Abstract
The present paper reports the presence of bacteria and yeasts tightly associated with spores of an isolate of Glomus mosseae. Healthy spores were surface disinfected by combining chloramine-T 5%, Tween-40, and cephalexin 2.5 g L−1 (CTCf). Macerates of these spores were incubated on agar media, microorganisms were isolated, and two yeasts were characterized (EndoGm1, EndoGm11). Both yeasts were able to solubilize low-soluble P sources (Ca and Fe phosphates) and accumulate polyphosphates (polyPs). Sequence analysis of 18S ribosomal deoxyribonucleic acid showed that the yeasts belong to the genera Rhodotorula or Rhodosporidium (EndoGm1) and Cryptococcus (EndoGm11). Results from inoculation experiments showed an effect of the spore-associated yeasts on the root growth of rice, suggesting potential tripartite interactions with mycorrhizal fungi and plants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In addition to the well-known interactions between plants and fungi, mycorrhizal roots offer excellent ecological niches for other microbes (Bonfante 2003).The fungal partner frequently hosts soil microorganisms, which could be of beneficial influence for fungal physiology and development (Artursson et al. 2006; Lumini et al. 2006; Javier and Germida 2003). The microorganisms may either be derived from the surrounding soil or could be carried by fungal spores. In fact, from an ecological viewpoint, many studies have shown that numerous soil microorganisms interact with hypogeous fungi promoting antagonistic, competitive, or synergistic activities (Buzzini et al. 2005).
Arbuscular mycorrhizal fungi (AMF) are not only an essential feature in the biology and ecology of most terrestrial plants, they also can interact with different classes of bacteria during their lifecycles (Bonfante et al. 2002). Walley and Germida (1996) observed bacteria associated with the outer spore wall layer of Glomus clarum NT4, but they considered their presence to be due to inefficient decontamination. Bianciotto et al. (1996) demonstrated that the cytoplasm of Gigaspora margarita harbors bacterium-like organisms in spores and mycelia. Bianciotto et al. (2003) identified a new species and bacterial genus Candidatus Glomeribacter gigasporarum in the cytoplasm of spores and symbiotic mycelia of the fungal species G. margarita, Scutellospora persica, and S. castanea. Some bacteria seem to be obligate, noncultivable endosymbionts, present through all the steps of the fungal lifecycle (Lumini et al. 2006). In addition, two filamentous fungi with different phenotypes and ascomycete-like ribosomal deoxyribonucleic acid (rDNA) sequences were isolated from crushed healthy spores or perforated dead spores of S. castanea (Hijri et al. 2002). It is therefore possible that fungi also have the ability to colonize AMF spores.
Partida-Martínez and Hertweck (2005) have proposed new ideas about the importance of endosymbiosis in fungi and have provided direct evidence to support the endosymbiont hypothesis from studies of the pathogenic fungus Rhizopus. They showed that the rhizoxin is not biosynthesized by the fungus itself but by endosymbiotic intracellular-living bacteria of the genus Burkholderia. However, the metabolic capabilities of baterial associations in the mycorrhizal system are not yet clear. In this paper, we report the presence of yeasts with a capacity to solubilize phosphate and accumulate polyphosphates (polyPs) associated with spores of Glomus mosseae and discuss the potential role of microorganisms living in association with AMF in their symbiotic interactions with plants.
Materials and methods
Collection of fungal spores
Certified inocula of G. mosseae (100 g of kaolinitic clay with spores and hyphae), from the mycorrhizal strain collection of the National Institute of Agricultural Sciences, Cuba, were inoculated into pot cultures where sorghum (Sorghum vulgare L.) was grown. After 90 days, shoots were severed, and the soil with mycorrhizal roots was used as source of spore inoculum (Fernández Martín et al. 2002). The homogenized soil material was stored at 4°C. The spores were harvested and separated by wet sieving and centrifugation in water/sucrose–solution gradient (720 g of sucrose and 20 mL L−1 of Tween-80 in water; slightly modified from Herrera-Peraza et al. 2004), and the spores were recovered from the sucrose interface. Debris and damaged or discolored spores were removed manually.
Surface decontamination of AMF spores
Two methods for spore surface decontamination were used: as per Nair et al. (1991) modified by Reis et al. (1999; N-R method) and the CTCf method developed during this study. The CTCf procedure consisted of five rinses changing the whole volume of the 2 mL vial with chloramine-T 5%. After the second rinse, 10 μL of Tween-40 was added into the vial, and the spores were subjected to further three rinses in chloramine-T. The final step of decontamination was done with 1 mL cephalexin solution (2.5 g L−1). The spores remained in the antibiotic solution for 16 h at 4°C and were then rinsed three times with sterile distilled water. A sieve of 40 μm was used as a filter at the top of the vial to avoid the loss of spores during rinsing. All materials were sterilized, and the process was carried out under aseptic conditions.
Spore inoculation onto agar media
Intact spores (∼30) sterilized as above (three replications for the N-R method; five replications for the CTCf method) were inoculated onto three growth media: LGI-P (Döbereiner et al. 1993), modified by adding 1.34 g L−1 of yeast extract, nutrient agar (NA; 28 g L−1; BIOCEN, Havana, cod. 4009), and starch–yeast extract–peptone (SYP; Caballero Mellado and Martínez Romero 1994). Surface-sterilized spores (∼30) using the CTCf method were also macerated in a sterilized porcelain mortar in small volumes of sterile distilled water (1 mL final volume), and suspensions of spore fragments were inoculated (0.1 mL) onto the three agar media. Microbial colonies were isolated after incubation for 7 days at 30°C and purified by four consecutive streaks in LGI-P media.
Microscopy and staining
Detailed examination of colonies and morphological characterization was performed using a stereoscopic microscope. Fluorescence microscopy was performed with an Olympus BX40 microscope with the cube WU (Excitation filter 380–385 nm, Barrier filter 420 nm) and a ×40 objective for polysaccharide and polyphosphate detection. β-1,4-Hexapyranose polysaccharides were stained in 1 mg mL−1 Calcofluor (CI 40622, Sigma) in water with NaOH (Melasniemi and Hernesmaa 2000). Microorganisms from LGI-P solid medium were spread on a slide, and two droplets of calcofluor solution were added. After 30 min, the sample was observed by fluorescence microscopy using UV excitation. 4′,6-Diamidino-2-phenylindole (DAPI; Hung et al. 2002), toluidine blue (Melasniemi and Hernesmaa 2000), and methylene blue (Grocetti et al. 2000) were used to detect polyPs.
Biochemical characteristics
The biochemical characteristics determined were: catalase (24 h cell growth), indole production, and starch hydrolysis (72 h cell growth; Harrigan and McCane 1968), acid production from arabinose, maltose, glucose, mannitol, or sucrose (Gordon et al. 1973), and nitrate reductase (72 h cell growth; Koneman et al. 1997).
Phosphate solubilization
Cells of 7-day-old cultures in SYP liquid medium were pelleted by centrifugation, washed, and suspended in 0.2 mL of National Botanical Research Institute (NBRI) medium without phosphate and then dropped onto NBRI phosphate growth (NBRI-P) solid medium (Nautiyal 1999) containing β-Ca3(PO4)2 or FePO4 (5g L-1) as a phosphate source. Phosphate solubilization was evaluated the formation of a clear halo and by subtracting colony diameter from the clear halo diameter. Phosphate solubilization was quantified by inoculating isolates onto the NBRI-P medium (Nautiyal 1999) containing β-Ca3(PO4)2 or FePO4. Soluble phosphate was measured in the supernatant using the phospho-molybdic method (Jackson 1958).
Effect of isolates on plant growth
Seeds of rice (Oryza sativa) were disinfected with 0.1% HgCl2, rinsed several times with sterile distilled water, and sowed onto semisolid agar (2%) LGI-P medium, modified by decreasing sucrose concentration (2%). The isolates were coinoculated by adding 100 μL of a 72-h culture of each microorganism (growing in liquid SYP medium) onto the LGI-P medium at the moment of seed implantation. Length, number, and dry weight of roots per plant were determined 10 days later; six plants per treatment were measured.
DNA extraction, polymerase chain reaction, and sequencing
All procedures were carried out as described by Persoh and Rambold (2002), using 60–80 μg of the purified microbial isolates for DNA extraction and employing the primers: ITS4: –TCC TCC GCT TAT TGA TAT GC– (White et al. 1990) and 1055F: –GGT GGT GCA TGG CCG– (Gunderson et al. 1986)
Results
Spore disinfection
The method of Nair modified by Reis et al. (1999) was not effective for disinfecting spores. Twenty-four, 48, 72, and 96 h after inoculation of “disinfected” spores onto the growth media, bacterial-like and fungal-like colonies appeared around the spores (Table 1). The results were consistent in three different trials. The CTCf method developed during this study was effective for spore disinfection. Seven days after inoculation of disinfected spores on different growth media, no growth of microorganisms on the media around the CTCf-disinfected spores was detected (Table 1). The same result was obtained five replicate trials.
Characteristics of microorganisms isolated from macerated spores disinfected using the CTCf method
After inoculation of macerated spores disinfected with the CTCf method, growth of different types of colonies was evident on the three media (Table 1). Seven days later, it was possible to differentiate about 30 types of colonies. The majority of the isolates were obtained on the LGI-P media. Nine of the isolates have survived storage on the LGI-P agar for 5 years. Bacteria-like microorganisms were microbiologically and biochemically characterized, but it has not been possible to identify them molecularly. Consequently, only the yeast-like microorganisms have been studied further here.
After 7 days growth, the yeast-like colonies were round with a continuous margin, opaque, convex, and easily emulsified with a cream-like consistency. Studies were focused on two isolates: EndoGm1 and EndoGm11. The colonies of EndoGm1 were orange or white in color (FF655D) with an average diameter of 1.9 ± 0.7 mm after 7 days; EndoGm11 colonies were beige or white in color (FFCC00) and 2.2 ± 0.6 in diameter (Table 2). Both isolates grew in the presence of 3% NaCl. Cells of the yeast-like colonies were hexagonal in shape with no determinable Gram reaction.
The isolate EndoGm11 was able to ferment all carbon sources tested (Table 2). Both yeast-like isolates were facultatively anaerobic and grew under natural air or in a chamber without oxygen.
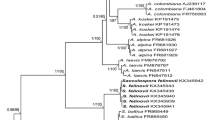
Budding and pseudomycelium formation were characteristic of EndoGm1 and EndoGm11. Additionally both fluoresced when treated with calcofluor white indicating the presence of chitin, a component of fungal cell walls (Pringle 1991; Melasniemi and Hernesmaa 2000). In the 18S rDNA analyses, 1,037 bases were sequenced for EndoGm1 and 1033 bases for EndoGm11. Sequences are registered at the National Center for Biotechnology Information database with accession number DQ643976 for EndoGm1 and DQ643977 for EndoGm11. Growth characteristics and sequence analyses concord that the two isolates are yeasts.
Comparison of 18S rDNA sequences indicated that EndoGm1 was closely related to the yeasts Rhodotorula glutinis (score = 1,275 bits [643], identity = 99%), Rhodosporidium babjevae (score = 1,092 bits [551], identity = 99%), Rhodosporidium diobovatum (score = 1,086 bits [548], identity = 99%), and Rhodotorula mucilaginosa (score = 1,076 bits [543], identity = 98%), all with an E value of 0.0. R. diobovatum is a teleomorph of R. glutinis (de Hoog et al. 2000). The isolate EndoGm11 shows homology to the yeasts Cryptococcus albidosimilis (score = 1,228 bits, identity = 100%), Cryptococcus liquefaciens (score = 1,122 bits, identity = 100%), and Cryptococcus diffluens (score = 1,120 bits, identity = 99%), all with an E value of 0.0. C. albidosimilis was isolated for the first time from Antarctic soils by Vishniac and Kurtzman (1992), C. diffluens from a diseased fingernail in Austria, and C. liquefaciens from Sake-moto in Japan (Fonseca et al. 2000). According to Fonseca et al. (2000), C. diffluens, C. liquefaciens, and C. albidosimilis use nitrate as a nitrogen source; however, EndoGm11 did not show nitrate reductase activity (Table 2).
Phosphate solubilization, polyPs formation, and effect of on root growth
The majority of the bacteria-like and yeast-like isolates that have been maintained to date produced a clear halo on a medium containing tricalcium phosphate or ferric phosphate. The size of the halo and the concentration of soluble phosphate produced after 72 h by EndoGm1 and EndoGm11 are presented in Table 3. The isolates also accumulated polyps, detected by staining with DAPI, toluidine blue, or methylene blue (Fig. 1). With DAPI, polyPs gave an intense yellow fluorescence against a bluish background of cells without polyPs (Hung et al. 2002). PolyPs-containing cells are visualized with toluidine blue by a dark blue or purple color (Melasniemi and Hernesmaa 2000). With methylene blue, polyps-containing cells appear dark blue against bluish background (Grocetti et al. 2000).
Dark cells with polyPs (arrows) after staining a EndoGm1 cells with toluidine blue and b EndoGm11 cells with methylene blue. d Yellow fluorescence (YF) in cells of EndoGm1 containing polyPs was revealed after staining with DAPI and not observed in a control preparation (c). Both yeast isolates revealed similar results with each polyPs staining
Inoculation of EndoGm1 and EndoGm11 from G. mosseae spores affected root growth of rice seedlings (Fig. 2). At the concentrations used in the in vitro experiments (2.107 cell per milliliter), EndoGm1 stimulated the amount of roots produced by rice plantlets, but EndoGm11 had an inhibitory effect on length and dry weight of roots. Indole production was detected in both isolates using the Kovacs reagent. Auxin promotes or inhibits plant growth depending on its concentration, and auxin concentration in the root environment could depend on production efficiency and size of the yeast population established on roots.
Discussion
The external disinfection of G. mosseae spores was successful with the CTCf method, and no microbial growth was observed after 7 days on the three media inoculated with clean intact spores. The main purpose of AMF spore disinfection has been to eliminate microorganisms while conserving spore viability for in vitro germination (Walley and Germida 1996), so that chloramine T at concentrations higher than 2% have been generally avoided. In the present work, chloramine T was used at a concentration of 5% for the CTCf method to assure elimination of any superficial microbial contaminants. The incorporation of the surfactant Tween-40 at low concentrations improved the effect of antimicrobial agents by increasing their contact on/adsorption to the cell wall (Buck 2003). Usually after treatment with chloramine T, AMF spores are subsequently rinsed in an antibiotic, the most frequently used being streptomycin–gentamycin solutions (Fortin et al. 2002). Streptomycin has been used at concentrations in the range 100 to 300 mg L−1 (Bianciotto et al. 2000; Reis et al. 1999). For the CTCf method, the antibiotic cephalexin was used at a concentration of 2,500 mg L−1. This high concentration of antibiotic, together with the combination of Tween plus Chloramine T at higher concentrations than previous methods, was one of the key factors responsible for the successful disinfection of G. mosseae spores and certainly helped to prepare conditions suitable for the detection and isolation of microorganisms tightly associated with the spores. The isolates obtained could be internal to the spores or imbricated in the spore walls.
Two of the isolates are yeast-like microorganisms. This is concluded from their growth characteristics, capacity for budding, formation of pseudeomycelia, presence of chitin in the cell wall, and 18S rDNA sequencing. The isolate EndoGm1 belongs to the genus Rhodotorula or Rhodosporidium, while EndoGm11 is a species of Cryptococcus with close similarity to C. albidosimilis, C. liquefaciens, and C. diffluens. The fact EndoGm11 lacks nitrate reductase activity while the three Cryptococcus species use nitrate (Fonseca et al. 2000) suggests that EndoGm11 from G. mosseae spores could be a different species of Cryptococcus.
The discovery of microorganisms tightly associated with spores of AMF raises the question about the role of these organisms in the fungal lifecycle. The spores of Glomus are reported to contain high amounts of lipids (Beilby and Kidby 1980). Lipids in the spores could also function as a carbon source for these microorganisms. It is highly probable that EndoGm1 is a species of Rhodotorula, and lipase activity has been demonstrated in this genus (Paskevicius 2001). In this context and if spore lipids are accessible, they could provide a carbon source to associated yeasts. We hypothesize that the yeasts associated with the G. mosseae spores have beneficial effects on the AMF–plant symbiosis. A positive effect of R. mucilaginosa on AMF has previously been reported (Fracchia et al. 2003). An extract of R. mucilaginosa increased hyphal length of G. mosseae and spore production in Gigaspora rosea. The yeast also had a beneficial effect on the mycorrhizal colonization of soybean and red clover (Fracchia et al. 2003).
Basidiomycetous yeasts of the genus Cryptococcus have previously been reported to be associated with plant roots and spores of mycorrhizal fungi collected from grassland (Renker et al. 2004). The authors did not isolate the microorganisms but used nuclear rDNA for detecting the yeasts. As the spores were not externally disinfected, it is not possible to determine whether the yeasts were externally and/or internally associated with spores of the fungi.
Recent studies have reported the presence of Rhodotorula and Cryptococcus yeasts in the truffle ecosystem (ectomycorrhizas). Zacchi et al. (2003) detected Cryptococcus albidus, Cryptococcus humicolus, R. mucilaginosa, Debaromyces hansenii, and Saccharomyces paradoxus in truffles associated with Quercus pubescens or Corylus avellana. Buzzini et al. (2005) isolated strains of Cryptococcus sp., D. hansenii, R. mucilaginosa, Trichosporon moniliiforme, and Candida saitoana from ascocarps of black and white truffles. Barbieri et al. (2005) found a microbial community associated with ascocarps of Tuber borchii Vittad.
Both yeast isolates obtained from G. mosseae spores solubilize nonsoluble phosphates. In the medium with tricalcium phosphate or ferric phosphate, the size of halos formed after 72 h ranged between 2 and 8 mm, which is similar to halo diameters reported by Nautiyal (1999) for microorganisms after 14 days. The concentration of P solubilized after 72 h by the yeasts from G. mosseae spores was of the same order of magnitude as for the majority of the outstanding phosphate-solubilizing strains reported by Nautiyal (1999). With FePO4 as a nonsoluble phosphate source, the growth of the microorganisms was less than with Ca3(PO4)2 (data not shown). A direct relationship between the capacity for clear halo formation in the medium with nonsoluble phosphate and the concentration of soluble phosphate that appear in the supernatant after the action of the microorganisms isolated from the spores did not occur in all instances. We have no explanation for this, but this situation was also reported in other previously published papers (Nautiyal 1999).
The yeasts isolated from G. mosseae spores showed the capacity to accumulate polyPs. polyPs are widespread in cells of living organisms including microorganisms, animals, and plants. The greatest quantities of polyPs are found in microbial cells where they play a significant role in increasing cell resistance to unfavorable environmental conditions and regulating different biochemical processes (Kulaev and Kulakovskaya 2000). The high capacity of EndoGm1 and EndoGm11 to solubilize nonsoluble phosphate, together with their ability to accumulate polyPs, may reflect an activity of these microorganisms in AMF-facilitated phosphorus supply to plants. These data, together with the effect of the yeasts on root growth, may contribute to the complex interactions of mycorrhizal fungi and plant roots. Our data suggest that the spores of G. mosseae represent an ecosystem shared not only by bacteria but also by yeasts and support the hypothesis of triple/tripartite interactions between AMF, plants, and associated microorganisms (Bonfante 2003).
References
Artursson V, Finlay RD, Jansson, JK (2006) Interactions between arbuscular mycorrhizal fungi and bacteria and their potential for stimulating plant growth. Appl Environ Microbiol 8:1–10
Barbieri E, Bertini L, Rossi I, Ceccaroli P, Saltarelli R, Guido Ch, Zambonelli A, Stocchi V (2005) New evidence for bacterial diversity in the ascoma of the ectomycorrhizal fungus Tuber borchii Vittad. FEMS Microbiol Lett 247:23–35
Beilby JP, Kidby DK (1980) Biochemistry of ungerminated and germinated spores of the vesicular- arbuscular mycorrhizal fungus, Glomus caledonius: changes in neutral and polar lipids. J Lipid Res 21:739–750
Bianciotto V, Bandi C, Minerdi D, Sironi M, Tichy HV, Bonfante P (1996) An obligately ensosymbiotic mycorrhizal fungus itself harbors obligately intracellular bacteria. Appl Environ Microbiol 62:3005–3010
Bianciotto V, Lumini E, Lanfranco L, Minerdi D, Bonfante P, Perotto S (2000) Detection and identification of bacterial endosymbionts in arbuscular mycorrhizal fungi belonging to the family Gigasporaceae. Appl Environ Microbiol 66:4503–4509
Bianciotto V, Lumini E, Bonfante P, Vandamme P (2003) ‘Candidatus Glomeribacter gigasporarum’ gen. nov., sp. nov., an endosymbiont of arbuscular mycorrhizal fungi. Int J Syst Evol Microbiol 53:121–124
Bonfante P (2003) Plants, mycorrhizal fungi and endobacteria: a dialog among cells and genomes. Biol Bull 204:215–220
Bonfante P, Bianciotto V, Minerdi D (2002) Interactions between endobacteria and arbuscular mycorrhizal fungi. In: Finan T, O’Brian M, Layzell D, Vessey K, Newton W (eds) Nitrogen fixation: global perspectives. CAB, Oxon, NY, pp 268–270
Buck KM (2003) The effect of germicides on microorganisms. In: Infection Control Today 09/2001. Available at: http://www.infectioncontroltoday.com/articles/191clean.html, viewed 9 July 2003
Buzzini P, Gasparetti C, Turchetti B, Cramarossa MR, Matini VA, Martini A, Pagnoni UM, Forti L (2005) Production of volatile organic compounds (VOCs) by yeasts isolated from the ascocarps of black (Tuber melanosporum Vitt.) and white (Tuber magnatum Pico) truffles. Arch Microbiol 184:187–193
Caballero Mellado J, Martínez Romero E (1994) Limited genetic diversity in the endophytic sugarcane bacterium Acetobacter diazotrophicus. Appl Environ Microbiol 60:1532–1537
de Hoog GS, Guarro J, Gene J, Figueras MJ (2000) Atlas of clinical fungi. 2nd ed, Centraalbureau voor Schimmelcultures, Uppsalalaan 8, 3584 CT Utrecht, The Netherlands
Döbereiner J, Reis VM, Paula MA, Olivares F (1993) Endophytic diazotrophs in sugar cane, cereals and tuber plants. In: Palacios R et al (ed) New Horizons in nitrogen fixation. Kluwer, The Netherland, pp 671–676
Fernández Martín F, Gómez Alvarez R, Venegas López LF, Martínez Silva MA, de la Noval Pons BM, Rivera Espinosa RA (2002) Producto inoculante micorrizógeno. Patent CU 22641 A1. Oficina Cubana de la Propiedad Industrial (OCPI), Available at: http://www.ocpi.cu/doc/2002/t7696.pdf, viewed 5 April 2006
Fonseca A, Scorzetti G, Fell JW (2000) Diversity in the yeast Crytococcus albidus and related species as revealed by ribosomal DNA sequence analysis. Can J Microbiol 46:7–27
Fortin JA, Bécard G, Declerck S, Dalpé Y, St-Arnaud M, Coughlan AP, Piché Y (2002) Arbuscular mycorrhiza on root–organ cultures. Can J Microbiol 80:1–20
Fracchia S, Sampedro I, Godeas A, Ocampo JA, García-Romera I (2003) Interaction between soil yeast Rhodotorula mucilaginosa and the arbuscular mycorrhizal fungi Glomus mosseae and Gigaspora rosea. Soil Biol Biochem 35:701–707
Gordon RE, Haynes WC, Pang CHN (1973) The genus Bacillus: agriculture handbook no. 427. ARS-USDA, Washington, DC
Grocetti R, Hugenholtz P, Bond P, Schuler A, Keller J, Jenkins D, Blackall L (2000) Identification of polyphosphate-accumulating organisms and design of 16S RNA-directed probes for their detection and quantification. Appl Environ Microbiol 66:1175–1182
Gunderson JH, McCutchan TF, Sogin ML (1986) Sequence of the small subunit ribosomal RNA gene expressed in the bloodstream stages of Plasmodium berghei: evolutionary implications. J Protozool 33:525–529
Harrigan WF, McCane ME (1968) Métodos de laboratorio en microbiología. Editorial Academia, Leon
Herrera-Peraza RA, Furrazola E, Ferrer RL, Fernández-Valle R, Torres-Arias Y (2004) Functional strategies of root hairs and arbuscular mycorrhizae in an evergreen tropical forest, Sierra del Rosario, Cuba. Revista CENIC Ciencias Biológicas 35: 113–123
Hijri M, Redecker D, MacDonald-Comber Petetot JA, Volgt K, Wostemeyer J, Sanders IR (2002) Identification and isolation of two ascomycete fungi from spores of the arbuscular mycorrhizal fungus Scutellospora castanea. Appl Environ Microbiol 68:4567–4573
Hung ChH, Peccia J, Zilles JL, Noguera DR (2002) Physical enrichment of polyphosphate-accumulating organisms in activated sluge. Water Environ Res 74:354–361
Jackson ML (1958) Soil chemical analysis. Printice-Hall, Englewood, NJ
Javier LJC, Germida J (2003) Bacteria associated with Glomus clarum spores influence mycorrhizal activity. Soil Biol Biochem 35:471–478
Koneman EW, Allen SD, Schreckenberger PC, Janda WM, Winn WC (1997) Color atlas and text book of diagnostic microbiology. Lippincott-Raven, Philadelphia PA, pp 171–252
Kulaev I, Kulakovskaya T (2000) Polyphosphate and phosphate pump. Annu Rev Microbiol 54:709–734
Lumini E, Ghignone S, Bianciotto V, Bonfante P (2006) Endobacteria or bacterial endosymbionts? To be or not to be. New Phytol 170:199–201
Melasniemi H, Hernesmaa A (2000) Yeast spores seem to be involved in biological phosphate removal: a microscopic in situ case study. Microbiology 146:701–707
Nair MG, Safir GR, Siqueira JO (1991) Isolation and identification of vesicular-arbuscular mycorrhiza-stimulatory compounds from clover (Trifolium repens) roots. Appl Environ Microbiol 57:434–439
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170:265–270
Partida-Martínez Laila P, Hertweck, C (2005) Pathogenic fungus harbours endosymbiotic bacteria for toxin production. Nature 437:884–888
Paskevicius A (2001) Lipase activity of yeast and yeast like fungi functioning under natural conditions. Biologija (Lithuania) 4:16–18
Persoh D, Rambold G (2002) Phacopsis—a lichenicolous genus of the family Parmeliaceae. Mycol Prog 1:43–56
Pringle JR (1991) Staining of bud scars and other cell wall chitin with calcofluor. Methods Enzymol 194:732–735
Reis VM, Paula MA, Döbereiner J (1999) Ocorrência de micorrizas arbusculares e da bacteria diazotrófica Acetobacter diazotrophicus em cana-de-açúcar. Pesqui Agropecu 34:1931–1941
Renker C, Blanke V, Borstler B, Heinrichs J, Buscot F (2004) Diversity of Cryptococcus and Dioszegia yeasts (Basidiomycota) inhabiting arbuscular mycorrhizal roots or spores. FEMS Yeast Res 4:597–603
Vishniac HS, Kurtzman CP (1992) Cryptococcus antarcticus sp. nov. and Cryptococcus albidosimilis sp. nov., basidioblastomycetes from Antarctic soils. Int J Syst Evol Microbiol 42:547–553
Walley F, Germida J (1996) Failure to decontaminate Glomus clarum NT4 spores is due to spore wall-associated bacteria. Mycorrhiza 6:43–49
White TJ, Bruns TD, Lee SB, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelgand DH, White T (eds) PCR protocols, a guide to methods and applications. Academic, San Diego, CA, pp 315–322
Zacchi L, Vaughan-Martini A, Angeline P (2003) Yeast distribution in a truffle-field ecosystem. Annals Microbiol 53:275–282
Acknowledgments
We are grateful to Dr. Ricardo Herrera in memorian for his support and help and to Prof Krishna Sivasithamparam for his critical review. LMA and EO appreciate very much stimulation and suggestions of Dr. Felix Fernández (INCA) with this research work and the technical assistance of Aniripsa Felipe-Guilarte. Our gratitude also goes to anonymous reviewers who helped very much to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirabal Alonso, L., Kleiner, D. & Ortega, E. Spores of the mycorrhizal fungus Glomus mosseae host yeasts that solubilize phosphate and accumulate polyphosphates. Mycorrhiza 18, 197–204 (2008). https://doi.org/10.1007/s00572-008-0172-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-008-0172-7