Abstract
Purpose
To identify an appropriate surgical approach for meningiomas of the foramen magnum and lower clivus and determine the factors influencing the surgical outcomes.
Method
The study involved 23 patients with foramen magnum or lower clival meningiomas (8 men, 15 women; average age, 56 years; range, 26–70 years) treated at Keio University Hospital between 1991 and 2008. Their clinical data were retrospectively reviewed with regard to the surgical approaches and outcomes. The average follow-up duration was 42.8 months, the mean tumour size, 25.9 mm (range, 12.0–50.0 mm).
Findings
The tumours most commonly originated in the anterolateral rim of the foramen magnum. In 12 cases with lateral compression of the brain stem, the tumours were resected via the suboccipital approach with C1 laminectomy. The transcondylar approach was adopted in 11 cases where the tumour was located on the anterior rim of the foramen magnum. Four patients required epidural drilling of the jugular tubercle. Gross total resection was achieved in 15 cases (62.5%), the resection rate being lowest in cases of tumour extension to the lower clivus. The transient and permanent morbidity rates were 30.4% and 17.4%, respectively. Logistic regression analysis revealed that extension to the lower clivus (noted in 6 of 23 patients) was a statistically significant, independent factor influencing the permanent morbidity rate (p = 0.005).
Conclusions
Selecting an appropriate surgical approach considering the tumour location resulted in a good surgical outcome. However, tumour extension to the lower clivus strongly influenced the morbidity and tumour radicality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Meningiomas account for 70% of all benign tumours arising in the foramen magnum [15, 28], and meningiomas in this region account for 0.2–3.2% of all meningiomas [1]. Recent developments in surgical techniques have improved the surgical outcomes associated with such tumours [3, 8, 13, 21–23, 27]. Several approaches for the resection of meningiomas have been proposed and modified; these include the lateral suboccipital [20], transcondylar [4], condylar fossa [14], extreme lateral [23], and transoral approaches [16]. Meningiomas in the lower clivus cannot be clinically distinguished from those in the foramen magnum because of the anatomical continuity between the two types of tumours. The latter tumours may occasionally extend superiorly into the lower clivus, and the surgical outcomes in such cases have not been discussed in detail. Therefore, we included lower clival meningiomas in the present series study on foramen magnum meningiomas (FMMs). We aimed to clarify the factors influencing the surgical and follow-up outcomes of meningioma patients who underwent subtotal resection.
Patients and methods
Patient population
We retrospectively examined 23 consecutive cases of meningiomas arising in the foramen magnum and lower one-third of the clivus. All the concerned patients (9 men and 15 women) were surgically treated at the Department of Neurosurgery, Keio University Hospital, between September 1991 and July 2008. The mean age of the patients was 55.5 years (range, 26–70 years). The most common presenting symptoms and signs were paraesthesia and hemiparesis, each of which was observed in eight patients. Paresis of the lower cranial nerves was observed in six patients (glossopharyngeal and vagal nerve palsy in three patients, accessory nerve palsy in one, and hypoglossal nerve palsy in two), hearing disturbance in three, and facial nerve palsy in one (Table 1). Three patients had no clinical symptoms before the operation. All patients except one underwent surgery as the initial treatment.
Neuroimaging and classification
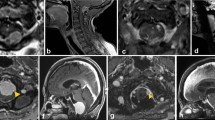
We retrospectively reviewed the patients’ clinical records and the findings of radiological examination [magnetic resonance (MR) imaging]. All the tumours originated from the region that has been defined as the foramen magnum by George et al. (anteriorly, from the lower one-third of the clivus to the upper edge of the axis; laterally, from the jugular tubercle to the C2 laminae; posteriorly, from the anterior edge of the squamous occipital bone to the C2 spinous process) [8, 9]. Using a modified version of the classification systems described by George et al. and Samii et al., we anatomically classified the meningiomas into the following three types according to their precise location in the foramen magnum and lower clivus: lateral, midline and lower clival (L-, M-, and C-type tumours, respectively; Fig. 1) [6, 8, 9, 20]. L-type meningiomas (14 cases) were attached to the anterolateral rim of the foramen magnum. M-type menigiomas (three cases) were attached to the anterior midline rim of the foramen magnum and caused deviation of the brain stem posteriorly. In C-type meningiomas (six cases), the bulk of the tumour was located on the lower one-third of the clivus. One patient with neurofibromatosis type 2 (case no. 6) had bilateral tumours on the rim of the foramen magnum. The mean size of the tumours was 26.5 mm (range, 12–50 mm). Extradural invasion was noted in three cases—around the dural ring of the vertebral artery (VA) in two cases and around the hypoglossal canal in one case.
Type of the tumour location: L-type meningiomas were attached to the anterolateral rim of the foramen magnum. M-type menigiomas were attached to the anterior midline rim of the foramen magnum and caused deviation of the brain stem posteriorly. In C-type meningiomas, the bulk of the tumour was located on the lower one-third of the clivus
Surgical approach
All the patients were positioned laterally. One of three surgical approaches, namely, the lateral suboccipital (condylar fossa) approach [14, 25], the transcondylar approach, or the presigmoid transpetrosal approach, was selected depending on the type and size of the tumour. C1 hemilaminectomy was also performed in the case of L- and C-type tumours. The jugular tubercle was epidurally resected in four cases where the tumours had extended into the midline lower clivus. Resection of the condyle was limited to the posterior quadrant. The VA was not transposed so as to avoid possible injury to the perforating arteries arising from the VA (Fig. 2). A staged operation via the presigmoid and transcondylar approaches was attempted in the case of one patient (case no. 19) with a large C-type tumour.
Case no. 14. a Preoperative MR image of an L-type tumour compressing the cervicomedullary junction. b Graphic representation of the operative view. A small perforating artery arising from the VA supplies blood to the spine (arrow). c Postoperative MR image showing the site after total tumour resection
Clinical and radiological follow-up
For all the patients, follow-up clinical and radiological examinations were performed every 6 months to check for tumour regrowth or recurrence. The morbidity at the time of discharge and the Karnofsky performance status (KPS) at the last follow-up examination (mean interval, 42.8 months) were evaluated. The Kruskal-Wallis test was used to analyse the relationship between surgical radicality and the tumour type. Logistic regression analysis was used to examine the influence of variables such as age, sex, tumour type, tumour size, dural invasion, the surgical approach, and the extent of tumour resection on the permanent morbidity rate.
Results
Gross total tumour resection (GTR) was achieved in 15 patients (65.2%), the GTR rate being 71.4% (10/14), 100% (3/3), and 33.3% (2/6) for L-, M-, and C-type tumours, respectively (Table 2). No significant difference in the resection rate was observed between tumour types in Kruskal-Wallis test. Incomplete resection was attributable to extradural invasion or adhesion of the tumour to the lower cranial nerves, the brain stem, or the VA (Figs. 3 and 4). There was no surgical mortality in this series. However, one patient with an M-type tumour (case no. 15), who was operated in 1992, i.e., before the introduction of the skull base approach, developed respiratory failure 3 days after the surgery and was bedridden. The KPS improved in 14 cases (60.8%) and deteriorated in 4 cases (17.4%). None of the patients who had undergone partial condylar resection complained of instability or restriction of neck movement. Long tract signs that were noted preoperatively largely improved after tumour resection. Hemiparesis, tetraparesis, paraesthesia, and hypaesthesia improved in 63% (5/8), 100% (1/1), 88% (7/8), and 100% (3/3) cases, respectively. However, one patient with a large C-type tumour (case 19) developed hemiparesis after the surgery. Transient and permanent cranial nerve deficit occurred in seven (30.4%) and four patients (17.4%), respectively. Dysphagia occurred in seven patients, two of whom developed permanent cranial nerve deficit; hoarseness occurred in three patients, 1 of whom developed permanent cranial nerve deficit; abducens nerve palsy occurred in four patients, two of whom developed permanent cranial nerve deficit. Permanent paresis of the lower cranial nerves and abducens palsy occurred only in the case of C-type tumours (Table 2). Further, of three patients who developed severe neurological deficits, two had C-type tumours. Logistic regression analysis revealed that tumour extension to the clivus (as in C-type tumours) was a statistically significant, independent factor influencing the permanent morbidity rate (p = 0.005), while age, sex, tumour size, dural invasion, surgical approach, and resection rate were not significant factors in this regard.
At the last follow-up examination, at a mean of 42.8 months after the surgery (range, 2–186 months), the mean KPS score was 89.5 (range, 30–100). All patients except two (91.7%) were caring for themselves. In 50% of the patients with C-type tumours, the KPS score was less than 80.0 (Table 2). Regrowth of the residual tumour was noted in only one of seven patients who underwent subtotal resection (case no. 11; recurrence rate, 4.3%). In this case, a little tumour was intentionally left because of its adhesion to the VA. Within 40 months thereafter, the residual tumour grew back to the size it was at the time of the first operation. The second operation was not successful because of the high degree of adhesion, and conventional radiation therapy was subsequently initiated. The tumour was histopathologically diagnosed as a clear cell meningioma with high proliferative activity (MIB-1 index, 10%; Fig. 5).
Discussion
Surgical approach
Ventrally located FMMs are confined to a space surrounded by the brain stem, lower cranial and cervical nerves, and vertebral artery; minimal retraction of these structures is warranted for tumour resection. In order to avoid severe surgical complications similar to those observed in case no. 15 in 1992, the transcondylar approach has widely been applied to reduce the damage that retraction causes to the brain stem. Among the early cases encountered in the present series, the transcondylar approach was widely indicated for tumours located beyond the midline of the ventral foramen magnum. Thereafter, this approach was reserved for relatively small M-type tumours that did not compress the brain stem laterally but did so posteriorly. In cases of lateral deviation of the brain stem (L-type tumours) and those of large M-type and C-type tumours, the surgical corridor was wide enough for tumour resection to be performed without condylectomy (Fig. 1). Samii et al. reported that in most cases, the time-consuming process of extensive drilling into the occipital condyle is not required to achieve adequate tumour exposure [20]. Moreover, the traditional suboccipital approach has proven effective for the treatment of FMMs in several previous series [2, 10, 11, 14, 17]. Suhardja et al. calculated and compared the area of the surgical window obtained when the lateral suboccipital approach and the transcondylar approach were used on cadaveric heads; they concluded that the latter approach afforded a significantly larger surgical field [5, 24]. However, the space created by the tumour was not considered in these previous studies. We emphasise that L-type tumours and large M-type and C-type tumours can be treated without condylectomy.
The necessity of VA transposition has been debated. Some surgeons suggested that transposition of the third part of the VA is essential [1, 13, 18, 19], while others noted that it is not necessary, especially in the case of intradural tumours [3, 4, 8]. Samii et al. reported that compromise of the small perforating arteries arising from the VA could be an important risk factor for postoperative cranial nerve palsy [20]. Therefore, the VA was not transposed in the present series in order to avoid injury to these arteries.
Clinical outcomes
In the present analysis, we identified tumour extension to the lower clivus as the sole factor influencing the permanent morbidity rate. When an appropriate surgical approach was selected, the tumour laterality did not influence the morbidity rate or the surgical radicality. George et al. classified FMMs with regard to three parameters: one in the axial plane, one in relation to the VA, and one in relation to the dura mater [6, 8, 9]. They compared the surgical outcomes in the case of tumours located anterior and lateral to the foramen magnum and found that the tumour location was not a statistically significant factor influencing the outcome. They also compared the surgical radicality in the case of anterior and lateral tumours [8] and concluded that this parameter is not influenced by the tumour type, but rather by extradural extension because of the invasive nature. Arnautovic et al. studied 18 FMM cases and identified radical tumour resection and a higher preoperative KPS score as significant factors influencing the clinical outcomes [1]. Further, Cushing et al. classified FMMs into the craniospinal and spinocranial types depending on their site of origin [7]. Thereafter, Samii et al. classified FMMs into four categories: spinocranial, craniospinal-clivus, craniospinal-lateral, and craniospinal-posterior [20]. They identified tumour extension to the clivus as an independent predictor of postoperative aspiration, because tumours exhibiting such invasion involved the lower cranial nerves and perforating arteries arising from the VA. Except with regard to tumours originating from the posterior margin of the foramen magnum (classified as craniospinal-posterior tumours), the classification system used by Samii et al. and their conclusions are very similar to those in the present study. In the case of tumours extending to the lower clivus, preservation of the lower cranial nerves and the arteries arising from the brain stem should be given priority over total tumour resection for improving the patient’s quality of life, because subtotally resected benign meningiomas have a very low recurrence rate. One of the reasons of the favourable outcome in this series might be due to preservation of small spinal arteries from the vertebral artery. We did not routinely perform intraoperative electrophysiological monitoring; however, in cases in which those must be sacrificed, the risk of brain stem ischemia should be evaluated beforehand by the electrophysiological monitorings [12, 26].
Conclusion
The use of an appropriate surgical approach to treat the FMMs resulted in good surgical outcomes with a mean KPS score of 89.5 and a mean recurrence rate of 4.3%. However, tumour extension to the lower clivus significantly increased the morbidity rate. Our results suggest that in the case of C-type meningiomas, which have a low recurrence rate, the degree of tumour resection should be carefully considered so as to avoid worsening the preoperative condition, except in cases of pathological malignancy.
References
Arnautovic KI, Al-Mefty O, Husain M (2000) Ventral foramen magnum meninigiomas. J Neurosurg 92:71–80
Bassiouni H, Ntoukas V, Asgari S, Sandalcioglu EI, Stolke D, Seifert V (2006) Foramen magnum meningiomas: clinical outcome after microsurgical resection via a posterolateral suboccipital retrocondylar approach. Neurosurgery 59:1177–1185 discussion 85–7
Bertalanffy H, Gilsbach JM, Mayfrank L, Klein HM, Kawase T, Seeger W (1996) Microsurgical management of ventral and ventrolateral foramen magnum meningiomas. Acta Neurochir Suppl 65:82–85
Bertalanffy H, Seeger W (1991) The dorsolateral, suboccipital, transcondylar approach to the lower clivus and anterior portion of the craniocervical junction. Neurosurgery 29:815–821
Boulton MR, Cusimano MD (2003) Foramen magnum meningiomas: concepts, classifications, and nuances. Neurosurg Focus 14:e10
Bruneau M, George B (2008) Foramen magnum meningiomas: detailed surgical approaches and technical aspects at Lariboisiere Hospital and review of the literature. Neurosurg Rev 31:19–32 discussion −3
Cushing H, Eisenhardt L (1938) Meningiomas, their classificaton, regional behavior, life history, and surgical results. Springfield, Baltimore
George B, Lot G, Boissonnet H (1997) Meningioma of the foramen magnum: a series of 40 cases. Surg Neurol 47:371–379
George B, Lot G, Velut S, Gelbert F, Mourier KL (1993) French language Society of Neurosurgery. 44th Annual Congress. Brussels, 8–12 June 1993. Tumors of the foramen magnum. Neurochirurgie 39(Suppl 1):1–89
Goel A, Desai K, Muzumdar D (2001) Surgery on anterior foramen magnum meningiomas using a conventional posterior suboccipital approach: a report on an experience with 17 cases. Neurosurgery 49:102–106 discussion 6–7
Gupta SK, Khosla VK, Chhabra R, Mukherjee KK (2004) Posterior midline approach for large anterior/anterolateral foramen magnum tumours. Br J Neurosurg 18:164–167
Kandenwein JA, Richter HP, Antoniadis G (2009) Foramen magnum meningiomas-experience with the posterior suboccipital approach. Br J Neurosurg 23:33–39
Kratimenos GP, Crockard HA (1993) The far lateral approach for ventrally placed foramen magnum and upper cervical spine tumours. Br J Neurosurg 7:129–140
Matsushima T, Natori Y, Katsuta T, Ikezaki K, Fukui M, Rhoton AL (1998) Microsurgical anatomy for lateral approaches to the foramen magnum with special reference to transcondylar fossa (supracondylar transjugular tubercle) approach. Skull Base Surg 8:119–125
Meyer FB, Ebersold MJ, Reese DF (1984) Benign tumors of the foramen magnum. J Neurosurg 61:136–142
Miller E, Crockard HA (1987) Transoral transclival removal of anteriorly placed meningiomas at the foramen magnum. Neurosurgery 20:966–968
Nanda A, Vincent DA, Vannemreddy PS, Baskaya MK, Chanda A (2002) Far-lateral approach to intradural lesions of the foramen magnum without resection of the occipital condyle. J Neurosurg 96:302–309
Pirotte B, David P, Noterman J, Brotchi J (1998) Lower clivus and foramen magnum anterolateral meningiomas: surgical strategy. Neurol Res 20:577–584
Roberti F, Sekhar LN, Kalavakonda C, Wright DC (2001) Posterior fossa meningiomas: surgical experience in 161 cases. Surg Neurol 56:8–20 discussion −1
Samii M, Klekamp J, Carvalho G (1996) Surgical results for meningiomas of the craniocervical junction. Neurosurgery 39:1086–1094 discussion 94–5
Seeger W (1978) Atlas of topograohical anatomy of the brain and surrounding structures. Springer-Verlag, Wien
Sekhar LN, Babu RP, Wright DC (1994) Surgical resection of cranial base meningiomas. Neurosurg Clin N Am 5:299–330
Sen CN, Sekhar LN (1990) An extreme lateral approach to intradural lesions of the cervical spine and foramen magnum. Neurosurgery 27:197–204
Suhardja A, Agur AM, Cusimano MD (2003) Anatomical basis of approaches to foramen magnum and lower clival meningiomas: comparison of retrosigmoid and transcondylar approaches. Neurosurg Focus 14:e9
Tange Y, Uto A, Wachi A, Koike J (2001) Transcondylar fossa approach to treat ventral foramen magnum meningioma-case report. Neurol Med Chir (Tokyo) 41:458–462
Wagner W, Peghini-Halbig L, Maurer JC, Perneczky A (1994) Intraoperative SEP monitoring in neurosurgery around the brain stem and cervical spinal cord: differential recording of subcortical components. J Neurosurg 81:213–220
Yasargil MG, Mrtara RW, Curcic M (1980) Meningiomas of basal posterior cranial fossa. Adv Tech Stand Neurosurg 7:1–115
Yasuoka S, Okazaki H, Daube JR, MacCarty CS (1978) Foramen magnum tumors. Analysis of 57 cases of benign extramedullary tumors. J Neurosurg 49:828–838
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
The authors provide a careful overview of their series of 23 patients treated with foramen magnum and lower clivus meningiomas at their institution. They note that the sole factor that influenced increased morbidity with resection was extension to the lower clivus. This is not surprising, given the risk for injury to lower cranial nerves, and brain stem perforators will increase with this extension. Those tumours limited to the rim of the foramen magnum present less of a surgical challenge.
I also agree with the authors that the extent of condyle resection should be tailored to the surgical corridor needed. I note that the Professor Kawase has evolved with his philosophy during the period that this series comprises, and has more recently used the full transcondylar approach only for relatively small M-type tumours that did not compress the brain stem laterally but did so anteriorly. In cases of lateral deviation of the brain stem (L-type tumours) and those of large M-type and C-type tumours,the surgical corridor is usually wide enough for tumour resection to be performed without condylectomy.
This reviewer commends the authors for sharing their valuable experience with the readership and providing the surgical pearls that they have gleaned from this experience.
WT Couldwell
Utah, USA
Rights and permissions
About this article
Cite this article
Kano, T., Kawase, T., Horiguchi, T. et al. Meningiomas of the ventral foramen magnum and lower clivus: factors influencing surgical morbidity, the extent of tumour resection, and tumour recurrence. Acta Neurochir 152, 79–86 (2010). https://doi.org/10.1007/s00701-009-0511-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-009-0511-2