Abstract
Purpose
Hyponatremia is the most common electrolyte disorder in hospitalized patients, and it might be an indicator of poor prognosis and might have negative effects on hospitalization length and quality of life in non-malignant as well as in malignant diseases. The aim of this study is to determine the impact of hyponatremia on the length and on the cost of hospitalization as well as on outcome in cancer patients.
Methods
The present study includes 105 consecutive cancer patients hospitalized at our institution from June 2013 to December 2013. Data regarding age, sex, staging, histology, chemotherapy, and serum sodium levels at admission, during hospitalization, and at discharge were recorded and statistically analyzed. Impact of hyponatremia on length and cost of hospitalization and on outcome was evaluated.
Results
A significant difference in overall survival since the date of admission was observed between eunatremic and hyponatremic patients (p = 0.0255). A statistically significant correlation was also found between the length of stay and the detection of hyponatremia. At multivariate analysis, hyponatremia at admission, severity of hyponatremia, and stage of disease resulted independent prognostic factors. Furthermore, a patient with moderate or severe hyponatremia cost, in rate terms, 128 and 299 % more than a normonatremic patient, respectively.
Conclusions
The occurrence of hyponatremia at the admission or during the hospitalization may represent a significant factor influencing the outcome and the length of hospitalization. Acting effective and timely on the normalization of sodium levels might have a positive effect on prognosis in this setting of patients, as well as on the length of stay in hospital, thus potentially resulting in savings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hyponatremia is the most common electrolyte disorder in hospitalized patients, occurring in 5–30 % of patients [1, 2].
It is commonly defined as a serum sodium level of 135 mEq/l and it can be classified in three different levels: mild (130–134 mEq/l), moderate (125–129 mEq/l), and severe (<125 mEq/l) [3–6].
A variety of risk factors have been reported for hospital-acquired hyponatremia, including older age, diabetes mellitus, chronic kidney disease, surgery, pulmonary infection, diuretic therapy, administration of antibiotics or opioids, and use of hypotonic intravenous fluids [7, 8].
Hyponatremia is associated with a poor outcome in several medical conditions, such as liver cirrhosis, congestive heart failure, and infectious diseases [9–11].
It is also associated with serious complications that have been linked to increased morbidity and mortality [12–14].
Although some of the adverse outcomes associated with hyponatremia can be related by the underlying medical illness, it should be noticed that hyponatremia itself could be associated with adverse outcomes independently of the comorbidities [15].
Gill et al. reviewed that about 14 % of hyponatremia in inpatients was caused by underlying tumor-related conditions [16].
The incidence and prevalence of hyponatremia in cancer patients vary greatly, depending on the cancer type, clinical setting, and serum sodium cutoff point.
The main cause of hyponatremia in cancer patients is the syndrome of inappropriate antidiuretic hormone secretion (SIADH), which is due to ectopic production of antidiuretic hormone (AVP) by extracellular fluid depletion or to renal toxicity induced by chemotherapy, especially platinum-based [17, 18].
SIADH is most commonly found in patients with small cell lung cancer (SCLC; 11–15 %), but it has also been reported in 3 % of patients with head and neck cancer and in other solid tumors and hematological malignancies [19–21].
It is debatable whether the occurrence of hyponatremia may correlate with the stage and the anatomical spread [22–25].
Recently, Hansen and colleagues suggested that low sodium serum values could be associated with the number of metastatic sites and could be a measure of the tumor burden [26].
Recent studies suggested that hyponatremia might be an indicator of poor prognosis, and we previously demonstrated its impact on prognosis in mesothelioma patients [27].
Furthermore, it might have negative effects on hospitalization length and quality of life in non-malignant as well as in malignant diseases. The degree of hyponatremia and its normalization could modify the outcome of hyponatremic cancer patients [17, 28].
The purpose of this study was to evaluate the impact of hyponatremia on the length of stay, with a pharmaco-economical evaluation, and on the prognosis in cancer patients.
Patients and methods
Study population
The study population included all consecutive cancer patients aged 18 years or older who were hospitalized at our Institution from June 2013 to December 2013. We collected data regarding age, sex, staging, histology, chemotherapy, and levels of serum sodium at admission and at discharge.
Statistical analysis
Clinical data were retrospectively collected from medical chart reviews and electronic records. Overall survival (OS) was defined as the interval between the date of hospital admission to last follow-up or death. Survival distribution was estimated by the Kaplan-Meyer method, and differences in probability of surviving were evaluated by log-rank test.
A Cox regression model was applied to the data with a univariate approach and used to assess the prognostic role of the variables. All significance levels were set at a 0.05 value.
Chi-square test and Fisher’s exact probability test were used to evaluate association between categorical variables. Statistical analysis was performed with MedCalc software version 10.4.8 for Windows.
The cost of hospitalization was calculated by summing the direct medical costs, indirect costs, and common business folded [29], supplied by the administrative office of our institution. Quantification considered the costs of the year 2012 (2013 is not already available).
Results
One hundred five patients were included in the analysis. Male to female ratio was 57:48, and median age was 65.8 years (range 26–83 years).
The majority of patients had lung adenocarcinoma (28.6 %), 34 cases (32.4 %) had gastrointestinal cancer, and ten patients (9.5 %) had a diagnosis of sarcoma. The remaining patients examined were affected, in smaller percentages, by other solid tumors. In almost all cases, stage of disease was advanced or metastatic. Table 1 summarizes patients’ characteristics.
A total of 38 % of patients presented at least one episode of mild or severe hyponatremia during hospitalization. Twenty patients (19 %) showed mild or severe hyponatremia at admission, while 20 patients (19 %) developed mild or severe hyponatremia during hospitalization (Table 2).
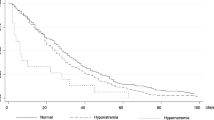
Median OS from the date of hospitalization was 50 days (range 3–282 days). A statistically significant difference in OS from the date of admission has been shown between eunatremic and hyponatremic patients (p = 0.0255) (Fig. 1). Furthermore, the presence of metastases was related to a worsened median OS (p = 0.0418).
A statistically significant correlation was found between the length of stay and the detection of hyponatremia (serum sodium <135 mEq/l) both at admission (p = 0.0009) (Fig. 2) and during hospitalization (p = 0.0001) (Fig. 3).
At multivariate analysis, including sodium level at admission, during hospitalization and at discharge, the length of stay and the presence of metastases, only hyponatremia at admission (hazard ratio (HR) = 2.64, 95 % confidence intervals = 1.72–4.35, p < 0.001), the severity of hyponatremia (HR = 1.35, 95 % confidence intervals (CI) = 1.04–1.88, p = 0.22), and the stage of disease (HR = 1.32, 95 % CI = 1.04–1.76, p = 0.23) resulted as independent factors.
We also conducted a drug economic analysis in order to understand the cost of the prolongation of hospitalization in patients with severe and moderate hyponatremia compared to patients with serum sodium level in normal range. A relevant problem of economic analysis of health interventions is the correct quantification of the costs [30].
The cost of hospitalization in our institute amounts to 381€/day per patient. The cost of hospitalization for a cancer patient, that was calculated to be 7 days on the average (corresponding to the national median length), amounted to €2,672. The cost for the days of hospitalization for a patient with moderate (131–135 mEq/l) and severe (<130 mEq/l) hyponatremia, calculated on the median of days of hospitalization (9 and 21, respectively), amounted to €3,435 in the first case and €8,015 in the second. Comparing the described costs, a patient with moderate or severe hyponatremia costs in rate terms 128 and 299 % more than a normonatremic patient, respectively. Comparing these data with the cost of hospitalization days for patients without hyponatremia in our sample, considering a median of 4 days (€1,527), the difference is 224 and 524 % more, respectively, in moderate and severe hyponatremic patient.
Discussion
Hyponatremia is recognized as the most common electrolyte disorder which occurs in hospitalized patients, and it is reported to be associated with higher morbidity and mortality [1, 2, 31].
Mild hyponatremia, which was thought to be relatively asymptomatic, also showed an excess of mortality compared with patients with a normal serum sodium level [15].
Serum sodium is a readily available, easily obtained, and routinely measured plasma electrolyte; however; hyponatremia is often under-diagnosed and untreated.
About 14 % of hyponatremia in inpatients is due to underlying tumor-related conditions [16]; therefore, it should be suspected and screened in all the patients who are diagnosed and are being treated for malignancies.
The systemic manifestations of many types of tumors and the toxicities of cancer therapy are involved in the pathogenesis of hyponatremia in cancer patients.
Although only few studies have evaluated the impact of hyponatremia on cancer patients outcome, literature data suggest that hyponatremia can be considered a negative prognostic factor in this setting of patients [17, 32].
Low serum sodium levels have recently been associated with poor OS in hepatocellular carcinoma [33], gastric cancer [34], mesothelioma [27], and small cell lung cancer [35].
In localized renal cell carcinoma, serum sodium level below median values has recently been associated with poor disease-free survival and OS after nephrectomy [36].
Cancer-related hyponatremia has been also hypothesized adversely affect the response to treatment [37, 38].
In a Belgian prospective study on hyponatremia in cancer patients, higher death rates were observed in hyponatremic patients compared with the entire examined cancer population. The authors reported that no death was apparently attributable to the electrolyte disorder itself but they suggested that hyponatremia might be a sign of general debility or of the gravity of underlying disease that increase the risk of death [17].
According to literature data, in our study, we observed a statistically significant difference in OS from the date of admission (p = 0.0255) between eunatremic and hyponatremic patients.
Furthermore, the presence of hyponatremia at admission, the severity of hyponatremia, and the stage resulted as independent prognostic factors (p = 0.0088).
Several studies analyzed the prognostic role of hyponatremia at hospital admission [31, 39].
Gill et al. recently found that severe hyponatremia was associated with a significantly higher mortality (27 vs. 9 %, p = 0.009) and a longer duration of hospitalization (16 vs. 13 days, p = 0.005) [16].
Our results showed a statistically significant correlation between the length of stay and the detection of hyponatremia (serum sodium <135 mEq/l) both at admission (p = 0.0009) and during hospitalization (p = 0.0001).
Our findings are consistent with those reported by Doshi and colleagues who, analyzing 3357 hospitalized cancer patients with cancer, showed a higher frequency of hyponatremia in this setting and a strong and independent association between mild and moderate hyponatremia and longer hospital stay (on average 4 to 8 additional days) with an increased 90-day mortality [39].
In a recent retrospective study conducted in a tertiary cancer center on 295 patients who underwent inpatient cancer rehabilitation, Nelson et al. found that a high percentage of patients have hyponatremia and it was associated with a prolonged rehabilitation length of stay (11 and 15 days for patients with mild and severe hyponatremia, respectively, vs. 10 days of eunatremic patients) [40].
There is a growing body of evidence that acting effectively and timely on the normalization of sodium levels could have a positive effect on prognosis of cancer patients.
The rate of serum sodium correction depends on the etiology, pathophysiology, acuity, severity, and clinical presentation of hyponatremia.
Hansen et al. in a retrospective study observed that SCLC patients who did not fully normalized serum sodium had a worse prognosis than hyponatremic patient who did. Moreover, they reported that also the baseline serum sodium levels were prognostic for outcome [26].
In a recent study, Petereit et al. reported that the median survival in lung cancer with hyponatremia was less than that in normonatremic patients and the correction of serum sodium above the level of 138 mEq/l was correlated with an improved survival [28].
In conclusions, our results seem to indicate that the occurrence of hyponatremia at the admission or during the hospitalization might represent a significant factor influencing the outcome. Moderate to severe hyponatremia in cancer patients delays cancer treatments and hospital discharge with a deep impact on the hospital bills.
References
Adrogué HJ, Madias NE (2000) Hyponatremia. N Engl J Med 342(21):1581–9
Upadhyay A, Jaber BL, Madias NE (2009) Epidemiology of hyponatremia. Semin Nephrol 29(3):227–38. doi:10.1016/j.semnephrol.2009.03.004
Patel GP, Balk RA (2007) Recognition and treatment of hyponatremia in acutely ill hospitalized patients. Clin Ther 29:211–229
Ghali JK (2008) Mechanisms, risks, and new treatment options for hyponatremia. Cardiology 111:147–157
Palmer BF, Gates JR, Lader M (2003) Causes and management of hyponatremia. Ann Pharmacother 37:1694–1702
Onitilo AA, Kio E, Doi SA (2007) Tumor-related hyponatremia. Clin Med Res 5:228–237
Beukhof CM, Hoorn EJ, Lindemans J, Zietse R (2007) Novel risk factors for hospital-acquired hyponatremia: a matched case–control study. Clin Endocrinol 66:367–72
Hoorn EJ, Lindemans J, Zietse R (2006) Development of severe hyponatraemiain hospitalized patients: treatment-related risk factors and inadequate management. Nephrol Dial Transplant 21:70–6
Luca A, Angermayr B, Bertolini G, Koenig F, Vizzini G, Ploner M, Peck- Radosavljevic M, Gridelli B, Bosch J (2007) An integrated MELD model including serum sodium and age improves the prediction of early mortality in patients with cirrhosis. Liver Transpl 13:1174–1180
Rossi J, Bayram M, Udelson JE, Lloyd-Jones D, Adams KF, Oconnor CM, Stough WG, Ouyang J, Shin DD, Orlandi C, Gheorghiade M (2007) Improvement in hyponatremia during hospitalization for worsening heart failure is associated with improved outcomes: insights from the acute and chronic therapeutic impact of a vasopressin antagonist in chronic heart failure (ACTIV in CHF) trial. Acute Card Care 9:82–86
Nair V, Niederman MS, Masani N, Fishbane S (2007) Hyponatremia in community-acquired pneumonia. Am J Nephrol 27:184–190
Bennani SL, Abouqal R, Zeggwagh AA et al (2003) Incidence, causes and prognostic factors of hyponatremia in intensive care. Rev Med Interne 24:224–9
Klein L, O’Connor CM, Leimberger CM et al (2005) Lower serum sodium is associated with increased short-term mortality in hospitalized patients with worsening heart failure: results from the outcomes of a prospective trial of intravenous milrinone for exacerbations of chronic heart failure (OPTIME-CHF) study. Circulation 111:2454–60
Biggins SW, Rodriguez HJ, Bacchetti P, Bass NM, Roberts JP, Terrault NA (2005) Serum sodium predicts mortality in patients listed for liver transplantation. Hepatology 41:32–9
Wald R, Jaber BL, Price LL, Upadhyay A, Madias NE (2010) Impact of hospital-associated hyponatremia on selected outcomes. Arch Intern Med 170(3):294–302
Gill G, Huda B, Boyd A, Skagen K, Wile D et al (2006) Characteristics and mortality of severe hyponatraemia—a hospital-based study. Clin Endocrinol (Oxf) 65:246–49
Berghmans T, Paesmans M, Body JJ (1999) A prospective study on hyponatraemia in medical cancer patients: epidemiology, aetiology and differential diagnosis. Support Care Cancer 8(3):192–7
Keenan AM (1999) Syndrome of inappropriate secretion of anti-diuretic hormone in malignancy. Semin Oncol Nurs 15:160–67
Sørensen JB, Andersen MK, Hansen HH (1995) Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) in malignant disease. J Intern Med 238:97–110
Talmi YP, Hoffman HT, McCabe BF (1992) Syndrome of inappropriate secretion of arginine vasopressin in patients with cancer of the head and neck. Ann Otol Rhinol Laryngol 101:946–949
Raftopoulos H (2007) Diagnosis and management of hyponatremia in cancer patients. Support Care Cancer 15:1341–1347
Onitilo AA, Ebenezer K, Suhail AR (2007) Tumour related hyponatraemia. Clin Med Res 5:228–23
Comis R, Miller M, Ginsberg S (1980) Abnormalities in water homeostasis in small cell anaplastic lung cancer. Cancer 45(9):2414–242
Kawahara M, Fukuoka M, Saijo N et al (1997) Prognostic factors and prognostic staging system for small cell lung cancer. Jpn J Clin Oncol 27:158–165
Østerlind K, Andersen PK (1986) Prognostic factors in small cell lung cancer: multivariate model based on 778 patients treated with chemotherapy with or without irradiation. Cancer Res 46:4189–4194
Hansen O, Sørensen P, Hansen KH (2010) The occurrence of hyponatremia in SCLC and the influence on prognosis: a retrospective study of 453 patients treated in a single institution in a 10-year period. Lung Cancer 68(1):111–4
Berardi R, Caramanti M, Fiordoliva I, et al. (2014) Hyponatremia is a predictor of clinical outcome for malignant pleural mesothelioma. Supp Care Cancer, Epub ahead of print
Petereit C, Zaba O, Teber I, Grohé C (2011) Is hyponatraemia a prognostic marker of survivalfor lung cancer? Pneumologie 65:565–71
Mantovani L. (2011) HEALTH TECHNOLOGY ASSESSMENT Principi, concetti, strumenti operativi. Il Sole 24 ore Spa ISBN: 978-88-324-7918-8 Chap. 3, Pag. 59
Drummond MF, O’Brien B, Stoddart GL, Torrance GW (1997) Methods for the economic evaluation of health care programmes. Oxford University Press, Oxford
Waikar SS, Mount DB, Curhan GC (2009) Mortality after hospitalization with mild, moderate, and severe hyponatremia. Am J Med 122(9):857–65
Castillo JJ, Vincent M, Justice E (2012) Diagnosis and management of hyponatremia in cancer patients. Oncologist 17(6):756–65
Huo TI, Lin HC, Hsia CY, Huang YH, Wu JC, Chiang JH, Chiou YY, Lui WY, Lee PC, Lee SD (2008) The MELD-Na is an independent short- and long-term prognostic predictor for hepatocellular carcinoma: a prospective survey. Dig Liver Dis 40:882–889
Kim HS, Yi SY, Jun HJ, Lee J, Park JO, Park YS, Jang J, Kim HJ, Ko Y, Lim HY, Kang WK (2007) Clinical outcome of gastric cancer patients with bone marrow metastases. Oncology 73:192–197
Gandhi L, Johnson BE (2006) Paraneoplastic syndromes associated with small cell lung cancer. J Natl Compr Canc Netw 4:631–638
Vasudev NS, Brown JE, Brown SR, Rafiq R, Morgan R, Patel PM, O’Donnell D, Harnden P, Rogers M, Cocks K, Anderson K, Paul A, Eardley I, Selby PJ, Banks RE (2008) Prognostic factors in renal cell carcinoma: association of preoperative sodium concentration with survival. Clin Cancer Res 14:1775–1781
Schutz FA, Xie W, Donskov F, Sircar M, McDermott DF, Rini BI, Agarwal N, Pal SK, Srinivas S, Kollmannsberger C, North SA, Wood LA, Vaishampayan U, Tan MH, Mackenzie MJ, Lee JL, Rha SY, Yuasa T, Heng DY, Choueiri TK (2013) The impact of low serum sodium on treatment outcome of targeted therapy in metastatic renal cell carcinoma: results from the international metastatic renal cell cancer database consortium. Eur Urol 65(4):723–30
Jeppesen AN, Jensen HK, Donskov F, Marcussen N, von der Maase H (2010) Hyponatremia as a prognostic and predictive factor in metastatic renal cell carcinoma. Br J Cancer 102(5):867–72
Doshi SM, Shah P, Lei X, Lahoti A, Salahudeen AK (2012) Hyponatremia in hospitalized cancer patients and its impact on clinical outcomes. Am J Kidney Dis 59(2):222–8
Nelson M, Palmer JL, Fu J, Williams JL, Yadav R, Guo Y (2014) Hyponatraemia in cancer patients on an inpatient rehabilitation unit. Eur J Cancer Care (Engl) 23(3):363–9
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sector.
Conflict of interest
All authors disclose no financial and personal relationships with other people or organizations that could inappropriately influence (bias) their work or that could be perceived as prejudicing the impartiality of the research reported. The authors have full control of all primary data and do agree to allow the journal to review their data if requested.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Berardi, R., Caramanti, M., Castagnani, M. et al. Hyponatremia is a predictor of hospital length and cost of stay and outcome in cancer patients. Support Care Cancer 23, 3095–3101 (2015). https://doi.org/10.1007/s00520-015-2683-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-015-2683-z