Abstract
The relationship between late-onset gestational diabetes mellitus [GDM] and oxidative stress is not well known, and the importance of the oxidant/antioxidant equilibrium in the clinical evolution and its complications require elucidation. The aim of the study was to evaluate the relationships between maternal levels of markers of oxidative stress in women with late-onset GDM that, potentially, may have considerable clinical implications in the pathogenesis and/or the evolution of GDM. Pregnant women (n = 78; 53 with GDM, 25 controls), between the 24th and 29th week of gestation, were enrolled. Both groups were analysed for demographic data, perinatal and obstetrics outcomes together with the levels of the marker’s oxidative stress and antioxidant status. Control versus patient results in the univariate analysis were the following: pre-gestational body mass index [BMI] 23.31 ± 4.2 vs. 27.13 ± 4.6 kg/m2 (P = 0.001); weeks at delivery 39.2 ± 3.05 vs. 38.9 ± 1.8 (P = 0.09); Caesarean delivery 12.5 vs. 43% (P = 0.004); macrosomia 4 vs. 9.4% (P = 0.6); lipoperoxides [LPO] 2.06 ± 1.00 vs. 3.14 ± 1.55 μmol/mg (P = 0.001); catalase 3.23 ± 1.41 vs. 2.52 ± 1.3 nmol/min/ml (P = 0.03); superoxide dismutase [SOD] 0.11 ± 0.04 vs. 0.08 ± 0.01 U/ml (P = 0.0003); glutathione peroxidase [GPX] 0.03 ± 0.006 vs. 0.025 ± 0.006 nmol/min/ml (P = 0.01); glutathione reductase [GSH] 0.004 ± 0.002 vs. 0.004 ± 0.004 nmol/min/ml (P = 0.9)]; and glutathione transferase [GST] 0.0025 ± 0.0012 vs. 0.0027 ± 0.00017 nmol/min/ml (P = 0.7). Multivariate analysis showed catalase might have a protective effect against GDM development and LPO seems to be a risk factor for the disease. These data suggest an increase in oxidative stress and a decrease in antioxidative defence in women with late-onset GDM and, as such, may have considerable clinical implications in the pathogenesis and/or the course of the pregnancy in these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of GDM is around 5% of all pregnancies [1] and its presence increases the risk of macrosomia, perinatal morbido-mortality [2] and the subsequent development of diabetes mellitus type 2 [DMT2] in the mother [3]. The pathophysiology of GDM remains unclear. Pregnant women with GDM have a notable reduction in insulin sensitivity [4] that continues to the end of the pregnancy [5]. Characteristics of GDM such as hyperglycaemia, insulin resistance and hyperlipidaemia are components of DMT2 and metabolic syndrome [MetS] and precede these clinical conditions, i.e. GDM can be considered a pre-diabetic state [6].
GDM is defined as a carbohydrate intolerance of variable severity, which begins, or is identified, during gestation. It is independent of the treatment employed for its control and its post-partum clinical evolution [7]. This definition includes women with overt diabetes initially diagnosed during pregnancy and women with true pregnancy-induced glucose intolerance. The presence of inflammation in women with overt diabetes within the group of women considered as having gestational diabetes may be a confounding factor since it is well documented that inflammation is often present in people with diabetes mellitus [8]. This can influence the outcomes since, as has been described, MetS was present in 20% of women with hyperglycaemia during pregnancy [9]. However, when we studied only women with late-onset gestational diabetes, i.e. those with normal glucose tolerance in early pregnancy and subsequent hyperglycaemia, we observed that only 10% of them had MetS [10].
Oxidative stress implicated in the pathogenesis of vascular diseases such as atherosclerosis, diabetes and hypertension is a result of an imbalance between the increase in the formation of reactive oxidative substances [ROS] [11, 12] and the synthesis of antioxidative defence mechanisms [13]. One current hypothesis is that oxidative stress, following the mechanisms associated with the production of superoxide, is a pathogenic factor that is conducive to insulin resistance, to β-cell dysfunction, to glucose intolerance and, finally, to the development of DMT2 [14]. ROS is implicated in the regulation of important physiological functions [15] such as the activation of a variety of transcription factors, for example, nuclear factor-kB [NF-kB] that is recognised as the principal regulator of the expression of the genetic response to stress [16]. NF-kB regulates the expression of multiple immunological and inflammatory genes, including the pro-inflammatory cytokines, proteins and enzymes implicated in the generation of ROS [17]. The injurious effects of oxidative stress in the cardiovascular system are numerous and varied but can be summarised as the reduction in the availability of nitric oxide [18, 19], increase in the inflammatory markers implicated in accelerated atherosclerosis [20–23] and modifications of lipids and lipoproteins [24, 25].
Hyperglycaemia is acknowledged as the principal factor responsible for the complications of DMT2, and oxidative stress can play an important role not only in the pathogenesis of DMT2 but also in GDM and the development of mother–foetus complications [26, 27]. However, the relationships between the GDM and oxidative stress and the importance of the oxidant/antioxidant balance have not been clarified.
Very few studies have observed an association between GDM and markers of oxidative stress. Peuchant et al. [28] observed that the levels of malondialdehyde [MDA] were increased and the levels of GPX decreased. Chaudhari et al. [29] detected elevated levels of MDA and decreased levels of SOD in patients with GDM. Rajdl et al. [30] observed elevated levels of thiobarbituric acid-reactive substances [TBARS] and decreased levels of GSH. However, Orhan et al. [31] did not find differences in the levels of GPX, catalase or TBARS. Toescu et al. [32] described levels of lipids and LPO higher in women with DMT2, DMT1 and GDM, while the total antioxidant capacity [TAC] was decreased. In a recent study [33], TAC was decreased in GDM and pre-eclampsia, and MDA levels were higher only in GDM group but LPO levels were not changed either in GDM or in pre-eclampsia when compared to normal pregnancies. These data suggest that oxidative stress can be implicated in the progression and/or pathogenesis of GDM and that the reduced antioxidant defences, demonstrated in a few studies with a small number of subjects with GDM, can reflect a protective response to existing oxidative stress.
None of these previous studies separately evaluated women with late-onset GDM, which is a mild form of gestational diabetes. So, the question remains whether or not oxidative stress is present in women with true pregnancy-induced hyperglycaemia.
The objective of the present study was to evaluate whether oxidative stress is present in women with late-onset GDM and to assess the importance of the oxidant/antioxidant equilibrium in the clinical evolution GDM in relation to the obstetric complications and the perinatal outcomes.
Materials and methods
The study protocol was approved by the ethical committee of the University Hospital Puerta del Mar and followed the guidelines of the Helsinki protocol. This was a prospective case–control study comparing a population with late-onset GDM with another sample of pregnant women with similar characteristics but without GDM. The patients diagnosed as having late-onset GDM were recruited between June 2007 and October 2008 from among those attending our high-risk-pregnancy clinic. The inclusion criteria were the following: normal glucose tolerance in early pregnancy, diagnosis of GDM in the 2nd or 3rd trimester of gestation according to the criteria of the National Diabetes Data Group [34], age range between 18 and 40 years, absence of any form of pre-pregnancy diabetes or family history suggestive of monogenic diabetes, BMI < 40 kg/m2 and absence of concomitant systemic disease (chronic or acute or infectious). The selection of control individuals was from among pregnant women with the same characteristics, but with normal glucose tolerance (normal O’Sullivan test conducted between the 24th and 26th week of gestation). The pregnant women candidates who fulfilled the selection criteria were informed of the characteristics and objectives of the study before they signed specially designed informed consent forms to participate in the study. A venous blood sample for biochemical analyses was taken in all cases following a minimum of 8-h fast. The blood sample was maintained at 4°C until centrifuged following which the plasma was distributed in aliquots and stored at −80°C until needed for laboratory analyses.
After delivery, in a second phase of the study, the clinical notes were consulted to record obstetric and perinatal data, which included complications during pregnancy, gestational age at delivery, route of delivery, weight and Apgar’s scores of the newborn.
Laboratory analyses
Glucose was determined in venous blood using the biochemical system Modular DPD (Roche Diagnostics). HbA1c was measured in the Cobas Integra 700 autoanalyzer (Roche Diagnostics) using an immunoturbidimetric method for completely haemolysed, anti-coagulated blood. The reference range for healthy individuals was 4.5–5.7%. Lipid profile including total-cholesterol (Total-chol), triglycerides (TG), LDL-cholesterol (LDL-chol) and HDL-cholesterol (HDL-chol) were quantified in the Modular DPD biochemical autoanalyzer using enzymatic colorimetry. Urinary albumin was determined on 24-h urine collection using an immunoturbidimetric test in the Cobas Integra 700 system (Roche Diagnostics).
Determination of plasma and serum oxidative stress markers
Lipoperoxides [LPO] levels were measured in serum using a commercial kit (Cayman Chemical, Ann Arbor, MI, USA). The activities of glutathione peroxidase [GPX], glutathione reductase [GSH], glutathione transferase [GST], catalase and superoxide dismutase [SOD] were measured in plasma with a commercial kit (Cayman Chemical, Ann Arbor, MI, USA). The intra- and inter-assay coefficients of variation for GPX, GSH, GST, catalase and SOD were 5.7 and 7.2%; 3.7 and 9.3%; 4.1 and 7.9%; 3.8 and 8.9%; 3.2 and 3.7%, respectively.
Statistical analyses
Descriptive statistics of the variables measured are presented as mean, median, standard deviation [SD], standard error [SE] and ranges for quantitative variables, and percentages for the qualitative variables. For each categorical variable of interest, associations between the dependent and independent variables were determined using the Mantel–Henszel χ2 test with Yates’ correction. The Fisher test was applied in the case that the variable contained less than 5 measurements.
The magnitude of association was calculated using the odds ratio [OR] with the precision described by the 95% confidence interval (95%CI) using the Cornfield approximation.
Comparisons between quantitative variables and groups were with the Student t-test and one-way analysis of variance [ANOVA] for the parametric variables. Non-parametric variables were evaluated with the Mann–Whitney test.
Correlations among variables were assessed with Spearman’s correlation coefficient.
Multivariate analysis was performed using non-conditional logistic regression. The stepwise technique was used to select the independent variables introduced into the model. The initial criterion for acceptance was a level of significance of P < 0.05.
Statistical analyses were performed with the Statistical Package for Social Sciences (SPSS version 14.0 for Windows).
Results
The principal demographic and clinical characteristics of the population studied are summarised in Table 1. The control group did not differ from the patient group with late-onset GDM in relation to age, history of pregnancy termination, of macrosomia, gravidity/parity and weeks of gestation pre-diagnosis. However, the levels of systolic blood pressure [BPS] and diastolic blood pressure [BPD] and pre-gestational BMI were greater in the group of patients with GDM.
In the laboratory measurements (Table 1), the levels of TG were higher in the patients with GDM, the HDL-chol levels were lower and, as expected, the levels of glucose and HBA1c were higher. The rest of the measured variables showed no significant differences between the groups.
With respect to the obstetric and perinatal variables, only the percentage of Caesarean deliveries was higher in the patients with GDM (Table 1), with no significant differences in the other variables studied.
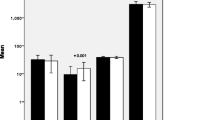
The levels of LPO were significantly higher in the patients with GDM compared to controls (Table 2). Of the enzymes involved in the redox cycle of glutathione, neither the levels of GSH nor of GST showed any significant differences between the GDM and control group of subjects. The levels of antioxidant enzymes such as catalase, SOD and GPX showed decreased levels in the patients with GDM compared to the controls (Table 2).
We did not observe significant relationships between the markers of oxidative stress, antioxidant status, metabolic control parameters and lipid profiles (results not shown).
In assessing the relationships between the different markers of oxidative stress, antioxidant status, obstetric and perinatal variables, we observed that the levels of GPX (0.024 ± 0.006; P = 0.012) and LPO (3.31 ± 2.1; P = 0.04) were significantly related to the percentage of Caesarean deliveries, while the percentage of macrosomia was related to the levels of GPX (0.032 ± 0.003; P = 0.01) (Table 3).
In the multivariate analysis with GDM as the dependent variable, the model included all the variables that showed statistical significance in the univariate analysis, as well as some other independent variables of possible clinical relevance. The results of this logistic regression analysis indicated that GDM was related to the markers of oxidative stress and of the antioxidant status (Table 4). Of note is that LPO appears to be a risk factor for GDM development while catalase might have a protective effect. Furthermore, we have analysed the differences between patients treated with diet alone and without insulin (n = 54; 67.9%) and with diet + insulin (n = 24; 30.7%). There were no differences in relation to demographic and clinical characteristics, laboratory variables and metabolic control between the groups (Table 5). The percentage of Caesarean deliveries and the gestational age at partum were higher in patients with GDM treated with insulin (Table 5), with no significant differences in the other variables studied. Neither were there differences in the markers of oxidative stress and antioxidants status between the groups (results not shown).
Discussion
The results of our study showed that there is an increase in oxidative stress and a decrease in the antioxidant defence in patients with late-onset GDM. These data are of note because oxidative stress could be implicated in the pathogenesis and clinical outcomes of the disease. Maternal obesity was the most important modifiable factor in the development of GDM [35], and the pre-gestational BMI was significantly higher in the patients with GDM. Similarly, we observed higher levels of TG and decreased levels of HDL-chol, as has been described by some investigators [36] but not confirmed by others [37]. Our results are not comparable to studies that included patients diagnosed from the start of the gestation, since our study dealt with patients with late-onset GDM. That we observed a higher rate of Caesarean deliveries in patients with GDM could be attributable, probably, to the consensus recommendations that delivery induction is advisable in patients with GDM, above all in those receiving insulin therapy [38]. This also could explain the finding of a gestational age at delivery significantly lower in women receiving insulin treatment. However, this attitude can increase the rate of Caesarean deliveries without affecting the risk of macrosomia [39] or improving the perinatal outcomes. This could have influenced our results since we did not observe any differences with respect to foetal weight, APGAR status, percentage of macrosomia or large-for-gestational age [LGA] values between the groups studied. More sensitive perinatal variables such as cord blood gasses, presence of meconial amniotic fluid, percentage admissions to the special care baby unit or neonate morbidity may need to be explored further before more definitive conclusions could be drawn.
The relationship between GDM and oxidative stress has not been well characterised, to date. The importance of the oxidant/antioxidant equilibrium in the clinical evolution and complications of GDM still remains to be elucidated. Several studies have observed associations between GDM and different markers of oxidative stress measured in plasma [28–33].
We observed elevated levels of LPO in the group of patients with GDM. Toescu et al. [32], in a prospective study that assessed changes in the lipid profile and markers of oxidative stress per trimester, observed that the lipid levels and that of LPO were more elevated in each trimester in women with DMT2, DMT1 and GDM. However, the observed changes were more severe in women with DMT2 and might be present before pregnancy. We did not observe significant associations between oxidative stress, antioxidant status and lipid profile, but our results are not comparable with previously published studies because we only dealt with late-onset GDM patients. Moreover, elevated levels of lipid peroxidation such as MDA [28, 29, 33] and TBARS [30], but not LPO [33], have been described in women with GDM, as well.
In our study, we observed that the levels of catalase were decreased in the patients with GDM. However, these results differ from other studies in which no significant differences had been described. In the study by Orhan et al. [31], which assessed the role of oxidative stress in pregnancies complicated by pre-eclampsia and DM, significant differences with respect to various complications of DM were observed, but not in the subgroup of patients with GDM. However, the study sample was very small. In our study, we observed decreased levels of SOD that were similar to the study by Chadhari et al. [29] who had conducted a case–control study with 20 women with GDM and 20 controls. They showed that levels of SOD were decreased in patients with GDM. We observed decreased levels of glutathione peroxidase in the GDM group, as did Peuchant et al. [28] in a study that evaluated oxidative and antioxidative status in 54 pregnant women. They reported reduced levels of GPX in patients with GDM compared to the healthy group. Thus, this reduced antioxidant defence (decreased levels of SOD, catalase and GPX) may, as has been previously suggested, reflect a protective response to an enhancement of oxidative stress.
It is important to note that the multivariate analysis showed that catalase might have a protective effect, and that LPO may be a risk factor for GDM development.
These limited data suggest that the increase in oxidative stress and the decrease in the antioxidant defence can be implicated in the progression and/or pathology of GDM. Hence, longitudinal studies over the course of gestation are needed to determine whether oxidative stress triggers the onset and the progression of GDM and whether improved glycaemic control will reduce the severity of oxidative stress. In accordance with the study by Karacay et al. [33], we found no differences between the markers of oxidative stress and antioxidant status in women with late-onset GDM treated with insulin.
With respect to obstetric and perinatal observations, we found that the levels of LPO were elevated and the GPX decreased in the women who had had a Caesarean section. This could be due to a greater oxidative capacity and lower antioxidative status in the gestational women with a higher metabolic disorder (for example, women with insulin treatment had a higher rate of Caesarean sections). Conversely, this finding does not appear to be related to neonate weight. As in cases of macrosomia in those who had a Caesarean delivery, the concentration of GPX is not decreased but, rather, is significantly elevated. The meaning of the relationship between the level of GPX and macrosomia, considering that there is no significant difference in the frequency of macrosomia between GDM and normal controls, is very difficult to establish. Whether this finding is related to GDM or with other circumstances should be addressed in more specific-designed research.
Our study has several limitations. First, the low number of cases studied. Secondly, the study had been limited to the gestational period and with no follow-up post-delivery. Thirdly, we found very little information in the literature to compare results to better formulate our conclusions. The strengths and the novelty of the present study are to have selected only women with late-onset GDM. As such, the vast majority of patients with undiagnosed overt diabetes have been excluded and the results can be clearly related to the true carbohydrate intolerance caused by pregnancy.
In conclusion, women with GDM have elevated levels of LPO and decreased antioxidants such as catalase and SOD. These findings suggest that there is an increase in oxidative stress and a lowering in antioxidative defence in GDM. This could have clinical implications in the progression of maternal GDM, on the gestational process, and, as well, could have a direct relationship with the number of Caesarean deliveries. Further prospective studies are required, with extensive post-partum follow-up, in order to evaluate the significance of these findings with greater precision.
Abbreviations
- ANOVA:
-
Analysis of variance
- BMI:
-
Body mass index
- DMT1:
-
Diabetes mellitus type 1
- DMT2:
-
Diabetes mellitus type 2
- BPD :
-
Diastolic blood pressure
- GDM:
-
Gestational diabetes mellitus
- GPX:
-
Glutathione peroxidase
- GSH:
-
Glutathione reductase
- GST:
-
Glutathione transferase
- LGA:
-
Large-for-gestational age
- LPO:
-
Lipoperoxides
- MDA:
-
Malondialdehyde
- MetS:
-
Metabolic syndrome
- NF-kB:
-
Nuclear factor-kB
- OR:
-
Odds ratio
- ROS:
-
Reactive oxidative substances
- SD:
-
Standard deviation
- SE:
-
Standard error
- SOD:
-
Superoxide dismutase
- BPS :
-
Systolic blood pressure
- TBARS:
-
Thiobarbituric acid reactive substances
- TAC:
-
Total antioxidant capacity
- TG:
-
Triglycerides
References
Ben-Haroush A, Yogev Y, Hod M (2004) Epidemiology of gestational diabetes mellitus and its association with type 2 diabetes. Diabetic Med 21:103–113
Ostlund I, Hanson U, Björklund A et al (2003) Maternal and fetal outcomes if gestational impaired glucose tolerance is not treated. Diabetes Care 26:2107–2111
Albareda M, Caballero A, Badell G et al (2003) Diabetes and abnormal glucose tolerance in women with previous gestational diabetes. Diabetes Care 26:1199–1205
Catalano PM, Huston L, Amini SB, Kalhan SC (1999) Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes. Am J Obstet Gynecol 180:903–916
Homko C, Sivan E, Chen X, Reece EA, Boden G (2001) Insulin secretion during and after pregnancy in patients with gestational diabetes mellitus. J Clin Endocrinol Metab 86:568–573
Xiang AH, Peters RK, Trigo E, Kjos SL, Lee WP, Buchanan TA (1999) Multiple metabolic defects during late pregnancy in women at high risk for type 2 diabetes mellitus. Diabetes 48:848–854
Metzger BE, The Organizing Committee (1991) Summary and recommendations of the 3rd international workshop-conference on gestational diabetes mellitus. Diabetes Care 40(Supp 2):197–201
Festa A, D’Agostino R Jr, Howard G, Mykkanen L, Tracy RP, Haffner SM (2000) Chronic subclinical inflammation as part of the insulin resistance syndrome: the insulin resistance atherosclerosis study (IRAS). Circulation 102:42–47
Bo S, Menato G, Gallo ML, Bardelli C et al (2004) Mild gestational hyperglycemia, the metabolic syndrome and adverse neonatal outcomes. Acta Obstet Gynecol Scand 83:335–340
Bartha JL, González-Bugatto F, Fernández-Macías R, González-González NL, Comino-Delgado R, Hervías-Vivancos B (2008) Metabolic syndrome in normal and complicated pregnancies. Eur J Obstet Gynecol Reprod Biol 137:178–184
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Maddux BA, See W, Lawrence JC, Goldfine AL, Goldfine ID, Evans JL (2001) Protection against oxidative stress-induced insulin resistance in rat L6 muscle cells by mircomolar concentrations of alpha-lipoic acid. Diabetes 50:404–410
Chen K, Thomas SR, Keaney JF (2003) Beyond LDL oxidation: ROS in vascular signal transduction. Free Radic Biol Med 35:117–132
Ceriello A, Motz E (2004) Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler Thromb Vasc Biol 24:816–823
Dröge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
Haddad JJ (2002) Oxygen homeostasis, thiol equilibrium and redox regulation of signalling transcription factors in the alveolar epithelium. Cell Signal 14:799–810
Barnes PJ, Karin M (1997) Nuclear factor-kB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 336:1066–1071
Du XL, Edelstein D, Dimmeler S, Ju Q, Sui C, Brownlee M (2001) Hyperglycemia inhibits endothelial nitric oxide synthase activity by posttranslational modification at the Akt site. J Clin Invest 108:1341–1348
Santilli F, Cipollone F, Mezzetti A, Chiarelli F (2004) The role of nitric oxide in the development of diabetic angiopathy. Horm Metab Res 36:319–335
Basta G, Schmidt AM, DeCaterina R (2004) Advanced glycation end products and vascular inflammation: implications for accelerated atherosclerosis in diabetes. Cardiovasc Res 63:582–592
Esposito K, Nappo F, Marfella R et al (2002) Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress. Circulation 106:2067–2072
Marfella R, Esposito K, Giunta R et al (2000) Circulating adhesion molecules in humans: role of hyperglycemia and hyperinsulinemia. Circulation 101:2247–2251
Ceriello A, Quagliaro L, Piconi L et al (2004) Effect of postprandial hypertriglyceridemia and hyperglycemia on circulating adhesion molecules and oxidative stress generation and the possible role of simvastatin treatment. Diabetes 53:701–710
Januszewski AS, Alderson NL, Metz TO, Thorpe SR, Baynes JW (2003) Role of lipids in chemical modification of proteins and development of complications in diabetes. Biochem Soc Trans 31:1413–1416
Jenkins AJ, Best JD, Klein RL, Lyons TJ (2004) Lipoproteins, glycoxidation and diabetic angiopathy. Diabetes Metab Res Rev 20:349–368
Baynes JW, Thrope SR (1999) Role of oxidative stress in diabetic complications. A new perspective on an old paradigm. Diabetes 48:1–9
Viana M, Herrera E, Bonet B (1996) Teratogenic effects of diabetes mellitus in the rat. Prevention by vitamin E. Diabetologia 39:1041–1046
Peuchant E, Brun JL, Rigalleau V et al (2004) Oxidative and antioxidative status in pregnant women with either gestational or type 1 diabetes. Clin Biochem 37:293–298
Chaudhari L, Tandon OP, Vaney N, Agarwal N (2003) Lipid peroxidation and antioxidant enzymes in gestational diabetics. Indian J Physiol Pharmacol 47:441–446
Rajdl D, Racek J, Steinerova A, Novotny Z, Stozicky F, Trefil L (2005) Markers of oxidative stress in diabetic mothers and their infants during delivery. Physiol Res 54:429–436
Orhan H, Önderoglu L, Yücel A, Sahin G (2003) Circulating biomarkers of oxidative stress in complicated pregnancies. Arch Gynecol Obstet 267:189–195
Toescu V, Nuttall SL, Martin U (2004) Changes in plasma lipids and markers of oxidative stress in normal pregnancy and pregnancies complicated by diabetes. Clin Sci 106:93–98
Karacay Ö, Sepici-Dincel A, Karcaaltincaba D et al (2010) A quantitative evaluation of total antioxidant status and oxidative stress markers in preeclampsia and gestational diabetic patients in 24–36 weeks of gestation. Diabetes Res Clin Pract 89:231–238
National Diabetes Data Group (1979) Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 28:1039–1057
Solomon CG, Willett WC, Carey VJ et al (1997) A prospective study of pregravid determinants of gestational diabetes mellitus. JAMA 278:1078–1083
Montelongo A, Lasuncion MA, Pallardo LF, Herrera E (1992) Longitudinal study of plasma lipoprotein and hormones during pregnancy in normal and diabetic women. Diabetes 41:1651–1659
Koukko E, Watts GF, Lowy C (1996) Serum lipid, lipoprotein and apolipoprotein changes in gestational diabetes mellitus: a cross-sectional and prospective study. J Clin Pathol 49:634–637
American Diabetes Association (1996) Gestational diabetes mellitus. Diabetes Care 27:88–90
Herranz L, Villaroel A, Riesco G et al (2003) Elective delivery in women with gestational diabetes mellitus. Diabetología 46:A248–A249
Acknowledgments
This study was financed, in part, by grants from the Andalusia Department of Health (CTS-368).
Conflict of interest
The authors declare that there is no conflict of interest that would prejudice the impartiality of this scientific work. Editorial assistance was by Dr Peter R Turner.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Tinoco, C., Roca, M., García-Valero, A. et al. Oxidative stress and antioxidant status in patients with late-onset gestational diabetes mellitus. Acta Diabetol 50, 201–208 (2013). https://doi.org/10.1007/s00592-011-0264-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-011-0264-2