Abstract
The role of arbuscular mycorrhizal fungi (AMF) in the control of migratory endoparasitic nematodes is nowadays largely admitted. Most studies were conducted under greenhouse conditions and a few used in vitro cultures with transgenic root organs. Here, we reported, for the first time, on the interaction between an AMF, Rhizophagus irregularis MUCL 41833 and Radopholus similis in roots of banana plantlets grown under in vitro culture conditions. The banana plantlets were pre-mycorrhized in an extraradical mycelium network arising from a Medicago truncatula donor seedling, before transfer to an autotrophic in vitro cultivation system and subsequent nematode inoculation. Both microorganisms were able to complete their life cycle in the absence as well as in presence of each other. The total R. similis population (i.e., summed over the roots and growth medium) as well as the surface of root necrosis was significantly reduced by 60 and 56 %, respectively, in the AMF-colonized banana plantlets. By contrast, nematodes had no visible impact on root colonization (i.e., percentage of arbuscules, intraradical spores/vesicles, and hyphae) by AMF and on the number of spores and hyphal length produced in the medium. These results clearly demonstrated that pre-mycorrhized banana plants could outcompete R. similis, while root colonization was not affected by the nematodes. They underline the interest of the novel in vitro cultivation system as a promising tool to investigate the biochemical factors and molecular mechanisms involved in the bio-protection conferred by AMF to a major root pathogen of banana.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The burrowing nematode Radopholus similis is one of the most damaging root pathogens of bananas that significantly reduce yield in commercial as well as in rural plantations worldwide (Gowen et al. 2005; Sarah et al. 1996). Until recently, its control relied predominantly on the use of nematicides (Quénéhervé 2009). However, the persistence of these chemicals in soil and their side effects on the environment and human health have encouraged the development of alternative control measures such as the application of fallow periods, the paring and hot water treatment of the corms, and the use of resistant cultivars and in vitro-produced plantlets (Gowen et al. 2005; Quénéhervé 2009). Biological control via the utilization of soil microorganisms is another option that is frequently proposed for the control of plant parasitic nematodes (Quénéhervé 2009; Vierheilig et al.1998).
Within the rhizosphere, arbuscular mycorrhizal fungi (AMF) are predominant microorganisms that form a symbiotic association with nearly 80 % of plant species (Smith and Read 2008). This association is largely based on the transfer of carbon from the plant to the fungus in exchange of minerals transferred from the AMF to the host plant. The cost–benefit balance of this bidirectional transport is most often reported as beneficial to the plant via improved biomass (Smith et al. 2011) and increased resistance to root pathogens (Azcón-Aguilar and Barea 1997; Akhtar and Siddqui 2008) and abiotic stresses (Smith and Read 2008), even though detrimental effects (e.g., biomass depression) have also been reported (Veiga et al. 2011).
Several studies have been conducted on the interactions between AMF and nematodes (see reviews by Hol and Cook 2005; Timper 2011; Veresoglou and Rillig 2012). In banana, most publications report a suppression in population buildup in roots of several genotypes infected by R. similis (Umesh et al. 1988; Elsen et al. 2003a, c, 2008), Meloidogyne incognita (Jaizme-Vega et al. 1997), or Pratylenchus coffeae (Elsen et al. 2003a, c, 2008), although other studies observed the absence of any visible effect on Pratylenchus goodeyi, Meloidogyne javanica, and R. similis population in banana roots (Jaizme-Vega and Pinochet 1997; Pinochet et al. 1997; Gañán et al. 2011). Overall, the AMF association has been reported to stimulate banana root and shoot growth (Umesh et al. 1988; Jaizme-Vega et al. 1997; Pinochet et al. 1997; Jaizme-Vega and Pinochet 1997; Gañán et al. 2011). It has been proposed that plant resistance/tolerance may be related to competition between AMF and nematodes for resources and space, through AMF increasing root branching and nutrient uptake, thereby improving biomass production and delaying the damage caused by plant parasitic nematodes (Jaizme-Vega and Pinochet 1997; Stoffelen et al. 2000; Gañán et al. 2011). In a recent study, Elsen et al. (2008) suggested that AMF were able to induce systemic resistance against R. similis and P. coffeae in banana roots, whilst modifications in root exudates has also been mentioned as a potential mechanism of control (Vos et al. 2012).
Although most of the studies on interactions between AMF and root pathogens were conducted in pots, a number have also been conducted under in vitro culture conditions in the last decade, using root organ cultures (ROC; Benhamou et al. 1994; Bharadwaj et al. 2011; Lioussanne et al. 2008). The ROC system offers several advantages (see review by Fortin et al. 2002) such as the absence of undesirable microorganisms and the possibility to follow nondestructively and continuously the interactions between the microbial protagonists. For instance, the impact of rhizosphere microorganisms on AMF (e.g., Rousseau et al. 1996) and the reverse (e.g., Benhamou et al. 1994) as well as the synergy between AMF and rhizospheric microorganisms (Martinez et al. 2004) have been investigated under ROC with physiological, cellular, and molecular tools (see review by Fortin et al. 2002). The possibility to grow roots in split-root systems separating AMF-colonized roots with and without pathogens was also reported as particularly efficient for gene expression and analysis of systemic resistance (Gallou 2011). Finally, the absence of patchiness in such systems could allow a better understanding of the mechanisms involved in the interaction between an AMF and a root pathogen. For nematodes, Elsen et al. (2001a, 2003b) were the first to investigate the interaction between Rhizophagus irregularis MUCL 41833 and P. coffeae and R. similis. They used ROC of carrot (Daucus carota L.) and observed a decrease in the number of females and males in the roots colonized by the AMF. However, some limitations are associated with ROC, which are the absence of photosynthetic tissues, of a normal hormonal balance, and of source-sink relationships (Fortin et al. 2002). In addition, the roots used in these interactions were most often transformed organs. In recent years, Voets et al. (2005, 2009), Dupré de Boulois et al. (2006), and Koffi et al. (2009) developed in vitro cultivation systems associating photosynthetically active plants (i.e., potato, Medicago truncatula, banana) with AMF. Extensive extra and intraradical mycelium producing thousands of spores was obtained. These systems also allowed studies with plant–pathogen interactions (Gallou et al. 2011).
In the present study, we report, for the first time, on the interaction between an AMF, R. irregularis MUCL 41833 and R. similis in roots of banana c.v. Grande Naine under in vitro culture conditions. The development of the AMF, the reproduction of the nematodes and the interactions between both organisms are investigated and discussed. R. irregularis MUCL 41833 has been previously used to study gene expression dynamics during AMF root colonization (Gallou et al. 2010) as well as the transcriptional regulation of defense genes and involvement of the WRKY transcription factor in root colonization (Gallou et al. 2012). It was also used in interaction studies with the biological control agent Trichoderma harzianum (De Jaeger et al. 2010) and with the hemibiotrophic pathogen Phytophthora infestans (Gallou et al. 2011).
Materials and methods
Plant materials
Tissue culture plantlets of the banana cultivar Grande Naine (Musa acuminata L., AAA genome, Cavendish group) were provided by the International Musa Germplasm collection at the INIBAP Transit Centre (K.U. Leuven, Belgium). The plant material was proliferated, regenerated, and rooted on the Murashige and Skoog (MS) medium (Murashige and Skoog 1962) as described by Koffi et al. (2009). The plantlets were further incubated in a growth chamber at 27/25 °C (day/night) with a photoperiod of 16 h day−1 and under a photosynthetic photon flux of 300 μmol m−2 s−1.
Seeds of M. truncatula Gaertn. cv. Jemalong A 17 (SARDI, Australia) were surface-disinfected, germinated, and incubated as described in Gallou et al. (2010). Seedlings were ready to use 4 days following germination.
Arbuscular mycorrhizal fungus
A strain of Rhizophagus irregularis (Błaszk., Wubet, Renker & Buscot) C. Walker & A. Schüßler comb. nov. MUCL 41833, was purchased from GINCO (http://www.mycorrhiza.be/ginco-bel/index.php). This AMF was used to develop the mycorrhizal donor plant (MDP) in vitro culture system used in the present study (see below). The AMF was grown in association with Ri T-DNA transformed carrot (D. carota L.) roots (Cranenbrouck et al. 2005) on Petri plates (90-mm diameter) filled with 35-ml modified Strullu-Romand (MSR) medium (Declerck et al. 1998). The Petri plates were incubated at 27 °C in the dark in an inverted position until thousand of spores were produced.
Nematode
A strain of R. similis (Cobb) Thorne originating from Indonesia was provided by the Nematology laboratory of K.U. Leuven. The nematodes were maintained under aseptic conditions at 27 °C in the dark, on Medicago sativa L. callus grown on the modified White’s medium (Elsen et al. 2001b).
A modified mycelium donor plant in vitro culture system for fast and homogenous colonization of banana plantlets
A modified MDP in vitro culture system was adapted from the system developed by Voets et al. (2009). The cover of a 55-mm diameter Petri plate (root compartment (RC)) was introduced in the base of a 145-mm diameter Petri plate (hyphal compartment (HC)). The RC was filled with 20-ml MSR medium lacking vitamins and sucrose, buffered with 10 mM 2-(N-morpholino)ethanesulfonic acid (MES) and adjusted to pH 6 before sterilization (121 °C for 15 min), while the HC received 100 ml of the same medium. The RC was lined to the border of the HC and a small opening (±2-mm diam.) was made in the base and the lid of the 145-mm Petri plates. One 4-day-old M. truncatula seedling was transferred into the RC as in Voets et al. (2009). The plantlet was subsequently inoculated with an approximate of 200 spores of the AMF (+AMF treatment). The culture plate was then carefully closed and covered with an opaque plastic bag to keep the AMF and M. truncatula roots in the dark, while the shoot developed under light condition (Voets et al. 2009). Twelve culture plates were set up and incubated in a growth chamber under controlled conditions (22/18 °C (day/night) with a photoperiod of 16 h day−1 and a photosynthetic photon flux of 300 μmol m−2 s−1). Starting from week 2, MSR medium (20 ml) without vitamins and sucrose buffered with 10 mM MES was added weekly to the RC. After 4 weeks, the extraradical mycelium extended into the HC, covering the whole surface within an additional of 4 weeks. At that time, six densely colonized culture plates were randomly selected. The number of spores in the HC was 229,051 ± 33,736 (Declerck et al. 2001) and with hyphal length of 83,635 ± 8,990 cm (Giovannnetti and Mosse 1980). In parallel, eight control culture plates (−AMF treatment) were set up following the same process as the above but without a M. truncatula seedling and AMF added to the RC. After 8 weeks, four culture plates were randomly selected.
Banana inoculation with the AMF
Four rooted banana plantlets (2 weeks old) were transferred into the HC of each culture plate of both treatments. To accommodate the plantlets to the system, the lid of the large Petri plates was replaced by another lid surmounted by a Full Gas Microsac (220 × 180 mm with filters 0.22 μm; SAOC Microsac, Eke, Belgium) sterilized at 25 kGy (Sterigenics, Belgium). Briefly, a hole of 110-mm diam was made in the lid of the 145-mm Petri plate. A ring of 110-mm diameter (made from the base of a Petri plate of 120-mm diameter) was glued with hot melt glue and then the Full Gas Microsac was attached to the ring with parafilm. The culture plates containing the banana plantlets were then sealed with parafilm. MSR medium (±50 ml) without vitamins and sucrose buffered with 10 mM MES (pH 6) and containing activated charcoal (0.25 g l−1) was added weekly to the HC and RC. The activated charcoal was used to adsorb phenolic compounds, improve rooting, and allowing the banana roots and AMF to grow under shaded conditions (Thomas 2008). The plantlets were incubated in a growth chamber at 27/25 °C (day/night) with a photoperiod of 16 h day−1 and a photosynthetic photon flux of 300 μmol m−2 s−1. Six culture plates with AMF (+AMF treatment) and four without AMF (−AMF treatment) were set up.
Interaction with R. similis
After 3 weeks of growth in the HC in the presence/absence of the AMF, the banana plantlets were removed from the culture plates and transferred in individual Petri plates (145 mm diam) following the method of Koffi et al. (2009). The Petri plates contained a thin layer (100 ml) of MSR medium lacking sucrose and vitamins, buffered with 10 mM MES (pH 6) and adjusted to pH 6 before sterilization. Twenty four Petri plates were set up with pre-mycorrhized banana plantlets (+AMF treatment) and 16 with non-mycorrhized banana plantlets (−AMF treatment). The Petri plates were covered with aluminum sheet, and the plantlets were incubated for 3 weeks in a growth chamber under the same conditions as the above.
After this period, eight plantlets from the +AMF treatment were randomly harvested to evaluate the AMF root colonization. The remaining plantlets from the +AMF treatment were randomly separated in two groups of eight plantlets (named+AMF−NEM and +AMF+NEM treatments), and the number of spores (Declerck et al. 2001) and the hyphal length (Giovannnetti and Mosse 1980) were evaluated. The banana plantlets in the −AMF treatment were also randomly separated in two groups of eight plantlets (named −AMF−NEM and −AMF+NEM treatments).
One group of mycorrhizal (+AMF+NEM) and non-mycorrhizal (−AMF+NEM) plantlets was inoculated with 200 nematodes. Mature females of R. similis were collected individually with a sterile micropipette from nematode cultures and placed in several drops of sterile water near the roots.
A total of four treatments with eight replicates were considered: mycorrhizal banana plantlets inoculated (+AMF+NEM) or not (+AMF−NEM) with nematodes and non-mycorrhizal banana plantlets inoculated (−AMF+NEM) or not (−AMF−NEM) with nematodes. The Petri plates were covered with aluminum sheet, and the plantlets were incubated for 8 weeks. MSR medium (±50 ml), without vitamins and sucrose buffered with 10 mM MES (pH 6) was added weekly to the Petri plates.
Assessment of extraradical development of the AMF
Spore number and extraradical hyphal length were estimated at 3, 5, 7, 9, and 11 weeks after banana transfer in the single Petri plates (week 3 corresponded to the time of nematode inoculation). Gridlines of 1 cm2 were marked on the bottom of each Petri plate. The number of spores was estimated in each square formed by the gridlines and summed over the entire Petri plate (Declerck et al. 2001). The presence of hyphae was noted at each point where they intersected a line (Giovannnetti and Mosse 1980). The total extraradical hyphae length was then calculated with the formula of Newman (1966).
Root colonization by the AMF
Root colonization was estimated on the plantlets in the +AMF+NEM and +AMF−NEM treatments. The roots were cautiously removed from the Petri plates with forceps, cleaned with deionized water from the MSR medium, weighed, and divided in two equal parts to estimate root colonization and/or nematode numbers. The roots were soaked in 10 % KOH for 30 min. They were washed several times with deionized water and bleached in a freshly prepared alkaline H2O2 solution for 30 min (Koske and Gemma 1989). The roots were subsequently stained with a solution of blue ink (ParkerP Quink®) diluted 1:50 in 1 % HCl (Vierheilig et al. 1998). All the procedures were done at 50 °C in a water bath. Root colonization was assessed by the method of McGonigle et al. (1990). A mean of 170–250 intersections were observe to determine the percentage of arbuscules, intraradical spores/vesicles, hyphae, and the total percentage of root colonization.
Root necrosis and nematode enumeration
Percentage of root necrosis was measured by scoring 5 × 10-cm longitudinally sliced functional primary roots selected randomly from the roots of the treatments +AMF+NEM and −AMF+NEM following the method of Speijer and De Waele (1997). Root systems were cleaned from the MSR medium, weighed, and separated into two equal parts for root colonization (see above) and/or nematode infestation. Nematodes were extracted from the roots of the treatments +AMF+NEM and −AMF+NEM by the maceration-sieving method (Speijer and De Waele 1997). The roots were cut into 1-cm pieces and macerated in a kitchen blender for 30 s (three 10-s periods separated by 5-s intervals). The suspension was passed through 250-, 100-, and 40-μm sieves placed in a bowl. Root residues in the sieves 250 and 100 μm were rinsed with tap water and discarded. The solution in the bowl was passed through a 25-μm sieve. Nematodes were collected in a beaker from the 40- and 25-μm sieves and the number of juveniles, female, and males, counted.
Plant growth parameters
Banana plantlets were harvested at week 11. The shoot and root fresh weights as well as the shoot dry weight were measured.
Statistical analyses
Data analysis was performed with the statistical package Statistica (Release 7, Statsoft, Tulsa, USA) and SAS v9.3 (The SAS Institute, NC, USA). Nematode populations and spore counts were log(x + 1)-transformed, while data for root necrosis, percentage arbuscules, intraradical spores/vesicles, and hyphae and the total percentage of root colonization were arcsin(x/100)-transformed prior to analysis. Shoot and root fresh weights were analyzed by two-way and three-way ANOVA for repeated measures (SAS procedure MIXED), while the AMF and nematode data were analyzed by one-way ANOVA. Tukey's honestly significant difference was conducted to identify significant differences (P ≤ 0.05) between the treatments.
Results
Two days after the transfer of the plantlets from the MDP in vitro culture plates onto the individual Petri plates, numerous hyphae started to emerge from the colonized banana roots. The first newly produced spores were observed at day 3. This was followed by a rapid increase in spore number and mycelium length in the following days. MSR medium was supplied every week, and the new layers of medium were rapidly colonized by the AMF, resulting in an increase in length of the hyphae and number of spores produced. At the time of nematode inoculation, i.e., 3 weeks after transfer of the banana plantlets to the individual Petri plates, eight randomly selected banana plantlets were harvested and root colonization, estimated. The total root colonization was 88.6 ± 1.9 %. The percentage of arbuscules, intraradical spores/vesicles, and hyphae was 86.6 ± 3.1, 64.2 ± 7.7, and 9.2 ± 2.1 %, respectively. The 16 remaining banana plantlets were separated in two groups (named +AMF−NEM and +AMF+NEM treatments). The number of spores and hyphal length were evaluated in both treatments. In the +AMF−NEM treatment, the number of spores and hyphal length produced was 29,035 ± 5,429 and 31,176 ± 5,871 cm, respectively, while in the +AMF+NEM treatment, these values were 29,017 ± 5,144 and 31,165 ± 4,174 cm for spores and hyphal length, respectively. Whatever the time of observation, no significant differences were noted in spore production and hyphal length between the +AMF−NEM and +AMF+NEM treatments (Table 1, repeated measures ANOVA). Spore production and hyphal length increased markedly until the end of the experiment to reach values close to 500,000 spores and 150,000 cm of hyphal length, in both treatments (Table 1).
At the end of the experiment (week 11, i.e., 8 weeks after inoculation of the nematodes), the plants were harvested and root colonization evaluated. The percentage of total root colonization was 92 ± 1.74 and 91.63 ± 1.58 % in the +AMF−NEM and +AMF+NEM treatments, respectively. Identically, the percentage of arbuscules and intraradical spores/vesicles were high with values of 86.7 ± 2.5 and 80.9 ± 4 %, and 87.95 ± 2.08 and 81.25 ± 4.88 % for the +AMF−NEM and +AMF+NEM treatments, respectively. Finally, the percentage of hyphae was 10.25 ± 0.96 and 9.11 ± 1.35 %, for the +AMF−NEM and +AMF+NEM treatments, respectively. None of these parameters significantly differed between the two treatments.
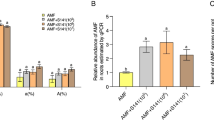
One hour after the inoculation in the Petri plates, some nematodes were observed to penetrate the MSR medium. No nematodes were observed on the surface of the culture medium the following day. Penetration of the roots and reproduction within the roots were observed in the presence (+AMF+NEM) as well as absence (−AMF+NEM) of AMF. All the nematode developmental stages were detected in the banana roots and MSR medium (Table 2). The number of females was significantly lower in the roots and MSR medium of the +AMF+NEM treatment as compared to the −AMF+NEM treatment, whereas the number of juveniles was only significantly reduced in the roots of the +AMF+NEM treatment. No significant difference was noted in the number of males in roots and medium between the two treatments. Similar results were obtained when the number of juveniles, females, and males and total number of nematodes were expressed per unit of root fresh weight. The number of juveniles, females, and total number of nematodes were significantly lower in the +AMF+NEM treatment as compared to the −AMF+NEM treatment (23 ± 7, 10 ± 3, and 36 ± 9 for juveniles, females, and total number of nematodes in the +AMF+NEM treatment and 67 ± 8, 24 ± 5, and 101 ± 12 for the −AMF + NEM treatment), while no difference was noted for the number of males between these two treatments (3 ± 1 and 10 ± 9 for the +AMF+NEM and −AMF+NEM treatments, respectively). Summed over the roots and medium, the total population of R. similis reached 905 ± 188 in the +AMF+NEM treatment and 2,287 ± 255 in the −AMF+NEM treatment. The population of R. similis was reduced by 60 % in the +AMF+NEM treatment as compared to the −AMF+NEM treatment (Table 2). Identically, the reproduction ratio was significantly lower in the +AMF+NEM treatment as compared to the −AMF+NEM treatment (Table 2).
After transfer from the MDP in vitro culture system into the individual Petri plates, banana plantlets continued to grow and developed new roots and leaves, whatever the treatment. Banana plantlets developed primary, secondary, and tertiary roots on the surface and into the culture medium. Newly produced roots were creamy white and became brown and dark with aging. Reddish-brown discolorations and small cavities, typical of necrosis, were observed on the roots inoculated with nematodes. Banana plantlets in the +AMF+NEM treatment had significantly less root necrosis as compared to the banana plantlets in the −AMF+NEM treatment (Table 2).
At harvest (i.e., week 11), no significant difference was noticed in shoot dry weight between the four treatments (two-way ANOVA). The values, expressed in grams, were 2.89 ± 0.03, 3.04 ± 0.04, 2.93 ± 0.07, and 3.00 ± 0.06, for the −AMF−NEM, −AMF+NEM, +AMF−NEM, and + AMF+NEM, respectively. Similarly, shoot fresh weight did not differ between the four treatments (two-way ANOVA). The values, expressed in grams, were 14.56 ± 0.18, 14.83 ± 0.21, 14.74 ± 0.25, and 14.94 ± 0.28 for − AMF−NEM, −AMF+NEM, +AMF−NEM, and + AMF+NEM, respectively. On the contrary, root fresh weight was significantly higher for the mycorrhizal plants in the presence (20.52 ± 0.31 g) as well as absence (20.49 ± 0.23 g) of nematodes (+AMF+NEM and +AMF−NEM treatments, respectively), as compared to the non-mycorrhizal plantlets grown in absence of nematodes (19.44 ± 0.27 g). The plantlets grown in the absence of AMF but in the presence of nematodes (−AMF+NEM) had intermediate root fresh weights (20.03 ± 0.23 g) as compared to the mycorrhizal treatments and the −AMF−NEM treatment. The data demonstrated a significant effect of AMF (two-way ANOVA, P = 0.007) and absence of effect of nematodes or combination of AMF and nematodes. Similar conclusions were obtained with a three-way generalized linear model (AMF×NEM×plant part) allowing for correlated residuals within the same plant treatment. The latter test was performed to reduce the risk of type 1 error resulting from separate statistical inference on root and shoot fresh weights.
Discussion
In this study, we report for the first time data on the interaction between an AMF (R. irregularis MUCL 41833) and a nematode (R. similis) on banana (c.v. Grande Naine) grown under in vitro culture conditions. Both microorganisms were able to complete their life cycle in the absence as well as presence of each other. R. irregularis MUCL 41833 produced a dense extraradical mycelium bearing several hundred thousands of spores and intense root colonization with abundant arbuscules and intraradical spores/vesicles. R. similis was able to penetrate and reproduce within the roots producing reddish-brown discolorations and small cavities typical of root necrosis. The nematodes had no visible impact on the AMF development, neither in the roots nor in the MSR medium. On the contrary, the total R. similis population (i.e., summed over the roots and growth medium) as well as the surface of root necrosis was significantly reduced by 60 and 56 %, respectively, in the AMF-colonized banana plantlets.
The MDP in vitro culture system developed by Voets et al. (2009) for the fast and homogenous colonization of photosynthetically active M. truncatula seedlings was successfully adapted to banana plantlets in this study. The plantlets were heavily colonized within a period of 3 weeks following their planting into the extraradical mycelium network. Their subsequent transfer from the MDP in vitro culture systems to individual Petri plates resulted in a profuse production of spores and extraradical hyphae (i.e., an average of 500,000 spores and 150,000-cm hyphae) within a period of 11 weeks. The total root colonization averaged 90 %. These values markedly exceeded those obtained by Koffi et al. (2009) using isolated spores of the same AMF as source of inoculum. This further supports that the extraradical mycelium composed of hundreds of actively growing hyphae extending from a mycorrhizal donor plants (i.e., M. truncatula) is the most adequate source of inoculum for the early colonization of banana plantlets and the subsequent development of the AMF within the roots and spreading within the medium. The high number of spores obtained within the period of 11 weeks was favored by the weekly addition of MSR medium to the Petri plates. This resulted in the superposition of successive layers of MSR medium in which the AMF spores and hyphae accumulated. This was earlier reported by Douds (2002) who observed a higher spore production after addition of a nutrient medium in the distal compartment of split-plate monoxenic in vitro culture systems.
The presence of R similis did not influence the development of the AMF. Neither arbuscules, intraradical spores/vesicles, hyphae, nor total root colonization were decreased in the pre-mycorrhized banana plantlets. Similarly, the extraradical hyphae and spore production dynamics were not influenced by the nematode. The same results were reported with pre-mycorrhized (with R. irregularis MUCL 41833) ROC of carrot grown in the presence of P. coffeae or R similis (Elsen et al. 2001a; Elsen et al. 2003b) and with pre-mycorrhized banana plants grown in the presence of M. incognita, P. coffeae, P. goodeyi, and R similis under greenhouse conditions (Pinochet et al. 1997; Elsen et al. 2003a, 2008; Jaizme-Vega et al. 1997). In contrast, the total population of R. similis (in roots and MSR medium) was significantly decreased in the presence of pre-mycorrhized banana plantlets, as earlier reported in interaction studies between R. similis and R. irregularis MUCL 41833 (Elsen et al. 2003a, b) and Glomus mosseae (Elsen et al. 2008) and between M. incognita and G. mosseae (Jaizme-Vega et al. 1997), although in contradiction with the results of Pinochet et al. (1997) and Gañán et al. (2011). Interestingly, in our study, this reduction was more pronounced in the roots than in the MSR medium. Indeed, the number of females and juveniles was significantly reduced in the roots, while in the MSR medium only the number of females was decreased. The decrease in number of females in roots and MSR medium was reported in an earlier study conducted in ROC in the presence of R. similis (Elsen et al. 2001a) and P. coffeae (Elsen et al. 2003b). Interestingly, no impact was noted on the number of juveniles in the excised roots of carrot and MSR medium (Elsen et al. 2001a; Elsen et al. 2003b). However, it is unclear whether the absence of visible effects on juveniles observed in ROC, contrary to the results on whole plantlets in our experiment, could be related to the nutritional status of the host (whole plant versus excised root), to the host species itself (carrot versus banana), to a higher mortality of the juveniles in our experiment or to any other environmental factor impacting the life cycle of nematodes and their maturation from juveniles to males or females. Banana pre-inoculation with the AMF further decreased significantly the surface of root necrosis. This confirmed the earlier observations made by Jaizme-Vega and Pinochet (1997) but contrasted with the results of Elsen et al. (2003a, c, 2008) reporting the absence of influence of AMF on nematode damage in roots. Interestingly, in our experiment, no significant difference was noticed in the root fresh weight of the mycorrhized banana plantlets grown in the presence or absence of R. similis. This suggests that the root system may have been partly protected from damage caused by the nematodes.
Overall, our results support the conclusion that early root colonization, i.e., preliminary to any contact with the pathogen, increases the ability of AMF to withstand the competition with nematodes while substantially decreasing the nematode population in roots and in the extraradical mycelium environment as well as reducing their incidence on roots. Francl (1993) has suggested a direct suppressive effect of AMF on root-feeding nematodes if both organisms compete for root space and feeding sites. In the present study, heavy root colonization was obtained (i.e., >90 %) which may have hampered nematode migration (Hol and Cook 2005) and reproduction (de la Pena et al. 2006). However, the absence of an inverse correlation (r = −0.22; P = 0.586) between total root colonization by the AMF and R. similis population densities in roots, both in the present experiment and in earlier reports in ROC (Elsen et al. 2001a; Elsen et al. 2003b), suggests that competition for root and feeding sites (i.e., local resistance) was presumably not a predominant factor impacting nematode infestation. Systemic resistance against nematodes has been previously reported in banana (Elsen et al. 2008) and grapevine (Hao et al. 2012). Curiously, in our experiment, the number of nematodes was strongly correlated with the percentage of arbuscules (r = 0.805, P = 0.0159), suggesting more nematodes in roots heavily colonized with arbuscules. However, no difference in arbuscule production was observed between the treatments with and without nematodes, suggesting that a systemic resistance could not be excluded as a possible mechanism involved in the nematode reduction.
In the recent decade, particular attention has been devoted to the activation of plant defense mechanisms in mycorrhizal plants. Recently, Hao et al. (2012) reported a suppressive effect on the ectoparasitic nematode Xiphinema index in pre-mycorrhized grapevine plantlets. This bioprotection was both local and systemic and was associated with the activation of several genes among which chitinase 1b, pathogenesis-related 10, and glutathione S-transferase. The authors suggested a priming of defense responses by the AMF and transmission of a plant-mediated signal to non-mycorrhizal tissues. Similarly, Li et al. (2006) reported an up-regulation and further enhancement of class III chitinase transcripts in mycorrhizal grapevine following infestation with M. incognita, in relation to an increased plant resistance against a subsequent infestation. Defense genes elicited during AMF root colonization may, thus, be of particular importance in the protection of plants against nematodes.
Competition in the root zone is another mechanism by which nematodes may be impacted by AMF before root infestation. It is well-known that AMF colonization modifies qualitatively and quantitatively the host root exudates (Jones et al. 2004). In a recent study, it was observed that root exudates of mycorrhizal banana plants reduced the attraction of nematodes and subsequent penetration of the host roots (Vos et al. 2012). If the total number of nematodes in the medium appeared not impacted by the presence of an AMF (i.e., the extraradical mycelium) in our experiment, we could not exclude that attraction to the roots and subsequent penetration was modified in the mycorrhized roots with subsequent impact on the population and reproduction of nematodes in the roots. Interestingly, the number of females in the root zone was significantly decreased in the presence of the AMF, which may further substantiate the subsequent lower reproduction within the roots. In addition, it is not excluded that cell wall modifications induced by AMF may have decreased the ability of R. similis to feed and migrate within the roots (Hol and Cook 2005). An increased production of phenolic compounds such as lignin and ferulic acid (Cordier et al. 1998; Akhtar and Siddiqui 2008), phenols, and cell wall-bound phenols (Zhu and Yao 2004) has been reported in mycorrhized roots which could hamper nematode penetration and affect their reproduction (Wuyts et al. 2006).
In the present study, we confirm the suppressive effect of an AMF (R. irregularis MUCL 41833) on the population of nematodes within the roots of banana and partially in the mycorrhizal root zone, using, for the first time, an autotrophic in vitro cultivation system. The reported data clearly demonstrate that pre-mycorrhized banana plants could outcompete R similis, while AMF root colonization was not affected by the nematodes. These results support the general observations from studies conducted under greenhouse conditions (e.g., Hao et al. 2012). The application of the autotrophic in vitro cultivation system represents a substantial improvement with regard to the ROC frequently used in studies of AMF interactions with pathogens among which nematodes (Elsen et al. 2001a; Elsen et al. 2003b), due to the presence of photosynthetic active tissues and absence of exogenous sugars in the growth medium. Furthermore, it offers a highly controlled alternative to investigate the implication of phenolic compounds (e.g., changes in lignin, phenols, flavonoids, condensed tannins, ferulic acid in roots, and/or AMF exudates) and of molecular (e.g., defense gene expression) mechanisms involved in the bio-protection conferred by AMF to a major root pathogen of banana.
References
Akhtar MS, Siddqui ZA (2008) In: Siddiqui ZA, Akhtar MS, Kazuyoshi (eds) Mycorrhizae: sustainable agriculture and forestry. Springer, Netherlands, pp 61–97
Azcón-Aguilar C, Barea JM (1997) Arbuscular mycorrhizas and biological control of soil-borne plant pathogens—an overview of the mechanisms involved. Mycorrhiza 6:457–464
Benhamou N, Fortin JA, Hamel C, St-Arnaud M, Shatilla A (1994) Resistance responses of Mycorrhizal Ri T-DNA-transformed carrot roots to infection by Fusarium oxysporum f. sp. Chrysanthemi. Phytopathology 84:958–968
Bharadwaj DP, Alström S, Lundquist PO (2011) Interactions among Glomus irregulare, arbuscular mycorrhizal spore-associated bacteria, and plant pathogens under in vitro conditions. Mycorrhiza. doi:10.1007/s00572-011-0418-7
Cordier C, Pozo MJ, Barea JM, Gianinazzi S, Gianinazzi-Pearson V (1998) Cell defense responses associated with localized and systemic resistance to Phytophthora parasitica induced in tomato by an arbuscular mycorrhizal fungus. Mol Plant Microbe Interact 11:1017–1028
Cranenbrouck S, Voets L, Bivort C, Renard L, Strullu DG, Declerck S (2005) Methodologies for in vitro cultivation of arbuscular mycorrhizal fungi with root organs. In: Declerck S, Strullu DG, Fortin A (eds) In vitro culture of mycorrhizas. Springer, Berlin, pp 341–375
De Jaeger N, Declerck S, de la Providencia IE (2010) Mycoparasitism of arbuscular mycorrhizal fungi: a pathway for the entry of saprotrophic fungi into roots. FEMS Microbiol Ecol 73:312–322
de la Peña E, Rodriguez-Echevarria S, van der Putten WH, Freitas H, Moens M (2006) Mechanism of control of root-feeding nematodes by mycorrhizal fungi in the dune grass Ammophila arenaria. New Phytol 169:829–840
Declerck S, Strullu DG, Plenchette C (1998) Monoxenic culture of the intraradical forms of Glomus sp. isolate from a troplical ecosystem: a proposed methology for germplasm collection. Mycologia 90:579–85
Declerck S, D’or D, Cranenbrouck S, Le Boulengé E (2001) Modelling the sporulation dynamics of arbuscular mycorrhizal fungi in monoxenic culture. Mycorrhiza 11:225–230
Douds DD (2002) Increased spore production by Glomus intraradices in the split-plate monoxenic culture system by repeated harvest, gel replacement, and resupply of glucose to the mycorrhiza. Mycorrhiza 12:163–167
Dupré de Boulois H, Voets L, Delvaux B, Jakobsen I, Declerck S (2006) Transport of radiocaesium by arbuscular mycorrhizal fungi to Medicago truncatula under in vitro conditions. Environ Microbiol 8:1926–1934
Elsen A, Declerck S, De Waele D (2001a) Effects of Glomus intraradices on the reproduction of the burrowing nematode (Radopholus similis) in dixenic culture. Mycorrhiza 11:49–51
Elsen A, Lens K, Nguyet DTM, Broos S, De Waele D (2001b) Development of aseptic culture systems of Radopholus similis for in vitro host-pathogen studies. J Nematol 33:147–151
Elsen A, Baimey H, Swennen S, De Waele D (2003a) Relative mycorrhizal dependency and mycorrhiza-nematode interaction in banana cultivars (Musa spp.) differing in nematode susceptibility. Plant Soil 256:303–313
Elsen A, Declerck S, De Waele D (2003b) Use of root organ cultures to investigate the interaction between Glomus intraradices and Pratylenchus coffeae. Appl Environ Microbiol 69:4308–4311
Elsen A, Beeterens R, Swennen R, De Waele D (2003c) Effects of an arbuscular mycorrhizal fungus and two plant-parasitic nematodes on Musa genotypes differing in root morphology. Biol Fertil Soils 38:367–376
Elsen A, Gervacio D, Swennen R, De Waele D (2008) AMF-induced biocontrol against plant parasitic nematodes in Musa sp.: a systemic effect. Mycorrhiza 18:251–256
Fortin JA, Bécard G, Declerck S, Dalpé Y, St Arnaud M, Coughlan AP, Piché Y (2002) Arbuscular mycorrhiza on root-organ cultures. Can J Bot 80:1–20
Francl LJ (1993) Interactions of nematodes with mycorrhizae and mycorrhizal fungi. In: Khan MW (ed) Nematode interactions. Chapman & Hall, London, pp 203–216
Gallou A (2011) Impact of Rhizophagus sp. (syn. Glomus sp.) and Trichoderma harzianum on the potato reistance against Rhizoctonia solani and Phytophthora infestans, two major potato pathogens. PhD thesis pp 414
Gallou A, De Jaeger N, Cranenbrouck S, Declerck S (2010) Fast track in vitro mycorrhization of potato plantlets allow studies on gene expression dynamics. Mycorrhiza 20:201–207
Gallou A, Mosquera HPL, Cranenbrouck S, Suárez JP, Declerck S (2011) Mycorrhiza induced resistance in potato plantlets challenged by Phytophthora infestans. Physiol Mol Plant Pathol 76:20–26
Gallou A, Declerck S, Cranenbrouck S (2012) Transcriptional regulation of defence genes and involvement of the WRKY transcription factor in arbuscular mycorrhizal potato root colonization. Funct Integr Genom 1:183–198
Gañán L, Bolaños-Benavides MM, Asakawa N (2011) Efecto de la micorrización sobre el crecimiento de plántulas de plátano en sustrato con y sin la presencia de nematodos. Acta Agronómica 60(4):297–305
Giovannnetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Gowen S, Quénéhervé P, Fogain R (2005) Nematodes of banana and plantains. In: Luc M, Sikora R, Bridge J (eds) Plant parasitic nematodes in subtropical and tropical agriculture, 2nd edn. CAB International, London, pp 611–643
Hao Z, Fayolle L, van Tuinen D, Chatagnier O, Gianinazzi S, Li X, Gianinazzi-Pearson V (2012) Local and systémic mycorrhiza-induced protection against the ectoparasitic nematodes Xiphinema index involved priming of defence gene responses in grapevine. J Exp Bot 63(10):3657–3672
Hol GWH, Cook R (2005) An overview of arbuscular mycorrhizal fungi–nematode interactions. Basic. Appl Ecol 6:489–503
Jaizme-Vega MC, Pinochet J (1997) Growth response of banana to three mycorrhizal fungi in Pratylenchus goodeyi infested soil. Nematropica 27:69–76
Jaizme-Vega MC, Tenoury P, Pinochet J, Jaumot M (1997) Interactions between the root-knot nematode Meloidogyne incognita and Glomus mosseae in banana. Plant Soil 196:27–35
Jones DL, Hodge A, Kuzyakov Y (2004) Plant and mycorrhizal regulation of rhizodeposition. New Phytol 163:459–480
Koffi MC, de la Providencia IE, Elsen A, Declerck S (2009) Development of an in vitro culture system adapted to banana mycorrhization. Afr J Biotechnol 8(12):2750–2756
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–505
Li HY, Yang GD, Shu HR, Yang YT, Ye BX, Nishida I, Zheng CC (2006) Colonization by the arbuscular mycorrhizal fungus Glomus versiforme induces a defense response against the root-knot nematode Meloidogyne incognita in the grapevine (Vitis amurensis Rupr.), which includes transcriptional activation of the class III chitinase gene VCH3. Plant Cell Physiol 47:154–163
Lioussanne L, Jolicoeur M, St-Arnaud M (2008) Mycorrhizal colonization with Glomus intraradices and development stage of transformed tomato roots significantly modify the chemotactic response of zoospores of the pathogen Phytophthora nicotianae. Soil Biol Biochem 40:2217–2224
Martinez A, Obertello M, Pardo A, Ocampo JA, Godeas A (2004) Interactions between Trichoderma pseudokoningii strains and the arbuscular mycorrhizal fungi Glomus mosseae and Gigaspora rosea. Mycorrhiza 14:79–84
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Newman EI (1966) A method of estimating the total length of root in a sample. J Appl Ecol 3:130–145
Pinochet J, Fernandez C, Jaizme-Vega M, Tenoury P (1997) Micropropagated banana infected with Meloidogyne responds to Glomus intraradices and phosphorus. HortScience 32:101–103
Quénéhervé P (2009) Integrated management of banana nematodes. In: Ciancio A, Mukerji KG (eds) Integrated management of fruit crops and forest nematodes. Springer, Netherlands, pp 3–61
Rousseau A, Benhamou N, Chet I, Piche Y (1996) Mycoparasitism of the extramatrical phase of Glomus intraradices by Trichoderma harzianum. Phytopathology 86:434–443
Sarah JL, Pinochet J, Stanton J (1996) The burrowing nematode of banana, Radopholus similis Cobb. Musa pest fact sheet No. 1. INIBAP, Montpellier, p 2
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Academic, San Diego
Smith SE, Jakobsen I, Grønlund M, Smith FA (2011) Roles of Arbuscular mycorrhizas in plant phosphorus nutrition: interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiol 156:1050–1057
Speijer PR, De Waele D (1997) Screening of Musa germplasm for resistance and tolerance to nematodes. International Network for the Improvement of Banana and Plantain (INIBAP) Technical Guidelines, 1. INIBAP, Montpellier, http://www.bioversityinternational.org/publications/publications/search.html
Stoffelen R, Verlinden R, Xuyen NT, Swennen R, De Waele D (2000) Host plant response of Eumusa and Australimusa bananas (Musa spp.) to migratory endoparasitic and root-knot nematodes. Nematology 2(896):896–907
Thomas TD (2008) The role of activated charcoal in plant tissue culture. Biotechnol Adv 26:618–631
Timper P (2011) Utilization of biological control for managing plant-parasitic namatodes. In: Davies K, Spiegel Y (eds) Biologicalcontrol of plant-parasitic nematodes: building coherence between microbial ecology and molecular mechanisms. Progress in Biological Control, US Government, 11:259–303. doi:10.1007/978-1-4020-9648-8_11
Umesh KC, Krishnappa K, Bagyaraj DJ (1988) Interaction of burrowing nematode, Radopholus similis - (Cobb-1893) Thorne 1949, and VA Mycorrhiza, Glomus fasciculatum (Thaxt.) gerd and trappe in banana (Musa acuminata colla.). Indian. J Nematol 18(1):6–11
Veiga RSL, Jansa J, Frossard E, van der Heijden MGA (2011) Can arbuscular mycorrhizal fungi reduce the growth of agricultural weeds? PLoS One 6(12):e27825. doi:10.1371/journal.pone.0027825
Veresoglou SD, Rillig MC (2012) Suppression of fungal and nematode plant pathogens through arbuscular mycorrhizal fungi. Biol Lett 23:214–217
Vierheilig H, Coughlan AP, Wyss U, Piche Y (1998) Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl Environ Microbiol 64:5004–5007
Voets L, Dupré de Boulois H, Renard L, Strullu D-G, Declerck S (2005) Development of an autotrophic culture system for the in vitro mycorrhization of potato plantlets. FEMS Microbiol Lett 248:111–118
Voets L, de la Providencia IE, Fernandez K, Ijdo M, Cranenbrouck S, Declerck S (2009) Extraradical mycelium network of arbuscular mycorrhizal fungi allows fast colonization of seedlings under in vitro conditions. Mycorrhiza. doi:10.1007/s00572-009-0233-6
Vos C, Van Den Broucke D, Lombi FM, De Waele D, Elsen A (2012) Mycorrhiza-induced resistance in banana acts on nematode host location and penetration. Soil Biol Biochem 47:60–66
Wuyts N, de Waele D, Swennen R (2006) Extraction and partial characterization of polyphenol oxidase from banana (Musa acuminata Grande naine) roots. Plant Physiol Biochem 44:308–314
Zhu HH, Yao Q (2004) Localized and systemic increase of phenols in tomato roots induced by Glomus versiforme inhibits Ralstonia solanacearum. J Phytopathol 152:537–542
Acknowledgments
The authors thank the International Musa Germplasm Collection at the International Transit Centre at K. U. Leuven, Belgium, for providing the in vitro-propagated banana plantlets and the nematodes. The present work was supported by an Ivory Coast Government Fellowship to M. C. Koffi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koffi, M.C., Vos, C., Draye, X. et al. Effects of Rhizophagus irregularis MUCL 41833 on the reproduction of Radopholus similis in banana plantlets grown under in vitro culture conditions. Mycorrhiza 23, 279–288 (2013). https://doi.org/10.1007/s00572-012-0467-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-012-0467-6