Abstract
Purpose
The aim of this double-blind randomized study was to compare the antiemetic efficacy of three 5-hydroxytryptamine type 3 antagonists in terms of the incidence and intensity of postoperative nausea and vomiting (PONV) in a homogenous group of female patients undergoing thyroidectomy.
Methods
The study cohort consisted of 203 American Society of Anesthesiologists PS I–II female patients randomized into four groups to receive at induction of anesthesia an intravenous (IV) bolus of 5 ml solution of one of the following: normal saline (placebo), granisetron 3 mg, ondansetron 4 mg, or tropisetron 5 mg. Nausea and vomiting were evaluated at five time points: during the first hour in the postanesthesia care unit (PACU) and 6, 12, 18, and 24 h postoperatively. Nausea intensity was measured using a visual analogue scale score (0–10).
Results
Patients in the placebo group displayed a high incidence of nausea in the PACU and at 6, 12, and 18 h postoperatively (44, 60, 50, and 34%, respectively) and of vomiting (26, 42, 30 and 10%). The administration of granisetron reduced significantly the incidence of nausea at 6, 12, and 18 h (26, 18, and 2%, respectively) and vomiting at 6 and 12 h (10 and 6%, respectively). Ondansetron reduced significantly the incidence of nausea and vomiting only at 6 h postoperatively (28 and 12%, respectively). The administration of tropisetron did not affect the incidence of PONV compared to placebo.
Conclusion
Among the female patients of this study undergoing thyroid surgery, granisetron 3 mg provided the best prophylaxis from PONV. Ondansetron 4 mg was equally effective, but its action lasted only 6 h, whereas tropisetron 5 mg was found ineffective.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postoperative nausea and vomiting (PONV) is one of the commonest adverse effects following surgery under general anesthesia, affecting 20–30% of unselected surgical inpatients [1, 2]. The etiology of PONV is multifactorial and depends on numerous risk factors, including previous history of PONV and motion sickness, site of surgery, gender, obesity, smoking, and opioid use [3–5]. PONV is associated with life-threatening complications, such as tension on suture lines, wound bleeding and dehiscence, increased intracranial pressure, pulmonary aspiration, dehydration, and electrolyte imbalance [6]. It is among the most unpleasant experiences associated with surgery under general anesthesia, and one of the most common reasons for poor patient satisfaction during the postoperative period [2, 6].
The traditional antiemetics, such as antihistamines (e.g., hydroxyzine), butyrophenones (e.g., droperidol), and dopamine receptor antagonists (e.g., metoclopramide), are associated with undesirable side effects (sedation, hypotension, dry mouth, dysphoria, restlessness, and extrapyramidal symptoms) [7]. In thyroid surgery, the selective 5-hydroxytryptamine type 3 (5-HT3) receptor antagonists granisetron, ondansetron, and tropisetron are considered superior to traditional antiemetic agents [8, 9]. However, to date, the efficacies of these three different serotonin antagonists have not been compared. The aim of this prospective study was to compare the prophylactic antiemetic properties (incidence and intensity) of granisetron, ondansetron, and tropisetron in a homogenous group of female patients undergoing thyroid surgery, a surgical procedure with a high incidence of PONV. The primary hypothesis was that the administration of any one of the aforementioned agents would cause a similar reduction in the incidence of PONV.
Patients and methods
The study was approved by the University Hospital of Heraklion’s Ethics Committee, and informed consent was obtained from all patients. The study was designed in 2005 and conducted from March 2006 to March 2009. Female euthyroid patients American Society of Anesthesiologists (ASA) PS I–II, aged 20–65 years, scheduled to undergo elective partial or total thyroidectomy were considered eligible for enrollment in the study. Exclusion criteria were the administration of antiemetic medication within 24 h prior to surgery, gastrointestinal disorders, and significant cardiovascular, respiratory, renal, hepatic, or endocrine diseases. Pregnant patients and patients menstruating at the time of surgery were also excluded.
Patients who agreed to participate to the study were instructed to limit fluid intake to clear fluid for up to 6 h preoperatively.
All patients were premedicated with midazolam 0.07 mg/kg intramuscularly 1–1.5 h before surgery. Upon arrival in the operating theater, the patient was started on an intravenous infusion of Ringers’ lactate solution and then connected to standard monitoring equipment (electrocardiogram, non-invasive blood pressure, saturation of peripheral oxygen). Intraoperative monitoring also included fraction of inspired oxygen, capnography, inspiratory and expiratory concentrations of inhaled agents, minimal alveolar concentration, airway pressures, and tidal and minute volumes.
To ensure that surgical technique and duration of operation remained constant in all groups, all operations were performed by the same teams of anesthetists and surgeons.
Patients were randomly assigned by an electronic randomization table to one of four groups to receive, in a blind manner immediately after induction of anesthesia, a 5 ml intravenous bolus of either 0.9% normal saline (placebo), 3 mg granisetron, 4 mg ondansetron, or 5 mg tropisetron.
Anesthesia was induced with a bolus of 2–3 mg/kg propofol and 2 μg/kg fentanyl followed by 0.15 mg/kg cis-atracurium to facilitate tracheal intubation. Anesthesia was maintained with 1.0 MAC of sevoflurane in 35% oxygen. Ventilation was mechanically controlled with a tidal volume of 6–8 ml/kg, and the respiratory rate was adjusted to maintain an end-tidal concentration of CO2 at 35–40 mmHg. Intermittent doses of 0.5–1.0 μg/kg fentanyl and 2–4 mg cis-atracurium were administered as needed. At the end of the surgical procedure, the neuromuscular blockade was reversed with 0.02 mg/kg atropine and 0.04 mg/kg neostigmine. Postoperative analgesia was provided by intravenous paracetamol 1 g three times daily and intramuscular pethidine 0.5–1 mg/kg every 4 h upon the patient’s request.
Postoperative nausea was assessed subjectively using a visual analogue scale (VAS) ruler by a doctor blind to the antiemetic/placebo administered to the patient. Postoperative assessment was at 1 h in the postanesthesia care unit (PACU) and at 6, 12, 18, and 24 h in the ward. The patients were also asked to report the number of vomiting episodes at these times. In the case of moderate/severe nausea, rescue medication consisting of metoclopramide 10 mg could be administered.
Subjective assessment of nausea
The assessment of the intensity of nausea was facilitated by a VAS ruler, which consisted of a 10-cm horizontal line. During the preoperative evaluation and recruitment, patients who agreed to enter the study received detailed instructions regarding the use of the ruler postoperatively. They were instructed that 0 cm was considered as no nausea and 10 cm corresponded to the worst imaginable nausea and that postoperatively they were expected to rate their subjective feeling of nausea by indicating the corresponding point on the ruler. In accordance with the method used in a previous study [10], the VAS scores reported by the patients were categorized as follows: no nausea if the patient reported a VAS score of 0–1 cm, mild nausea if a VAS score of 1+ to 4 cm was reported, moderate nausea if a VAS score of 4+ to 7 cm was reported, and severe nausea if a VAS score of 7+ to 10 cm was reported.
Statistical analysis
The efficiency of each of the three serotonin antagonists was investigated by comparing the incidence of nausea of any degree, the incidence of moderate/severe nausea (VAS >4 cm) [10] needing rescue medication, and the incidence of vomiting episodes among the four groups. Statistical analysis was performed using analysis of variance (ANOVA) for the quantitative parameters with normal distribution, and chi-square tests for qualitative variables, with Bonferroni correction for six pairs of comparisons. Demographic data were expressed as the number or mean ± standard deviation (SD). The incidence of nausea of any degree, moderate/severe nausea, and vomiting episodes among the four groups of patients at the different time periods was expressed as a percentage (%). Statistical analysis was performed with GraphPad Prism ver. 5.01 (GraphPad Software, San Diego, CA). Since the incidence of PONV after thyroid surgery is reported to be approximately 70% and assuming that a decrease to 35% is of clinical significance, it was calculated that 60 patients in each group were needed to provide 80% power at the 5% level of significance. Power analysis was performed with DSS RESEARCH [11].
Results
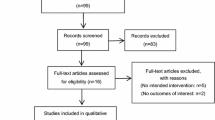
A total of 245 female patients were initially randomized into the four groups, namely, the placebo group (n = 62 patients, ondansetron group (n = 61), granisetron group (n = 61), and tropisetron group (n = 61). Data were lost for four, three and one patient in the placebo, ondansetron, and granisetron group, respectively. Four patients refused to continue with the study after randomization, and four patients were excluded because of inadvertent propofol administration. Twenty-five patients (Fig. 1) were not included in the final analysis due to the administration of a serotonin antagonist as rescue medication for PONV during the study period. In the final analysis, 50, 51, 50, and 52 patients were included in the placebo, ondansetron, granisetron, and tropisetron groups, respectively. Details on the patients’ enrolment are shown in Fig. 1, and demographic, surgery, and anesthesia information are shown in Table 1. There were no differences among groups in terms of age, weight, height, ASA physical status, duration of anesthesia, total dose of fentanyl and volume of Ringer’s lactate solution infused, days from last menstruation, and history of PONV or motion sickness. The majority of the patients reported mild pain that was sufficiently managed with paracetamol. There were no differences in the number of patients who required pethidine for postoperative analgesia among the four groups. The only significant difference found was in the number of smokers between the ondansetron and placebo groups.
Incidence of nausea and vomiting
The incidence of PONV in the placebo group was quite high, varying from 44% in the PACU to 60 and 50% during the first 6 and 12 h postoperatively, then decreasing after 12 h to 34%. In the PACU, no significant difference was observed between the placebo and granisetron, ondansetron or tropisetron groups regarding the incidence of nausea and vomiting (Table 2). However, 50% of patients who received tropisetron reported nausea, which was a significantly higher percentage than that in the granisetron group (24%).
At 6 h postsurgery, significantly fewer patients in the granisetron and ondansetron groups reported nausea or experienced vomiting compared to those in the placebo group (Table 2). No differences between the placebo and tropisetron groups were observed. At 12 and 18 h, only granisetron was found to be superior to the placebo in the prevention of PONV since fewer patients in this group reported nausea and experienced vomiting (Table 2). At 24 h, no significant differences among the four groups were observed.
During the whole period of the study, the administration of tropisetron did not significantly decrease the incidence of PONV compared to the placebo.
Discussion
We have examined the efficacy of the prophylactic administration of ondansetron, granisetron, and tropisetron as compared to placebo in a homogenous population of women undergoing thyroidectomy. The main finding of this study was that the administration of ondansetron and granisetron immediately after the induction of anesthesia were equally effective in preventing PONV during the first 6 h postoperatively. However, granisetron exhibited longer lasting effects, and its action extended up to 18 h postoperatively. It was more effective than tropisetron in reducing the incidence of nausea in the PACU (Table 2). Tropisetron did not significantly decrease the incidence of PONV compared to placebo during the whole study period.
Ondansetron, the first 5-HT3 receptor antagonist to be used for the management of PONV [12] has an elimination half life of approximately 3 h [13]. It has been studied extensively at varying dosages, ranging from 1 to 16 mg, with some authors considering 8 mg to be the minimum effective dose [14, 15], whereas others have found the 4 mg dose to be effective [16–18]. We chose to test the 4 mg dose as we have had satisfactory outcomes with this dose in our clinical practice. However, this low dose may be the reason for the short-lived beneficial effects of ondansetron against nausea and vomiting, which were observed only at 6 h postoperatively compared to placebo. The difference in the incidence of vomiting by patients given placebo and those receiving ondansetron at 12 h was quite high (30 vs. 10%, p = 0.013, odds ratio 4, 95% confidence interval 1.17–8.5), but it did not reach statistical significance with the Bonferroni correction (Table 2). On the other hand, the ondansetron group included significantly more smokers than the placebo group (Table 1) which might have affected our results since smoking has a prophylactic effect against PONV [19, 20].
One of the limitations of this study is the elevated dose of granisetron. The 3 mg dose used in our study is considered high for treating PONV since no dose–response relationship has been identified for this drug [21, 22]. We chose this dose because it was the standard dose for patients on chemotherapy in our hospital at the time of the study design. The longer lasting effects of granisetron relative to ondansetron can be attributed to the high dose of 3 mg as well as its longer elimination half life (10.6 h) [23].
The dose of 5 mg for tropisetron, which is the maximum recommended dose for chemotherapy-induced nausea, was chosen in this study because the 2 mg dose was found to be less effective in women undergoing breast surgery [24] as well as in our practice. Its administration did not significantly decrease the incidence of PONV compared to placebo at any time period in our study. These results contrast those of Jokela et al. [9], who compared the efficacy of the preoperative oral administration of tropisetron to that of ondansetron or metoclopramide in patients undergoing thyroid and parathyroid surgery. These authors found that the incidence of PONV at 2 h postoperatively was lower after premedication with tropisetron and that both serotonin antagonists were more effective compared to metoclopramide during the first 24 h. As the authors did not include a placebo group, it is difficult to compare the different findings. Furthermore, the much higher incidence of PONV in the PACU in our study can be a result of the use of neuromuscular reversal agents. In the study of Akin et al. [25], who did include a placebo group, tropisetron was found to be superior to placebo in preventing PONV following thyroid surgery. The sample size of this study was rather small (35 patients in each group) and included male and female patients, while other important demographic factors that influence PONV were not reported.
The discussion around the timing of prophylactic administration of the 5-HT3 antagonists has not been resolved, and relevant studies are sparse. Strong evidence of an increased efficacy after administration at the end of surgery exists only for ondansetron [26, 27]. The evidence of increased efficacy of the delayed administration of granisetron is indirect and based on a study published in 2005 [22] that showed remarkable results with low-dose granisetron administration varying from 0.1 to 0.3 mg at the end of surgery. There are no relevant studies for tropisetron. In our study, we decided to administer the drugs at the induction of anesthesia, since our unpublished data had shown no influence of the timing of the administration on the effectiveness of tropisetron.
Most antiemetic guidelines do not discriminate between the different agents. However, recent evidence suggests that the efficacy of serotonin antagonists is affected by genetic polymorphism in the cytochrome P450 system [12]. The three serotonin antagonists studied here are metabolized by different isoenzymes of the liver P450 system [28–30]. The efficiency of tropisetron is reduced in patients who are ultrarapid metabolizers due to polymorphism of the CYP2D6 isoenzyme, whereas the effects on the efficiency of ondansetron are less pronounced [31, 32]. The efficacy of granisetron is unaffected by CYP2D6 polymorphism [32]. Data on the prevalence of ultrarapid metabolizers in the Greek population are lacking, thus we cannot prove that the observed differences between the three serotonin antagonists are the result of possible genetic polymorphism of the CYP2D4 isoenzyme.
Thyroid surgery carries a high risk for PONV, and the inclusion of a placebo group may be considered unethical [33]. However, we decided to include a placebo group to estimate the relative efficacy and side effects of the antiemetic interventions. When two antiemetics are shown to be equally effective, we do not know if both work, or if both are ineffective, or whether the trial was not able to show possible differences [34]. With the inclusion of a placebo group for reference, such difficulties and ambiguities can be avoided. The only significant difference among the three antiemetics studied here was the lower incidence of nausea in the granisetron group compared to tropisetron in the PACU, but the reported incidence of PONV in the tropisetron group was never significantly different to placebo.
In conclusion, the administration of granisetron 3 mg at the induction of anesthesia resulted in better prophylaxis from PONV 6–18 h postoperatively compared to placebo and was more efficient than tropisetron in the PACU. The administration of 4 mg ondansetron was effective only at 6 h postoperatively. The longer lasting effect of granisetron is likely due to the elevated dose used in this study. Tropisetron 5 mg did not reduce the incidence of PONV significantly compared to placebo.
References
Gan TJ. Postoperative nausea and vomiting: can it be eliminated? JAMA. 2002;287:1233–6.
Watcha MF, White PF. Postoperative nausea and vomiting: its etiology, treatment and prevention. Anesthesiology. 1992;77:162–84.
Andrews PLR. Physiology of nausea and vomiting. Br J Anaesth. 1992;69:2S–19S.
Camu F, Lauwers MH, Verbessen D. Incidence and aetiology of postoperative nausea and vomiting. Eur J Anaesthesiol Suppl. 1992;6:25–31.
Gan TJ. Risk factors for postoperative nausea and vomiting. Anesth Analg. 2006;102:1884–98.
Gan T, Meyer T, Apfel CC, Chung F, Davis PJ, Habib AS, Hooper VD, Kovac AL, Kranke P, Myles P, Philip BK, Samsa G, Sessler DI, Temo J, Tramer MR, Kolk GV, Watcha M. Consensus guidelines for managing postoperative nausea and vomiting. Anesth Analg. 2007;105:1615–28.
Watcha MF. Nausea and vomiting: choice of drugs and treatment. Curr Opin Anaesthesiol. 1996;9:300–5.
Fujii Y. The benefits and risks of different therapies in preventing postoperative nausea and vomiting in patients undergoing thyroid surgery. Curr Drug Saf. 2008;3:27–34.
Jokela R, Koivuranta M, Kangas-Saarela T, Purhonen S, Alahuhta S. Oral ondansetron, tropisetron or metoclopramide to prevent postoperative nausea and vomiting: a comparison in high-risk patients undergoing thyroid or parathyroid surgery. Acta Anaesthesiol Scand. 2002;46:519–24.
Boogaerts JG, Vanacher E, Seidel I, Albert A, Bardian FM. Assessment of postoperative nausea using a visual analogue scale. Acta Anaesthesiol Scand. 2000;44:470–4.
http://www.dssresearch.com/toolkit/sscalc/size_p2.asp 2010 Decision Support Systems, LP. All Rights Reserved.
Ho KY, Gan TJ. Pharmacology, pharmacogenetics, and clinical efficacy of 5-hydroxytryptamine type 3 receptor antagonists for postoperative nausea and vomiting. Curr Opin Anaesthesiol. 2006;19:606–11.
Roila F, Del Favero A. Ondansetron clinical pharmacokinetics. Clin Pharmacokinet. 1995;29:95–109.
Tramer MR, Reynolds DJ, Moore RA, McQuay HJ. Efficacy, dose-response, and safety of ondansetron in prevention of postoperative nausea and vomiting: a quantitative systematic review of randomized placebo-controlled trials. Anesthesiology. 1997;87:1277–89.
Bouly A, Nathan N, Feiss P. Prevention of postoperative nausea and vomiting by ondansetron. Ann Fr Anesth Reanim. 1992;11:496–501.
Korttila K, Clergue F, Leeser J, Feiss P, Olthoff D, Payeur-Michel C, Wessel P, Nave S, Hahne W, Brown R. Intravenous dolasetron and ondansetron in prevention of postoperative nausea and vomiting: a multicenter, double-blind, placebo-controlled study. Acta Anaesthesiol Scand. 1997;41:914–22.
Pearman MH. Single dose IV ondansetron in the prevention of postoperative nausea and vomiting. Anaesthesia. 1994;49:11–5.
Figueredo ED, Canosa LG. Ondansetron in the prophylaxis of postoperative vomiting: a meta-analysis. J Clin Anesth. 1998;10:11–21.
Chimbira W, Sweeney BP. The effect of smoking on postoperative nausea and vomiting. Anaesthesia. 2000;55:540–4.
Ionescu D, Badescu C, Acalovschi I. Nicotine patch for the prevention of postoperative nausea and vomiting: a prospective randomized trial. Clin Drug Investig. 2007;27:559–64.
Fujii Y, Saiton Y, Tanaka H, Toyooka H. Prophylactic antiemetic therapy with granisetron in women undergoing thyroidectomy. Br J Anaesth. 1998;8:526–8.
D’Angelo R, Philip B, Gan TJ, Kovac A, Hantler C, Doblar D, Melson T, Minkowitz H, Dalby P, Coop A. A randomized, double-blind, close ranging, pilot study of intravenous granisetron in the prevention of postoperative nausea and vomiting in patients undergoing abdominal hysterectomy. Eur J Anaesthesiol. 2005;22:774–9.
Carmichel J, Cantwell BMJ, Edwards CM, Zussman BD, Thompson S, Rapeport WG, Harris AL. A pharmacokinetic study of granisetron (BRL43694A), a selective 5-HT3 receptor antagonist: correlation with antiemetic response. Cancer Chemother Pharmacol. 1989;24:45–59.
Chan MT, Chui PT, Ho WS, King WW. Single-dose tropisetron for preventing postoperative nausea and vomiting after breast surgery. Anesth Analg. 1998;87:931–5.
Akin A, Esmaoglu AE, Gunes I, Boyacu A. The affects of the prophylactic tropisetron propofol combination on postoperative nausea and vomiting in patients undergoing thyroidectomy under desflurane anesthesia. Mt Sinai Med. 2006;73:560–3.
Sun R, Klein KW, White PF. The effect of timing of ondansetron administration in outpatients undergoing otolaryngologic surgery. Anesth Analg. 1997;84:331–6.
Tang J, Wang B, White PF, Watcha MF, Qi J, Wender RH. The effect of timing of ondansetron administration on its efficacy, cost-effectiveness, and cost-benefit as a prophylactic antiemetic in the ambulatory setting. Anesth Analg. 1998;86:274–82.
Fischer V, Baldeck JP, Tse FL. Pharmacokinetics and metabolism of the 5-hydroxytryptamine antagonist tropisetron after single oral doses in humans. Drug Metab Dispos. 1992;20:603–7.
Zofran (ondansetron hydrochloride) injection premixed (prescribing information). Research Triangle Park, NC; GlaxoSmithKline; 2006. Available at: http://www.drugs.com/monograph/ondansetron-hydrochloride.html.
Kytril (granisetron hydrochloride) injection (prescribing information). Nutley, NJ: Roche Pharmaceuticals; 2006. Available at: https://www.medicinescomplete.com/mc/ahfs/current/login.htm.
Kaiser R, Sezer O, Papies A, Bauer S, Schelenz C, Tremblay PB, Possinger K, Roots I, Brockmoller L. Patient-tailored antiemetic treatment with 5-hydroxytryptamine type 3 receptor antagonists according to cytochrome P-450 2D6 genotypes. J Clin Oncol. 2002;20:2805–11.
Janicki PK, Schuler HG, Jarzembowski TM, Rossi M 2nd. Prevention of postoperative nausea and vomiting with granisetron and dolasetron in relation to CYP2D6 genotype. Anesth Analg. 2006;102:1127–33.
Aspinall LR, Goodman NW. Denial of effective treatment and poor quality of clinical information in placebo controlled trials of ondansetron for postoperative nausea and vomiting: a review of published trials. Br Med J. 1995;311:844–6.
Tramer MR. A rational approach to the control of postoperative nausea and vomiting: evidence from systematic reviews. Part I. Efficacy and harm of antiemetic interventions and methodological issues. Acta Anaesthesiol Scand. 2001;45:4–13.
Conflict of interest
There are no conflicts of interest related to the study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Metaxari, M., Papaioannou, A., Petrou, A. et al. Antiemetic prophylaxis in thyroid surgery: a randomized, double-blind comparison of three 5-HT3 agents. J Anesth 25, 356–362 (2011). https://doi.org/10.1007/s00540-011-1119-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-011-1119-2