Abstract
Cinnamon (Cinnamomum zeylanicum), fenugreek (Trigonella foenum-graecum), shallot (Allium hirtifolium Boiss), and clove (Syzygium aromaticum) are well-known and widely used medicinal plants in the world. There is some experimentally proven evidence demonstrated the beneficial effects of these herbs on metabolic disorders like diabetes. Despite the fact that there are limited studies in the field of herb-herb interactions, some of the diabetic patients prefer to use these herbs in a combination form. The aim of this study was to evaluate the possible effects of an equal mixture of these plants (named Tetraherbs) in streptozotocin (STZ)-induced diabetic rats. Severe diabetic rats (FBS above 350 mg/dL) were treated orally with ethanolic extracts of cinnamon (EEC), fenugreek (EEF), shallot (EES), and clove (EECL) separately at the dose of 75 mg/kg, or in equal combination formula (Tetraherbs) at doses of 100–300 mg/kg once per day for 28 days. At the end of the study, their effects on glucose levels, plasma lipids, liver enzymes activity, and histology of pancreas were evaluated. The blood glucose lowering activity, as well as pancreatic β cell regeneration of Tetraherbs, was significantly higher than the plants when they used separately. However, there was no significant difference in lipid-lowering and hepatoprotective potentials of the herbs whether used singly or in combination. In conclusion, the results of the present study provide evidence show combination form of the studied plants increased their glucose-lowering activity by positive interaction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes mellitus is defined as a group of chronic metabolic disorders characterized by persistent hyperglycemia resulting from a complete or relative lack of insulin secretion or action (Cole and Kramer 2016). The evidence demonstrated that persistent hyperglycemia may cause long-term complications including microvascular, macrovascular, and neuropathic alterations (Hami et al. 2016; Hassanzadeh-Taheri et al. 2016; Lotfi et al. 2016; Zarezadeh et al. 2017). According to the latest report of International Diabetes Federation, globally 8.8% of people aged 20–79 years had diabetes and approximately 5 million died from the disease in 2015, equivalent to one death every 6 seconds. If these trends continue, 642 million people, or one adult in ten, will have diabetes by 2040 (Ogurtsova et al. 2017).
The main strategies for the prevention and management of diabetes are lifestyle modifications including nutritional changes, physical activity, and use of different glucose-lowering medications (Gillies et al. 2007). However, despite these approaches, failure in control of glucose hemostasis occurs frequently among diabetic patients (Kokil et al. 2015). Moreover, due to some reasons such as popular belief about side effects of oral hypoglycemic drugs, and their low efficacy in managing all aspects of diabetes, scientists encouraged to explore and evaluate new methods to control the disease and its complications (Ekor and Pistelli 2013; Abtahi-Evari et al. 2017; Hassanzadeh-Taheri et al. 2018a).
In more recent years, the concepts of complementary and alternative medications (CAMs) have expanded worldwide tremendously. Among the multiple categories of CAMs, herbal medicine and nutraceuticals gained more popularity (Pandey et al. 2004). According to Iranian traditional medicine, the first step for treatment of any kind of disease is a lifestyle change, and the second step is the more advanced treatments such as using drugs or surgery. In sequence, recommended treatments often consist of proper diet, drugs (from single plants or mineral), followed by the combination of different herbs (Dabaghian et al. 2012). There are several polyherbal formulas for the treatment of diabetes in Iranian traditional medicine. Some of the medicinal herbs such as cinnamon bark (Cinnamomum zeylanicum), fenugreek seed (Trigonella foenum-graecum), shallot bulb (Allium hirtifolium Boiss), and clove flower bud (Syzygium aromaticum) are frequently used in different antidiabetic polyherbal formulas (Medagama and Bandara 2014; Katiyar et al. 2015; Yadav et al. 2015).
The hypoglycemic effects of cinnamon and fenugreek have been proposed by some molecular mechanisms such as decreasing gluconeogenesis, antioxidant effects, increasing glucose uptake, and direct insulin-like activities (Raju et al. 2001; Lu et al. 2012). Also, it is suggested that clove has antidiabetic potential through insulin-like activity, decreasing expression of phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6 phosphatase, and human peroxisome proliferator-activated receptor (PPAR)-γ ligand-binding activity (Kuroda et al. 2012). Moreover, there are some scientific reports regarding hypoglycemic effects of shallot in animal models (Mahmoodi et al. 2013a).
Due to different mechanisms of action and inconsistent reports of hypoglycemic activity of each plant, the aim of the present study was to investigate the probable synergistic or toxic effects of an equal mixture of ethanolic extracts of these four plants named here Tetraherbs on blood glucose, plasma lipids, liver enzymes, and pancreas histology in streptozotocin-induced diabetic rats.
Materials and methods
Preparation of plant extracts
The inner bark of cinnamon, the seed of fenugreek, the bulb of shallot, and the flower buds of clove were purchased from the traditional market in Birjand, Iran. The genus and species of the plants were authenticated by an expert botanist (Department of Botany, University of Birjand, Iran). The plants’ parts were washed, dried in shade and room temperature, powdered, and then stored in a cool and dry condition until use.
To prepare ethanolic extracts, a 100 g of each powdered plants organs was macerated in 1000 mL of 80% ethanol with constant stirring for 48 h at room temperature. Afterwards, the mixture was passed through a filter paper (Blue Ribbon, Grade 589, Germany), concentrated under a vacuum evaporator, and lyophilized using a freeze-dryer (Dena Vacuum Industry, model FD-5005-BT, Iran) (Hassanzadeh-Taheri et al. 2018b).
Diabetes induction
All of the animal experiments were approved and conducted in accordance with the guide for the laboratory animals’ care and usage of Birjand University of Medical Sciences, Birjand, Iran (IR.BUMS.1394.373).
Two-month-old male Wistar rats were housed in a temperature-controlled room (25 °C) with a 12-h dark/light cycle and free access to commercial laboratory animal food and tap water during the study period. Diabetes was induced by a single intraperitoneal (IP) injection of streptozotocin (STZ) (Sigma, 55 mg/kg dissolved in citrate buffer, pH 4.5) in the 14-h fasted rats (Vafaei-Nezhad et al. 2016). Age-matched control animals received an IP injection of vehicle (citrate buffer). After 72 h of the injection, animals with plasma glucose concentration above than 350 mg/dL were considered as severe diabetic (Fard et al. 2015).
Experimental design
All diabetic animals were left untreated for 4 weeks to develop a complicated form of diabetes (Davidson et al. 2011). Diabetic rats were randomly divided into nine equal (n = 8) groups while eight age-matched healthy rats were allocated as a normal control group. All groups were orally treated once per day with an equal volume of solutions (1 mL) for consecutive 28 days. Normal control group (NC) and diabetic model group (DM) were treated with saline 0.9%. Metformin (Merck, dissolved in saline) was administrated as a reference drug to another diabetic group at the dose of 500 mg/kg (MET500) (Waisundara et al. 2009). The seven remained diabetic groups were treated with ethanolic extracts of cinnamon (EEC), fenugreek (EEF), shallot (EES), and clove (EECL) at the dose of 75 mg/kg singly, or in equal combination (Tetraherbs) at the doses of 100 mg/kg (Tetraherbs 100), 200 mg/kg (Tetraherbs 200), and 300 mg/kg (Tetraherbs 300), respectively.
Biochemical analysis
At 29th day and after 24 h of the last treatment, the rats were anesthetized by IP injection of ketamine: xylazine (65:10 mg/kg) (Hassanzadeh-Taheri et al. 2018) and immediately their blood specimens were collected. The blood samples were centrifuged (3000g for 10 min) and the plasma collected. Fasting blood glucose and lipid profile including total cholesterol (TC), triglyceride (TG), high-density lipoprotein cholesterol (HDL-c), and low-density lipoprotein cholesterol (LDL-c) were assessed and liver enzymes including aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were evaluated using Pars Azmoon standard kits (Iran) and Roche Hitachi 912,auto-analyzer (Japan).
Oral glucose tolerance test in diabetic rats
In a complementary study, 30 diabetic and 6 healthy rats were divided into other six equal groups. Oral glucose tolerance test (OGTT) was carried out as follows:
-
Group I - Normal control received 0.9% saline
-
Group II - Diabetic control received 0.9% saline
-
Group III- Diabetic rats received metformin (500 mg/kg)
-
Group IV - Diabetic rats treated with 100 mg/kg of the Tetraherbs
-
Group V - Diabetic rats treated with 200 mg/kg of the Tetraherbs
-
Group VI- Diabetic rats treated with 300 mg/kg of the Tetraherbs
Thirty minutes after treatment with the solvent, reference drug (MET) and Tetraherbs, an oral glucose solution (Sigma, US) was administered at the dose of 3 g/kg (Ghiravani et al. 2016). Blood glucose was measured before and at 30, 60, 90, and 120 minutes after oral glucose loading using a glucometer (Accu-Check Active, Roche).
Histology
Immediately after blood collection, the animals were transcardially perfused with cold 0.9% saline, followed by a fixative solution containing 4% paraformaldehyde and 0.1 M glutaraldehyde in 0.1 M phosphate buffer (pH 7.4). Their pancreases were rapidly dissected, post-fixed in the same fixative solution for 48 h at 4 °C. The tissues were processed by routine histological methods and embedded in paraffin blocks. The specimens were sectioned (5-um thickness) using a sliding microtome (Leitz, Italy), mounted on poly-L-lysine coated slides, and stained with Hematoxylin and Eosin (H-E) dyes. For each rat, three random slides (9 sections) were analyzed under a light microscope (UPLAN FI, Japan) in a blinded manner. The morphometric parameters of pancreatic islets including the number of islets, area, and diameters of islets as well as the number of cells per islet were measured quantitatively by using Image J software (1.44p; National Institute of Health, USA).
Statistical analysis
Data were expressed as mean ± SEM. Experimental groups were compared using the ANOVA variance analysis test, and a post-hoc analysis using Tukey’s test was performed. Values of p < 0.05 were considered statistically significant. SPSS for Windows (SPSS, Chicago, IL, USA) was used to perform all statistical analyses.
Results
Hypoglycemic activity
In diabetic rats, STZ injection caused 4-fold increase in blood glucose levels. Before starting the treatments, all the animals had well-developed signs of diabetes (polyuria, polyphagia, polydipsia, and weight loss). Table 1 shows the changes in body weight as well as fasting blood glucose (FBG) levels in all studied groups.
The hypoglycemic activity of Tetraherbs was assessed by measuring FBG levels in initial and the last day of the treatments. As shown in Table 1, the FBG concentration of STZ-induced diabetic rats was significantly higher (p < 0.001) than the NC group in both 1st and 28th days of study. Moreover, before treatment (at 1st day), the FBG levels of the nine diabetic groups (DM, MET, EEC, EEF, EES, EECL, Tetraherbs 100, 200, and 300) were almost identical and there was no significant difference between them (p > 0.05). However, at the end of the study, all the investigations significantly decreased (p < 0.001) FBG levels in diabetic animals when compared to the DM group. Both of Metformin and Tetraherbs (300 mg/kg) exhibited the highest glucose-lowering activity in diabetic animals significantly more than the other investigations. There was no significant difference between MET500 and Tetraherbs 300 in FBG level (111.62 ± 16.09, 144.75 ± 36.32, respectively).
Effect of Tetraherbs on OGTT
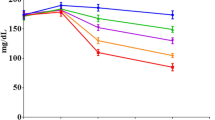
A graph of blood glucose levels (mg/dL) versus time (min) was constructed, and the area under the curve (AUC) was calculated according to the trapezoidal rule. The AUCs of each group were compared with the DM group, to represent glucose utilization by tissues.
Figure 1 illustrates the blood glucose levels of STZ-induced diabetic and non-diabetic rats. The diabetic groups had a significant elevation in blood glucose level throughout the total measurement period (120 min) compared to the NC group.
The AUC of each group was compared with the DM group. The results are presented in Table 2. Compared to the DM group, MET (p = 0.007), Tetraherbs 200 (p = 0.04), and Tetraherbs 300 (p = 0.014) could significantly decrease AUC in diabetic rats. These reductions were 54.87, 33.49, and 54.94%, respectively. Treatment with Tetraherbs at the dose of 100 mg/kg could not effectively (13.43%; p = 0.32) reduce AUC.
Hypolipidemic activity
At the end of the study, diabetic rats had significantly elevated plasma TG, TC, and LDL-c levels compared to the NC group (p < 0.001, each). However, there was no significant difference in HDL-c levels between the DM and NC groups.
In diabetic animals, the mean of TG concentration was about 62% higher than normal rats (65.00 ± 13.69 vs. 40.33 ± 3.16, p = 0.003). Treatment with EEC (46.16 ± 2.31, p = 0.04) and Tetraherbs 200 (44.57 ± 10.50, p = 0.019) decreased TG elevation in diabetic rats significantly.
In the DM group, the TC level was about 2-fold higher than the NC group (133.33 ± 25.64 vs. 55.66 ± 13.27, p < 0.001). All the investigations could decrease TC elevation in diabetic rats significantly (p < 0.05). There was no significant difference in TC-lowering activity between the herbs whether used singly or in combination.
Similar to cholesterol, LDL-c levels increased about 2-fold in diabetic rats compared to the NC group (59.66 ± 21.25 vs. 28.83 ± 4.21, p < 0.001). All the herbs significantly decreased LDL-c levels in diabetic rats when used singly. However, their combination (Tetraherbs) only at the dose of 200 mg/kg could ameliorate LDL-c levels in diabetic rats significantly (p < 0.001) (Table 3).
Liver enzymes activity
The effects of extracts on AST and ALT levels in diabetic rats are shown in Table 4. Compared to the NC group, plasma levels of AST and ALT significantly increased (p < 0.001) in diabetic rats. Diabetic animals had 130% greater AST levels than normal animals (260.83 ± 38.81 vs. 113.16 ± 16.33, p < 0.001). While all the investigations decreased the elevated AST levels in diabetic rats significantly (p < 0.05), the AST levels of the MET and EES groups were still remained higher than the NC group.
ALT levels of diabetic animals significantly elevated (2.5-fold) compared to the NC group (132.50 ± 11.79 vs. 49.33 ± 5.71, p < 0.001). Like AST, all the investigations could ameliorate elevated ALT levels in diabetic rats when compared to the DM group. However, only EEF and Tetraherbs at the doses of 200 mg/kg could normalize ALT levels in diabetic rats.
Pancreas histology
Figure 2 illustrates the pancreatic sections of the studied groups. Figure 2a demonstrates a normal Langerhans’ islet with normal appearance. However, sections of the diabetic groups showed the damaged islet, represented by a reduction in the size, shrinkage, marked reduction in cell density, and remaining cells showed small degenerated features such as dark and pycnotic nuclei, and also fibrosis and vacuolation were observed in the islets (Fig. 2b). Only single treatment of EEC showed some signs of the islet improvement (Fig. 2d). Treatment with Tetraherbs in a dose-dependent manner exhibited a slight improvement in size, cell density, and fibrosis of Langerhans’ islets in diabetic rats compared to the DM group (Fig. 2h–k). The results of the quantitative morphometric analysis are presented in Table 5. Tetraherbs in a dose-dependent manner significantly improved Langerhans’ islet cell density, islets number, islets area, and diameter in diabetic rats compared to the DM group.
a–k Photomicrographs of H-E stained slides in the islets of Langerhans of each group, × 400 magnification, scale bar = 25 um. a Normal control rats. b Diabetic model rats. c Diabetic rats treated with metformin. d Diabetic rats treated with 75 mg/kg of ethanol extract of cinnamon. e Diabetic rats treated with ethanolic extract of fenugreek. f Diabetic rats treated with ethanolic extract of shallot. g Diabetic rats treated with ethanolic extract of clove. h Diabetic rats treated with 100 mg/kg of Tetraherbs. j Diabetic rat treated with 200 mg/kg of Tetraherbs. k Diabetic rat treated with 300 mg/kg of Tetraherbs. The pancreatic specimen of the diabetic model group showed an apparent decrease in cell density and fibrosis (arrowheads), and few cells looked dark and degenerated (arrows). The severity of these alterations generally reduced in metformin-treated diabetic rats. The extracts of fenugreek, shallot, and clove did not cause any significant improvement in pancreatic architecture compared to untreated diabetic rats. Tetraherbs at the doses of 100 and 200 mg/kg could not effectively ameliorate the pancreatic alterations. However, the general morphology of pancreatic islets in diabetic animals treated with 300 mg/kg of Tetraherbs was improved and showed quite a normal appearance
Discussion
In the present study, STZ-induced diabetic rats were treated orally with the ethanolic extract of cinnamon (EEC), fenugreek (EEF), shallot (EES), and clove (EECL) singly at the dose of 75 mg/kg or in equal combination together (Tetraherbs) at the three doses (100–300 mg/kg) for 28 consecutive days. At the end of the study, their effects on glucose levels, plasma lipids, liver enzymes activity, and pancreas histology were evaluated. The blood glucose-lowering activity, as well as pancreatic β cell regeneration of combination formula, was significantly higher than the plants used singly. However, there was no significant difference in lipid-lowering and hepatoprotective activities of the herbs whether used singly or in combination.
In the most of experimental studies, interventions are performed routinely in subacute form (Shukla et al. 2011). In rodents, subacute investigation refers to repeated exposures for 1 month or less, and chronic interventional assay refers to repeated exposures for more than 3 months (Hassanzadeh-Taheri et al. 2018b). Therefore, in the present study, we performed the investigation during 28 days to find the effective dose and also long-term effects.
In diabetic rats, STZ caused significant hyperglycemia, weight loss, and histological degeneration of pancreas as shown in previous reports (Mythili et al. 2004). Although, Tetraherbs dose-dependently decreased the elevated FBG levels in diabetic rats, its effect was lower than metformin at the doses of 100 and 200 mg/kg. However, Tetraherbs showed significant improvement in OGTT at the doses of 200 and 300 mg/kg and this effect was similar to that of metformin in diabetic rats. These findings were inconsistent to previous studies reporting hypoglycemic effects of these medicinal plants or their active ingredients including of Cinnamaldehyde, Eugenol, Trigonelline, and Isoalliin (as a single drug) in animal models or diabetic patients (Jalal et al. 2007; Lu et al. 2012; Adefegha and Oboh 2012; Farrokhfall et al. 2014). Nevertheless, there are conflicting data about cinnamon and its main ingredients such as Cinnamaldehyde and Procyanidins. Some studies reported that cinnamon lowers glucose level by increasing glucose transporters (GLUT) in myocytes and adipocytes (Lu et al. 2012), increasing insulin secretion (Hosni et al. 2017), downregulation of gluconeogenesis gene expression (Cheng et al. 2012), and also its hypolipidemic effects were reported (Ping et al. 2010). On the other hand, there is evidence demonstrated that cinnamon did not change fasting blood glucose, cholesterol, and LDL-c levels (Akilen et al. 2012). Hypoglycemic effects of fenugreek and its main ingredient Trigonelline have been investigated and several studies reported its beneficial effects on FBG, OGTT, lipids, and HBA1c (Hemoglobin A1c) in both animal and human studies by regeneration of beta cells, increasing of insulin secretion, and antioxidant effects (Deng 2012). Mechanisms of action of clove and its major ingredients including Eugenol, Eugenol acetate, and Caryophyllene have been evaluated in both in vitro and animal experiments and it has been proposed that inhibition of α-glucosidase and α-amylase, increasing muscle glycolysis, improving mitochondrial function, and especially decreasing gluconeogenesis are responsible for its beneficial effects on glucose, urea, liver enzymes, and lipid levels (Prasad et al. 2005; Jeong et al. 2014; Tu et al. 2014).
Unlike the other plants of Tetraherbs, shallot has not investigated extensively. Phytochemical studies showed that shallot has a high amount of sulfur compounds such as Diallyl disulfide, S-allyl cysteine, and Diallyl trisulfide which, generally claimed to be responsible for antihypertensive, hypolipidemic, and antithrombotic potentials of this plant (Kubec and Dadakova 2009). One study reported that shallot lowered FBG slightly and improved OGTT while had no effects on LDL-C and HDL levels and even increased total cholesterol and urea in rat (Mahmoodi et al. 2013a).
The effects of Tetraherbs on lipid profile in diabetic rats exhibited significant ameliorating effects on TG, TC, and LDL-c particularly at the dose of 200 mg/kg; however, the other doses had no efficient effects. Generally, the drug interaction would generally produce one of three different effects: synergism, antagonism, and additive effect. The dose-response relation is a key topic in pharmacology (Gu et al. 2015). Hence, it seems that the studied plants, in high dose, have antagonistic interaction together mainly in lipid lowering-activity. Metformin slightly decreased TC and LDL-c, while it had no noticeable effects on TG elevation. These findings were inconsistent with previous studies with the same experimental design (Hule et al. 2011).
In a study conducted by Zarvandi et al., polyherbal formulation consisted of 39.1% fenugreek (main compound) could efficiently decrease FBG, TG, and LDL-c levels in diabetic patients (Zarvandi et al. 2017).
The results of histopathological examination of pancreatic islets showed that in single form of the herbs, only cinnamon could efficiently improve damaged islets. However, as expected, administration of Tetraherbs in a dose-dependent manner efficiently ameliorated pancreatic alterations. The exact mechanism by which Tetraherbs regenerated damaged islets has not elucidated in this study, but some hypothesis can be put forward. Similar to our findings, some evidence showed that phytochemicals can regenerate β cells in STZ-induced diabetic rats. Earlier studies showed that single administration of fenugreek, cinnamon, shallot, and clove could regenerate or prevent pancreatic beta cells destructions induced by Alloxan or STZ in rodents (Li et al. 2013; Mahmoodi et al. 2013b; Srinivasan et al. 2014; Joshi et al. 2015). In most of these studies, antioxidant properties were proposed as probable mechanism involved in β cell regenerations.
AST and ALT are two main aminotransferases that have leaked into circulation and served as hepatocytes injury markers. In this study, single or concurrent administration of the studied herbs decreased the elevated levels of AST and ALT in diabetic rats. The hepatoprotective effects of the single use of the plants by decreasing of ALT and AST have been reported in some previous studies (Sushma and Devasena 2010; Mbarki et al. 2017).
Recently, polyherbal formulations have become an interesting area of research and several formulations have been studied to attenuate hyperglycemia and its related complications. Some of these polyherbal formulations showed favorable results. The most prevalent plants used in these products were Momordica charantia, G. sylvestre, T. foenum-graecum, Curcuma longa, and Syzygium cumini. However, one of the disadvantages of active ingredients of plants is low bioavailability (Kalia and Gauttam 2013). Fortunately, some of the plants such as cinnamon have convoy effects, and therefore may increase absorption of other drugs into target organs by inhibition of P-glycoproteins and p450 enzymes (Sadati et al. 2016). Hypoglycemic effects and improvement of histopathological changes in the pancreas were superior in Tetraherbs compared to single administration of each plant. These results may be due to convoy activity of the herbs involved in the Tetraherbs.
Conclusion
The evidence from this study supports the use of Tetraherbs as an antidiabetic remedy in Iranian traditional medicine. The antidiabetic activity of Tetraherbs is supported by biochemical and histopathologic analyses.
References
Abtahi-Evari S-H, Shokoohi M, Abbasi A et al (2017) Protective effect of Galega officinalis extract on Streptozotocin-induced kidney damage and biochemical factor in diabetic rats. Crescent J Med Biol Sci 4:108–114
Adefegha SA, Oboh G (2012) In vitro inhibition activity of polyphenol-rich extracts from Syzygium aromaticum (L.) Merr. & Perry (Clove) buds against carbohydrate hydrolyzing enzymes linked to type 2 diabetes and Fe(2+)-induced lipid peroxidation in rat pancreas. Asian Pac J Trop Biomed 2:774–781. https://doi.org/10.1016/S2221-1691(12)60228-7
Akilen R, Tsiami A, Devendra D, Robinson N (2012) Cinnamon in glycaemic control: systematic review and meta analysis. Clin Nutr 31:609–615. https://doi.org/10.1016/j.clnu.2012.04.003
Cheng DM, Kuhn P, Poulev A, Rojo LE, Lila MA, Raskin I (2012) In vivo and in vitro antidiabetic effects of aqueous cinnamon extract and cinnamon polyphenol-enhanced food matrix. Food Chem 135:2994–3002. https://doi.org/10.1016/j.foodchem.2012.06.117
Cole L, Kramer P (2016) Human physiology, biochemistry and basic medicine - chapter 2.2. Academic Press
Dabaghian FH, Kamalinejad M, Shojaei A, Abdollahi Fard M (2012) Presenting anti-diabetic plants in Iranian traditional medicine. J Diabetes Endocrinol 3:70–76. https://doi.org/10.5897/JDE12.004
Davidson EP, Coppey LJ, Holmes A, Dake B, Yorek MA (2011) Effect of treatment of high fat fed / low dose streptozotocin-diabetic rats with Ilepatril on vascular and neural complications. Eur J Pharmacol 668:497–506. https://doi.org/10.1016/j.ejphar.2011.07.016
Deng R (2012) A review of the hypoglycemic effects of five commonly used herbal food supplements. Recent Pat Food Nutr Agric 4:50–60. https://doi.org/10.2174/2212798411204010050
Ekor M, Pistelli L (2013) Frontiers The growing use of herbal medicines issues relating to adverse reactions and challenges in monitoring safety. Pharmacology Front Pharmacol v.4:177. doi:10.3389%2Ffphar.2013.00177
Fard MH, Naseh G, Lotfi N et al (2015) Effects of aqueous extract of turnip leaf (Brassica rapa) in alloxan-induced diabetic rats. Avicenna J Phytomedicine 5:148–156
Farrokhfall K, Khoshbaten A, Zahediasl S, Mehrani H, Karbalaei N (2014) Improved islet function is associated with anti-inflammatory, antioxidant and hypoglycemic potential of cinnamaldehyde on metabolic syndrome induced by high tail fat in rats. J Funct Foods 10:397–406. https://doi.org/10.1016/j.jff.2014.07.014
Ghiravani Z, Hosseini M, Taheri MMH, Fard MH, Abedini MR (2016) Evaluation of hypoglycemic and hypolipidemic effects of internal septum of walnut fruit in alloxan-induced diabetic rats. African J Tradit Complement Altern Med 13:94–100. https://doi.org/10.4314/ajtcam.v13i2.12
Gillies CL, Abrams KR, Lambert PC, Cooper NJ, Sutton AJ, Hsu RT, Khunti K (2007) Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: systematic review and meta-analysis. Bmj 334:299–299. https://doi.org/10.1136/bmj.39063.689375.55
Gu J, Zhang X, Ma Y, Li N, Luo F, Cao L, Wang Z, Yuan G, Chen L, Xiao W, Xu X (2015) Quantitative modeling of dose–response and drug combination based on pathway network. J Cheminform 7:19. https://doi.org/10.1186/s13321-015-0066-6
Hami J, Vafaei-nezhad S, Ghaemi K, Sadeghi A, Ivar G, Shojae F, Hosseini M (2016) Stereological study of the effects of maternal diabetes on cerebellar cortex development in rat. Metab Brain Dis 31:643–652. https://doi.org/10.1007/s11011-016-9802-5
Hassanzadeh-Taheri M, Hassanpour-Fard M, Doostabadi M, Moodi H, Vazifeshenas-Darmiyan K, Hosseini M (2018a) Co-administration effects of aqueous extract of turnip leaf and metformin in diabetic rats. J Tradit Complement Med 8:178–183. https://doi.org/10.1016/j.jtcme.2017.05.010
Hassanzadeh-Taheri M, Hosseini M, Hassanpour-Fard M, Ghiravani Z, Vazifeshenas-Darmiyan K, Yousefi S, Ezi S (2016) Effect of turnip leaf and root extracts on renal function in diabetic rats. Orient Pharm Exp Med 16:279–286. https://doi.org/10.1007/s13596-016-0249-3
Hassanzadeh-Taheri, Mohammadmehdi, Hosseini M, et al (2018b) Acute and sub-acute oral toxicity evaluation of Astragalus hamosus seedpod ethanolic extract in Wistar rats. 24:23–30. doi: https://doi.org/10.15171/PS.2018.05
Hassanzadeh- Taheri M, Hassanzadeh- Taheri M, Jahani F, Hosseini M (2018) Effects of yoghurt butter oils on rat plasma lipids, haematology and liver histology parameters in a 150-day study. Int J Dairy Technol 71:140–148
Hosni AA, Abdel-Moneim AA, Abdel-Reheim ES, Mohamed SM, Helmy H (2017) Cinnamaldehyde potentially attenuates gestational hyperglycemia in rats through modulation of PPARγ, proinflammatory cytokines and oxidative stress. Biomed Pharmacother 88:52–60
Hule AK, Shah AS, Gambhire MN, Juvekar AR (2011) An evaluation of the antidiabetic effects of Elaeocarpus ganitrus in experimental animals. Indian J Pharm 43:56–59. https://doi.org/10.4103/0253-7613.75671
Jalal R, Bagheri SM, Moghimi A, Rasuli MB (2007) Hypoglycemic effect of aqueous shallot and garlic extracts in rats with fructose-induced insulin resistance. J Clin Biochem Nutr 41:218–223. https://doi.org/10.3164/jcbn.2007031
Jeong KJ, Kim DY, Quan HY, Jo HK, Kim GW, Chung SH (2014) Effects of eugenol on hepatic glucose production and AMPK signaling pathway in hepatocytes and C57BL/6J mice. Fitoterapia 93:150–162. https://doi.org/10.1016/j.fitote.2013.12.023
Joshi DV, Patil RR, Naik SR (2015) Hydroalcohol extract of Trigonella foenum-graecum seed attenuates markers of inflammation and oxidative stress while improving exocrine function in diabetic rats. Pharm Biol 53:201–211. https://doi.org/10.3109/13880209.2014.913296
Kalia A, Gauttam V (2013) Development of polyherbal antidiabetic formulation encapsulated in the phospholipids vesicle system. J Adv Pharm Technol Res 4:108–117. https://doi.org/10.4103/2231-4040.111527
Katiyar D, Singh V, Gilani SJ, Goel R, Grover P, Vats A (2015) Hypoglycemic herbs and their polyherbal formulations: a comprehensive review. Med Chem Res 24:1–21. https://doi.org/10.1007/s00044-014-1080-3
Kokil GR, Veedu RN, Ramm GA, Prins JB, Parekh HS (2015) Type 2 diabetes mellitus: limitations of conventional therapies and intervention with nucleic acid-based therapeutics. Chem Rev 115:4719–4743. https://doi.org/10.1021/cr5002832
Kubec R, Dadakova E (2009) Chromatographic methods for determination of S-substituted cysteine derivatives—a comparative study. J Chromatogr A 1216:6957–6963. https://doi.org/10.1016/j.chroma.2009.08.032
Kuroda M, Mimaki Y, Ohtomo T, Yamada J, Nishiyama T, Mae T, Kishida H, Kawada T (2012) Hypoglycemic effects of clove (Syzygium aromaticum flower buds) on genetically diabetic KK-A y mice and identification of the active ingredients. J Nat Med 66:394–399. https://doi.org/10.1007/s11418-011-0593-z
Li R, Liang T, Xu L, Li Y, Zhang S, Duan X (2013) Protective effect of cinnamon polyphenols against STZ-diabetic mice fed high-sugar, high-fat diet and its underlying mechanism. Food Chem Toxicol 51:419–425. https://doi.org/10.1016/j.fct.2012.10.024
Lotfi N, Hami J, Hosseini M, Haghir D, Haghir H (2016) Diabetes during pregnancy enhanced neuronal death in the hippocampus of rat offspring. Int J Devl Neurosci Int J Dev Neurosci 51:28–35. https://doi.org/10.1016/j.ijdevneu.2016.04.009
Lu T, Sheng H, Wu J, Cheng Y, Zhu J, Chen Y (2012) Cinnamon extract improves fasting blood glucose and glycosylated hemoglobin level in Chinese patients with type 2 diabetes. Nutr Res 32:408–412. https://doi.org/10.1016/j.nutres.2012.05.003
Mahmoodi M, Hosseini J, Hosseini Zijoud S-M et al (2013a) The effect of Persian shallot (Allium hirtifolium Boiss.) extract on blood sugar and serum levels of some hormones in diabetic rats. Pak J Pharm Sci 26:397–402
Mahmoodi M, Zarei S, Rezaeian M, Arababadi MK, Ghasemi H, Khoramdelazad H, Rezayati N, Hasanshahi G, Hosseini-Zijoud SM (2013b) Persian shallot (Allium hirtifolium Boiss) extract elevates glucokinase (GCK) activity and gene expression in diabetic rats. Am J Plant Sci 4:1393–1399
Mbarki S, Alimi H, Bouzenna H, Elfeki A, Hfaiedh N (2017) ScienceDirect phytochemical study and protective effect of Trigonella foenum graecum (Fenugreek seeds) against carbon tetrachloride-induced toxicity in liver and kidney of male rat. Biomed Pharmacother 88:19–26. https://doi.org/10.1016/j.biopha.2016.12.078
Medagama A, Bandara R (2014) The use of Complementary and Alternative Medicines (CAMs) in the treatment of diabetes mellitus: is continued use safe and effective? Nutr J 13:1–9. https://doi.org/10.1186/1475-2891-13-102
Mythili MD, Vyas R, Akila G, Gunasekaran S (2004) Effect of streptozotocin on the ultrastructure of rat pancreatic islets. Microsc Res Tech 63:274–281. https://doi.org/10.1002/jemt.20039
Ogurtsova K, da Rocha Fernandes JD, Huang Y, Linnenkamp U, Guariguata L, Cho NH, Cavan D, Shaw JE, Makaroff LE (2017) IDF Diabetes Atlas: global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract 128:40–50. https://doi.org/10.1016/j.diabres.2017.03.024
Pandey A, Tripathi P, Pandey R et al (2004) Alternative therapies useful in the management of diabetes: a systematic review. Altern Ther Health Med 10:44–50. https://doi.org/10.4103/0975-7406.90103
Ping H, Zhang G, Ren G (2010) Antidiabetic effects of cinnamon oil in diabetic KK-Ay mice. Food Chem Toxicol 48:2344–2349. https://doi.org/10.1016/j.fct.2010.05.069
Prasad RC, Herzog B, Boone B, Sims L, Waltner-Law M (2005) An extract of Syzygium aromaticum represses genes encoding hepatic gluconeogenic enzymes. J Ethnopharmacol 96:295–301. https://doi.org/10.1016/j.jep.2004.09.024
Raju J, Gupta D, Rao AR, Yadava PK, Baquer NZ (2001) Trigonella foenum graecum (fenugreek) seed powder improves glucose homeostasis in alloxan diabetic rat tissues by reversing the altered glycolytic, gluconeogenic and lipogenic enzymes. Mol Cell Biochem 224:45–51. https://doi.org/10.1023/A:1011974630828
Sadati SN, Ardekani MRS, Ebadi N, Yakhchali M, Dana AR, Masoomi F, Khanavi M, Ramezany F (2016) Review of scientific evidence of medicinal convoy plants in traditional Persian medicine. Pharmacogn Rev 10:33–38. https://doi.org/10.4103/0973-7847.176546
Shukla S, Chatterji S, Mehta S, Rai PK, Singh RK, Yadav DK, Watal G (2011) Antidiabetic effect of Raphanus sativus root juice. Pharm Biol 49:32–37. https://doi.org/10.3109/13880209.2010.493178
Srinivasan S, Muthukumaran J, Muruganathan U, Venkatesan RS, Jalaludeen AM (2014) Antihyperglycemic effect of syringic acid on attenuating the key enzymes of carbohydrate metabolism in experimental diabetic rats. Biomed Prev Nutr 4:595–602. https://doi.org/10.1016/j.bionut.2014.07.010
Sushma N, Devasena T (2010) Aqueous extract of Trigonella foenum graecum (fenugreek) prevents cypermethrin-induced hepatotoxicity and nephrotoxicity. Hum Exp Toxicol 29:311–319. https://doi.org/10.1177/0960327110361502
Tu Z, Moss-Pierce T, Ford P, Jiang TA (2014) Syzygium aromaticum L. (Clove) extract regulates energy metabolism in myocytes. J Med Food 17:1003–1010. https://doi.org/10.1089/jmf.2013.0175
Vafaei-Nezhad S, Hami J, Sadeghi A, Ghaemi K, Hosseini M, Abedini MR, Haghir H (2016) The impacts of diabetes in pregnancy on hippocampal synaptogenesis in rat neonates. Neuroscience 318:122–133. https://doi.org/10.1016/j.neuroscience.2016.01.025
Waisundara VY, Hsu A, Tan BKH, Huang D (2009) Baicalin reduces mitochondrial damage in streptozotocin-induced diabetic Wistar rats. Diabetes Metab Res Rev 25:671–677. https://doi.org/10.1002/dmrr.1005
Yadav D, Aa C, Garg V et al (2015) In vitro toxicity and antidiabetic activity of a newly developed polyherbal formulation ( MACST/001 ) in streptozotocininduced diabetic Wistar rats. PubMed Commons Protoplasma 250:6–7. https://doi.org/10.1007/s00709
Zarezadeh M, Vazifeshenas- Darmiyan K, Afshar M et al (2017) Effects of extract of Crocus sativus petal on renal function in diabetic rats. J Maz Univ Med Sci 27:11–24
Zarvandi M, Rakhshandeh H, Abazari M, Shafiee-Nick R, Ghorbani A (2017) ScienceDirect safety and efficacy of a polyherbal formulation for the management of dyslipidemia and hyperglycemia in patients with advanced-stage of type-2 diabetes. Biomed Pharmacother 89:69–75. https://doi.org/10.1016/j.biopha.2017.02.016
Acknowledgements
This study was financially supported by Birjand University of Medical Sciences (Grant number: 1098). We would like to acknowledge the anonymous referees for helpful comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All the experiments involving animals were performed according to the guide line of the Institutional Animal Ethical Committee, Birjand University of Medical Sciences (the permit code is Ir.bums.REC.1394.373).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kiani, Z., Hassanpour-Fard, M., Asghari, Z. et al. Experimental evaluation of a polyherbal formulation (Tetraherbs): antidiabetic efficacy in rats. Comp Clin Pathol 27, 1437–1445 (2018). https://doi.org/10.1007/s00580-018-2755-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-018-2755-9