Abstract
Decreased myocardial uptake of I-123 metaiodobenzylguanidine (MIBG) is an important finding for diagnosis of Parkinson’s disease (PD). This study compared I-123 MIBG SPECT and planar imaging with regard to their diagnostic yield for PD. 52 clinically diagnosed PD patients who also had decreased striatal uptake on FP-CIT PET/CT were enrolled. 16 normal controls were also included. All underwent cardiac MIBG planar scintigraphy and SPECT separately. Myocardial I-123 MIBG uptake was interpreted on planar and SPECT/CT images separately by visual and quantitative analysis. The final diagnosis was made by consensus between two readers. Kappa analyses were performed to determine inter-observer agreement for both methods. Sensitivity, specificity, and accuracy were compared with McNemar’s test. The sensitivity, specificity, and accuracy were 84.6, 100, and 88.2 % for planar images and 96.2, 100 and 97.1 % for SPECT, respectively, with a significant difference between the two imaging methods (p < 0.031). All inter-observer agreements were almost perfect (planar scintigraphy: κ = 0.82; SPECT: κ = 0.93). Heart-to-mediastinum ratios from PD patients with negative planar and positive SPECT scans (group A) and patients with positive planar and positive SPECT scans (group B) were 1.69 ± 0.16 (1.59–1.85) and 1.41 ± 0.15 (1.20–1.53), respectively, and showed significant difference (p = 0.023). Lung-to-mediastinum ratios for groups A and B were 2.16 ± 0.20 (1.96–2.37) and 1.6 ± 0.19 (1.3–1.78), respectively, and were significantly higher in the former (p = 0.001). I-123 MIBG SPECT has a significantly higher diagnostic performance for PD than planar images. Increased lung uptake may cause false-negative results on planar imaging.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In idiopathic Parkinson’s disease (PD) patients, autonomic failure is an important clinical feature. I-123 metaiodobenzylguanidine (MIBG), a norepinephrine analogue, is taken up and stored in the sympathetic nerve endings. In PD patients with autonomic failure, MIBG cardiac scintigraphy shows myocardial postganglionic sympathetic dysfunction (Druschky et al. 2000).
Cardiac scintigraphy with I-123 MIBG has been used as a diagnostic modality to differentiate PD with postganglionic autonomic dysfunction from other Parkinsonian syndromes such as multiple system atrophy with central or preganglionic sympathetic dysfunction (Druschky et al. 2000; Saiki et al. 2004; Izawa et al. 2012). Decreased MIBG uptake was seen in PD patients even at the earliest stage of Hoehn and Yahr and in PD patients without significant clinical autonomic dysfunction, suggesting that postganglionic damage may occur in the early stage of PD (Braune et al. 1999).
Tomographic images, rather than planar, are ideal for evaluation of specific organ activity (Fukuoka et al. 2011). However, to our knowledge, there is limited literature comparing I-123 MIBG planar scintigraphy and SPECT in the diagnosis of PD.
The aim of this study was to compare diagnostic accuracy between I-123 MIBG planar and SPECT imaging for detection of abnormal heart uptake in PD.
Materials and methods
This study was approved by the institutional review board (Catholic Medical Center Office of Human Research Protection Program), and written informed consent was obtained.
Study population
All patients were prospectively recruited in consecutive manner, and the study was conducted between May 2012 and May 2013. Diagnosis of PD was made according to the UK Parkinson’s Disease Society Brain Bank Clinical Diagnosis Criteria (Daniel and Lees 1993) on the basis of clinical neurologic examination conducted by S.W.C. and I.U.S., neurologists with 20 and 12 years of experience, respectively.
Additionally, we included only patients with PD associated with the presence of nigrostriatal dopaminergic denervation on FP-CIT PET/CT, based on decreased putaminal uptake compared with the caudate nucleus (Oh et al. 2012). We evaluated the severity of patients using Hoehn and Yahr (H&Y) staging.
The evaluation procedure consisted of detailed medical history taking physical and neurological examinations, neuropsychological assessments, laboratory tests, magnetic resonance imaging (MRI) of the brain, and I-123 MIBG scintigraphy. None of the patients had any history or symptoms of cardiac disease or diabetes, showed abnormal Q waves at electrocardiography, or had any cerebrovascular lesions (as assessed by neuroimaging).
61 patients with PD were recruited during the study period. We excluded four patients with markedly fluctuating cognition with pronounced variations in attention and alertness, or recurrent vivid hallucinations suggesting the presence of diffuse Lewy body disease. We also excluded 3 patients who had signs of atypical Parkinsonism or secondary causes of parkinsonism (e.g., Wilson’s disease, neuroleptic drug users, and psychiatric diseases) that would, in the judgment of the investigators, interfere with their safe participation in the study. Additionally, we also excluded two patients with Parkinsonism that showed orthostatic hypotension, gaze palsy of eye movement, or poor response to dopaminergic therapy.
A total 52 patients with PD (20 men and 32 women; mean age 68.5 years, range 48–87 years) and 16 age-matched healthy controls (6 men and 10 women; mean age 69.5 years, range 45–84 years) were included in the study.
Healthy subjects did not have any history or symptoms of movement disorders including PD, tremor, dystonia, or other cognitive dysfunctions according to a dementia screening questionnaire, and they did not have any history of other neurological impairments such as sleep disorders, head trauma, epilepsy, stroke, or brain surgery. They did not have any history of medial illness or medication. The demographic and clinical characteristics of the subjects are summarized in Table 1.
MIBG Scintigraphy and SPECT/CT
MIBG scintigraphic imaging was obtained 20 min after intravenous injection of 111 MBq of I-123 MIBG using a dual-head gamma camera (Infinia, GE Healthcare, Milwaukee, WI, USA) equipped with a low-energy, high-resolution, parallel-hole collimator. Planar images were obtained with a 128 × 128 matrix. Projection imaging was taken for 60 s each in six increments over 180 circular orbits beginning from 45 at right anterior oblique projection and ending at the 45 left posterior oblique projection. Regional left ventricular MIBG uptake images were obtained after filtered back projection and recorded at a digital resolution of 64 × 64 pixels.
Image analysis
Myocardial uptake was interpreted separately for planar and SPECT/CT images. Image analysis was performed by two experienced nuclear medicine physicians in consensus. Scintigraphic data were independently analyzed by two nuclear medicine specialists (J.K.O. and Y.A.C., with 6 and 18 years of experience in nuclear medicine, respectively), who were unaware of the patients’ disease and autonomic function status. Any discrepancies were reviewed and resolved by consensus. Both visual and quantitative analyses were performed. For visual analysis, myocardial uptake was divided into three categories: normal uptake, decreased uptake, and no uptake. Images with decreased or no cardiac uptake were considered as positive results for PD.
For quantitative analysis, MIBG uptake was quantified by calculating the heart-to-mediastinum count (H/M) ratio and lung-to-mediastinum (L/M) ratio. Regions of interest were drawn over the mediastinum, heart, and lung on anterior views of the thorax. Care was taken to exclude lung or liver from the myocardial region and to exclude large vessels and lung from the mediastinum region (Fig. 1). To obtain the H/M and L/M ratios, average counts per pixel in the myocardium and lung were divided by average counts per pixel in the mediastinum (Kashihara et al. 2006).
Anterior planar image of I-123 MIBG scan (a). Heart I-123 MIBG uptake was calculated as the heart-to-mediastinum count ratio, using regions of interest (ROI) positioned over the heart (H) and upper mediastinum (M). Lung I-123 MIBG uptake was calculated as the lung-to-mediastinum count ratio, using ROIs positioned over the lung (L) and upper mediastinum (M). Axial (b) and coronal (c) SPECT images of the same patient. Intense heart activity is well defined from lung activity
In MIBG myocardial SPECT images, ROIs were drawn around the septal, anterior, lateral, and inferior walls of the heart to measure size of the area and uptake, from which mean uptake/area ratio was calculated for each ROI. However, in those cases where myocardial uptake was not visible and each wall could not be distinguished, a normal template was constructed from SPECT images of healthy controls and used as a spatial reference to draw ROIs.
Statistical analysis
All values are expressed as mean ± SD. Intergroup differences in various variances including H/M and L/M ratio of I-123 MIBG uptake were assessed by Student’s t test for continuous variables. Sensitivity, specificity, positive predictive value, and negative predictive value were estimated with 95 % confidence intervals. Statistical analysis of the difference in diagnostic yields between planar and SPECT was performed using McNemar’s test. Inter-observer agreements between the two readers with regard to planar and SPECT were assessed by κ coefficients. Inter-observer agreement was considered to be slight for κ < 0.21, fair for κ = 0.21–0.40, moderate for κ = 0.41–0.60, substantial for κ = 0.61–0.80, and almost perfect for κ = 0.81–1.00 (Nakajo et al. 1983).
The regional difference within LV was evaluated using one-way ANOVA. The level of statistical significance was set at p < 0.05. All statistical analyses were performed using MedCalc for Windows, version 15.4 (MedCalc Software, Mariakerke, Belgium).
Results
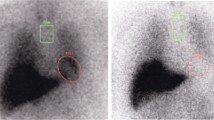
Among 52 patients with Parkinson’s disease, 2 showed normal findings on both planar scintigraphy and SPECT, and the other 50 all showed decreased heart uptake. Of these 50 patients, 44 also showed decreased heart uptake on planar scintigraphy (Fig. 2). However, the remaining 6 showed normal findings on planar scintigraphy (Fig. 3). There were no patients who showed positive results only on planar scintigraphy. All 16 healthy volunteers showed negative MIBG scan findings (Table 1).
A patient with PD who showed false-negative findings on MIGB planar scintigraphy. Planar image (a) shows relatively well visualization of heart uptake, and was considered a negative study. Diffuse lung uptake is also noted. In axial (b) and coronal (c) SPECT images of the same patient, the heart activity is decreased and poorly differentiated from increased lung activity, especially in the lateral wall (arrow), and considered as a positive study
The sensitivity, specificity, and accuracy were 84.6, 100, and 88.2 % for planar imaging and 96.2, 100, and 97.1 % for SPECT, respectively, with a significant difference between the two (p < 0.016) (Table 2). Analysis of inter-observer agreement for planar scintigraphy resulted in k statistics of 0.82 for the two reviewers; this improved to 0.93 for SPECT, suggesting almost perfect agreement between the reviewers (Table 3).
Of 50 patients who showed positive SPECT findings, 6 patients with negative planar image were classified as group A, and the remaining 44 with positive planar image as group B. There were no significant differences in age, H&Y stage, and duration of disease between the two groups. The H/M ratios from groups A and B were 1.69 ± 0.16 (1.59–1.85) and 1.41 ± 0.15 (1.20–1.53), respectively, with a significant difference between the two (p = 0.023). The L/M ratios were 2.16 ± 0.20 (1.96–2.37) and 1.6 ± 0.19 (1.3–1.78), respectively, and were significantly higher in group A (p = 0.001).
The mean H&Y stage of all 52 PD patients was 1.94 ± 0.8. There was no significant difference in H&Y stage and the duration of disease between PD patients with normal MIBG scan and those with abnormal findings.
On quantitative analysis with SPECT, the mean uptake/area ratio from PD patients was 2.95 ± 1.0 (1.2–6.4), 2.55 ± 0.85 (1.0–5.7), 2.54 ± 0.84 (1.1–5.5), and 2.3 ± 0.82 (0.89–5.2) in septal, inferior, anterior, and lateral walls, respectively. The uptake/area ratio was significantly higher in the septum and significantly lower in the lateral wall (p = 0.001) (Fig. 4).
The mean uptake/area ratio from control group was 6.1 ± 1.2 (5.2–8.3), 6.03 ± 1.0 (5.0–8.0), 6.24 ± 1.21 (5.1–8.5), and 6.3 ± 1.1 (4.9–7.9) in septal, inferior, anterior, and lateral walls, respectively.
Discussion
The sensitivity, specificity, and accuracy were 84.6, 100, 88.2 % for planar imaging and 96.2, 100, and 97.1 % for SPECT, respectively. SPECT showed significantly better results.
6 PD patients with false-negative planar scan showed significantly higher lung uptake. The heart activity was overestimated due to increased lung activity anterior and posterior to the heart in planar imaging. With tomographic images, heart activity could be accurately measured and delineated from lung activity. Therefore, when the planar image is positive, but there seems to be increased lung uptake, an additional SPECT should be performed.
Concerning previous diagnostic performance estimates, a meta-analysis of MIBG scan in PD reported that pooled sensitivity and specificity were 88 and 85 %, respectively (Treglia et al. 2012). The specificity in the present study is higher than that reported in the aforementioned study. This may be attributed to the fact that the patients with positive FP-CIT PET/CT were included in this study.
There has been very little literature on the utility of MIBG SPECT in diagnosis of PD. The sensitivity and specificity of planar scintigraphy for the diagnosis of PD have been reported to be approximately 73.3–100 % and 54.5–100 %, respectively (Chung et al. 2009; Sawada et al. 2009; Jost et al. 2010; Sudmeyer et al. 2011; Watanabe et al. 2011), which are similar to the results of this study.
I-123 MIBG is also taken up by the lungs through a saturable, energy-requiring sodium-dependent transport mechanism, similar to that of biogenic amines such as serotonin and norepinephrine (Slosman et al. 1989). Transport of these biogenic amines requires normal endothelial cell integrity. Thus, I-123 MIBG is regarded as an indicator of pulmonary endothelial function (Slosman et al. 1990). Elevated pulmonary artery systolic pressure caused by LV diastolic dysfunction may be involved in the mechanism of increased lung I-123 MIBG uptake (Nakae et al. 2013).
Increased MIBG uptake is known to be associated with improved prognosis in heart failure patients (Gerson et al. 2013). However, in the present study, there were no cardiopulmonary symptoms in the patients with increased lung uptake, and the reason for lung uptake could not be identified. Further research is required on this matter.
The availability of three-dimensional information allows the position of any abnormality to be more accurately located by SPECT. If the object under study contains a region of low activity within surrounding areas of increased or normal activity, such a cold lesion will appear to have normal activity due to superimposed tissue in front of and behind it. However, when the object is viewed with SPECT imaging, any slice through the cold lesion will show good contrast because the overlying normal tissues will appear in a different slice, and they will no longer be superimposed. SPECT is particularly useful for imaging organs where cold lesions may be present within normal tissue.
In the 6 PD patients with false-negative planar imaging, lung uptake was significantly increased. Diffuse lung uptake appears to be responsible for the overestimation of heart activity. The SPECT images of these patients provided good visualization of decreased myocardial uptake, as three-dimensional location of abnormality was available. In planar images, a region of decreased activity superimposed with anteriorly or posteriorly adjacent area of increased activity may appear to have normal activity. However, in SPECT images, the areas with increased and decreased activity are no longer superimposed, thus providing good contrast. SPECT is particularly useful in detection of cold lesions that is surrounded by areas of diffuse activity. However, the low count acquisition rate is the downside of SPECT.
In the current study, the pattern of decreased cardiac uptake in PD patients was dominant in the lateral wall, with relative sparing of the septum. These findings are in concordance with a previous C-11 HED PET study (Wong et al. 2012) and F-18 fluorodopamine PET studies (Goldstein et al. 2000; Li et al. 2002), which reported relatively lower sympathetic innervation in the lateral wall with sparing of the septum. Such pattern of cardiac sympathetic denervation is considered to be a result of the lateral wall and to a lesser extent, posterior (inferior) wall supplied by the left lateral cardiac nerve, which has the least amount of overlapping innervation (Janes et al. 1986). Studies that have evaluated adrenergic denervation in patients with heart disease have shown regional differences in myocardial catecholamine uptake, with decreased uptake in the ischemic zone of ischemic heart disease patients, increased uptake in hypertrophic septa of hypertrophic cardiomyopathy patients, and increased uptake in proximal anterior and septal walls of patients with transplanted hearts (Nakajima et al. 1990; Schwaiger et al. 1991; Simula et al. 2000).
We did not find significant differences in Hoehn and Yahr scores or duration of disease between the patients with preserved cardiac sympathetic innervation and those with severe cardiac sympathetic denervation. These results are in concordance with the study by Wong et al. (2012). Several studies have reported that I-123 MIBG uptake correlated with Hoehn and Yahr stage (Saiki et al. 2004; Kashihara et al. 2006). However, the correlation between MIBG scan findings and prognosis is unclear as of yet.
There are several limitations in the present study. Firstly, patients were diagnosed clinically without neuropathological investigations to confirm the presence of Lewy body pathology as the patients were still alive. Therefore, some patients may have been misdiagnosed as PD. However, we attempted to reduce these confounders by only including patients who fulfilled the clinical diagnostic criteria and radiologic findings by experienced neurologists and radiologists.
Secondly, although patients with history of cardiac disease were excluded, the possibility of cardiomyopathy cannot be completely ruled out. However, the possibility of decreased heart uptake due to undiagnosed coronary heart disease is low, as all patients with history of heart disease, symptoms of angina, or abnormal electrocardiogram findings were excluded.
Finally, this study did not include delayed images. This paper focused on the comparison between planar image and SPECT, and performing both lead to extending exam times. In order to prevent the discomfort of PD patients with mobilization difficulty, only early images were acquired. Future studies with delayed images may provide a comparison between the added information from myocardial washout rate and SPECT.
In diagnosis of PD, MIBG SPECT is superior to planar imaging. SPECT can accurately assess not only total heart activity, but also regional uptake pattern, providing additional information in differentiation between decreased MIBG uptake due to PD and heart disease.
References
Braune S, Reinhardt M, Schnitzer R, Riedel A, Lücking CH (1999) Cardiac uptake of [123I]MIBG separates Parkinson’s disease from multiple system atrophy. Neurology 53(5):1020–1025
Chung E, Lee W, Yoon W, Kim B, Lee G (2009) MIBG scintigraphy for differentiating Parkinson’s disease with autonomic dysfunction from Parkinsonism-predominant multiple system atrophy. Mov Disord 24(11):1650–1655
Daniel SE, Lees AJ (1993) Parkinson’s Disease Society Brain Bank, London: overview and research. J Neural Transm Suppl 39:165–172
Druschky A, Hilz MJ, Platsch G, Radespiel-Tröger M, Druschky K, Kuwert T, Neundörfer B (2000) Differentiation of Parkinson’s disease and multiple system atrophy in early disease stages by means of I-123-MIBG-SPECT. J Neurol Sci 175(1):3–12
Fukuoka M, Taki J, Mochizuki T, Kinuya S (2011) Comparison of diagnostic value of I-123 MIBG and high-dose I-131 MIBG scintigraphy including incremental value of SPECT/CT over planar image in patients with malignant pheochromocytoma/paraganglioma and neuroblastoma. Clin Nucl Med 36(1):1–7
Gerson M, Dwivedi A, Abdallah M, Shukla R, Jacobson A (2013) Significance of I-123 metaiodobenzylguanidine (123I-MIBG) lung activity in subjects with heart failure in comparison to healthy control subjects. Journal of nuclear cardiology 20(4):592–599
Goldstein DS, Holmes C, Li ST, Bruce S, Metman LV, Cannon RO (2000) Cardiac sympathetic denervation in Parkinson disease. Ann Intern Med 133(5):338–347
Izawa MO, Miwa H, Kajimoto Y, Kondo T (2012) Combination of transcranial sonography, olfactory testing, and MIBG myocardial scintigraphy as a diagnostic indicator for Parkinson’s disease. Eur J Neurol 19(3):411–416
Janes RD, Brandys JC, Hopkins DA, Johnstone DE, Murphy DA, Armour JA (1986) Anatomy of human extrinsic cardiac nerves and ganglia. Am J Cardiol 57(4):299–309
Jost W, Del Tredici K, Landvogt C, Braune S (2010) Importance of 123I-metaiodobenzylguanidine scintigraphy/single photon emission computed tomography for diagnosis and differential diagnostics of Parkinson syndromes. Neurodegener Dis 7(5):341–347
Kashihara K, Ohno M, Kawada S, Okumura Y (2006) Reduced cardiac uptake and enhanced washout of 123I-MIBG in pure autonomic failure occurs conjointly with Parkinson’s disease and dementia with Lewy bodies. J Nucl Med 47(7):1099–1101
Li S-T, Dendi R, Holmes C, Goldstein D (2002) Progressive loss of cardiac sympathetic innervation in Parkinson’s disease. Ann Neurol 52(2):220–223
Nakae I, Hayashi H, Mitsunami K, Horie M (2013) Clinical significance of lung iodine-123 metaiodobenzylguanidine uptake assessment in Parkinson’s and heart diseases. Ann Nucl Med. doi:10.1007/s12149-013-0741-5
Nakajima K, Bunko H, Taki J, Shimizu M, Muramori A, Hisada K (1990) Quantitative analysis of 123I-meta-iodobenzylguanidine (MIBG) uptake in hypertrophic cardiomyopathy. Am Heart J 119(6):1329–1337
Nakajo M, Shapiro B, Copp J, Kalff V, Gross MD, Sisson JC, Beierwaltes WH (1983) The normal and abnormal distribution of the adrenomedullary imaging agent m-[I-131]iodobenzylguanidine (I-131 MIBG) in man: evaluation by scintigraphy. J Nucl Med 24(8):672–682
Oh M, Kim J, Shin K-H, Park S, Kim H, Moon D, Oh S, Chung S, Lee C (2012) Subregional patterns of preferential striatal dopamine transporter loss differ in Parkinson disease, progressive supranuclear palsy, and multiple-system atrophy. J Nucl Med 53(3):399–406
Saiki S, Hirose G, Sakai K, Kataoka S, Hori A, Saiki M, Kaito M, Higashi K, Taki S, Kakeshita K, Fujino S, Miaki M (2004) Cardiac 123I-MIBG scintigraphy can assess the disease severity and phenotype of PD. J Neurol Sci 220(1–2):105–111
Sawada H, Oeda T, Yamamoto K, Kitagawa N, Mizuta E, Hosokawa R, Ohba M, Nishio R, Yamakawa K, Takeuchi H, Shimohama S, Takahashi R, Kawamura T (2009) Diagnostic accuracy of cardiac metaiodobenzylguanidine scintigraphy in Parkinson disease. Eur J Neurol 16(2):174–182
Schwaiger M, Hutchins GD, Kalff V, Rosenspire K, Haka MS, Mallette S, Deeb GM, Abrams GD, Wieland D (1991) Evidence for regional catecholamine uptake and storage sites in the transplanted human heart by positron emission tomography. J Clin Investig 87(5):1681–1690
Simula S, Lakka T, Laitinen T, Remes J, Kettunen R, Kuikka J, Hartikainen J (2000) Cardiac adrenergic denervation in patients with non-Q-wave versus Q-wave myocardial infarction. Eur J Nucl Med 27(7):816–821
Slosman DO, Donath A, Alderson PO (1989) 131I-metaiodobenzylguanidine and 125I-iodoamphetamine. Parameters of lung endothelial cell function and pulmonary vascular area. Eur J Nucl Med 15(4):207–210
Slosman DO, Polla BS, Donath A (1990) 123I-MIBG pulmonary removal: a biochemical marker of minimal lung endothelial cell lesions. Eur J Nucl Med 16(8–10):633–637
Sudmeyer M, Antke C, Zizek T, Beu M, Nikolaus S, Wojtecki L, Schnitzler A, Muller HW (2011) Diagnostic accuracy of combined FP-CIT, IBZM, and MIBG scintigraphy in the differential diagnosis of degenerative parkinsonism: a multidimensional statistical approach. J Nucl Med 52(5):733–740. doi:10.2967/jnumed.110.086959
Treglia G, Cason E, Stefanelli A, Cocciolillo F, Di Giuda D, Fagioli G, Giordano A (2012) MIBG scintigraphy in differential diagnosis of Parkinsonism: a meta-analysis. Clin Auton Res 22(1):43–55. doi:10.1007/s10286-011-0135-5
Watanabe M, Takeda T, Nakamagoe K, Tamaoka A (2011) Sequential imaging analysis using MIBG scintigraphy revealed progressive degeneration of cardiac sympathetic nerve in Parkinson’s disease. Eur J Neurol 18(7):1010–1013. doi:10.1111/j.1468-1331.2010.03211.x
Wong K, Raffel D, Koeppe R, Frey K, Bohnen N, Gilman S (2012) Pattern of cardiac sympathetic denervation in idiopathic Parkinson disease studied with 11C hydroxyephedrine PET. Radiology 265(1):240–247
Conflict of interest
All authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Oh, JK., Choi, EK., Song, IU. et al. Comparison of I-123 MIBG planar imaging and SPECT for the detection of decreased heart uptake in Parkinson disease. J Neural Transm 122, 1421–1427 (2015). https://doi.org/10.1007/s00702-015-1409-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-015-1409-1