Abstract
Purpose
Despite the recommendations for cancer survivors to engage in either moderate or vigorous physical activity, light-intensity physical activity may also have beneficial effects on mental health. The purpose of this study was to examine the associations between light, moderate, and vigorous physical activity and depressive symptoms in breast cancer survivors over 1 year post-treatment.
Methods
Participants (N = 201) were a sample of breast cancer survivors who self-reported depressive symptoms and wore an accelerometer for seven consecutive days to measure physical activity, on five occasions every 3 months post-treatment for cancer.
Results
Based on the results of hierarchical linear modeling, relative to others (i.e., between-person effects) and to oneself (i.e., within-person effects), higher levels of light- and moderate-intensity physical activity, but not vigorous-intensity physical activity, were associated with lower scores of depressive symptoms.
Conclusions
In the first year post-treatment, increases in light- and moderate-intensity physical activity, but not vigorous-intensity physical activity, were associated with lower scores of depressive symptoms in relation to other study participants (i.e., between-person effects) and when participants were compared to their own typical levels of physical activity (i.e., within-person effects). The findings may have implications for physical activity recommendations following treatment for breast cancer as light-intensity physical activity may play a role in mitigating depressive symptoms over the first year.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Breast cancer and its associated treatment induce significant psychological distress for many women receiving this diagnosis [1]. An increase in depressive symptoms is one of the most prevalent manifestations of psychological distress that breast cancer survivors (BCS) will experience during their cancer treatment and the year after [2,3,4]. Specifically, depressive symptoms refer to the level of dysphoric symptoms (e.g., feelings of guilt, helplessness and hopelessness, sadness, worthlessness) that emerge from the experience of ordinary events [5, 6]. Depending on their intensity and frequency, these symptoms can manifest in a form ranging from “low mood” to “clinical depression” [7]. Although prevalence estimates vary, it is reported that about one third and up to 50% of cancer survivors go through a state of low mood or minor depression after receiving their diagnosis [8, 9]. Moreover, the prevalence of clinical depression is estimated to range between 8 and 24% [10], and it might be higher (20–50%) during the first year following a cancer diagnosis [2, 10, 11]. As such, this is a time to identify potential factors that mitigate depressive symptoms.
Through both epidemiological and experimental studies, researchers have concluded that moderate- and vigorous-intensity physical activity (PA) is a non-pharmacological treatment to mitigate depressive symptoms in both apparently healthy [12,13,14,15] and cancer survivor populations [12, 16,17,18]. The reported anti-depressant effect for PA among cancer survivors is modest, with reported between-group post-treatment effect sizes (Cohen’s d) ranging from −0.13 to −0.22 [16, 17], with stronger effects of PA reported for BCS when compared to survivors from other cancers [16, 19]. The American College of Sports Medicine (ACSM; [20]) as well as the American Cancer Society (ACS; [21]) PA guidelines highlight the need for cancer survivors to be active at moderate and vigorous intensities. As such, cancer survivors are encouraged to perform ≥150 min of moderate-to-vigorous aerobic PA/week or ≥75 min per week of vigorous aerobic PA/week and include at least 2 days of strength training exercises [21].
Although the ACSM and ACS emphasize the importance of regular moderate-to-vigorous PA for health and well-being, it is also essential to consider the potential anti-depressant effect of light-intensity PA. In absolute terms, light-intensity PA refers to activities that are associated with objectively assessed accelerometer-based cut-points ranging from 100 to 1951 cpm [22]. In relative terms, light-intensity PA refers to activities that are rated by participants as ≤11 on the rating of perceived exertion scale [23] and/or that are performed at a heart rate corresponding to ≤63% of maximum heart rate of the participant [24]. Examples of light-intensity PA include walking (≤ 2 mph), stretching, Tai Chi, yoga, and Qigong [25], as well as housework, and occupation and domestic-related activities (e.g., gardening, light housework, washing dishes). It is worthwhile testing the potential anti-depressant effect of light-intensity PA because cancer survivors report enjoying light-intensity PA such as walking [26], yoga [27], and have more plans to engage in light-intensity PA (compared to moderate or vigorous PA; [28,29,30]).
One factor that complicates conclusions regarding light-intensity PA is that PA has predominantly been assessed using a self- or interviewer-administered questionnaire. For a number of reasons, this is a significant shortcoming. First, some PA questionnaires do not include questions that specifically seek information about light-intensity PA [31]. Second, the “intensity” feature of PA questionnaires is likely problematic; it is prone to misinterpretation and PA misclassification (e.g., reporting light as moderate-intensity PA) for various populations [32, 33]. Finally, supporting this contention, test-retest reliability values are consistently lower for light- than for vigorous-intensity PA items [33, 34]. Taken together, higher-intensity PA is more likely than lower intensity PA to be adequately captured by questionnaires. The utility of PA questionnaires to examine the association between different PA intensities and mental health among cancer survivors is therefore limited. The use of a device-based PA measure, such as an accelerometer, would be preferable for delineating the effect of different PA intensities (i.e., light, moderate, vigorous) on symptoms of depression.
Few experimental studies adequately report the intensity of the PA performed by the participants during PA interventions or specifically test the effect of light-intensity PA on depressive symptoms among apparently healthy adults [13] and cancer survivors [17, 19], making it impossible to determine the effect of light-intensity PA on depressive symptoms among cancer survivors. Some observational and experimental studies show that both light- (e.g., yoga, stretching, walking) and moderate- (aerobic and strength training) intensity PA are positively associated with lower levels of depressive symptoms and improved mental health among apparently healthy adults [18, 35, 36]. One cross-sectional study conducted among colorectal cancer survivors also reported that higher levels of light-intensity PA were associated with better well-being, but only for those performing no moderate-to-vigorous intensity PA [37]. In contrast, Phillips et al. [28] reported an inconsistent and non-statistically significant association between objectively assessed light-intensity or lifestyle PA and quality of life indicators (i.e., well-being, anxiety, depressive symptoms, fatigue) among BCS. While, Phillips et al. [28] collected data at two time points to examine PA and depressive symptoms prospectively, due to the design of the study, they were unable to examine both between and within-person changes in the trajectories of PA and depressive symptoms in the year following treatment for breast cancer. Phillips et al. [28] suggested that researchers examine the association between changes in different activity intensities on depressive symptoms at multiple time points after treatment.
The purpose of this study was to examine the extent to which changes in objectively assessed light, moderate, and vigorous PA are associated with changes in depressive symptoms among BCS during the first year post-treatment. This is an important time in the survivorship trajectory since depressive symptoms may be increased [2], and yet treatment plans are often conducive to interventions in the early post-treatment period [38]. The relationship between PA and depressive symptoms was tested at both the inter- (i.e., between) and intra- (i.e., within) individual levels. Specifically, we sought to examine the effects of various intensities of PA on depressive symptoms more generally at the group level (i.e., between people) and to explore the association between PA intensity and depressive symptoms relative to the woman’s own average PA levels (at each level of intensity). Given the overall effect of PA on depressive symptoms for BCS [16], we hypothesized that changes in objectively measured light, moderate, and vigorous PA would be associated with changes in depressive symptoms at both the between and within-person levels. In summary, we expected that in general, women with higher amounts of PA at moderate-vigorous intensities would report fewer depressive symptoms (e.g., between-person effects), and women would report lower depressive symptoms during data collections when they reported higher amounts and intensity of PA compared to their own average (e.g., within-person effects).
Methods
Participants and procedures
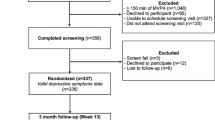
Following appropriate hospital and university research ethics board approvals, women nearing completion of systemic treatment for breast cancer were approached in cancer clinics or through posted information and informed of the study. Interested participants contacted the research assistant and, if eligible, provided written informed consent. Informed consent was obtained from all individual participants included in the study. To meet inclusion criteria for the current study, participants (N = 201) had to be at least 18 years of age, provide written informed consent in either English or French, recently (i.e., 0–20 weeks) completed primary treatment (i.e., surgery, chemotherapy, radiation therapy) for stage I to III breast cancer, be treated for a first cancer diagnosis, and report no health concerns that may inhibit participation in PA. Involvement in this study included completing self-report questionnaires and wearing an accelerometer to measure light, moderate, and vigorous PA for 7 consecutive days during five data collection periods (i.e., at baseline, then every 3 months for four more data collections over 1 year).
Measures
Demographics
Participants were asked to self-report their age, marital status, breast cancer stage, when/what treatment was received (i.e., surgery, chemotherapy, and radiation), income, and highest level of education attained. Furthermore, a trained lab technician measured and recorded baseline height and weight, which were used to calculate body mass index (BMI; i.e., weight in kilograms divided by height in meters squared).
Physical activity
PA was measured using a GT3X accelerometer (Actigraph, Pensicola, Florida). During each data collection period, women were asked to wear the accelerometer at all times over 7 days; they were asked to remove the accelerometer when participating in water activities. Movement data were recorded every minute and the number of daily minutes of light (100 to 1951 counts/min), moderate (1952–5724 counts/min), and vigorous (> 5725 counts/min) PA was calculated using established cut-points [23]. PA scores reflect the average number of minutes per day spent in light, moderate, and vigorous movement.
Depressive symptoms
The 10-item Center for Epidemiologic Studies Depression Scale [39, 40] was used in this study. Following the stem “During the past week (7 days), about how often did you feel…”, participants were asked to rate 10-items (e.g., I felt depressed; I felt fearful) on a Likert-type rating scale ranging from 0 (Rarely or none of the time [<1 day]) to 3 (All of the time [5–7 days]). Depressive symptom scores were calculated by taking the average of these 10 items at each of the five data collections. Cronbach’s alpha coefficients were α = 0.82–0.86 across data collections.
Data analysis
Descriptive statistics such as means and correlation analysis were calculated to describe the sample population. Multilevel modeling was implemented with SAS 9.4 (SAS Institute, Cary, NC, USA), using the PROC MIXED program. A two-level hierarchical model was used to assess whether changes in PA were associated with changes in the level of depressive symptoms. First-level units were within-person variables (i.e., repeated measures; time variable) and second-level units were between-individual predictors (i.e., frequency of days and duration of time the accelerometer was worn, as well as age, level of education, BMI, and time since treatment). Consistent with previous research [41, 42], accelerometer data was not included in the analyses if the participants had extreme counts (i.e., > 20,000) or if data were not available for a minimum of 600 min on 4 or more days. Correlations within-individual over repeated measures of time were controlled using a random intercept model. In the random intercept model, it was assumed that at each time point the effect of the explanatory variables on the response variable is equal. The random intercept model was used to examine the association between changes in PA and depressive symptoms over time. The slope coefficients for light-, moderate-, and vigorous-intensity PA were of central relevance to the objective of this study as they represented the between-person associations between various PA intensities and depressive symptoms while controlling the within-person correlation. Furthermore, this parsimonious model includes within-person and between-person variables to examine whether the changes in individual’s PA levels, relative to their own typical/average amounts of light-, moderate-, and vigorous-intensity PA, are independent of the between-person effects of PA in predicting scores of depressive symptoms. For the within-person analysis, each participant’s average score (across time points) of light-, moderate-, and vigorous-intensity physical activity was used as the comparison to assess individual changes in light-, moderate-, and vigorous-intensity physical activity relative to their own behavior. To calculate the between-person and within-person effects of individual’s PA levels on depressive symptoms, the main predictors light-, moderate-, and vigorous-intensity PA were centered on each individual’s mean of each predictor over time. A number of covariates (e.g., age, education, time since surgery, chemotherapy and radiation, BMI, and marital status) were also included in the model to assess unique relationships between light, moderate, and vigorous PA on scores of depressive symptoms after accounting for demographic correlates and time spent engaged in other PA intensities.
Results
Preliminary analyses
The study sample comprised women with stage 1 (n = 83), 2 (n = 78), or 3 (n = 38) breast cancer (n = 2; did not indicate). On average, participants were 55 years of age (SD = 11), had an average household income of $102,041 (SD = 185,217), an average BMI of 26.06 (SD = 6.15) kg/m2 and were 3.5 months (SD = 2.3) post-surgery, chemotherapy, and/or radiation. Most participants reported having a university degree or higher education (n = 101; 50.7%) and being married (n = 128; 64.3%).
There were 10 data points across all data collection points (5 data collections × 7 days = 35 total days of data) that were slightly lower than the 600 min/day cut-point for usable data. This represented 0.01% of all data, and removing these individuals made no difference to the descriptive or main findings. As such, they were retained in the analysis and there was between 98.6 and 100.0% of usable accelerometer data (time 1 = 98.6%, time 2 = 100.0%, time 3 = 99.3%, time 4 = 98.6%, time 5 = 99.3%). Since the multilevel modeling approach is capable of handling missing data on level 1, missing data for PA were not replaced. Across the five data collections, mean accelerometer wear times were 821.8 (SD = 83.0) to 899.4 (SD = 90.2) min/day, and the median number of days worn per week was seven.
Descriptive statistics of average daily minutes of light, moderate, and vigorous PAs and depressive symptom scores are reported in Table 1. Across all time points, participants completed approximately 128 min of light intensity, 15 min of moderate intensity, and 2 min of vigorous-intensity PA per day and had depressive symptom scores of approximately 0.70 (on a scale from 0 to 3). Average scores of light daily activity ranged from 129.10 (SD = 40.32) at time 1 to 126.63 (SD = 38.08) at time 5. Similarly, average daily minutes of moderate activity and vigorous activity ranged from 15.01 (SD = 11.58) to 13.33 (SD = 10.62) and 2.21 (SD = 4.10) to 2.13 (SD = 3.10), respectively. Average depressive symptom scores of the 10 items ranged from 0.65 (SD = 0.51) at time 3, to 0.77 (SD = 0.61) at time 2. Pearson correlation coefficients between study variables are reported in Table 2.
Main analyses
While variability in the overall mean depressive symptom scores was evident over time (see Table 1), the within-person analyses showed that over time (i.e., data collection time points, Table 3), depressive symptoms decreased (β = −.73; p = .03), and across time points, there was a small negative correlation between average daily minutes of light, moderate, and vigorous PA and depressive symptom scores.
The standardized regression coefficients from the random intercept model are reported in Table 3. The intra-individual association between minutes of light and moderate PA and depressive symptoms was statistically significant. Furthermore, results show that changes in average minutes of both light and moderate PA predicted changes in depressive symptom scores at both the between-person (σ 2 = 0.14, S.E = 0.01, p < 0.01) and within-person (σ 2 = 0.11, S.E = 0.006; p < 0.01) level. Specifically, results show that an increase in light and moderate PA, relative to others (i.e., between-person effects) and oneself (i.e., within-person effects), was associated with a decrease in depressive symptom scores. Most of the covariates had an effect on depressive symptom scores at the between-person level. Specifically, being older, having more education, more time since surgery, chemotherapy, and/or radiation, being married, and time since baseline measure, were all negatively associated with depressive symptoms scores (see Table 3). It is noteworthy that relative to their own average level of light PA, every one standard deviation increase in light PA (b = −0.96, SE = 0.34, p < 0.01) is associated with a decrease by 0.96 standard deviations in the individual’s average depressive symptom scores after controlling for moderate- and vigorous-intensity PA and covariates. Similarly, one standard deviation of increase in moderate PA (b = −1.30, SE = 0.34, p < 0.01) is associated with a 1.30 standard deviation units decrease in the individual’s average score of depressive symptoms after controlling for light- and vigorous-intensity PA and covariates. Our results did not show any statistically significant predictive effect of vigorous PA on depressive symptom scores (b = −0.21, SE = 0.33, p = 0.54).
Discussion
The purpose of this study was to examine the association between various intensities of PA and depressive symptoms over the first year after breast cancer treatment. Specifically, we sought to examine the extent to which light, moderate, and vigorous PA predicts depressive symptoms in BCS over time. Based on the findings, increases in objectively measured light- and moderate-intensity PA, but not vigorous-intensity PA, predicted lower depressive symptoms at both the between and within- person levels. In other words, relative to those who engaged in less light and moderate PA, BCS who engaged in more light and moderate PA reported lower depressive symptom scores (i.e., between-person effects). Furthermore, when BCS increased their light- and moderate-intensity PA, compared to their own typical amount of activity, their depressive symptom scores were lower (i.e., within-person effects).
Consistent with previous research, we found a negative association between moderate PA and depressive symptoms among cancer survivors over time [12,16,17 19]. For example, Brown et al. [16] found that PA provides a small reduction in depressive symptoms among cancer survivors. However, in their meta-analysis, Brown et al. [16] examined the overall effect of PA on depressive symptoms in PA interventions, and mean levels of PA intensity across the studies. We extended their findings by specifically examining the relationships of various intensities of PA on depressive symptoms both within and between BCS. Furthermore, we used an objective measure of PA (i.e., accelerometers) to obtain average minutes of light, moderate, and vigorous PA which lends credibility to our results regarding the effects of different PA intensities on depressive symptoms over time. In line with existing research, age was negatively associated with depressive symptoms [e.g., 43]. Specifically, we found that younger breast cancer survivors reported higher levels of depressive symptoms than older survivors. This is aligned with general trends in mental health among adults [44] suggesting that depression symptoms decrease over time. Additionally, cancer is considered an aging disease [45] and if diagnosed at a younger age, the diagnosis may interfere with many aspects of development such as family and work responsibilities.
To support health benefits, researchers suggest cancer survivors should accrue at least 150 min of moderate or 75 min of vigorous aerobic PA every week [17]. However, in addition to moderate PA, we found that light-intensity PA was also negatively associated with levels of depressive symptoms. This finding indicates that increases in light-intensity PA are associated with reductions in depressive symptoms over time. Increasing light (and moderate) intensity PA may precede lower scores of depressive symptoms and be one suitable method for encouraging PA behavior among BCS. Specifically, researchers have identified that within cancer survivor populations, light [32] and moderate [28] intensity PA are preferred to vigorous PA and are beneficial for mental health. For clinical practice, this means that the type of PA that cancer survivors prefer and are likely to do (i.e., all light PA, such as walking) [28] could decrease their depressive symptoms. Targeting BCS to increase their light-intensity PA could be one avenue for improving mental health in the first year post-treatment (e.g., through walking groups) as any increase in light PA above the women’s own average levels appears to be beneficial. Furthermore, PA intensity has been found to inversely influence affective responses to PA [46], and affective responses to PA are predictive of PA behavior [47]. As such, engaging in lower intensity PA may lower scores of depressive symptoms and foster additional PA behavior [48]. That is, undertaking light-intensity PA may serve as a starting point for being more physically active, which could potentially lead to higher-intensity PA and additional subsequent health benefits. However, further research is warranted to test this contention.
Despite the benefits of using a prospective design and an objective measure of PA, study limitations should also be acknowledged. Although the association between light and moderate PA systematically varied over time, the observational design of the current study prohibits inferences of causality. Further work using an experimental design is necessary to test the extent to which light- and moderate-intensity PA influence depressive symptoms in BCS. Furthermore, while the objective marker of minutes of light, moderate, and vigorous PA is a strength of the current study, depressive symptoms were measured through a self-report questionnaire which may have influenced results. In addition, the generalizability of the findings is limited to BCS in their first year post-treatment. While our findings indicate a linear relationship between light and moderate PA and lower scores of depressive symptoms in BCS, our findings do not provide insight regarding the optimal dose of light or moderate PA that is associated with lower scores of depressive symptoms. Although a suggested dose of moderate PA is recommended in the PA guidelines (e.g., 150 min/week), further understanding of the minimum/optimal amount of light-intensity PA that could be beneficial for BCS is needed to make recommendations. In addition, the short form CES-D precludes clinical diagnosis of depression [39], and thus research is warranted to examine changes in clinically depressed BCS.
Stakeholders who create guidelines may wish to consider more than just physical health outcomes for BCS as mental health is an important focus that appears to improve systematically with participation in lower intensity PA. Overall, light- and moderate-intensity PA were predictive of BCS depressive symptoms overtime. Of particular interest in this study was the unique predictive effect that light-intensity PA had on depressive symptoms in BCS during the first year post-treatment. Therefore, health promotion specialists may wish to encourage light-intensity PA due to the beneficial association it has with depressive symptoms in BCS. The findings provide a foundation for researchers to further examine the potentially beneficial effects of light-intensity PA for mental health and determine whether light-intensity PA could be included in PA guidelines for BCS. Further research examining the causal effects of light-intensity PA on depressive symptoms is warranted, as well as research examining the extent to which light-intensity PA may lead to physical health outcomes via reducing depressive symptoms in BCS.
References
Stanton AL (2006) Psychosocial concerns and interventions for cancer survivors. J Clin Oncol 24:5132–5137
Bower JE (2008) Behavioral symptoms in patients with breast cancer and survivors. J Clin Oncol 26:768–777
Luecken LJ, Compas BE (2002) Stress, coping, and immune function in breast cancer. Ann Behav Med 24:336–344
Sellick SM, Crooks DL (1999) Depression and cancer: an appraisal of the literature for prevalence, detection, and practice guideline development for psychological interventions. Psycho-Oncology 8:315–333
Radloff LS (1977) The CES-D scale a self-report depression scale for research in the general population. Appl Psychol Meas 1:385–401
Wrosch C, Miller GE (2009) Depressive symptoms can be useful: self-regulatory and emotional benefits of dysphoric mood in adolescence. Journal of Personality and Social Psychology 96(1181)
Andrews PW, Thomson JA Jr (2009) The bright side of being blue: depression as an adaptation for analyzing complex problems. Psychol Rev 116:620–654
Derogatis LR, Morrow GR, Fetting J, Penman D, Piasetsky S, Schmale AM, Henrichs M, Carnicke CL (1983) The prevalence of psychiatric disorders among cancer patients. J Am Med Assoc 249:751–757
Reich M, Lesur A, Perdrizet-Chevallier C (2008) Depression, quality of life and breast cancer: a review of the literature. Breast Cancer Res Treat 110:9–17
Krebber AM, Buffart LM, Kleijn G, Riepma IC, Bree R, Leemans CR, Becker A, Brug J, Straten A, Cuijpers P, Verdonck-de Leeuw IM (2014) Prevalence of depression in cancer patients: a meta-analysis of diagnostic interviews and self-report instruments. Psycho-Oncology 23:121–130
Burgess C, Cornelius V, Love S, Graham J, Richards M, Ramirez A (2005) Depression and anxiety in women with early breast cancer: five year observational cohort study. British Medical Jouranl 330:702
Sabiston CM, Brunet J (2012) Reviewing the benefits of physical activity during cancer survivorship. Am J Lifestyle Med 6:167–177
Conn VS (2010) Depressive symptom outcomes of physical activity interventions: meta-analysis findings. Ann Behav Med 39:128–138
Cooney GM, Dwan K, Greig CA, Lawlor DA, Rimer J, Waugh FR, McMurdo M, Mead GE (2013) Exercise for depression. Cochrane Libr 12:CD004366
Rebar AL, Stanton R, Geard D, Short C, Duncan MJ, Vandelanotte C (2015) A meta-meta-analysis of the effect of physical activity on depression and anxiety in non-clinical adult populations. Health Psychol Rev 9:366–378
Brown JC, Huedo-Medina TB, Pescatello LS, Ryan SM, Pescatello SM, Moker E, LaCroix JM, Ferrer RA, Johnson BT (2012) The efficacy of exercise in reducing depressive symptoms among cancer survivors: a meta-analysis. PLoS One 7:e30955
Craft LL, VanIterson EH, Helenowski IB, Rademaker AW, Courneya KS (2011) Exercise effects on depressive symptoms in cancer survivors: a systematic review and meta-analysis. Cancer Epidemiol Biomark Prev 21:3–19
Teychenne M, Ball K, Salmon J (2008) Physical activity and likelihood of depression in adults: a review. Prev Med 46:397–411
Fong DY, Ho JW, Hui BP, Lee AM, Macfarlane DJ, Leung SS, Cerin E, Chan WY, Leung IP, Lam SH, Taylor AJ (2012) Physical activity for cancer survivors: meta-analysis of randomised controlled trials. Br Med J 344:e70
Panel E (2010) American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. American College of Sports Medicine:1409–1426
Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, Bandera EV, Hamilton KK, Grant B, McCullough M, Byers T (2012) Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin 62:242–274
Freedson PS, Melanson E, Sirard J (1998) Calibration of the computer science and applications, Inc. accelerometer. Medicine and Science in Sports and Exercise 30:777–781
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14:377–381
Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee IM, Nieman DC, Swain DP (2011) American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc 43:1334–1359
Ainsworth BE, Haskell WL, Herrmann SD, Meckes N, Bassett DR, Tudor-Locke C, Greer JL, Vezina J, Whitt-Glover MC, Leon AS (2012) The compendium of physical activities tracking guide. Healthy Lifestyles Research Center, College of Nursing & Health Innovation, Arizona State University. Arizona State University, phoenix.
Jones LW, Courneya KS (2002) Exercise counseling and programming preferences of cancer survivors. Cancer Pract 10:208–215
Nicole Culos-Reed S, Carlson LE, Daroux LM, Hately-Aldous S (2006) A pilot study of yoga for breast cancer survivors: physical and psychological benefits. Psycho-Oncology 15:891–897
Phillips SM, Awick EA, Conroy DE, Pellegrini CA, Mailey EL, McAuley E (2015) Objectively measured physical activity and sedentary behavior and quality of life indicators in survivors of breast cancer. Cancer 121:4044–4052
Vallance J, Lavallee C, Culos-Reed N, Trudeau M (2013) Rural and small town breast cancer survivors’ preferences for physical activity. International journal of behavioral medicine 20:522–528
Whitehead S, Lavelle K (2009) Older breast cancer survivors’ views and preferences for physical activity. Qual Health Res 19:894–906
Norton K, Norton L, Sadgrove D (2010) Position statement on physical activity and exercise intensity terminology. J Sci Med Sport 13:496–502
Altschuler A, Picchi T, Nelson M, Rogers JD, Hart J, Sternfeld B (2009) Physical activity questionnaire comprehension-lessons from cognitive interviews. Med Sci Sports Exerc 41:336–343
Matthews CE, Steven CM, George SM, Sampson J, Bowles HR (2012) Improving self-reports of active and sedentary behaviors in large epidemiologic studies. Exerc Sport Sci Rev 40:118–126
van Poppel MN, Chinapaw MJ, Mokkink LB, Van Mechelen W, Terwee CB (2010) Physical activity questionnaires for adults. Sports Med 40:565–600
Buman MP, Hekler EB, Haskell WL, Pruitt L, Conway TL, Cain KL, Sallis JF, Saelens BE, Frank LD, King AC (2010) Objective light-intensity physical activity associations with rated health in older adults. Am J Epidemiol 172:1155–1165
Chu IH, Buckworth J, Kirby TE, Emery CF (2009) Effect of exercise intensity on depressive symptoms in women. Ment Health and Phys Act 2:37–43
Thraen-Borowski KM, Trentham-Dietz A, Edwards DF, Koltyn KF, Colbert LH (2013) Dose–response relationships between physical activity, social participation, and health-related quality of life in colorectal cancer survivors. J Cancer Surviv 7:369–378
Vallance JK, Friedenreich CM, Lavallee CM, Culos-Reed N, Mackey JR, Walley B, Courneya KS (2016) Exploring the feasibility of a broad-reach physical activity behavior change intervention for women receiving chemotherapy for breast cancer: a randomized trial. Cancer Epidemiology Biomarkers and Prevention 25:391–398
Andresen EM, Malmgren JA, Carter WB, Patrick DL (1994) Screening for depression in well older adults: evaluation of a short form of the CES-D. American Journal of Preventive Medicine 10:77–84
Zhang W, O’Brien N, Forrest JI, Salters KA, Patterson TL, Montaner JS, Hogg RS, Lima VD (2012) Validating a shortened depression scale (10 item CES-D) among HIV-positive people in British Columbia. Canada PLoS One 7:e40793
Sabiston CM, Brunet J, Vallance JK, Meterissian S (2014) Prospective examination of objectively assessed physical activity and sedentary time after breast cancer treatment: sitting on the crest of the teachable moment. Cancer Epidemiol Biomark Prev 23:1324–1330
Troiano RP, Berrigan D, Dodd KW, Masse LC, Tilert T, McDowell M (2008) Physical activity in the United States measured by accelerometer. Med Sci Sports Exerc 40:181–188
Linden W, Vodermaier A, MacKenzie R, Greig D (2012) Anxiety and depression after cancer diagnosis: prevalence rates by cancer type, gender, and age. J Affect Disord 141:343–351
Jorm AF (2000) Does old age reduce the risk of anxiety and depression? A review of epidemiological studies across the adult life span. Psychological medicine 30:11–22
Campisi J (2013) Aging, cellular senescence, and cancer. Annu Rev Physiol 10:685–705
Ekkekakis P, Parfitt G, Petruzzello SJ (2011) The pleasure and displeasure people feel when they exercise at different intensities. Sports Med 41:641–671
Ekkekakis P, Hargreaves EA, Parfitt G (2013) Invited guest editorial: envisioning the next fifty years of research on the exercise–affect relationship. Psychol Sport Exerc 14:751–758
Segar ML, Richardson CR (2014) Prescribing pleasure and meaning: cultivating walking motivation and maintenance. American Journal of Preventitive Medicine 47:838–841
Acknowledgments
This research was supported by a Canadian Institutes of Health Research grant. Catherine Sabiston is supported by the Canada Research Chairs program. Benjamin Sylvester is supported by the Social Sciences and Humanities Research Council of Canada.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Sylvester, B.D., Ahmed, R., Amireault, S. et al. Changes in light-, moderate-, and vigorous-intensity physical activity and changes in depressive symptoms in breast cancer survivors: a prospective observational study. Support Care Cancer 25, 3305–3312 (2017). https://doi.org/10.1007/s00520-017-3745-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-017-3745-1