycle on periodontal health-a clinical and microbiological study. Oral Health Prev Dent 10:185–192
Becerik S, Ozçaka O, Nalbantsoy A, Atilla G, Celec P, Behuliak M, Emingil G (2010) Effects of menstrual cycle on periodontal health and gingival crevicular fluid markers. J Periodontol 81:673–681
Fantuzzi G (2005) Adipose tissue, adipokines, and inflammation. J Allergy Clin Immunol 115:911–919
Kullo IJ, Hensrud DD, Allison TG (2002) Comparison of numbers of circulating blood monocytes in men grouped by body mass index (<25, 25 to <30,≥ 30). Am J Cardiol 89:1441–1443
Lundin M, Yucel-Lindberg T, Dahllöf G, Marcus C, Modéer T (2004) Correlation between TNFalpha in gingival crevicular fluid and body mass index in obese subjects. Acta Odontol Scand 62:273–277
Rosa EF, Corraini P, Inoue G, Gomes EF, Guglielmetti MR, Sanda SR, Lotufo JPB, Romito GA, Pannuti CM (2014) Effect of smoking cessation on non-surgical periodontal therapy: results after 24 months. J Clin Periodontol 41:1145–1153
Bergstrom J (2014) Smoking rate and periodontal disease prevalence: 40-year trends in Sweden 1970–2010. J Clin Periodontol 41:952–957
Acknowledgments
This study was approved by Medical Ethics Committee, University of Malaya Medical Centre (UMMC), and supported by University of Malaya [Grant No. PG140-2013A] and RG303/11HTM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by Medical Ethics Committee, University of Malaya Medical Centre (UMMC).
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The work was supported by University of Malaya [Grant No. PG140-2013A] and RG303/11HTM, Department of Periodontology, Kuala Lumpur, Malaysia.
Informed consent
For a systematic review and meta-analysis, formal consent is not required.
Appendix 1
Appendix 1
List of excluded studies after full-text examination. Reason for exclusion is shown in parenthesis.
-
a)
Rocha, Miriam L., et al. “Effect of alendronate on periodontal disease in postmenopausal women: a randomized placebo-controlled trial.” Journal of periodontology 75.12 (2004): 1579–1585. (Use of adjunct with NSPT)
-
b)
Offenbacher, Steven, et al. “Results from the Periodontitis and Vascular Events (PAVE) Study: a pilot multicentered, randomized, controlled trial to study effects of periodontal therapy in a secondary prevention model of cardiovascular disease.” Journal of periodontology 80.2 (2009): 190–201. (Obesity associated with cardiovascular disease)
-
c)
Lakkis, Dima, et al. “Response to periodontal therapy in patients who had weight loss after bariatric surgery and obese counterparts: a pilot study.”Journal of periodontology 83.6 (2012): 684–689. (No inclusion of non-obese control)
-
d)
Shimada Y, Komatsu Y, Ikezawa-Suzuki I, Tai H, Sugita N, Yoshie H (2010) The effect of periodontal treatment on serum leptin, interleukin-6, and C-reactive protein. J Periodontol 81:1118–1123. (Focused question not answered)
-
e)
Shimoe M, Yamamoto T, Iwamoto Y, Shiomi N, Maeda H, Nishimura F, Takashiba S (2011) Chronic periodontitis with multiple risk factor syndrome: a case report. J Int Acad Periodontol 13:40–47. (Case report)
-
f)
Sora ND, Marlow NM, Bandyopadhyay D, Leite RS, Slate EH, Fernandes JK (2013) Metabolic syndrome and periodontitis in Gullah African Americans with type 2 diabetes mellitus. J Clin Periodontol 40:599–606. (Focused question not answered)
-
g)
Gorman A, Kaye EK, Nunn M, Garcia RI (2012) Changes in body weight and adiposity predict periodontitis progression in men. J Dent Res 91:921–926. (Focused question not answered)
-
h)
Gursoy UK, Marakoglu I, Ersan S (2006) Periodontal status and cytoplasmic enzyme activities in gingival crevicular fluid of type 2 diabetic and/or obese patients with chronic periodontitis. J Int Acad Periodontol 8:2–5. (Focused question not answered)
-
i)
Keller JJ, Wu CS, Chen YH, Lin HC (2013) Association between obstructive sleep apnoea and chronic periodontitis: a population-based study. J Clin Periodontol 40:111–117. (Irrelevant citation)
-
j)
Kshirsagar AV, Moss KL, Elter JR, Beck JD, Offenbacher S, Falk RJ (2005) Periodontal disease is associated with renal insufficiency in the Atherosclerosis Risk In Communities (ARIC) study. Am J Kidney Dis 45:650–657. (Irrelevant citation)
-
k)
Mohangi GU, Singh-Rambirich S, Volchansky A (2013) Periodontal disease: Mechanisms of infection and inflammation and possible impact on miscellaneous systemic diseases and conditions. SADJ 68: 462, 464–467. (Irrelevant citation)
-
l)
Leung WK ML, Wong MC, Corbet EF, Siu SC, Kawamura M. (2008) Health behaviour, metabolic control and periodontal status in medically treated Chinese with type 2 diabetes mellitus. Annals of the Royal Australasian College of Dental Surgeons:102–110. (Focused question not answered)
-
m)
Ekuni D, Yamamoto T, Koyama R, Tsuneishi M, Naito K, Tobe K (2008) Relationship between body mass index and periodontitis in young Japanese adults. J Periodontal Res 43:417–421. (Focused question not answered)
-
n)
Kapellas K, Maple-Brown LJ, Jamieson LM, Do LG, O’Dea K, Brown A, Cai TY, Anstey NM, Sullivan DR, Wang H, Celermajer DS, Slade GD, Skilton MR (2014) Effect of periodontal therapy on arterial structure and function among aboriginal Australians: a randomized, controlled trial. Hypertension 64:702–708. (Irrelevant citation)
-
o)
Pradeep AR, Kumari M, Kalra N, Priyanka N (2013) Correlation of MCP-4 and high-sensitivity C-reactive protein as a marker of inflammation in obesity and chronic periodontitis. Cytokine 61:772–777. (Review article)
-
p)
Pradeep AR, Priyanka N, Prasad MV, Kalra N, Kumari M (2012) Association of progranulin and high-sensitivity CRP concentrations in gingival crevicular fluid and serum in chronic periodontitis subjects with and without obesity. Dis Markers 33:207–213. (Focused question not answered)
-
q)
Al-Zahrani MS, Zawawi KH, Altaf FM (2013) The effect of obesity and periodontitis on the expression of antimicrobial peptides in gingival tissues. Saudi Med J 34:525–530. (Focused question not answered)
-
r)
Alabdulkarim M, Bissada N, Al-Zahrani M, Ficara A, Siegel B (2005) Alveolar bone loss in obese subjects. J Int Acad Periodontol 7: 34–38. (Irrelevant citation)
-
s)
Rao Deepika PC, Saxena RM (2013) Comparison of glycosylated hemoglobin levels in severe periodontitis patients and healthy controls: a study in an Indian population. Quintessence Int 44: 319–325. (Irrelevant citation)
-
t)
Susanto H, Nesse W, Dijkstra PU, Hoedemaker E, van Reenen YH, Agustina D, Vissink A, Abbas F (2012) Periodontal inflamed surface area and C-reactive protein as predictors of HbA1c: a study in Indonesia. Clin Oral Investig 16: 1237–1242. (Irrelevant citation)
-
u)
Ueda H, Yagi T, Amitani H, Asakawa A, Ikeda S, Miyawaki S, Inui A (2013) The roles of salivary secretion, brain-gut peptides, and oral hygiene in obesity. Obes Res Clin Pract 7:e321–329. (Irrelevant citation)
-
v)
Zimmermann GS, Bastos MF, Dias Goncalves TE, Chambrone L, Duarte PM (2013) Local and circulating levels of adipocytokines in obese and normal weight individuals with chronic periodontitis. J Periodontol 84:624–633. (Focused question not answered)
-
w)
Gursoy UK, Marakoglu I, Oztop AY (2008) Relationship between neutrophil functions and severity of periodontitis in obese and/or type 2 diabetic chronic periodontitis patients. Quintessence Int 39:485–489.(Irrelevant citation)
-
x)
Haffajee AD, Socransky SS (2009) Relation of body mass index, periodontitis and Tannerella forsythia. J Clin Periodontol 36:89–99. (Focused question not answered)
-
y)
Palle AR, Reddy CM, Shankar BS, Gelli V, Sudhakar J, Reddy KK (2013) Association between obesity and chronic periodontitis: a cross-sectional study. J Contemp Dent Pract 14:168–173. (Focused question not answered)
-
z)
Chaffee BW, Weston SJ (2010) Association between chronic periodontal disease and obesity: a systematic review and meta-analysis. J Periodontol 81 (12):1708–1724. (Review article)
-
aa)
Chitsazi MT, Pourabbas R, Shirmohammadi A, Ahmadi Zenouz G, Vatankhah AH (2008) Association of Periodontal Diseases with Elevation of Serum C-reactive Protein and Body Mass Index. J Dent Res Dent Clin Dent Prospects 2: 9–14. (Focused question not answered)
-
bb)
Fan WH, Liu DL, Xiao LM, Xie CJ, Sun SY, Zhang JC (2011) Coronary heart disease and chronic periodontitis: is polymorphism of interleukin-6 gene the common risk factor in a Chinese population? Oral Dis 17: 270–276. (Irrelevant citation)
-
cc)
Kumar A, Masamatti SS, Virdi MS (2012) Periodontal diseases in children and adolescents: a clinician’s perspective part 2. Dent Update 39:639–642, 645–636, 649–652. (Irrelevant citation)
-
dd)
Moravec LJ, Boyd LD (2011) Bariatric surgery and implications for oral health: a case report. J Dent Hyg 85:166–176. (Case report)
-
ee)
Bawadi HA, Khader YS, Haroun TF, Al-Omari M, Tayyem RF (2011) The association between periodontal disease, physical activity and healthy diet among adults in Jordan. J Periodontal Res 46: 74–81. (Irrelevant citation)
Rights and permissions
About this article
Cite this article
Akram, Z., Safii, S.H., Vaithilingam, R.D. et al. Efficacy of non-surgical periodontal therapy in the management of chronic periodontitis among obese and non-obese patients: a systematic review and meta-analysis. Clin Oral Invest 20, 903–914 (2016). https://doi.org/10.1007/s00784-016-1793-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-016-1793-4