Abstract
Nanosized carbon materials are offering great opportunities in various areas of nanotechnology. Carbon nanotubes and graphene, due to their unique mechanical, electronic, chemical, optical and electrochemical properties, represent the most interesting building blocks in various applications where analytical chemistry is of special importance. The possibility of conjugating carbon nanomaterials with biomolecules has received particular attention with respect to the design of chemical sensors and biosensors. This review describes the trends in this field as reported in the last 6 years in (bio)analytical chemistry in general, and in biosensing in particular.

Carbon nanotubes and graphene in analytical applications
Similar content being viewed by others
Avoid common mistakes on your manuscript.
General description on carbon nanotubes and graphene
In 1991, when carbon nanotubes (CNTs) were discovered [1], scientists were fascinated with this new form of carbon which changed the paradigm of the three basic forms of carbon: diamond, graphite and amorphous carbon. While research on CNTs was continuing, in 2004 another interesting material, graphene (G) [2] that was later gaining more attention was reported to be obtained at University of Manchester in the UK.
CNTs and G are two carbon allotropes with an identical composition which have a meshwork of sp2—hybridized carbon atoms [3] but have different structures. CNTs posses a cylindrical nanostructure formed by rolling up graphene sheets with a quasi-one-dimensional structure and G is an unrolled nanotube into a flat two-dimensional sheet. CNTs can be divided into single-walled CNTs (SWCNTs) and multi-walled CNTs (MWCNTs). The size and structure of CNTs and G can be analyzed by transmission electron microscopy (TEM) and atomic force microscopy (AFM) [4], as can be seen in the Fig. 1.
Owing to their identical composition, the properties of both materials would also be similar, although is not always the case, and the differences in their structure open new ways for further developments in biosensors. For example, G shows a better mechanical adhesion than CNTs, because CNT is a tubular structure and the contact area is minimized due to the curvature, whereas the contact area is maximal in graphene [5]. Furthermore, small changes in the charge environment caused by the adsorption of biomolecules can give measurable changes in their properties [3].
Based on the remarkable electronic and optical properties, CNTs and G have been extensively explored for chemical and biological sensing applications beside their use as catalyst supports. Tuning the graphene forms production according to (bio)sensing applications is of crucial importance. For electrochemical applications, the approach with chemical/thermal reduction of graphene oxide looks promising. In recent reports, graphene has been produced through the electrochemical reduction of graphene oxide [6–10]. The electrochemically reduced graphene oxide exhibit much better performance for electrochemical applications than chemically reduced one [8, 11].
A key step in CNT and G-based biosensing is the immobilization of biomolecules on their surface for their further use as recognition elements in various kinds of transducers including field-effect transistors (FETs). Two approaches for their functionalization have been reported: (1) noncovalent interaction including physical adsorption or entrapment of biomolecules on/within the material surface and (2) covalent interaction with the functional groups produced via chemical reactions. Thereby, the functionalized CNTs and G exhibit high sensitivity and selectivity in electrochemical, optical, and electronic biosensing systems. Furthermore, thanks to their small size, high electrical conductivity, and high surface-to-volume ratio, devices with fast response and high sensitivity have been developed [3, 5, 12–14]
CNTs and G, used in electrical biosensors [3, 14–20], have shown a high capacity for charge transfer, which makes them suitable to reach lower LODs and higher sensitivity values. These nanomaterials are used as modifiers of transducers ensuring efficient immobilization of biomolecules or other synthetic receptors which are the principal components to improve the performance of biosensors. Particularly, graphene is offering high benefices relative to CNTs [21, 22] in terms of Förster resonance energy transfer (FRET) quenching efficiency for biosensing applications, although if considering FET-style biosensors CNTs could present better results [3, 14]. New opportunities for generating or even amplifying the analytical signals by using these carbon nanoforms in biosensing are being opened [12, 15–17, 23, 24].
Another aspect to be considered is the dispersion of CNTs and G in various matrixes. Thanks to their large surface energy and strong interaction, both materials are difficult to be dispersed in polymeric matrices [25, 26]. Nevertheless, the use of suitable dispersing agents such as surfactants and polymers can preserve their original structures and intrinsic electrical properties, and improve their solubility in aqueous medium. Under these conditions these carbon nanomaterials can be transformed/integrated into various stronger, conductor and flexible devices with interest to be used in various applications. Of particular interest, for CNTs and G applications in analytical sciences, is the research related to electroanalysis field. The large specific surface area, good biocompatibility and a high adsorption capacity are some of the properties which make these materials suitable to be used in enzymatic sensors, immunosensors and genosensors (DNA sensors).
Considering the huge amount of publications concerning to analytical applications of CNTs and graphene, this review shows some of the most significant contributions appeared in the literature since 2006, particularly in the development of optical and electrochemical sensing systems. For example, some novel (bio)sensing and separation applications related to the electrocatalytic effects of CNTs and graphene, as well as the possibility to obtain composites based on their mixing with polymers or other chemical substances, and also their capability to be used as electrode modifiers, in FETs devices and as detectors in separation techniques such as high performance liquid chromatography (HPLC) or micro-capillary electrophoresis (MCE) are presented in the following sections for each one of these materials. Table 1 displays several examples from CNT and G-based biosensing. Details on the analytes, detection methods, detection limits (LODs) and kinds of applications are also included.
Carbon nanotubes
(Bio)sensing applications
CNT integration into biology based devices is an important trend in the current nanotechnology based analytical sciences [19, 22]. CNTs are offering significant advantages over many existing materials due to their high surface area, the facility for accumulating biomolecules, their excellent conductivity, minimization of surface fouling and electrocatalytic activity that they have. In particular, the unique properties of CNTs make them extremely attractive for the fabrication of electrochemical (bio)sensors [19, 20, 27–29]. Recent studies have demonstrated that CNTs can enhance the electrochemical reactivity of biomolecules and promote the electron–transfer reactions of proteins. The high conductivity of CNTs permits their use as highly sensitive nanoscale sensors and biosensors.
Different (bio)sensing platforms based on the use of CNTs as electrode modifiers have been developed. Our group has integrated CNTs onto a glassy carbon (GC) electrode by using a matrix based in MWCNTs, tetrahydrofuran (THF) mixed with poly(vinyl chloride) (PVC) and a glutaraldehyde (GA) solution, for β-Nicotinamide adenine dinucleotide (NADH) detection [30]. This CNTs matrix promotes better the electron transfer of NADH minimizing the fouling effect. GC electrode modified with CNTs shows remarkable electrochemical and mechanical advantages compared to bare GC electrode offering future alternatives for biosensors applications due to the ability of the developed design for the covalent binding of biological molecules. Another example based on (bio)sensing platforms using CNTs has been reported by Reuel et al. [31]. They developed a sensor array employing recombinant lectins as glycan recognition sites tethered via Histidine tags to Ni2+ complexes which act as fluorescent quenchers for semiconducting SWNTs embedded in a chitosan hydrogel spot. This detection platform is based on near-infrared (near-IR) fluorescent detection which allows measuring of binding kinetics of model glycans in real time in a similar way as the surface plasmon resonance (SPR). The absolute detection limit for the current platform was found to be 2 μg of glycosylated protein or 100 ng of free glycan to 20 μg of lectin.
On the other hand, CNTs mixed with magnetic nanoparticles (MNPs) functionalized with enzymes can provide magneto-switchable bioelectrocatalysis by using an external magnetic field. MNP-enzyme-CNT conjugate can simplify magneto-switching and open the door to a wide range of novel electrocatalytic and bioelectrocatalytic applications for magnetocontrolled redox enzymes. Based on this concept, a novel biosensor has been developed. This uses a MNPs-tyrosinase conjugate in operational synergy with MWCNTs, where an on—off external magnetic field is applied to a screen-printed electrode used as a transducing platform (see Fig. 2) [20]. The response of the biosensor to catechol is evaluated obtaining a limit of detection (LOD) around 7.61 μM (S/N = 3) with a relative standard deviation (RSD) of 4.91 % (n = 3).
Scheme of a tyrosinase-modified magnetic nanoparticles (MNPs) (size: 100 nm) conjugate in operational synergy with multi-wall carbon nanotubes (MWCNTs) by applying an on-off external magnetic field under a screen printing electrode (SPE). The insets show the current–time recordings for the catechol response from the SPE modified with a MWCNTs; b the bioconjugate of MNPs and tyrosinase; and c the bioconjugate of MNPs, tyrosinase, with MWCNTs, as well as the corresponding calibration plot. (Adapted from reference [20])
Another study based on magnetic particles (MPs) has been performed for the specific detection of ferrocene labels used for the immunodetection of dopamine in artificial and real samples [32]. Here, CNTs are adsorbed onto the surface of the beads and used as wiring tools for electrochemical biosensing. These CNT/MP complexes attached onto the electrode surface allow straightforward electrochemical sensing of the MP surface by exploiting CNT wiring.
Nanomaterials are also offering new opportunities in the development of new -based (bio)sensing systems for applications in food industry, environmental monitoring, clinic diagnostics and safety and security. In this context, an impedimetric detection method of a DNA sequence related to Influenza A (H1N1) virus using CNTs platform and AuNPs so as to improve the sensitivity and rapidity of analysis is reported [33]. This device uses colloidal gold for labelling of DNA oligonucleotides and the impedimetric signal of AuNPs onto screen printed carbon nanotubes electrode is measured and correlated to the DNA target concentration.
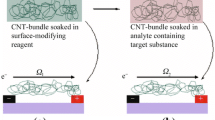
CNTs can be used as efficient transducers in solid-contact ion-selective electrodes [34]. CNTs must be connected with a suitable receptor to selectively detect a specific analyte. Aptamers are good receptor candidates for the selective and high-proficiency detection of a wide range of molecular targets, including bacteria [35]. In this way, a rapid and sensitive strategy for detection of living bacteria at ultralow concentrations using a CNT based potentiometric aptasensor has been reported by Rius’s group [35]. It takes advantages of CNTs with respect to the traditional culture detection method. The authors demonstrate that aptamer-based SWCNT potentiometric sensors are highly selective and can be successfully used to detect living microorganisms in an assay in real time (see Fig. 3). The most important advantage of this biosensor is that simple positive/negative tests can be carried without cross reaction with other types of bacteria. The easy way with which measurements are performed using a potentiometric sensor opens the door to simpler microbiological analysis.
A Schematic representation of the interaction between the target bacteria and the aptamer–SWCNT hybrid. B Potentiometric responses of the SWCNT electrode functionalized with aptamer for different concentrations of Salmonella Typhi (ST). Inset shows the fast response time (in seconds) of the inoculation step at 0.2 CFU mL-1 C Electromotive force (EMF) response versus log of concentration of ST. (Adapted with permission from reference [93]). For more details, see text
Furthermore, the development of simultaneous multiplex assays is showing a great efficiency in clinical applications, which shortens the analysis time and consequently decreases detection cost in comparison with the traditional single analyte assays. For example, a novel sandwich-type electrochemical aptasensor has been fabricated for simultaneous sensitive detection of platelet-derived growth factor (PDGF) and thrombin based on dual signal amplification of SWCNTs and multi-labeled graphene sheets using as redox probes toluidine blue (Tb) and ferrocene (Fc) attached with reduced graphene oxide (rGO) sheets respectively, which are subsequently coated with platinum nanoparticles (PtNPs) to form the PtNPs-redox probes-rGS nanocomposites. Thus, a signal amplification strategy based on bienzyme (glucose oxidase and horseradish peroxidase) modified PtNPs-redox probes-rGS nanocomposites as the tracer labels for secondary aptamers (Apt II) through sandwiched assay was described. AuNPs functionalized SWCNTs (AuNPs@SWCNTs) as the biosensor platform enhance the surface area to capture a large amount of primary aptamers (Apt I), thus amplifying the detection response. The results showed that the multi-labeled PtNPs-redox probes-rGS nanocomposites display satisfying electrochemical redox activity and highly electrocatalytic activity of PtNPs and bienzyme, which exhibit a LOD of 8 pM for PDGF and of 11 pM for thrombin [36].
Moreover, electrochemical detection using CNTs modified electrodes as detecting systems in separation techniques such as HPLC or capillary electrophoresis (CE) has also been reported [37–39]. For example, Pingarron’s group has modified a GC electrode with MWCNTs for amperometric detection of tetracycline (TC) antibiotics (tetracycline, oxytetracycline, chlortetracycline and doxycycline). They have demonstrated the possibility to carry out multiresidue analysis in samples containing tetracyclines and sulfadrugs (see Fig. 4). This HPLC with electrochemical detection was applied to the analysis of fish farm pool water and underground well water samples spiked with the four TCs at 0.2 μM. Thus, solid-phase extraction (SPE) was accomplished for the preconcentration of the analytes and clean-up of the samples [38].
A Cyclic voltammograms of 0.1 mM (a) tetracycline (TC) and (b) chlortetracycline (CTC) at a glassy carbon electrode modified with MWCNTs (MWCNT-GCE) (solid line); at a unmodified glassy carbon electrode (dashed line); and a background voltammogram in 0.05 M phosphate buffer solution of pH 2.0 (dotted line), ν = 50 mV s−1. B Chromatograms obtained from a (c) standard solution containing 10−4 M each of oxytetracycline (1, OTC), tetracycline (2, TC), chlortetracycline (3, CTC) and doxycycline (4, DC) with amperometric detection at a MWCNT-GCE. Mobile phase, 18:82 acetonitrile/0.05 M phosphate buffer of pH 2.5; flow rate, 1.0 mL min−1; Eapp = +1.20 V; (d) a water sample containing 20 mM each of sulfadiazine (1), sulfamerazine (2), OTC (3), TC (4) and sufamethoxazol (5), with amperometric detection at a MWCNT-GCE. (Adapted with permission from reference [38])
Separation applications
Preconcentration is an important step in various analytical process technologies used for trace-level determination; thus a variety of sample pretreatment techniques are being developed. Among the most promising technique for application in analytical chemistry is the SPE, which has provided an efficient tool using a large number of materials. In this context CNTs have been also reported to be used as adsorbents in SPE, and as stationary phases coupled with gas and liquid chromatography (LC) [37, 40–46]. CNTs provide an active surface for the adsorption/desorption of organic molecules that make them suitable as SPE adsorbents.
Analysis and speciation for cobalamins (various forms of vitamin B12) has been performed in seafoods by using LC that contains a MWCNT-packed mini-column system for on-line sample introduction [40]. Such system was tested to be an excellent alternative to trace-level determination.
On the other hand, gas chromatography (GC) has improved the selectivity of compounds detection by using modified CNTs as stationary phase. Speltini et al., [47] studied the separation of esters and chloroaromatic hydrocarbons by using chemically modified MWCNTs as stationary phase. Amino-terminated alkyl MWCNTs (MWCNTs-R-NH2) were synthesized by chemical modification of the nanotube skeleton by nucleophilic substitution with 2,2′-(ethylenedioxy) diethylamine. The so-prepared stationary phase was used for analysis of a synthetic mixture containing different classes of analytes, such as esters, ketones, alcohols, alkanes, and aromatic hydrocarbons. Good chromatographic profiles were obtained, with satisfactory resolution and peak shape, even for the most retained analytes. MWCNTs-R-NH2 had higher selectivity and resulted in enhanced resolution and better sorption–desorption behavior than non-functionalized MWCNTs (nf-MWCNTs).
Both SWCNTs and MWCNTs have been explored as high performance sorbents for μ-solid phase extraction in packed and self assembled formats. For example, Sae-Khow and Mitra [48] have reported the implementation of the μ-solid phase extraction in the needle of a syringe for integrating sampling, analyte enrichment and sample introduction into a single device. This device was constructed by using a syringe attached to a removable capillary probe (0.53 mm in ID) containing CNTs (as shown in Fig. 5). The CNTs were used in self-assembled (open tubular) as well as in packed formats. The analytical signals of the HPLC chromatograms of the original samples and samples enriched by μ-solid phase extraction with MWCNTs resulted significantly higher than the direct injection of a standard solution. MWCNTs provided the lowest LODs for naphthalene which was 0.1 ng mL−1 compared with 10 ng mL−1 from C-18 (S/N = 3). These results clearly indicate that CNTs have outstanding enrichment capabilities, and can be successfully used for trace analysis.
A Scheme of the μ-solid phase extraction device; B SEM images of (a) CNTs in self-assembled in the capillary probe, (b) SWNTs, and (c) MWNTs used for packing the μ-solid phase extraction probe; C HPLC chromatograms of (a) original samples and (b) samples enriched by μ-solid phase extraction with MWCNTs. (Adapted with permission from reference [48]). For more details, see text
Recently, CNTs are applied for preconcentration of heavy metals [42, 49], organics [50], and biological impurities [51] due to the large specific surface area. Cui and co-authors modified MWCNTs for preconcentration of Pb(II). The MWCNTs were grafted by the tris-(2-aminoethyl) amine (TAA), which has been proved as a good chelating reagent for metal ions [52]. TAA-grafted MWCNTs (MWCNTs-TAA) were employed as extractants for Pb(II) through a SPE process using a microcolumn packed with MWCNTs-TAA. The results show that the dispersibility of the MWCNTs-TAA is obviously increased compared with pristine MWCNTs and show good selectivity for adsorption of Pb(II) ions. A maximum adsorption capacity of 38 mg g−1 of Pb(II), a LOD 0.32 ng mL−1, an enrichment factor of 60 and RSD of 3.5 % (n = 6) were obtained.
Graphene
(Bio)sensing applications
A significant number of publications reporting graphene-based biosensors [12, 53–57] which have been used for improving the sensitivity and selectivity of biosensing systems based on the unique chemical, optical, electrical and electrochemical properties of G [3, 16, 21, 58–62] have appeared.
G is reported to decrease overpotentials of electrochemical reactions, ensure a better reversibility of some redox reactions, and bring novel labelling opportunities including multidetection capabilities.
The graphene’s electrical and optical properties are affected by several important factors such as number of layers, the used substrate, adsorbed impurities, flatness, defects, size of sheet, edge types and functionalization. These factors need to be considered and controlled where possible during the fabrication of graphene-based biosensing. The first factor, the number of layers, has special importance due to the fact that by its increasing the complexity of the electronic band structures, thereby the electrical and optical properties should change [3].
Graphene have been employed in various optical and electrochemical biosensors. In these applications, detection techniques such as fluorescence (FL), electrochemiluminescence (ECL) and FETs in addition to the use of G either as transducer or as biomolecular labels have been reported [16]. For example, Chen and co-authors [58] have studied the fluorescence quenching capacity of G and its potential for (bio)sensing. They have evaluated a graphene oxide (GO)-based photoinduced charge transfer (PCT) label-free near-infrared fluorescent biosensor for dopamine (DA), as shows the Fig. 6. The multiple noncovalent interactions between GO and dopamine (DA) resulted in effective self-assembly of DA on the surface of GO, and significant fluorescence quenching, allowing development of a simple label-free PCT-based near-IR fluorescence biosensor for selective and sensitive detection of DA in biological fluids with a LOD of 94 nM and a RSD of 2.0 %.
A Schematic representation for the graphene oxide (GO)-based photoinduced charge transfer fluorescent biosensor for dopamine (DA). B Fluorescence spectra of GO (25 μg mL-1) in presence of different DA concentrations (0, 0.25, 0.5, 1.0, 2.0, 3.0, 5.0, 10, 20, 30, 40, and 50 μM) under excitation at 450 nm and with a 5.0 mM Tris–HCl solution, pH = 5.0; C Quenched fluorescence intensity at 660 nm (ΔI) vs DA concentration ([DA]). (Adapted with permission from reference [58])
A novel self-assembled homogenous immunoassay has been developed by Liu et al. [63], which explores nanoscale-graphene sheets as excellent FL acceptors and CdTe QDs as donors for ultrasensitive detection of trace amounts of the target glycoprotein α-fetoprotein (AFP), a potential diagnostic biomarker for hepatocellular carcinoma. They demonstrated that the radiative quenching efficiency was distance independent on a wide dynamic range due to the effect of the two-dimensional G-based material, which significantly broke the distance limit in traditional Förster Resonance Energy Transfer (FRET) or Photoinduced Electron Transfer (PIET)-based biosensors.
Chemically modified graphene (CMG) is an ideal nanomaterial for the construction of FET transistors for sensing charged molecules. Therefore, CMG-based FETs can be employed for DNA sensing since DNA has a charged phosphate backbone [64]. The generation of holes is attributed to the negative-charge molecular gating from the phosphate ions of the complementary DNA. The change in conductivity due to hybridization/dehybridization varied from 60 % to 200 % for different graphene-DNA (G-DNA) samples. Immersing the G-DNA device in a solution of non-complementary DNA did not change the conductivity. Even though the DNA hybridization/dehybridization measurements were made in dry nitrogen conditions, they were effective in producing the negative-charge-gating. The results elucidate the high sensitivity of CMG nanostructures which function effectively as a label-free DNA detector and a molecular transistor. With these studies, Mohanty and Berry have demonstrated the interfacing of CMGs with biological systems to build a novel live-bacterial-hybrid device and a DNA-hybridization device with excellent sensitivity. CMGs, with their two-dimensional nanostructures and adjustable surface chemistry, can strongly interface with the biological systems without geometric restrictions and without compromising the integrity of the microbial attachment.
Other studies related with the detection of DNA hybridization based on CMG platforms has been developed by Pumera’s group [65, 66]. They have compared for the first time different graphene platforms modified with hairpin-DNA (hpDNA) probes for the sensitive detection of single nucleotide polymorphism (SNP) correlated to the development of Alzheimer’s disease. Graphene as transducer and electrochemical impedance spectroscopy (EIS) as a highly sensitive detection technique have been used. The LOD for each of the three different platforms was 50 nM for graphene nanoribbons consisting mostly of single- and double-layered graphene (G-SL), 6.6 pM for triple and four layer graphenes (G-FL), and 66 μM for multilayer graphene nanoribbons (G-ML) [65].
Several (bio)sensing systems have been reported to date involving several kinds of “graphene”, e.g., GO, chemically- rGO and graphene produced by direct in-liquid exfoliation (dG) [67]. For instance, Lu et al. [68] have reported a novel glucose biosensor by combining exfoliated graphite nanoplatelets (xGnPs) decorated with platinum (Pt) and palladium (Pd) nanoparticles, glucose oxidase (GOx), and Nafion. Pt-xGnP and Pd-xGnP-based glucose biosensors showed LODs of 1.0 and 4.0 μM (S/N = 3); detection limits of around 20 and 10 mM and sensitivities of 61.5 ± 0.6 and 47.9 ± 3.2 μA (mM·cm2)−1 upon the addition of 0.5 mM glucose solution in 50 mM phosphate buffer solution at 700 mV, respectively.
Shan and co-authors [69] developed another novel method for glucose detection. They have exploited the high surface area and electrical conductivity of rGO to attempt direct electron transfer (DET) between the GOx and the electrode. The polyethylenimine-functionalized ionic liquid (PFIL) has been used to construct the biosensor. High biocompatibility and exchangability of the counter-anions in PFIL, e.g., with negatively charged GOx, are favorable in order to immobilize biomolecules. Thereby, a graphene-PFIL solution (2 μL) was dropped and dried onto a GC electrode at room temperature for 24 h. Then, the graphene-PFIL modified GC electrode was impregnated in GOx solution (2 mg mL−1) for 24 h at 4 °C to obtain the graphene-GOx-PFIL modified GC electrode. This electrochemical biosensor achieved the DET of GOx maintaining its bioactivity and showing a potential application in the construction of novel glucose biosensors with linear glucose response up to 14 mM.
The determination of phenolic compounds is very important for evaluating the toxicity of environmental samples. Song et al. [70] developed a biosensor based on GO conjugated with tyrosinase assembled AuNPs for the determination of catechol. A screen-printed electrode was modified by using covalent attachment between 1-pyrenebutanoic acid, succinimidyl ester (PASE) adsorbing on the GO sheets and amines of tyrosinase-protected gold nanoparticles (Tyr–AuNPs). They detected catechol with a high sensitivity, good reproducibility and acceptable stability with a linear range of 8.3 × 10−8 to 2.3 × 10−5 M with a correlation coefficient (R2) of 0.9980 and a LOD of 2.4 × 10−8 M (S/N = 3).
On the other hand, G has also received significant attention as new and advantageous nanomaterial for electrochemical (bio)sensing in microfluidic devices. Martin and co-workers [71] evaluated GO amperometric detectors in microfluidic devices for detection of dopamine, catechol and nitroaromatic explosives, such as 2,4,6-trinitrotoluene (TNT), 2,4-dinitrotoluene (DNT) and 1,3-dinitrobenzene (DNB). They have reported that GO as electrochemical detector in the tested microchip doesn’t show any advantage in terms of sensitivity or selectivity over graphite microparticles, as showed in Fig. 7.
A Scheme of the microchip: (a) the glass chip, (b) separation channel, (c) buffer reservoir, (d) run buffer reservoir, (e) sample reservoir, (f) platinum cathode for separation, (g) detection electrode, (h) Ag/AgCl reference electrode, and (i) platinum counter electrode. B Electrophoregrams of a mixture of (a) dopamine and (b) catechol at bare glassy carbon (GC) (A), graphite microparticles (B), and GO (C) modified electrodes. Conditions: Dopamine and catechol at 400 and 100 μm respectively; 25 mM MES (pH 6.5); separation voltage, +1500 V; injection voltage, +1500 V; injection time, 5 s; detection potential, +0.6 V. Carbon material film loading, 1 μg. C Electrophoregrams of a mixture of (a) 1,3-dinitrobenzene (DNB), (b) 2,4,6-trinitrotoluene (TNT) and (c) 2,4-dinitrotoluene (DNT) (20 ppm for all) at bare GC (A), graphite microparticles (B), and GO (C) modified electrodes. Conditions: 15 mM borate buffer (pH 9.2) with 20 mM sodium dodecyl sulphate (SDS); separation voltage, +2000 V; injection voltage, +2000 V; injection time, 3 s; detection potential, −0.5 V. Carbon material film loading, 1 μg. (Adapted with permission from reference [71])
The optical properties of G have also been of great interest in (bio)sensing applications [72, 73]. Graphene oxide has been explored as acceptor of quantum dots (QDs) FRET donors into liquid phase and as acceptor of QDs FRET individual donors [63, 74]. According to Kim et al. [75] “the strong quenching by GO is likely due to the residual graphitic domains in the basal plane that survived the severe chemical oxidation”. For GO or reduced GO, as acceptors, the FRET effect can be independent of the emission spectra of the donor. Recently, a simple FRET evidence for the ultrahigh QD quenching efficiency by GO compared to graphite, carbon nanofibers and carbon nanotubes has been demonstrated [21]. The results evidence the fact that GO is the most powerful acceptor of QDs FRET donors.
The ultrahigh quenching efficiency of G opens the way to several interesting applications in the field of biosensing [23, 58, 67, 76], principally in bioimaging, labeling and sensing. For example, Lu and co-authors [76] have demonstrated optical testing of dye-labeled DNA. GO could bind dye-labeled ssDNA and completely quench the fluorescence of the dye. The binding between the dye-labeled DNA and target molecule alter the conformation of dye-labeled DNA, and disturbs the interaction between the dye-labeled DNA and GO, thus the target molecule releases itself from the GO and restores the quenched fluorescence (see Fig. 8). This design could result in a fluorescence-enhanced detection that is sensitive and selective to the target molecule.
A Schematic representation of the target-induced fluorescence change of the ssDNA-fluorescein-based dye (FAM)-GO complex. B Fluorescence spectra of the fluorescein-based dye-labeled aptamer-GO (dye-labeled aptamer 50 nM) in presence of human thrombin (0-100 nM). Excitation: 480 nm. (Adapted with permission from reference [76]). For more details, see text
Another platform for detecting biomolecules based on graphene has been performed by Chang et al. [77]. The biosensing platform was constructed according to the non-covalent assembly of fluorescent dye labeled aptamer on graphene for thrombin detection, using fluorescein amidite (FAM) as dye and 5′-FAM-GGTTGGTGTGGTTGG-3′ as DNA sequence of thrombin aptamer. In such a configuration, graphene quenched the fluorescence signal due to a transfer of fluorescence resonance energy from dye to graphene. Thereby, the conformation of aptamer on graphene can be changed by quadruplex formation induced by thrombin. The weak binding between quadruplex-thrombin complexes and graphene surface makes the dye to move far away from the graphene surface, inducing the fluorescence recovery. In this way, a highly sensitive and specific FRET aptasensor for thrombin detection was obtained (see Fig. 9). A linear range of 62.5–187.5 pM and a LOD for thrombin of around 31.3 pM, which is two orders of magnitude lower than those by using CNT fluorescent biosensors was reported [78].
A Schematic representation of graphene Fluorescence Resonance Energy Transfer (FRET) aptasensor for the thrombin detection; B Fluorescence recovery of graphene FRET aptasensor in the presence of different concentrations of thrombin (0, 31.3, 62.5, 93.8, 125, 156.3, 187.5, 218.8, and 250 pM) in 20 mM PBS buffer (pH = 7.0); C Relative fluorescence changes with thrombin concentration from 0 to 250 pM, where F0 and F are the fluorescence intensity without and with thrombin, respectively. FAM-aptamer concentration: 20 nM. Excitation wavelength: 470 nm. (Adapted with permission from reference [77]). For more details, see text
Separation applications
Due to its large specific surface area, high adsorption capacity and good chemical and thermal stability graphene is widely applied in analytical techniques such as chromatography [79–81], mass spectrometry [82–84] and atomic absorption spectrometry [85, 86].
Recently, Liu et al. [79] have used G as adsorbent in SPE for water samples monitoring with interest for environmental applications. Eight chlorophenols (2-Chlorophenol (2-CP), 3-chlorophenol (3-CP), 2,4-dichlorophenol (2,4-DCP), 3,4-dichlorophenol (3,4-DCP), 2,4,6-trichlorophenol (2,4,6-TCP), 4-Chlorophenol (4-CP), 2,3,5-trichlorophenol (2,3,5-TCP) and 2,3-Dichlorophenol (2,3-DCP)) as model analytes were extracted on a graphene-packed SPE cartridge, and then eluted with alkaline methanol. HPLC with multi-wavelength ultraviolet (UV) detection has been used to determine the concentrations of the elutes, obtaining a high sensitivity (LODs = 0.2, 0.2, 0.2, 0.2, 0.4, 0.1, 0.4, and 0.2 ng mL−1 (S/N = 3), respectively) and good reproducibility (RSDs = 5.9, 4.6, 5.3, 2.9, 4.8, 5.4, 7.7, and 2.2 % for run-to-run assays, respectively). By using graphene higher recoveries than other adsorbents including C18 silica, graphitic carbon and CNTs, owing to the large surface area and unique chemical structure have been achieved (as shown in Fig. 10). A similar method based on the separation and fluorescent detection by HPLC has been developed to extract neurotransmitters from rat brain, using graphene as a sorbent for SPE [81].
A AFM image of grahene oxide (GO) sheets on a mica substrate; B Effect of eluent type on the recoveries of 2-Chlorophenol (2-CP), 3-chlorophenol (3-CP,), 2,4-dichlorophenol (2,4-DCP), 3,4-dichlorophenol (3,4-DCP), 2,4,6-trichlorophenol (2,4,6-TCP), 4-Chlorophenol (4-CP), 2,3,5-trichlorophenol (2,3,5-TCP) and 2,3-Dichlorophenol (2,3-DCP); C Comparison of graphene versus C18 silica, graphitic carbon, SWCNTs, and MWCNTs, as adsorbents for the solid-phase extraction of eight chlorophenols. (Adapted with permission from reference [79]). For more details, see text
A novel graphene-assisted matrix solid-phase dispersion (GA-MSPD) method has been developed by Liu et al. [87] for extraction of polybrominated diphenyl ethers (PBDEs) and their methoxylated (MeO–) and hydroxylated (OH–) analogs from different kinds of spiked environmental samples, including soil, tree bark and fish. They found that grinding the solid sample with chemically converted graphene (CCG) powder yielded a tight contact and sufficient dispersion of the sample matrix due to the large surface area and flexible nanosheet morphology of CCG. The resultant blend was eluted using a two-step elution strategy: PBDEs and MeO-PBDEs were eluted firstly by hexane/dichloromethane and analyzed by gas chromatography–electron capture detection (GC–ECD), and then OH-PBDEs were eluted by acetone and determined by liquid chromatography–electrospray-tandem mass spectrometry (LC–ESI-MS/MS) (see Fig. 11). The method LODs of five PBDEs, ten MeO-PBDEs and ten OH-PBDEs (in soil samples) were in the range of 5.9–28.7, 14.3–46.6, and 5.3–212.6 pg g−1 dry weight, respectively. According to the authors [79, 87] graphene in comparison with other absorbents including C18 silica, graphitic carbon, florisil and CNTs gives improvements as SPE adsorbent owing to its high sorption capacity, good compatibility with various organic solvents, good reusability, no impact of sorbent drying, and fine reproducibility.
A Scheme of the graphene-assisted matrix solid-phase dispersion (GA-MSPD) method to extract polybrominated diphenyl ethers (PBDEs) and their methoxylated (MeO–) and hydroxylated (OH–) analogs from environmental matrices; B Comparison of chemically converted graphene (CCG) with C18, Florisil and MWCNTs as sorbents (20 mg) for extraction of PBDEs, MeO-PBDEs and OH-PBDEs from soils; C Comparison of GA-MSPD with other extraction techniques (ASE and Soxhlet extraction). (Adapted with permission from reference [87]). For more details, see text
Solid-phase extraction has also been proposed for separation and preconcentration of trace metal ions in environmental samples combined with flame atomic absorption spectrometry. Ma’s group [85, 86] has developed a new method for the preconcentration of trace amounts of lead (Pb) in water samples based on the use of a column packed with graphene as the sorbent, prior to its determination by flame atomic absorption spectrometry. G, as sorbent material used, is considered the core of SPE because it determines the selectivity and sensitivity of the method owing to its ultrahigh specific surface area and high adsorption capacity. They also showed that G is superior to other adsorbents including C18 silica, graphitic carbon, and single- and multi-walled CNTs for the extraction of Pb [85].
More recently, GO bonded fused-silica fiber has been applied in solid phase microextraction (SPME) of polycyclic aromatic hydrocarbons (PAHs) in water samples coupled with GC, achieving good results. The GO/SPME fiber provide good stability towards organic solvent, acidic and alkali solutions and high temperature, wide linearity range (from 0.05 to 200 mg L−1) with a R2 = 0.9954 and low LODs (less than 0.08 mg L−1) for extracting six PAHs couple with GC. The repeatability and fiber to fiber reproducibility were less than 6.13 and 15.87 %, respectively [80].
Conclusion & future prospects
CNTs and graphene offer significant advantages as analytical tools for biosensing systems. Graphene and chemically modified G sheets possess a high electrical conductivity, high surface area, and outstanding mechanical properties comparable with or even better than CNTs. The advantages of either CNTs or G would sometimes depend on the application field.
A wide variety of researches have demonstrated that GO can be used in a similar mode as CNTs as platforms for fast, sensitive, and selective biosensing. CNT and G-based composites seem to be suitable candidates to satisfy the requirements in biomedical devices, chemical analysis, time-resolved spectroscopy, environmental related sensing, and in optical devices. Composites infused with graphene are stronger, stiffer, and less prone to failure than composites infused with CNTs or other nanoparticles, according to reported studies [12, 88, 89]. This means that graphene, in relation to robustness, could be a key enabler in the development of next-generation of nanocomposite materials.
Despite the large amount of recent publications on CNTs and graphene-based biodetection, the advantages of those novel biosensing platforms over conventional bioassay tools remain to be clarified. Although high detection sensitivities have been achieved by many graphene-based biosensors, their reliability and reproducibility must be carefully studied following the corresponding electrochemical or optical techniques. However, graphene due to the low cost and large production scale is widely believed to be the material with interest to design and fabricate future devices. Of special interest seems to be the biosensing applications of graphene oxide in relation to optical techniques [72].
CNTs have been extensively and successfully used for sample treatment and preconcentration to overcome certain difficulties in analytical chemistry, such as unsatisfactory LODs and removing matrix interferences. It is expected that CNTs will be placed in the leading role as sorbent for SPE and replacing probably previous sorbents. Nevertheless graphene is also being demonstrated as a possible replacer of CNTs in the same field [90]. However, still much work is needed so as to definitively decide if either graphene or CNT is the ideal candidate for such applications.
The differences between CNTs and graphene could be associated with the surface chemistry, conductivity and their redox properties. A better understanding of the structure/property relationship with interest for fundamental research but also for developing effective biosensing platforms based on these carbon nanomaterials is still necessary. In general, the development of CNTs and graphene-based platforms with interest for analytical chemistry applications is still growing, and significant efforts are still showing to bring graphene to the same stature as CNTs. The future of graphene is bright and its amazing potential will surely be the focus of many researchers during the coming years.
References
Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354:56–58
Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, Grigorieva IV (2012) Electric field effect in atomically thin carbon films. Science 666:666–669
Yang W, Ratinac KR, Ringer SP, Thordarson P, Gooding JJ, Braet F (2010) Carbon nanomaterials in biosensors: should you use nanotubes or graphene? Angew Chem Int Ed 49:2114–2138
Merkoçi A (2005) Carbon nanotubes in analytical sciences. Microchim Acta 152:157–174
Biswas C, Lee YH (2011) Graphene versus carbon nanotubes in electronic devices. Adv Funct Mater 21:3806–3826
Wang Z, Zhou X, Zhang J, Boey F, Zhang H (2009) Direct electrochemical reduction of single-Layer graphene oxide and subsequent functionalization with glucose oxidase. JPhys Chem C 113:14071–14075
Peng X-Y, Liu X-X, Diamond D, Lau KT (2011) Synthesis of electrochemically-reduced graphene oxide film with controllable size and thickness and its use in supercapacitor. Carbon 49:3488–3496
Guo H-L, Wang X-F, Qian Q-Y, Wang F-B, Xia X-H (2009) A green approach to the synthesis of graphene nanosheets. ACS Nano 3:2653–2659
Dilimon VS, Sampath S (2011) Electrochemical preparation of few layer-graphene nanosheets via reduction of oriented exfoliated graphene oxide thin films in acetamide–urea–ammonium nitrate melt under ambient conditions. Thin Solid Films 519:2323–2327
Ramesha GK, Sampath S (2009) Electrochemical reduction of oriented graphene oxide films: an in situ raman spectroelectrochemical study. J Phys Chem C 113:7985–7989
Wang J, Yang S, Guo D, Yu P, Li D, Ye J, Mao L (2009) Comparative studies on electrochemical activity of graphene nanosheets and carbon nanotubes. Electrochem Commun 11:1892–1895
Artiles MS, Rout CS, Fisher TS (2011) Graphene-based hybrid materials and devices for biosensing. Adv Drug Deliv Rev 63:1352–1360
Pérez López B, Merkoçi A (2009) Improvement of the electrochemical detection of catechol by the use of a carbon nanotube based biosensor. Analyst 134:60–64
Chen Y, Vedala H, Kotchey GP, Audfray A, Cecioni S, Imberty A, Sébastien V, Star A (2012) Electronic detection of lectins using nanostructures: graphene versus carbon nanotubes. ACS Nano 6:760–770
Loo AH, Bonanni A, Pumera M (2012) Impedimetric thrombin aptasensor based on chemically modified graphenes. Nanoscale 4:143–147
Pumera M (2011) Graphene in biosensing. Mater Today 14:308–315
Kuila T, Bose S, Khanra P, Kumar A (2011) Recent advances in graphene-based biosensors. Biosensors Bioelectron 26:4637–4648
Angione MD, Pilolli R, Cotrone S, Magliulo M, Mallardi A, Palazzo G, Sabbatini L, Fine D, Dodabalapur A, Cioffi N, Torsi L (2011) Carbon based materials for electronic bio-sensing. Mater Today 14:424–433
Guix M, Pérez-López B, Sahin M, Roldán M, Ambrosi A, Merkoçi A (2010) Structural characterization by confocal laser scanning microscopy and electrochemical study of multi-walled carbon nanotube tyrosinase matrix for phenol detection. Analyst 135:1918–1925
Pérez-López B, Merkoçi A (2011) Magnetic nanoparticles modified with carbon nanotubes for electrocatalytic magnetoswitchable biosensing applications. Adv Funct Mater 21:255–260
Morales-Narváez E, Pérez-López B, Pires LB, Merkoçi A (2012) Simple förster resonance energy transfer evidence for the ultrahigh quantum dot quenching efficiency by graphene oxide compared to other carbon structures. Carbon 50:2987–2993
Wang BX, Wang C, Qu K, Song Y, Ren J, Miyoshi D, Sugimoto N, Xiaogang Q (2010) Ultrasensitive and selective detection of a prognostic indicator in early-stage cancer using graphene oxide and carbon nanotubes. Adv Funct Mater 20:3967–3971
Jiang H (2011) Chemical preparation of graphene-based nanomaterials and their applications in chemical and biological sensors. Adv Mater 7:2413–2427
Wang K, Liu Q, Dai L, Yan J, Ju C, Qiu B, Wu X (2011) A highly sensitive and rapid organophosphate biosensor based on enhancement of CdS – decorated graphene nanocomposite. Anal Chim Acta 695:84–88
Subrahmanyam KS, Vivekchand SRC, Govindaraj RCNR (2008) A study of graphenes prepared by different methods: characterization, properties and solubilization. J Mater Chem 18:1517–1523
Bahun GJ, Wang C, Adronov A (2006) Solubilizing single-walled carbon nanotubes with pyrene-functionalized block copolymers. J Polym Sci Part A: Polym Chem 44:1941–1951
Vashist SK, Zheng D, Al-Rubeaan K, Luong JHT, Sheu F-S (2011) Advances in carbon nanotube based electrochemical sensors for bioanalytical applications. Biotech Adv 29:169–188
Jacobs CB, Peairs MJ, Venton BJ (2010) Review: carbon nanotube based electrochemical sensors for biomolecules. Anal Chim Acta 662:105–127
Wang J, Lin Y (2008) Functionalized carbon nanotubes and nanofibers for biosensing applications. Trends Anal Chem 27:619–626
Pérez-López B, Sola J, Alegret S, Merkoçi A (2008) A carbon nanotube PVC based matrix modified with glutaraldehyde suitable for biosensor applications. Electroanal 20:603–610
Reuel NF, Ahn J-H, Kim J-H, Zhang J, Boghossian A, Mahal LK, Strano MS (2011) Transduction of glycan-lectin binding using near-infrared fluorescent single-walled carbon nanotubes for glycan profiling. J Am Chem Soc 133:17923–17933
Baldrich E, Muñoz FX (2011) Carbon nanotube wiring: a tool for straightforward electrochemical biosensing at magnetic particles. Anal Chem 83:9244–9250
Bonanni A, Pividori MI, Del Valle M (2010) Impedimetric detection of influenza A (H1N1) DNA sequence using carbon nanotubes platform and gold nanoparticles amplification. Analyst 135:1765–1772
Crespo GA, Macho S, Rius FX (2008) Ion-selective electrodes using carbon nanotubes as ion-to-electron transducers. Anal Chem 80:1316–1322
De-los-Santos-Álvarez N, Lobo-Castañón MJ, Miranda-Ordieres AJ, Tuñón-Blanco P (2008) Aptamers as recognition elements for label-free analytical devices. Trends Anal Chem 27:437–446
Bai L, Yuan R, Chai Y, Zhuo Y, Yuan Y, Wang Y (2012) Simultaneous electrochemical detection of multiple analytes based on dual signal amplification of single-walled carbon nanotubes and multi-labeled graphene sheets. Biomater 33:1090–1096
Agüí L, Yáñez-Sedeño P, Pingarrón JM (2008) Role of carbon nanotubes in electroanalytical chemistry: a review. Anal Chim Acta 622:11–47
Vega D, Agüí L, González-Cortés A, Yáñez-Sedeño P, Pingarrón JM (2007) Voltammetry and amperometric detection of tetracyclines at multi-wall carbon nanotube modified electrodes. Anal BioanalChem 389:951–958
Zhao J, Zhang Y, Wu K, Chen J, Zhou Y (2011) Electrochemical sensor for hazardous food colourant quinoline yellow based on carbon nanotube-modified electrode. Food Chem 128:569–572
Viñas P, López-García I, Bravo MB, Hernández-Córdoba M (2011) Multi-walled carbon nanotubes as solid-phase extraction adsorbents for the speciation of cobalamins in seafoods by liquid chromatography. Anal BioanalChem 401:1393–1399
André C, Aljhani R, Gharbi T, Guillaume YC (2011) Incorporation of carbon nanotubes in a silica HPLC column to enhance the chromatographic separation of peptides: theoretical and practical aspects. J SepScience 34:1221–1227
Ravelo-Pérez LM, Herrera-Herrera AV, Hernández-Borges J, Rodríguez-Delgado MA (2010) Carbon nanotubes: solid-phase extraction. J Chromatogr A 1217:2618–2641
Cui Y, Liu S, Hu Z-J, Liu X-H, Gao H-W (2011) Solid-phase extraction of lead(II) ions using multiwalled carbon nanotubes grafted with tris(2-aminoethyl)amine. Microchim Acta 174:107–113
Safavi A, Maleki N, Doroodmand MM (2010) Single-walled carbon nanotubes as stationary phase in gas chromatographic separation and determination of argon, carbon dioxide and hydrogen. Anal Chim Acta 675:207–212
Wu R-G, Yang C-S, Wang P-C, Tseng F-G (2009) Nanostructured pillars based on vertically aligned carbon nanotubes as the stationary phase in micro-CEC. Electrophoresis 30:2025–2031
Sombra L, Moliner-Martínez Y, Cárdenas S, Valcárcel M (2008) Carboxylic multi-walled carbon nanotubes as immobilized stationary phase in capillary electrochromatography. Electrophoresis 29:3850–3857
Speltini A, Merli D, Dondi D, Paganini G, Profumo A (2012) Improving selectivity in gas chromatography by using chemically modified multi-walled carbon nanotubes as stationary phase. Anal BioanalChem 403:1157–1165
Sae-Khow O, Mitra S (2009) Carbon nanotubes as the sorbent for integrating micro-solid phase extraction within the needle of a syringe. J Chromatogr A 1216:2270–2274
Chen S, Zhu L, Lu D, Cheng X, Zhou X (2010) Separation and chromium speciation by single-wall carbon nanotubes microcolumn and inductively coupled plasma mass spectrometry. Microchim Acta 169:123–128
Rastkari N, Ahmadkhaniha R, Yunesian M (2009) Single-walled carbon nanotubes as an effective adsorbent in solid-phase microextraction of low level methyl tert-butyl ether, ethyl tert-butyl ether and methyl tert-amyl ether from human urine. J Chromatogr B 877:1568–1574
Upadhyayula VKK, Deng S, Mitchell MC, Smith GB (2009) Application of carbon nanotube technology for removal of contaminants in drinking water: A review. Sci Total Environ 408:1–13
Huang X, Chang X, He Q, Cui Y, Zhai Y, Jiang N (2008) Tris(2-aminoethyl) amine functionalized silica gel for solid-phase extraction and preconcentration of Cr(III), Cd(II) and Pb(II) from waters. J Hazard Mater 157:154–160
Yang J, Deng S, Lei J, Ju H, Gunasekaran S (2011) Electrochemical synthesis of reduced graphene sheet-AuPd alloy nanoparticle composites for enzymatic biosensing. Biosens Bioelectron 29:159–166
Wang C, Li J, Amatore C, Chen Y, Jiang H, Wang X-M (2011) Gold nanoclusters and graphene nanocomposites for drug delivery and imaging of cancer cells. Angew Chem Int Ed 50:11644–11648
Kong F-Y, Li X-R, Zhao W-W, Xu J-J, Chen H-Y (2012) Graphene oxide–thionine–Au nanostructure composites: preparation and applications in non-enzymatic glucose sensing. Electrochem Commun 14:59–62
Fan Y, Liu J-H, Yang C-P, Yu M, Liu P (2011) Graphene–polyaniline composite film modified electrode for voltammetric determination of 4-aminophenol. Sens Actuators B 157:669–674
Pumera M, Ambrosi A, Bonanni A, Chng ELK, Poh HL (2010) Graphene for electrochemical sensing and biosensing. TrAC Trends in Anal Chem 29:954–965
Chen J-L, Yan X-P, Meng K, Wang S-F (2011) Graphene oxide based photoinduced charge transfer label-free near-infrared fluorescent biosensor for dopamine. Anal Chem 83:8787–8793
Alwarappan S, Erdem A, Liu C, Li C-Z (2009) Probing the electrochemical properties of graphene nanosheets for biosensing applications. J Phys Chem C 113:8853–8857
Choi BG, Im J, Kim HS, Park H (2011) Flow-injection amperometric glucose biosensors based on graphene/Nafion hybrid electrodes. Electrochim Acta 56:9721–9726
Roy S, Soin N, Bajpai R, Misra DS, McLaughlin JA, Roy SS (2011) Graphene oxide for electrochemical sensing applications. J Mater Chem 21:14725–14731
Chen D, Tang L, Li J (2010) Graphene-based materials in electrochemistry. Chem Soc Rev 39:3157–3180
Liu M, Zhao H, Quan X, Chen S, Fan X (2010) Distance-independent quenching of quantum dots by nanoscale-graphene in self-assembled sandwich immunoassay. Chem Commun 46:7909–7911
Mohanty N, Berry V (2008) Resolution biodevice and DNA transistor: interfacing graphene derivatives with nanoscale and microscale biocomponents. Nano Lett 8:4469–4476
Bonanni A, Pumera M (2011) Graphene platform for Hairpin-DNA-based impedimetric genosensing. ACS Nano 5:2356–2361
Giovanni M, Bonanni A, Pumera M (2012) Detection of DNA hybridization on chemically modified graphene platforms. Analyst 137:580–583
Stergiou DV, Diamanti EK, Gournis D, Prodromidis MI (2010) Comparative study of different types of graphenes as electrocatalysts for ascorbic acid. Electrochem Commun 12:1307–1309
Lu J, Do I, Drzal LT, Worden RM, Lee I (2008) Nanometal-decorated exfoliated graphite nanoplatelet based glucose biosensors with high sensitivity and fast response. ACS Nano 2:1825–1832
Shan C, Yang H, Song J, Han D, Ivaska A, Niu L (2009) Direct electrochemistry of glucose oxidase and biosensing for glucose based on graphene. Anal Chem 81:2378–2382
Song W, Li D-W, Li Y-T, Li Y, Long Y-T (2011) Disposable biosensor based on graphene oxide conjugated with tyrosinase assembled gold nanoparticles. Biosens Bioelectron 26:3181–3186
Chua CK, Ambrosi A, Pumera M (2011) Graphene based nanomaterials as electrochemical detectors in Lab-on-a-chip devices. Electrochem Commun 13:517–519
Morales-Narváez E, Merkoçi A (2012) Graphene oxide as an optical biosensing platform. Adv Mater 25:3298–3308
Jung JH, Cheon DS, Liu F, Lee KB, Seo TS (2010) A graphene oxide based immuno-biosensor for pathogen detection. Angew Chem Int Ed 49:5708–5711
Chen Z, Berciaud S, Nuckolls C, Heinz TF, Brus LE (2010) Energy transfer from individual semiconductor nanocrystals to graphene. ACS Nano 4:2964–2968
Kim J, Cote LJ, Kim F, Huang J (2010) Visualizing graphene based sheets by fluorescence quenching microscopy. J Am ChemSoc 132:260–267
Lu C-H, Yang H-H, Zhu C-L, Chen X, Chen G-N (2009) A graphene platform for sensing biomolecules. Angew Chem In Ed 48:4785–4787
Chang H, Tang L, Wang Y, Jiang J, Li J (2010) Graphene fluorescence resonance energy transfer aptasensor for the thrombin detection. Anal Chem 82:2341–2346
Yang R, Tang Z, Yan J, Kang H, Kim Y, Zhu Z, Tan W (2008) Noncovalent assembly of carbon nanotubes and single-stranded DNA: an effective sensing platform for probing biomolecular interactions. Anal Chem 80:7408–7413
Liu Q, Shi J, Zeng L, Wang T, Cai Y, Jiang G (2011) Evaluation of graphene as an advantageous adsorbent for solid-phase extraction with chlorophenols as model analytes. J Chromatogr A 1218:197–204
Xu L, Feng J, Li J, Liu X, Jiang S (2012) Graphene oxide bonded fused-silica fiber for solid-phase microextraction-gas chromatography of polycyclic aromatic hydrocarbons in water. J Sep Sci 35:93–100
Huang K-J, Yu S, Li J, Wu Z-W, Wei C-Y (2011) Extraction of neurotransmitters from rat brain using graphene as a solid-phase sorbent, and their fluorescent detection by HPLC. Microchim Acta 176:327–335
Gulbakan B, Yasun E, Shukoor MI, Zhu Z, You M, Tan X, Sanchez H, Powell DH, Dai H, Tan W (2010) A dual platform for selective analyte enrichment and ionization in mass spectrometry using aptamer-conjugated graphene oxide. J Am Chem Soc 132:17408–17410
Tang LAL, Wang J, Loh KP (2010) Graphene-based SELDI probe with ultrahigh extraction and sensitivity for DNA oligomer. J Am Chem Soc 132:10976–10977
Dong X, Cheng J, Li J, Wang Y (2010) Graphene as a novel matrix for the analysis of small molecules by MALDI-TOF MS. Anal Chem 82:6208–6214
Wang Y, Gao S, Zang X, Li J, Ma J (2012) Graphene-based solid-phase extraction combined with flame atomic absorption spectrometry for a sensitive determination of trace amounts of lead in environmental water and vegetable samples. Anal Chim Acta 716:112–118
Wang YK, Zang XH, Gao ST, Li JC, Ma JJ (2012) Application of graphene as a sorbent for the preconcentration and determination of trace amounts of lead in water samples prior to flame atomic absorption spectrometry. Microchim Acta 177:497–504
Liu Q, Shi J, Sun J, Wang T, Zeng L, Zhu N, Jiang G (2011) Graphene-assisted matrix solid-phase dispersion for extraction of polybrominated diphenyl ethers and their methoxylated and hydroxylated analogs from environmental samples. Anal Chim Acta 708:61–68
Bai H, Li C, Shi G (2011) Functional composite materials based on chemically converted graphene. Adv Mater 23:1089–1115
Kuilla T, Bhadra S, Yao D, Kim NH, Bose S, Lee JH (2010) Recent advances in graphene based polymer composites. Prog Polym Sci 35:1350–1375
Kauffman DR, Star (2010) A graphene versus carbon nanotubes for chemical sensor and fuel cell applications. Analyst 135:2790–2797
Zhang Q, Yang S, Zhang J, Zhang L, Kang P, Li J, Xu J, Zhou H, Song X-M (2011) Fabrication of an electrochemical platform based on the self-assembly of graphene oxide-multiwall carbon nanotube nanocomposite and horseradish peroxidase: direct electrochemistry and electrocatalysis. Nanotechnology 22:494010–494017
Alarcón-Angeles G, Pérez-López B, Palomar-Pardave M, Ramírez-Silva M, Alegret S, Merkoci A (2008) Enhanced host–guest electrochemical recognition of dopamine using cyclodextrin in the presence of carbon nanotubes. Carbon 46:898–906
Zelada-Guillén GA, Riu J, Düzgün A, Rius FX (2009) Immediate detection of living bacteria at ultralow concentrations using a carbon nanotube based potentiometric aptasensor. Angew Chem Int Ed 48:7334–7337
Acknowledgments
We acknowledge funding from the MICINN (Spain) for MAT2011 -25870 project and E.U.’s support under FP7 contract number 246513 “NADINE’ and Torres Quevedo scholarship given to Briza Pérez-López.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pérez-López, B., Merkoçi, A. Carbon nanotubes and graphene in analytical sciences. Microchim Acta 179, 1–16 (2012). https://doi.org/10.1007/s00604-012-0871-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-012-0871-9