Abstract
The effects of self-etching adhesives on primary teeth were evaluated. Reports in the literature suggest differences between the first and second dentition regarding the composition and the morphology and, therefore, a possible difference in the performance of dental adhesives. The purpose of this study was to evaluate the effects of self-etching adhesives on primary dentin. Eight dentin disks were obtained from the occlusal surface of primary molars. The disks were divided between four dental adhesives [Prompt L-PoP (LP), Clearfil SE BOND (SE), Etch&Prime 3.0 (EP) and Prime&Bond NT (PB) + H3PO4 (Control) and “restored” with a composite (Pertac II)]. After sectioning, fixation and HMDS drying, specimens were polished and Field Emission SEM examinations were carried out. Clearly visible hybrid layer formation was found for PB, LP and EP. An undoubtedly detectable interdiffusion zone was not evident after the use of SE. A clearly visible adhesive layer was recognizable for PB, EP and SE, but not continuously detectable for LP. Debonded regions were observed for all systems evaluated, but distinct differences in the failure mode were detected. The evaluated dental adhesives did not generate completely sealed interfaces between the composite resin and the dentin of primary teeth in vitro.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Significant improvements of dental bonding agents have allowed a paradigm change towards a minimally invasive concept in operative dentistry and have made resin composite progressively more the material of choice for the restoration of primary and permanent teeth. Lately, in an attempt to simplify and shorten the application of dental adhesives, systems have been introduced which are based on self-etching adhesives. These systems use chemically modified acidic monomers, which are able to simultaneously demineralize and penetrate dental hard tissues. While bond strength studies have been carried out and the effects of these new bonding agents on permanent teeth have been studied extensively, the same research in the field of pediatric dentistry is deficient.
Besides the obvious differences of primary teeth in terms of numeric appearance, size and shape, morphological and structural differences are evident [9]. For a long time, the morphology and composition of dentin from deciduous and permanent teeth were regarded as being similar and the knowledge about permanent teeth was transferred to primary teeth [9]. The results of recently published investigations, however, do permit the conclusion that significant differences in the chemical and morphologic structure of the dentin of primary teeth exist [2, 19].
For permanent teeth the optimal conditioning time is well known and each manufacturer indicates a detailed description in their instructions. Unfortunately, the instructions for use when treating primary teeth are not yet firmly established and currently manufacturers do not recommend a different protocol for their products when used for bonding to primary teeth, even though this has been postulated [12].
Hence, the aim of this in vitro study was to evaluate and examine the resin-dentin interface of primary teeth after the application of self-etching adhesives and provide information about this interaction.
Materials and methods
Eight 800±200 μm-thick dentin discs were obtained from eight human primary second molars that had been extracted for orthodontic reasons by sectioning the occlusal surface with a water-cooled slow-speed Isomet (Buehler, Lake Bluff, IL) diamond disk. The dentin disks were randomly assigned to the four different bonding agents (n=2). The adhesive systems were applied to the top dentin surface according to the sequences described in Table 1.
A 1-mm thick layer of a composite resin (Pertac II, 3 M ESPE Seefeld, Germany) was then applied over the adhesive system and light cured for 40 sec. The “restored” disks were cross-sectioned through the center of the disk with a water-cooled slow-speed Isomet(Buehler, Lake Bluff, IL) diamond disk. The specimens were immediately immersed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer at pH 7.4 for 12 h at 4°C. After fixation, the disks were rinsed with 20 mL of 0.2 M sodium cacodylate buffer at pH 7.4 for 1 h with three changes, followed by distilled water for 1 min. They were then dehydrated in ascending grades of ethanol (25% for 20 min, 50% for 20 min, 75% for 20 min, 95% for 30 min, and 100% for 60 min). After the final ethanol step, the specimens were dried by immersion in hexamethyldisilazane (HMDS, Ted Pella, Redding, CA) for 10 min, placed on a filter paper inside a covered glass vial, and air-dried at room temperature. The dentin disks were embedded in self-curing epoxy resin (Epo-thin, Buehler, Lake Bluff, IL) and stored at room temperature for 24 h. After setting, the epoxy casts were polished in an automatic Pedemax-2 polishing machine with silicon carbide papers (Buehler, Lake Bluff, IL) of decreasing abrasiveness (600-, 1200- and 4000-grit) and fabric tissue with increasingly fine alumina suspension (Buehler, Lake Bluff, IL) to a particle size of 0.1 μm. The top of the epoxy cast was sectioned with an Isomet (Buehler, Lake Bluff, IL) diamond-saw in order to separate the specimen from the epoxy block. The specimens were ultra-sonicated in 100% ethanol for 5 min, thoroughly dried, demineralized in 1 N HCl for 60 sec, and deproteinized in 2% NaOCl for 10 min.
After drying, the specimens were mounted on aluminum stubs (Ted Pella, Redding, CA) with carbon disks (Ted Pella, Redding, CA) and colloidal quick drying silver paint (Ted Pella, Redding, CA). Then, the specimens were coated with gold-palladium, by means of a sputter-coater (E-5100, Polaron Equipment, Watford, England) at 10 mA for 1 min. The dentin slabs (four for each bonding agent) were observed under a Field Emission Scanning Electron Microscope (FE-SEM) (Hitachi S-4700, Hitachi, Tokyo, Japan) at an accelerating voltage of 5 kV and a working distance of 12–13 mm.
Results
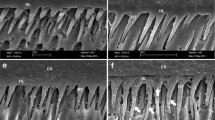
The FE-SEM micrographs for the four bonding systems are illustrated in Figs. 1, 2, 3, 4, 5, 6, 7 and 8. For all systems evaluated, distinct characteristics were observed. Clearly visible hybrid layer formation was found for PB, LP and EP. An undoubtedly detectable interdiffusion zone was not evident after the use of SE. On the other hand, a clearly visible adhesive layer was recognizable for PB, EP and SE, but not continuously detectable for LP. Debonding was observed for all systems, but differences in the failure mode were detected.
Figures 1 and 2 illustrate the hybridization and penetration abilities of LP. The demineralized and deproteinized resin-dentin interdiffusion zone shows the reticular aspect of the interpenetrating resin. Penetration of the resin resulted in a blood-vessel-like substructure. In addition, distinct tubule hybridization can be identified. Note that an adhesive layer is not always detectable. Areas of gap-formation became visible at the adhesive-composite interface (Fig. 3).
Interface after the application of Prompt L-Pop. Note the funnel configuration of the resin tags and the absence of an adhesive layer between the composite and the hybrid layer. The reticular configuration of the hybrid layer suggests the presence of resin-embedded collagen fibers. A Adhesive, C Composite, F Filler particle, HL Hybrid Layer, T Tag
The application of SE to primary dentin (Fig. 4) resulted in no distinct formation of a dense resin-dentin interdiffusion zone. On the other hand, a thick adhesive layer was observed. Dentinal tubular orifices were not widely opened. Areas of the tag formation showed a “moth-eaten” appearance, suggesting that the buffer capacity of the dentin limited further penetration of the acidic primer into the tubules. The use of SE also induced the formation of gaps (Fig. 5). Note that as soon as a distinct adhesive layer is perceptible, a sealed interface is detectable.
Resin-dentin interface after the application of Clearfil SE Bond. No visible hybrid layer is detectable. Areas of the tag formation show a “moth-eaten”appearance, suggesting that the buffer capacity of the dentin limited further penetration of the acidic primer into the tubules (white circle). A Adhesive, C Composite, T Tag
All evaluated specimens for EP revealed a very unusual cohesive failure in composite (Fig. 6). Note that the dentin-adhesive interface, as well as the adhesive-composite interface, is intact and shows no signs of failure.
The application of PB (+ H3PO4) resulted in very pronounced acid-resistant resin-dentin interdiffusion area (Fig. 7). In addition, massive appearing resin tags as well as multiple lateral tags/anastomosis are observable, resulting in a network-like appearance. Resin-tag formations in the opened tubules are encircled by hybridized tubule-orifice walls. The orifices were widened to the extent that funnel-shaped resin tags were constantly identified. The formation of a diminutive hybrid layer into the wall of the lateral tubule branches could also be detected (lateral tubule hybridization). Areas of the evaluated specimens also showed debonded sections (Fig. 8). Possibly exposed resin impregnated collagen fibers as well as entrances to tubules are apparent. Debonding resulted in the fracture of resin tags showing unsealed dentin tubules. Note the differences in failure and tag remnants in some of the orifices.
Very pronounced hybrid layer after applying Prime&Bond NT. In addition, massive appearing resin tags and multiple lateral tags/anastomosis (arrows) are observable, resulting in a network like appearance. Resin-tag formations in the opened tubules are encircled by hybridized tubule-orifice walls (asterisk). The orifices were widened to the extent that funnel-shaped resin tags were constantly identified. The formation of a diminutive hybrid layer into the wall of the lateral tubule branches could also be detected (lateral tubule hybridization). C Composite, HL Hybrid Layer, T Tag
Gap formation at the resin-dentin interface after applying Prime&Bond NT. Debonding resulted in the fracture of resin tags in the “neck-area” (arrows), resulting in possibly unsealed dentin tubules (arrow heads). Note the differences in failure and tag remnants (pointer) in some of the orifices. C Composite, T Tag
Discussion
This in vitro study was designed to examine the interfacial micromorphology of bonded restorations with dental bonding agents to primary dentin. The purpose of this examination was to observe the morphological characteristics of the resin-dentin-interface and to gain information on their hybridization and penetration abilities. With the use of Field-Emission Scanning Electron microscopy (FE-SEM), an excellent characterization of the ultramorphological interface is possible due to an enhanced resolution and reduced voltage, which lessens damage to specimen surface.
We are aware of the possibility that embedding and polishing the restored disks followed by demineralization and deproteinization may have induced artifacts. Also, the dehydration techniques and the high Fe-SEM vacuum may also have provoked changes to the interfaces analyzed. Yet, the challenging conditions during SEM examinations are an excellent test to reveal the weakest link of the evaluated interfaces (like in the case of EP) [18]. Because of the limited availability of sound primary teeth, we only evaluated four dentin sections for each bonding agent. Nevertheless, several studies were able to show that this sample size is sufficient to allow proper conclusions [15, 17, 20].
Self-etching adhesives have been developed to shorten bonding protocols and to reduce the sensitivity of the bonding-technique. Especially in the field of pediatric dentistry, such improvements would be desirable. The reactive components of these “Condiprimer-adhesives” [5] are esters from bivalent alcohols with methacrylic acid and phosphoric acid or derivates. The phosphate residue causes the conditioning of enamel and dentin, similar to the effect of phosphoric acid, while the methacrylate component of the molecule is available for copolymerization with the adhesive or the restorative material [6].
The evaluated LP-specimens revealed large areas of separation, despite the extents of the interdiffusion zone. Overall, the observation is in agreement with recently published studies. While Agostini et al. [1] report of complete bond failures with LP to dentin of primary teeth, da Silva Telles et al. [3] state that this adhesive was unable to create a visible hybrid layer, resulting in large and more frequent interfacial gaps than the control restorations bonded with an conventional adhesive system. However, the specimens in our in vitro study clearly indicated all signs of a successful hybridization but point towards a possible different problem. In the specimens evaluated, debonding occurred at the adhesive-composite interface, other than the reported failure at the resin-dentin joint [3]. The variation might be due to differences in the bonding protocol. In our study, LP was used according to the manufacturer’s instructions, which calls for a glossy surface after the application of the adhesive. This sometimes meant an application of multiple layers (up to four) of the adhesive prior to the polymerization. This routine has been shown to clearly benefit the performance of LP to permanent dentin [4].
Overall, a shiny surface is regarded as a sign that resin was adequately and sufficiently applied. However, the SEM-evaluation revealed that even the application of multiple layers did not result in a thick and uniform resin layer on top of the hybrid layer and it appeared as if the composite was mainly in direct contact with the hybrid layer. According to Inoue et al. [7], a sufficient monomer application is of great importance not only for the complete infiltration and penetration into the demineralized surface, but also for creating a sufficiently thick adhesive layer on top of the hybrid layer. Such a distinct resin layer can be regarded as a flexible, intermediate “shock-absorber”. Among others, our observations might be responsible for the debonded areas seen for LP.
Just recently, 3 M ESPE launched a modified version of the adhesive tested in our study. This product is now marketed under the name Adper Prompt L-Pop.
Even though SE has been reported to produce high tensile bond strength to primary dentin [1], areas of the evaluated specimens also showed failure at the adhesive interface. Debonding occurred where an adhesive layer was not clearly visible. Following the elastic bonding concept, it is conceivable that the allegedly insufficient adhesive layer was unable to withstand the polymerization forces that occurred during the polymerization of the composite. Filled low-viscosity resin is thought to have a strain capacity sufficient to relieve stresses between the shrinking composite restoration and the rigid dentin substrate, thereby improving the conservation of the dentin bond [23]. This effect might be due to a low Young’s modulus of elasticity of the filled adhesive, which may act as an inherent elastic buffering layer that absorbs the resin composite’s curing contraction stress [22]. The polymerization shrinkage stresses may be alleviated up to 50% if such an intermediate layer is present [10]. Note that as soon as a visible adhesive layer was recognizable (on the right of the arrow head), the adhesive interface was intact.
All evaluated EP specimens showed a very unusual cohesive failure in composite. The portion of the composite that actually remained attached to the adhesive contained fewer filler particles compared with the overlying composite. It is likely that the adhesive applied was 5 μm thick but was oxygen-inhibited. When the composite was added, the adhesive solubilized the composite matrix, mixing the two into a new material that was like an adhesive but different from the original. It probably had a relatively low modulus of elasticity compared with the overlying composite. The preparation of the specimens may have caused shrinkage to induce interfacial stresses where there is a sudden change in modulus that was large enough to cause crack initiation and propogation.
Maybe poorly cured adhesive mixed with the composite, making a second layer of “adhesive” that shifted stress to the filled portion making it appear to be a cohesive failure in the composite. In fact, what failed was not really the composite but perhaps a new interfacial structure (personal communication with Dr. D. Pashley).
Disadvantages after the usage of self-etching adhesives with a very low pH have recently been reported. The acidity of these systems seems to interfere with the polymerization of the composite. It is also possible that the high acidity of unpolymerized monomers remaining after light curing in a relatively high concentration at the oxygen-inhibited layer causes a separation at the adhesive-composite interface. The unreacted acid groups have been hypothesized to attack the polymerization initiation system of the composite material, especially in case of prolonged contact of the acidic adhesive monomers with the uncured composite material [8]. It has to be emphasized, that all evaluated EP specimens showed the above reported cohesive failure in composite. While Agostini et al. [1] were not able to explain the complete bond failures of EP to primary dentin in their study, our findings might offer a possible explanation.
Evaluated PB specimens also showed gap formation at the dentin-adhesive interface. Usually, it is believed that when a bond fails at the bottom or top of the hybrid layer, the two weak links of the micromechanical attachment [14], the intimate attachment of the resin tags to the walls of the tubule orifices keep the dentin tubules sealed [21]. In our observations, however, the resin tags did not break off at the level of the hybrid layer but closer to the “neck-area” of the tags. Therefore, it can be taken into consideration that the direct connection to the pulp is no longer sealed. Usually, the top 5–10 μm of the tags are believed to contribute the most to retention and sealing effectiveness of modern dental adhesives, whereas their actual length is of minor importance [7].
One explanation for this observation could be the use of phosphoric acid, which perhaps over-etched the dentin. Nör et al. [11] and Olmez et al. [13] have reported, that acid conditioning of primary dentin results in the formation of a 25–30% thicker hybrid layer compared with permanent teeth. They therefore concluded that primary dentin is more reactive to acidic conditioners and suggested a different protocol with shorter acid conditioning times when bonding to primary teeth. However, for all adhesives used in this study, no modification in the protocol for bonding to primary teeth was recommended by the manufacturers. The resulting increased thickness of the hybrid layer and the subsequent lack of complete penetration of adhesive resin into previously demineralized dentin may have contributed to the debonding seen for PB specimens.
Conclusion
The evaluated dental adhesives did not generate completely sealed interfaces between the resin composite and the dentin of primary teeth in vitro. Morphological differences among the adhesives were found. Our results clearly reveal that failure mode analysis by low magnification, as usually performed in combination with bond strength studies, does not always expose the true nature of the failure mode. As already reported for permanent teeth [16], ultra-morphological observations of non-demineralized bonding sites of primary teeth should be the goal for future investigations.
References
Agostini FG, Kaaden C, Powers JM (2001) Bond strength of self-etching primers to enamel and dentin of primary teeth. Pediatr Dent 23:481–486
Bordin-Aykroyd S, Sefton J, Davies EH (1992) In vitro bond strengths of three current dentin adhesives to primary and permanent teeth. Dent Mat 8:74–78
da Silva Telles PD, Aparecida M, Machado M, Nor JE (2001) SEM study of a self-etching primer adhesive system used for dentin bonding in primary and permanent teeth. Pediatr Dent 23:315–320
Frankenberger R, Perdigão J, Rosa BT, Lopes M (2001) “No-bottle” vs “multi-bottle” dentin adhesives—a microtensile bond strength and morphological study. Dent Mater 17:373–380
Haller B (2000) Recent developments in dentin bonding. Am J Dent 13:44–50
Hannig M, Reinhardt KJ, Bott B (1999) Self-etching primer vs. phosphoric acid: an alternative concept for composite-to-enamel bonding. Oper Dent 24:172–180
Inoue S, Van Meerbeek B, Vargas M, Yoshida Y, Lambrechts P, Vanherle G (2000) Adhesion mechanism of self-etching adhesives. In: Tagami J, Toledano M, Prati C (eds) Proceedings of 3rd International Kuraray Symposium on Advanced Adhesive Dentistry. Grafiche Erredue, Como
Inoue S, Vargas MA, Abe Y, Yoshida Y, Lambrechts P, Vanherle G, Sano H, Van Meerbeek B (2001) Microtensile bond strength of eleven contemporary adhesives to dentin. J Adh Dent 3:237–245
Johnson DC (1987) Comparison of primary and permanent teeth. In: Avery JK (ed) Oral developments and histology. Decker, Philadelphia, pp 180–202
Kemp-Scholte CM, Davidson CL (1990) Complete marginal seal of Class V resin composite restorations effected by increased flexibility. J Dent Res 69:1240–1243
Nör JE, Feigal RJ, Dennison JB, Edwards CA (1996) Dentin bonding: SEM comparison of the resin-dentin interface in primary and permanent teeth. J Dent Res 75:1396–1403
Nör JE, Feigal RJ, Dennison JB, Edwards CA (1997) Dentin bonding: SEM comparison of the dentin surface in primary and permanent teeth. Pediatr Dent 19:246–252
Olmez A, Oztas N, Basak F, Erdal S (1998) Comparison of the resin-dentin interface in primary and permanent teeth. J Clin Pediatr Dent 22:293–298
Pashley DH, Ciucchi B, Sano H, Horner JA (1993) Permeability of dentin to adhesive agents. Quintessence Int 24:618–631
Perdigão J, Swift EJ, Heymann HO, Malek MA (1998) Effect of a re-wetting agent on the performance of acetone-based dentin adhesives. Am J Dent 11:207–213
Perdigão J, Lopes M, Gomes G (2000) Ultramorphology of the hybrid layer—a TEM study of non-decalcified interfaces (Abstract). J Dent Res 79:1543
Ramos JC, Perdigão J (1997) Bond strengths and SEM morphology of dentin-amalgam adhesives. Am J Dent 10(3):152–158
Sano H, Takatsu T, Ciucchi B, Horner JA, Matthews WG, Pashley DH (1995) Nanoleakage: leakage within the hybrid layer. Oper Dent 20:18–25
Sumikawa DA, Marshall GW, Gee L, Marshall SJ (1999) Microstructure of primary tooth dentin. Pediat Dent 21:439–444
Tanumiharja M, Burrow MF, Tyas MJ, Carpenter J (2000) Field-emission scanning electron microscopy of resin-dentin interface morphology of seven dentin adhesive systems. J Adhes Dent 2:259–269
Tay FR, Gwinnett AJ, Pang KM, Wei SH (1994) Structural evidence of a sealed tissue interface with a total-etch wet-bonding technique in vivo. J Dent Res 73:629–633
Van Meerbeek B, Dhem A, Goret-Nicaise M, Braem M, Lambrechts P, Vanherle G (1993) Comparative SEM and TEM examination of the ultrastructure of the resin-dentin interdiffusion zone. J Dent Res 72:495–501
Van Meerbeek B, Willems G, Celis JP, Roos JR, Braem M, Lambrechts P, Vanherle G (1993) Assessment by nano-indentation of the hardness and elasticity of the resin-dentin bonding area. J Dent Res 72:1434–1442
Acknowledgements
The authors would like to thank Dr. Jorge Perdigão (University of Minnesota) for his invaluable help and assistance during this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaaden, C., Schmalz, G. & Powers, J.M. Morphological characterization of the resin-dentin interface in primary teeth. Clin Oral Invest 7, 235–240 (2003). https://doi.org/10.1007/s00784-003-0235-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-003-0235-2