Abstract
The aim of this study was to investigate the efficacy of concurrent treatment with vitamin K2 and risedronate compared with treatment with risedronate alone in patients with osteoporosis and to explore subsets of patients for which concurrent treatment is particularly efficacious. Women with osteoporosis aged 65 years or older were recruited from 123 institutes in Japan and allocated to take either vitamin K2 (45 mg/day) and risedronate (2.5 mg/day or 17.5 mg/week) or risedronate (2.5 mg/day or 17.5 mg/week) alone. The primary end point was the incidence of any fracture (vertebral and nonvertebral). The secondary end points were bone mineral density, height, undercarboxylated osteocalcin concentration, quality of life, and safety. Over a 2-year follow-up, vertebral or nonvertebral fractures occurred in 117 or 22 sites respectively among 931 patients in the risedronate and vitamin K2 group and in 104 or 26 sites respectively among 943 patients in the risedronate alone group. The rates of any incident fracture were similar between the two groups (incidence rate ratio 1.074, 95 % confidence interval 0.811–1.422, p = 0.62), implying that the primary end point was not met. There were no differences in the degree of increase in bone mineral density between the two groups. Undercarboxylated osteocalcin concentration decreased from 5.81 ± 3.93 ng/mL to 2.59 ± 1.52 ng/mL at 6 months in the risedronate and vitamin K2 group, whereas the change in the risedronate alone group was minimal (from 5.96 ± 4.36 ng/mL to 4.05 ± 3.40 ng/mL at 6 months) (p < 0.01). The treatment discontinuation rate was higher in the risedronate and vitamin K2 group than in the risedronate alone group (10.0 % vs 6.7 %). No unknown adverse drug reactions were reported. In conclusion, concurrent treatment with vitamin K2 and risedronate was not efficacious compared with monotherapy with risedronate in terms of fracture prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bisphosphonates have been used as first-line drugs to treat osteoporosis. Their key mode of action is inhibition of osteoclastic bone resorption through inhibition of the mevalonate pathway [1], achieving a decrease in bone turnover followed by an increase in secondary mineralization of bone. This process turns bone catabolism to anabolism and finally prevents fracture [2]. However, the rate of risk reduction of fractures through the use of bisphosphonates has ranged from 70 to 50 %, which means that the prevention of fracture has not yet been completely achieved by bisphosphonate treatment alone [3–7]. This insufficient reduction in the risk of fracture by bisphosphonate treatment may be partly explained by the concept that the pathogenesis of osteoporosis is complex.
Carmel et al. [8] reported that a sufficient level of 25-hydroxyvitamin D in serum (33 ng/mL or greater) is required for bisphosphonates to exert a favorable effect on bone. We previously found an association between undercarboxylated osteocalcin (ucOC) concentration and incident fracture among osteoporosis patients receiving bisphosphonate treatment [9]. Vitamin K is considered to maintain bone strength through γ-carboxylation of glutamic acid residues in matrix proteins such as osteocalcin. In vitamin K insufficiency or deficiency, ucOC is released from osteoblasts into the circulation. Thus, the serum concentration of ucOC has been recognized as being a sensitive marker of vitamin K status in bone [10]. Serum ucOC concentration decreases after vitamin K1 (phylloquinone) or vitamin K2 (menaquinone) treatment, and vitamin K administration was consistently reported to reduce fracture risk [11, 12]. Furthermore, Okano et al. [13] reported that vitamin K1) can be converted to vitamin K2 in various cells, including osteoblasts, through geranylgeranylation in the side chain and that menaquinone 4 was considered to be an active form of vitamin K because menaquinone 4 was reported to bind to nuclear steroid and xenobiotic receptor [14]. Since bisphosphonate inhibits farnesyl diphosphate synthase activity, bisphosphonate possibly inhibits the vitamin K activation process in the same bone cells. Thus, we attempted to investigate the efficacy of concurrent treatment with vitamin K2 and risedronate compared with treatment with risedronate alone and to explore subsets of patients for whom concurrent treatment is particularly efficacious in a randomized prospective study [15].
Materials and methods
Japanese Osteoporosis Intervention Trial-03 is a prospective, multicenter, open-labeled, randomized trial (University Hospital Medical Information Network Clinical Trials Registry identification number UMIN000000991), and details of its design have been reported elsewhere [15]. The protocol was approved by the central ethics committee for the Japanese Osteoporosis Intervention Trial (Rikushi Morita as Chairman) and was reviewed by the institutional review board of each participating institution as necessary. The trial was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained before patient enrollment and after the patient had been provided with a thorough explanation of the trial’s objectives, duration, and procedure.
Patients were recruited from 123 institutions nationwide in Japan from January 2008 to February 2010. Those who met all the eligibility criteria were randomly allocated to either the vitamin K2 (45 mg/day) and risedronate (2.5 mg/day or 17.5 mg/week) group or the risedronate alone (2.5 mg/day or 17.5 mg/week) group in a 1:1 randomization ratio. Random allocation was implemented by a Web-based computerized system for the modified minimization method that adjusts that data for an imbalance in eight factors: age, number of prevalent vertebral fractures, bone mineral density (BMD), serum ucOC level, presence of alcohol intake, past or current smoking, history of parents’ femoral neck fracture, and institution. The algorithm for random allocation was concealed from the investigators and patients. The planned follow-up duration was 2 years.
Eligibility criteria
Eligible participants were women who met the criteria to start pharmacological therapy defined by the Japanese guidelines for the prevention and treatment of osteoporosis (2014 edition) [16], were aged 65 years or older, had any of the risk factors suggested by the Adequate Treatment of Osteoporosis (A-TOP) research group, could walk unassisted, and had the ability to answer questionnaires. The A-TOP risk factors included age of 70 years or older, having one or more prevalent vertebral fractures between T4 and L4, BMD below minus three standard deviations of the young adult mean, and an ucOC level of 4.5 ng/mL or more. Exclusion criteria were prior treatment with warfarin, secondary osteoporosis or metabolic bone diseases other than osteoporosis, contraindication for administration of vitamin K2 and risedronate, hyperparathyroidism or hypoparathyroidism, mental disorders with the potential to result in unreliable self-reported data, prevalent vertebral fracture at six or more sites, severe degenerative deformation of the spine between T4 and L4, which may interfere with judgment of incident fracture or the precise measurement of lumbar BMD, critical disorders of the heart, liver, or kidney, and prior treatment with bisphosphonates within 6 months.
Assessments
For the assessment of prevalent vertebral fractures (T4–L4), anteroposterior and lateral radiographs of the thoracic and lumbar spine within 3 months before the date of informed consent were initially examined by investigators independent of the central committee according to the semiquantitative method [17]. After the first X-ray films had been collected, one evaluator for prevalent fractures or two independent evaluators for incident fractures, an orthopedist, and a radiologist reviewed those films simultaneously after the patient’s information had been masked according to the same semiquantitative method. When a diagnosis of prevalent vertebral fractures differed from that made by the investigators, the diagnosis made by the independent evaluators was preferentially adopted. If inconsistencies arose between the two independent readers, the two evaluators negotiated between themselves to reach a consensus. Incident vertebral fractures were assessed at 6, 12, and 24 months in the same manner as for prevalent vertebral fractures with use of radiographs obtained within 1 month of each visit. Also, assessors were completely blinded to treatment assignment.
To assess prevalent nonvertebral fractures, all fractures excluding vertebral, facial, and skull fractures that had occurred after the age of 50 years were recorded. All incident nonvertebral fractures were assessed by radiographs at the time of fracture with the exception of vertebral, facial, and skull fractures. Information such as the date, site, and circumstance of the fracture was recorded simultaneously. After X-ray films taken at each institution had been collected, they were reviewed by two independent evaluators.
At the baseline and at 6, 12, and 24 months, BMD was measured at one or more of the following sites: proximal part of the femur, femoral neck, lumbar vertebrae, distal part of the radius, and second metacarpal bone. The BMD measurements were done by a method that could be used in each institution. Quality of life was assessed by self-administered questionnaires [the Japanese Osteoporosis Quality of Life (JOQOL) questionnaire and the EuroQol five dimensions (EQ-5D) questionnaire] at the baseline and at 6, 12, and 24 months [18]. Baseline laboratory tests included those for ucOC, homocysteine, and 25-hydroxyvitamin D, and other routine biochemical examinations. The levels of ucOC were measured at 6 and 24 months, and those of homocysteine were measured at 24 months. Serum samples were sent to the central laboratories (SRL, Tokyo, Japan, and Toyo Kensa Center, Tokyo, Japan, for ucOC and 25-hydroxyvitamin D, and MBL, Tokyo, Japan, for homocysteine). Adverse events were assessed according to MedDRA version 6.1J.
Statistical considerations
The primary end point was the incidence of any fracture (vertebral and nonvertebral) analyzed by the person-year method. The accumulation of person-years at risk began at the randomization of each subject and ended at the date of the last visit (or the date of last radiographs for analysis of vertebral fractures), the date of lost to follow-up, or the date of death. The secondary end points were changes in BMD, height, ucOC concentration, JOQOL questionnaire score, EQ-5D questionnaire score, and safety. The current report focus on end points other than the JOQOL and EQ-5D questionnaire scores, and results regarding JOQOL and EQ-5D questionnaire scores will be reported elsewhere.
On the basis of past studies [19–21] and an attrition proportion of 15 %, a sample size of 910 participants per group and a 2-year follow-up were determined to provide 80 % power to detect the minimum clinically meaningful effect of a 35 % risk reduction of fracture with a two-sided significance level of 5 %. Interim analysis for safety was conducted at 1 year after enrollment of one third of the planned number of patients. Interim analysis for efficacy was not planned.
For the primary analysis we fit a multivariate Poisson regression model that included the adjustment factors for random allocation as covariates to the incidence of vertebral and nonvertebral fractures to estimate the hazard ratio between the two arms with its 95 % confidence interval and p value. We also fit the same Poisson regression models to incident fractures at each site as a secondary analysis. BMD, height, and ucOC concentration were described by means ± standard errors for each visit and means ± standard errors of changes from the baseline. Differences in changes from the baseline between the two arms were compared by a t test. All analyses were based on the intention-to-treat principle. Subgroup analyses of patients stratified according to the following factors that accompanied the interaction tests were also planned before provision of data: adjustment factors for random allocation, body mass index, 25-hydroxyvitamin D status, estimated glomerular filtration rate, grade of vertebral fracture, JOQOL questionnaire score, EQ-5D questionnaire score, and comorbidity. Two exploratory analyses were performed after provision of data: subgroup analysis according to homocysteine concentration and multivariate Poisson regression models and generalized additive models for the associations between the ucOC concentration at 6 months and incidence rate of vertebral fracture. In the latter analysis, we excluded incidents and person-years before 6 months to avoid bias due to outcome-by-outcome analysis. The regression models included age, BMD at the baseline, prevalent vertebral fractures, and A-TOP risk factors as covariates. All p values were two-tailed without multiplicity adjustment, and p < 0.05 indicated statistical significance. Data management and all statistical analyses were conducted by the Japan Clinical Research Support Unit (Tokyo, Japan) and academic biostatisticians with use of SAS version 9.2 (SAS Institute, Cary, NC, USA).
Results
Baseline characteristics
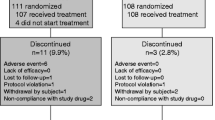
We randomized 1983 patients from 123 institutes in Japan. Finally, 931 patients in the risedronate and vitamin K2 group and 943 in the risedronate alone group were included the full analysis set after exclusion of those who did not receive the planned treatment and those who were not eligible (Fig. 1). The baseline characteristics of the patients in the full analysis set were well balanced between the two groups (Table 1). The mean age of the patients was 75.3 years. About 54 % of patients had BMD less than the young adult mean minus three standard deviations, and about 70 % of patients had not receive osteoporosis treatment during the previous year. Half of the patients had an ucOC level less than 4.5 ng/mL.
Adherence
The proportion of participants who discontinued treatment was higher in the risedronate and vitamin K2 group (10.0 % vs 6.7 %; Fig. 1). Adherence to risedronate or vitamin K2 therapy was excellent in both groups. The proportions of patients who took risedronate on 2, 3, or more days in 6, 12, 18, and 24 months were 89.2, 87.7, 87.0, and 82.1 % respectively in the risedronate and vitamin K2 group and 90.5, 88.3, 91.7, and 83.9 % respectively in the risedronate alone group. The proportions of patients who took vitamin K2 on 2, 3, or more days in 6, 12, 18, and 24 months were 84.3, 78.8, 79.8, and 74.1 % respectively in the risedronate and vitamin K2 group.
Fracture
The 2-year follow-up rate was 70.9 % (69.2 % in the risedronate and vitamin K2 group and 73.1 % in the risedronate alone group). There was no significant difference in the proportion of patients who were not assessed for vertebral fracture between the two groups (Fig. 1).
Table 2 shows the incidence rates of fracture over a period of 2 years. A total of 216 patients experienced any fracture. In comparison with no-fracture patients, the fracture patients were significantly older (77.6 years vs 75.0 years) and had lower BMD (T score of −3.7 vs −3.1) and 25-hydroxyvitamin D concentration (19.0 ng/mL vs 20.6 ng/mL) at the baseline. However, there were no significant differences at the baseline in ucOC concentration (5.8 ng/mL vs 5.9 ng/mL), homocysteine concentration (13.9 nmol/mL vs 13.2 nmol/mL), and estimated glomerular filtration rate (70.3 mL/min/1.73 m2 vs 70.5 mL/min/1.73 m2). Incident vertebral fractures occurred in 94 patients in the risedronate and vitamin K2 group and in 83 patients in the risedronate alone group. Incident nonvertebral fractures occurred in 20 patients in the risedronate and vitamin K2 group (3 rib, 2 radius, 2 ulna, 1 breastbone, 1 femur, 1 fibula, 1 forearm, 1 humerus, 1 knee joint, 1 kneecap, 1 middle phalanx, 1 patella, 1 proximal interphalangeal joint, 1 shoulder, 1 tibia, and 1 wrist) and in 24 patients in the risedronate alone group (5 rib, 4 radius, 3 femur, 3 kneecap, 2 breastbone, 2 proximal part of phalanx, 2 tibia, 1 coccyx, 1 finger, 1 humerus). There were no significant differences in the rate of any incident fracture between the two groups (incidence rate ratio 1.074, 95 % confidence interval 0.811–1.422, p = 0.62), implying that the primary end point was not met. Figure 2 shows the subgroup analysis by the prespecified stratification factors. Interaction tests indicated that a significant effect modification was not observed.
Subgroup analysis of effects on any incident fracture according to prespecified subgroups. BMD bone mineral density, BMI body mass index, eGFR estimated glomerular filtration rate, IRR incidence rate ratio, ucOC undercarboxylated osteocalcin, 25(OH)D 25-hydroxyvitamin D, SD standard deviation, SQ semiquantitative, YAM young adult mean
Secondary end points
Figure 3 depicts changes in BMD, height, and ucOC and homocysteine concentrations. There were no significant differences in the changes in BMD between the two groups; BMD according to the T score increased from −3.17 ± 1.65 at the baseline to −2.95 ± 1.70 at 24 months in the risedronate and vitamin K2 group and from −3.21 ± 1.58 at the baseline to −3.06 ± 1.66 at 24 months in the risedronate alone group (p = 0.21 for difference in change at 24 months). In contrast, ucOC concentration decreased from 5.81 ± 3.93 ng/mL at the baseline to 2.59 ± 1.52 ng/mL at 6 months and was stable after 6 months (2.63 ± 1.87 at 24 months) in the risedronate and vitamin K2 group, whereas the change in ucOC concentration in the risedronate alone group was less (5.96 ± 4.36 ng/mL at the baseline to 4.05 ± 3.40 ng/mL at 6 months) and continued until 24 months (3.74 ± 3.31 ng/mL) (p < 0.01 for difference in change at 24 months). Patients with an ucOC concentration at 6 months of less than 4.5 ng/mL and of 4.5–6 ng/mL accounted for 92.0 and 5.6 %, respectively, of the patients in the risedronate and vitamin K2 group, and 70.4 and 10.3 %, respectively, of patients in in the risedronate alone group. The degree of change in height was slightly greater in the risedronate and vitamin K2 group. The concentration of homocysteine, which is a marker of bone quality, was 13.5 ± 5.2 mmol/mL at the baseline and 12.1 ± 5.0 mmol/mL at 24 months in the risedronate and vitamin K2 group and was 13.1 ± 4.4 mmol/mL at the baseline and 11.9 ± 4.4 mmol/mL at 24 months in the risedronate alone group.
Safety
Adverse events according to MedDRA occurred in 80 patients (83 reports) in the risedronate and vitamin K2 group and in 52 patients (54 reports) in the risedronate alone group, and 97 of the 137 reports were of adverse drug reactions for which causal relationships with treatment cannot be ruled out. Table 3 provides information on the 97 reports of adverse drug reactions. All adverse drug reactions were known, and the commonest adverse drug reactions were gastrointestinal disorders (4.05 % in the risedronate and vitamin K2 group and 2.56 % in the risedronate alone group). Serious adverse events occurred in 14 patients (14 reports) in the risedronate and vitamin K2 group (including 7 patients who died) and in 16 patients (16 reports) in the risedronate alone group (including 12 patients who died); these included three adverse drug reactions related to the protocol-specific treatment (two patients in the risedronate and vitamin K2 group and one patient in the risedronate alone group).
Discussion
In this study of 1874 Japanese women with osteoporosis, we compared incidence rates of vertebral and nonvertebral fractures over 2 years, changes in BMD, height, and ucOC concentration, and safety with the concurrent treatment with vitamin K2 and risedronate and monotherapy with risedronate. The rates of any incident fracture were similar between the two groups, and the subgroup analysis revealed no effect modification among patients with ucOC concentration of 4.5 ng/mL or more and other subsets. Decreases in ucOC concentration were observed with both concurrent treatment and monotherapy, and the proportion of patients with an ucOC concentration less than 4.5 ng/mL at 6 months was 92.0 % in the risedronate and vitamin K2 group and 70.4 % in the risedronate alone group. However, there were no significant differences in changes in BMD and height between those receiving concurrent treatment and monotherapy.
Overall this study was conducted as planned. The incidence rate of vertebral fracture in the risedronate alone group, 0.068 per year, was close to that in our previous trial of alendronate [17] and the value for sample size calculation, a 2-year risk of 12.3 % with risedoronate monotherapy [19]. Therefore, the nonsignificant results of this study are not due to lack of statistical power. Although treatment discontinuation and gastrointestinal disorders were more frequent in the risedronate and vitamin K2 group, these did not influence fracture end points since the proportions were quite low and adherence to risedronate or vitamin K therapy was excellent in both groups. Therefore, our present results clearly indicate that the combination treatment was not superior to the monotherapy in terms of fracture prevention.
Although combination therapy is common in clinical practice, evidence for use of vitamin K combined with bisphosphonates is sparse. We hypothesized that bisphosphonates possibly interfere with vitamin K activation and that vitamin K and bisphosphonates have additive or synergistic effects. However, the effects of vitamin K combined with bisphosphonates may not be comparable to those of vitamin K monotherapy. Systematic reviews estimated that vitamin K monotherapy increased BMD by 1.80 % [22] and reduced the incidence of vertebral fractures by 37 % [23], although the results of the largest trial were negative [24]. Thus, fracture prevention effects of vitamin K monotherapy would be partially mediated through an increase in BMD. On the other hand, in our study an increase in BMD was observed in both arms, and the degree of increase was not significantly different between the two arms. Our findings were close to observations in a randomized clinical trial of risedronate combined with vitamin K that reported a nonsignificant difference in the change in BMD at 1 year [25]. Taken together, the effects on BMD of vitamin K combined with bisphosphonates are different from those of monotherapy. That may explain the lack of a significant difference in incidence rates of fracture in this study.
It is reasonable to expect that the efficacy of concurrent treatment with vitamin K is dependent on the nutritional status of a patient. The major source of dietary intake of vitamin K in Japanese people is fermented soybean foods, leading to a substantial variability in vitamin K intake [26]. In this study population, the average concentration of ucOC was around 6 ng/mL, and half of the patients had a ucOC concentration less than 4.5 ng/mL. However, our study revealed no significant differences in incidence rates of fracture in both the primary analysis and the subgroup analysis of ucOC concentration; that is, there was no evidence supporting the efficacy of concurrent treatment with vitamin K among patients with a low ucOC level.
In conclusion, concurrent treatment with vitamin K2 and risedronate was not more efficacious than monotherapy with risedronate in terms of fracture prevention.
References
Luckman SP, Hughes DE, Coxon FP, Graham R, Russell G, Rogers MJ (1998) Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J Bone Miner Res 13:581–589
Russell RG, Watts NB, Ebetino FH, Rogers MJ (2008) Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int 19:733–759
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC, Man Z, Mautalen C, Mesenbrink P, Hu H, Caminis J, Tong K, Rosario-Jansen T, Krasnow J, Hue TF, Sellmeyer D, Eriksen EF, Cummings SR (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med 356:1809–1822
Liberman UA, Weiss SR, Bröll J, Minne HW, Quan H, Bell NH, Rodriguez-Portales J, Downs RW Jr, Dequeker J, Favus M (1995) Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med 333:1437–1443
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet. 348:1535–1541
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH 3rd, Brown J, Eriksen EF, Hoseyni MS, Axelrod DW, Miller PD (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. JAMA 282:1344–1352
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J, Glüer CC, Krueger K, Cohen FJ, Eckert S, Ensrud KE, Avioli LV, Lips P, Cummings SR (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. JAMA 282:637–645
Carmel AS, Shieh A, Bang H, Bockman RS (2012) The 25(OH)D level needed to maintain a favorable bisphosphonate response is ≥33 ng/mL. Osteoporos Int 23:2479–2487
Shiraki M, Yamazaki Y, Shiraki Y, Hosoi T, Tsugawa N, Okano T (2010) High level of serum undercarboxylated osteocalcin in patients with incident fractures during bisphosphonate treatment. J Bone Miner Metab 28:578–584
Shiraki M, Tsugawa N, Okano T (2015) Recent advances in vitamin K-dependent Gla-containing proteins and vitamin K nutrition. Osteoporos Sarcopenia 1:22–38
Shiraki M, Shiraki Y, Aoki C, Miura M (2000) Vitamin K2 (menatetrenone) effectively prevents fractures and sustains lumbar bone mineral density in osteoporosis. J Bone Miner Res 15:515–521
Cheung AM, Tile L, Lee Y, Tomlinson G, Hawker G, Scher J, Hu H, Vieth R, Thompson L, Jamal S, Josse R (2008) Vitamin K supplementation in postmenopausal women with osteopenia (ECKO trial): a randomized controlled trial. PLoS Med 5:e196
Okano T, Shimomura Y, Yamane M, Suhara Y, Kamao M, Sugiura M, Nakagawa K (2008) Conversion of phylloquinone (vitamin K1) into menaquinone-4 (vitamin K2) in mice: two possible routes for menaquinone-4 accumulation in cerebra of mice. J Biol Chem 283:11270–11279
Tabb MM, Sun A, Zhou C, Grün F, Errandi J, Romero K, Pham H, Inoue S, Mallick S, Lin M, Forman BM, Blumberg B (2003) Vitamin K2 regulation of bone homeostasis is mediated by the steroid and xenobiotic receptor SXR. J Biol Chem 278(45):43919–43927
Tanaka S, Miyazaki T, Uemura Y, Kuroda T, Miyakawa N, Nakamura T, Fukunaga M, Ohashi Y, Ohta H, Mori S, Hagino H, Hosoi T, Sugimoto T, Itoi E, Orimo H, Shiraki M (2014) Design of a randomized clinical trial of concurrent treatment with vitamin K2 and risedronate compared to risedronate alone in osteoporotic patients: Japanese Osteoporosis Intervention Trial-03 (JOINT-03). J Bone Miner Metab 32:298–304
Orimo H, Hayashi Y, Fukunaga M, Sone T, Fujiwara S, Shiraki M, Kushida K, Miyamoto S, Soen S, Nishimura J, Oh-Hashi Y, Hosoi T, Gorai I, Tanaka H, Igai T, Kishimoto H (2001) Diagnostic criteria for primary osteoporosis: year 2000 revision. Osteoporosis Diagnostic Criteria Review Committee: Japanese Society for Bone and Mineral Research. J Bone Miner Metab 19:331–337
Orimo H, Nakamura T, Fukunaga M, Ohta H, Hosoi T, Uemura Y, Kuroda T, Miyakawa N, Ohashi Y, Shiraki M (2011) Effects of alendronate plus alfacalcidol in osteoporosis patients with a high risk of fracture: the Japanese Osteoporosis Intervention Trial (JOINT)—02. Curr Med Res Opin 27:1273–1284
Kumamoto K, Nakamura T, Suzuki T, Gorai I, Fujinawa O, Ohta H, Shiraki M, Yoh K, Fujiwara S, Endo N, Matsumoto T (2010) Validation of the Japanese Osteoporosis Quality of Life Questionnaire. J Bone Miner Metab 28:1–7
Kushida K, Fukunaga M, Kishimoto H, Shiraki M, Itabashi A, Inoue T, Kaneda K, Morii H, Nawata H, Yamamoto K, Ohashi Y, Orimo H (2004) A comparison of incidences of vertebral fracture in Japanese patients with involutional osteoporosis treated with risedronate and etidronate: a randomized, double-masked trial. J Bone Miner Metab 22:469–478
Iwamoto J, Sato Y, Takeda T, Matsumoto H (2009) High-dose vitamin K supplementation reduces fracture incidence in postmenopausal women: a review of the literature. Nutr Res 29:221–228
Kushima Y, Ishii K, Shimizu K, Sasaki A, Yamamoto K et al (2007) Effect of combination therapy with risedronate and vitamin K2 for osteoporosis: multicentre collaborative study. Preliminary report of a 1-year study. Osteoporos Jpn 15:234–237
Fang Y, Hu C, Tao X, Wan Y, Tao F (2012) Effect of vitamin K on bone mineral density: a meta-analysis of randomized controlled trials. J Bone Miner Metab 30:60–68
Stevenson M, Lloyd-Jones M, Papaioannou D (2009) Vitamin K to prevent fractures in older women: systematic review and economic evaluation. Health Technol Assess 13:1–134
Inoue T, Fujita T, Kishimoto H, Makino T, Nakamura T, Nakamura T, Sato T, Yamazaki K (2009) Randomized controlled study on the prevention of osteoporotic fractures (of study): a phase IV clinical study of 15-mg menatetrenone capsules. J Bone Miner Metab 27:66–75
Kasukawa Y, Miyakoshi N, Ebina T, Aizawa T, Hongo M, Nozaka K, Ishikawa Y, Saito H, Chida S, Shimada Y (2014) Effects of risedronate alone or combined with vitamin K2 on serum undercarboxylated osteocalcin and osteocalcin levels in postmenopausal osteoporosis. J Bone Miner Metab 32:290–297
Kaneki M, Hodges SJ, Hosoi T, Fujiwara S, Lyons A, Crean SJ, Ishida N, Nakagawa M, Takechi M, Sano Y, Mizuno Y, Hoshino S, Miyao M, Inoue S, Horiki K, Shiraki M, Ouchi Y, Orimo H (2001) Japanese fermented soybean food as the major determinant of the large geographic difference in circulating levels of vitamin K2: possible implications for hip-fracture risk. Nutrition 17:315–321
Acknowledgments
The authors thank the leading investigators involved in this study at the following medical facilities: Yoshida Orthopedic Clinic, Mie Memorial Hospital, Sapporo Kiyota Orthopedic Hospital, Irei Seikeigeka, and National Hospital Organization Fukui Hospital. Thanks are extended to all investigators involved in this study at the following medical facilities: Ako Hakuhou Hospital, Akiyama Orthopedic Clinic, Arai Clinic, Chiba Foundation for Health Promotion and Disease Prevention, Chousei Hospital, Fujii Internal Clinic, Fujisaki Orthopedic Clinic, Fujiwara Memorial Hospital, Fukase Orthopedic Clinic, Fuki City Hospital, Fukujukai Clinic, Fukui Kousei Hospital, Fukuroi City Hospital (integrated), Fukuroku Orthopedic Clinic, Fukutoku Orthopedic Surgery Clinic, Gomyo Orthopedic Clinic, Gyoda General Hospital, Hakodate Red Cross Hospital, Hakodate Central Hospital, Hara Orthopedic Clinic, Haramoto Clinic, Hayashi Orthopedic Clinic, Hayashi Surgery Clinic, Hirose Clinic, Hitachi Yokohama Hospital, Ichinomiya-nishi Hospital (closed), Inabe General Hospital, Ishida Orthopedic Clinic, JA Gifu Tohno Kosei Hospital, Japanese Red Cross Kyoto Daini Hospital, Japanese Red Cross Nagoya Daini Hospital, Joto Central Hospital Orthopedic Clinic, Kamiyouga Setagaya Street Clinic, Katsuura Orthopedic Clinic, Katsuga Orthopedic Clinic, Kawashima Orthopedic Hospital/Kawasima Clinic, Kenmochi Clinic, Kitasato University Hospital, Kodama Clinic, Takano Geka Seikeigeka, Kurosawa Hospital, Kurume University Hospital, Kyoto City Hospital, Kyoto Katsura Hospital, Matsui Orthopedic Clinic, Murase Hospital, Matsukuchi Orthopedic Clinic, Matsuzaka Central General Hospital, Miyazaki Orthopedic Clinic, Mori Orthopedic Clinic, Minami Hospital, Morooka Orthopedic Clinic, Nashinoki Clinic, National Hospital Organization Utano Hospital, National Hospital Organization Miyazaki Higashi Hospital, Nishimura Orthopedic Clinic, Nozaki Clinic, Ota City Hospital, Ota Orthopedic Clinic, Ootaki Hospital, Otoda Orthopedic Internal Clinic, Okumura Orthopedic Clinic, Ota General Hospital, Obase Hospital, Orito Orthopedic Clinic, Osaka City University Hospital, Research Institute and Practice for lnvolutional Disease, Saiseikai Kawaguchi General Hospital, Sanada Clinic, Sakura Clinic, Sappro Orthopedic Clinic, Saiseikai Matsuyama Hospital, Saito Orthopedic Clinic, Seirei Hamamatsu Hospital, Senkawa-Shinoda Orthopedic Clinic, Shibata Clinic, Shiozawa Orthopedic Clinic, Shinseikai Hospital, Shin Sapporo Orthopedic Hospital, Soejima Orthopedic Clinic, Suga Surgical Orthopedic Clinic, Sugioka Orthopedic Clinic, Takahashi Orthopedic Clinic, Takakuwa Orthopedic Nagayama Clinic, Tokyo Medical and Dental University, University Hospital of Medicine, Tottori University School of Medicine, Tokyo Women’s Medical University Medical Center Obstetrics and Gynecology, Toyokawa City Hospital, Tsuji Clinic, Tsukabara Clinic, Tsuruga City Hospital, Yamao Clinic, Yamane Hospital, Yasue Hospital, Yamamoto Orthopedic Clinic, Yanagisawa Orthopedic Clinic, Yodakubo National Health Insurance Hospital, Ui Orthopedic Clinic, Ugaji Orthopedic Clinic, Unnan City Hospital, University of Toyama Hospital, Wada Women’s Clinic, Watanabe Clinic, Zenshukai Hospital, Toyama Prefecture Saiseikai Takaoka Hospital, Shibayama Seikeigeka, Makiyama Central Hospital, Toumi City Mimaki Spa Clinic, Okuyama Orthopedic Clinic, Yoshida Clinic, Hamazaki Clinic, Tama-sougo Hospital (closed), Noda Clinic, Furuoka Orthopedic Clinic, Iseikai Hospital, and Murakami Orthopedic Clinic. The authors also express thanks to the chairman (Rikushi Morita) and the members of the Ethics Committee: Mamiko Matsumura, Tetsuro Inoue, Isao Yoshimura, Mitsuyoshi Nakajima (died), Tooru Ebihara, Shinya Hattori, and Kousaku Uchida. The authors also express sincere thanks to members of the A-TOP research group: Shigeto Morimoto (Geriatric Medicine, Kanazawa Medical University), Akira ltabashi (Saitama Center for Bone Research, Kubojima Clinic), Toshihiko Yamashita (formerly of the Department of Orthopedic Surgery, Sapporo Medical University School of Medicine), Hideaki Kishimoto (Department of Orthopedic Surgery, San-in Rosai Hospital), Hideki Mizunurna (Department of Obstetrics and Gynecology, Hirosaki University School of Medicine), Naoto Endo (Division of Orthopedic Surgery, Niigata University Medical and Dental Hospital), Yoshiki Nishizawa (formerly of the Department of Metabolism, Endocrinology, and Molecular Medicine, Osaka City University Graduate School of Medicine), and Kunio Takaoka (formerly of the Department of Orthopedic Surgery, Osaka City University Medical School) for their help in establishing the study design, and Naoto Fujinawa (I’cros), Kazumasa Tanzawa (formerly of ING Corporation), and Akan Katagiri (Japanese Clinical Research Support Unit) for their contribution to data management. Japanese Osteoporosis Intervention Trial-03 was sponsored by the Public Health Research Foundation. The authors also thank Eisai, Teijin Pharma Asahi Kasei, Takeda Pharmaceutical, GE Healthcare Japan, Toyo Medic, Banyu Pharmaceutical, and Dainippon Sumitomo Pharma for their generous donations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Teruhiko Miyazaki is an employee of the Public Health Research Foundation. All other authors have no conflicts of interest.
About this article
Cite this article
Tanaka, S., Miyazaki, T., Uemura, Y. et al. Comparison of concurrent treatment with vitamin K2 and risedronate compared with treatment with risedronate alone in patients with osteoporosis: Japanese Osteoporosis Intervention Trial-03. J Bone Miner Metab 35, 385–395 (2017). https://doi.org/10.1007/s00774-016-0768-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-016-0768-5