Abstract
The objective of this study was to determine the seasonal pattern of group A streptococcal pharyngitis in children attended at a hospital emergency department in the Mediterranean island of Mallorca (Spain), and its association with meteorologic factors and schooling. We conducted a retrospective review of the medical records of children aged 1–15 years with a diagnosis of Streptococcus pyogenes pharyngitis between January 2006 and December 2011. The number of S. pyogenes pharyngitis was correlated to temperature, humidity, rainfall, atmospheric pressure, wind speed, solar radiation, and schooling, using regression and time series techniques. A total of 906 patients (median, 4 years old) with S. pyogenes pharyngitis, confirmed by throat culture, were attended during the study period. A seasonal pattern was observed with a peak activity in June and a minimum in September. Mean temperature, solar radiation, and school holidays were the best predicting variables (R 2 = 0.68; p < 0.001). S. pyogenes activity increased with the decrease of mean temperature (z = −2.4; p < 0.05), the increase of solar radiation (z = 4.2; p < 0.001), and/or the decrease in school holidays (z = −2.4; p < 0.05). In conclusion, S. pyogenes pharyngitis had a clear seasonality predominating in springtime, and an association with mean temperature, solar radiation, and schooling was observed. The resulting model predicted 68 % of S. pyogenes pharyngitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Group A streptococcus (GAS) is the most frequent cause of acute bacterial pharyngitis in children 5–15 years of age. In general, 15 to 30 % of all cases of acute pharyngitis in this age group and 10 % in adults are usually caused by this microorganism. In Spain, no data on the global incidence of GAS pharyngitis has been reported but there are approximately four million of pediatric consultations for acute pharyngitis annually in the primary care centers with a prevalence of GAS pharyngitis from 10 to 45 % (Giraldez-Garcia et al. 2011). Attempts to measure the global burden of GAS diseases, including GAS pharyngitis, are difficult by a lack of reliable information. In fact, the only GAS disease for which global disease burden estimates has been made is rheumatic heart disease. Population-based studies documenting the incidence of GAS pharyngitis are very scarce (Carapetis et al. 2005). In New Zealand, Kuwait, and northern India, sore throat with a positive throat swab for GAS occurred once every 1–2 years in each child. In Melbourne (Australia), an incidence of serologically proven GAS pharyngitis of 0.14 cases per child-year has been reported (Danchin et al. 2004). As a matter of fact, interest in the epidemiology of throat infections caused by Streptococcus pyogenes decreased with the decline of its complications, particularly acute rheumatic fever. Nonetheless, the resurgence of suppurative and nonsuppurative sequelae of S. pyogenes infections during the 1980s and 1990s generated a new interest in its epidemiology (Givner et al. 1991; Kaplan et al. 2001; Lamagni et al. 2008; Danchin et al. 2007).
The classic studies of S. pyogenes pharyngitis suggested a role of cold weather and crowding as important factors in the development and spread of this infection. In general, a higher incidence of streptococcal sore throat during the late winter and early spring was reported in these studies (Ross et al. 1971). More recently, reports from different parts of the world have added new information on seasonality. In the northern hemisphere, a report from a cold area of the USA (Minneapolis) in 2001 showed a bimodal pattern with peaks of GAS pharyngitis in autumn (October) and winter (January) (Kaplan et al. 2001). In the UK, a marked seasonal pattern in severe GAS disease was described in the period 2003–2004, with an initial peak in early winter (January) and a strong peak in early spring (March) (Lamagni et al. 2008). In the southern hemisphere, peaks in spring (November) and autumn (June) were described in 2005 from a cold area of Argentina (Rubinstein et al. 2005). In 2007, a study from Australia showed a bimodal seasonal pattern of S. pyogenes pharyngitis with peaks in winter/spring and autumn (Danchin et al. 2007).
The seasonality of GAS infections, which is variable according to different geographic areas, suggests the influence of meteorologic factors. However, there are other factors involved in the development of GAS pharyngitis. The influence of crowding and season on the occurrence of this infection has been well studied, but reports on the influence of meteorologic conditions are lacking.
School environment is known to foster the transmission of infectious diseases as a result of the close contact of children and sharing of supplies and equipment. The increase of respiratory and gastrointestinal infections is well documented especially in preschool infants attending day care centers (Barros et al. 1999). The spread of viral infections such as measles, chickenpox, or influenza in schools are examples of the effect of schooling in the transmission of infectious diseases. School closure, on the other hand, has been demonstrated to decrease respiratory morbidity during an influenza outbreak in Israel (Heymann et al. in 2004). Likewise, an inverse association was observed between the number of varicella cases and school holidays in Mallorca (Spain) (Hervás et al. 2015). However, the impact of schooling on bacterial infections is less known, and the association between GAS pharyngitis and schooling has not been evaluated.
The aim of the present study was to determine the seasonal pattern of group A streptococcal (GAS) pharyngitis in a Mediterranean region of Spain, and the possible influence of some meteorologic factors and schooling in this pattern. The meteorologic conditions are closely associated variables and analysis of a small number of these factors cannot exclude false associations or establish the degree of association between the variables involved. Therefore, we studied the effects of a large number of meteorologic factors (temperature, humidity, rainfall, atmospheric pressure, wind speed, and solar radiation). To our knowledge, this is the first study applying multiple regression and time series techniques for the analysis of the effects of weather conditions and schooling on the development of GAS pharyngitis.
Materials and methods
The study was conducted on the island of Mallorca (Spain), a temperate Mediterranean region with mild winters (mean temperature, 10.2 °C), warm summers (mean temperature, 24.3 ° C), and moderate rainfalls (51 days/year, annual mean precipitation of 421 mm). To measure the activity of GAS, we used the number of GAS-positive throat cultures in children attended at the pediatric emergency department of Son Espases University Hospital at Palma de Mallorca. From January 1, 2006, to December 31, 2011, children aged 1–15 years attended with a diagnosis of pharyngitis and clinical findings suggestive of GAS, routinely had pharyngeal swabs taken for culture in blood agar plates.
The hospital where this study was performed is a tertiary care center of the public network, attending to a population of 399,093 inhabitants (60,659 < 15 years of age). The public health sector covers 100 % of the population, and there is no evidence of health disparities in the region. A map illustrating the study area (hospital’s catchment area) as well as the locations of the hospital and the meteorologic station is included (Fig. 1). The district colored in dark gray concentrates over 80 % of the population covered by the hospital.
Meteorologic data
During the study period, data on temperature (°C), humidity (%), rainfall (mm), atmospheric pressure (hPa), wind speed (km/h), and solar radiation (10 kJ/m2) were obtained from the Spanish Agency of Meteorology. This agency collects daily information from a weather station located at latitude 39.55° N, longitude 2.73° E and 4 m height, at 15 km from the hospital. The population studied lives within a radius of 30 km around the weather station, and exposure is very unlikely to have taken place somewhere else. Therefore, the meteorologic measurements were obtained very close to the area where the patients became infected.
Data analyses
We obtained daily values of meteorologic variables and GAS pharyngitis cases. For analysis, we used their weekly means (using 52 observations for analyses). In other words, data were first aggregated in weeks and after that, a mean was obtained for the corresponding weeks of the different years (e.g., week 1 corresponds to the mean of the first week of years 2006 through 2011). Continuous variables are expressed as mean ± standard deviation (SD) or median and interquartile (IQR) as required. Regression and time series techniques were used to model the relationship between the number of GAS pharyngitis cases and the meteorologic factors. Analysis was started with normal distribution analyses, variable log transformation, simple and multivariate regression, partial regression calculation, and collinearity tolerance testing. The regression model was analyzed for autocorrelation using Durbin Watson tests. Autocorrelation involves the existence of correlation between a variable (e.g., number of GAS cases) and the same variable at particular lag periods. If autocorrelation was observed or could not be excluded, an autoregressive moving average (ARIMA) model was built. When ARIMA is built using exogenous variables, the model is known as ARMAX (Diggle 1990). For this purpose, correlograms, partial autocorrelation, and autocorrelation graphs were used to determine the autoregressive and moving average order. Variable selection was obtained by a forward adjustment of the meteorologic factors. The final model was considered adequate if portmanteau Q test for residuals resembled white noise, and residuals were normally distributed and showed no systematic trends. To analyze the influence of schooling on GAS pharyngitis cases, the variable school holiday during the week was introduced in the model. All statistical analyses were performed using STATA 12.1 (TX, USA). Statistical significance was set at p < 0.05.
Results
General epidemiology
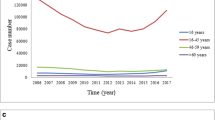
For the study, we used the number of cases of pharyngitis with positive throat culture for GAS. From January 1, 2006, to December 31, 2011, there were a total of 906 patients with GAS-positive pharyngitis. Figure 2 shows the total number of cases during the 6 years of study. The median age was 4 years (IQR 2–7 years), and there was a male predominance (498/906, 55 %). A seasonal pattern was observed with a peak activity during June and a minimum activity in September (Fig. 3). Table 1 shows the mean value of the weekly meteorologic factors analyzed during the whole study period.
Bivariate and multivariate analyses
The weekly average number of GAS pharyngitis cases presented a normal distribution after logarithmic transformation (log). Simple correlations between the number of logGAS pharyngitis cases and the studied factors were evaluated for different lag periods (Table 2). Multivariate analyses showed that solar radiation, temperature and school holidays were independently associated with the logGAS activity (Table 3). The increase in solar radiation, the decrease in mean temperature, and the absence of school holidays could explain 37, 17, and 14 % of GAS activity, respectively (Table 4). Autocorrelation could not be excluded from the model after analysis with Durbin-Watson test (using 4 predictors and 52 observations, d = 1.48). As the accuracy of the regression model could not be confirmed, the analysis was continued with time series techniques.
Time series analyses
The autocorrelation and partial autocorrelation graphs used for ARMAX model fitting are shown in Fig. 4. In the ARMAX model, a third-order moving average showed the best fit for GAS activity. Using a forward adjustment, mean temperature and 1-week lag solar radiation were the best predicting meteorologic variables (see Table 5). To show this association, mean temperature and solar radiation are plotted together with GAS pharyngitis cases (see Fig. 3). GAS activity increased with the decrease of mean temperature (z = −2.4; p < 0.05) and/or the increase of solar radiation (z = 4.2; p < 0.001). A school holiday period 2 weeks earlier, also diminished the number of GAS pharyngitis cases (z = −2.4; p < 0.05). Overall, the resulting model (with schooling, mean temperature, and solar radiation) predicted 68 % (p < 0.001) of GAS activity (see Fig. 5), and residuals showed no nonrandom noise, were normally distributed, and had no systematic trends. When schooling was not considered, the model with these two meteorologic factors predicted 59 % of GAS activity.
Discussion
In this study, carried out in a temperate Mediterranean region, we have observed a definite seasonal pattern of GAS-positive pharyngitis in children aged 1–15 years. GAS pharyngitis was observed in similar rates from October to February, rising significantly in the spring period, starting in March and peaking in June. During the summertime, the cases of GAS pharyngitis declined, reaching a minimum in September. This springtime preferential seasonality is different to that of the winter and early spring months in the classic reports of GAS sore throat (Ross et al. 1971).
GAS is spread by direct person-to-person contact, most likely via droplets of saliva or nasal secretions. Crowding such as occurs in households, schools, or military facilities favor an interpersonal spread of the microorganisms. A decrease of temperature and more indoor activities during the winter could explain GAS pharyngitis in this season. However, a study by Krause et al. (1962) could not demonstrate that the change in climate, from a cold area of the USA (Wyoming) to a warm one (Florida), had any effect on the length of the carrier state or the numbers of organisms isolated from the pharynx of military personnel. In our study, the paradoxical predominance of GAS pharyngitis during the springtime could indicate the existence of other factors in the development of the disease.
Experimental studies have demonstrated the influence of environmental temperature upon capsule production in GAS, increasing at sub-body temperature and decreasing when temperature increases (Kang et al. 2012). The GAS capsule is an adhesin involved in initial colonization at the lower temperatures typical of the mucosal surfaces of the throat and skin and serves as an antiphagocytic factor for tissue invasion (Wessels et al. 1994). In addition, a global differential gene expression in response to growth temperature alteration ranging from 29 to 37 °C has been reported in GAS (Smoot et al. 2001). These studies demonstrate changes in various GAS virulence factors at different temperatures and support the possibility of peaks of GAS pharyngitis in the springtime, as in our study, or even in the summertime as reported from India (Nandi et al. 2001).
We observed a direct correlation between solar radiation a week earlier and GAS activity. Clearly, ultraviolet (UV) light has a bactericidal effect but its indirect effects on vitamin D production and/or the host defenses are not fully understood. Solar radiation has been linked to the immune system, and immunostimulant or immunosuppressive effects have been suggested. Sufficient data have been generated from the animal models that indicate a suppression of viral immune responses induced by sunlight exposure (Norval et al. 2006; Norval et al. 1999). Nevertheless, the possibility of immunosuppressive effects of UV radiation in human bacterial infections is not known. One of the major effects of UV radiation is the synthesis of vitamin D in the skin, an immunomodulator with immunostimulant and immunosuppresive actions. Vitamin D insufficiency has been related to the development of infections by Mycobacterium tuberculosis, and also to recurrent GAS tonsillitis in adults (Ralph et al. 2013; Nseir et al. 2012). Experimental studies suggest that vitamin D plays a role in the production of cathelicidin and defensins, which are involved in the natural defense against upper respiratory infections. However, suppression of the adaptive immune system and the inflammatory action of the nuclear factor-kB pathway caused by vitamin D could prove detrimental for some infections (Gombart et al. 2009). Recently, Love et al. (2012) has found that vitamin D has the paradoxical effect of increasing GAS resistance to macrophage-mediated killing, i.e., a detrimental effect on the host. Therefore, the role of UV radiation and vitamin D in GAS infections has not been established. The direct association between GAS pharyngitis cases and solar radiation observed in our study suggests the possibility of immunosuppresive effects of UV radiation and/or vitamin D in this infection.
We hypothesize that the seasonality of GAS sore throat is a consequence of different processes. Firstly, GAS colonization must occur, which can be favored by environmental or behavioral factors that increase the contact of the individuals with this microorganism. The school period is a time of increased contact among children that favors transmission of GAS. The literature usually assumes that incubation period for GAS pharyngitis is 2 to 5 days. However, the pathogenic events from colonization to development of pharyngitis are not fully understood (Martin et al. 2004; Martin 2010). During an outbreak of GAS pharyngitis in a Swedish day care center, Falck et al. (1992) observed that GAS infection and carriage occurred in 61 % of the children within 16 days of identification of the index case. In our study, a school holiday period 2 weeks earlier (8–14 days) resulted in a decrease of GAS pharyngitis cases. Considering that schooling may have an impact on GAS colonization, our study suggests that colonization 2 weeks earlier could be a critical period for infection if some conditions occur. In other words, these results suggest the possibility of a longer time period from colonization to the development of GAS pharyngitis. According to our findings, once GAS colonization occurs, an increase in solar radiation and/or vitamin D production during the previous week could cause an increase in the host immune susceptibility to GAS infection. In this situation, the increase in GAS virulence when mean temperature lowers could eventually lead to clinical pharyngitis.
The development of meteorologic models to predict viral infections have been widely used. In viral respiratory infections, these models use to give high predictive rates, as we have reported for RSV (88 %) (Hervás et al. 2012). However, there is a lack of studies on the association of bacterial infections and meteorologic factors. In a study with pneumococcus, an inverse association of invasive pneumococcal disease with ambient temperature yielded correlation (r) values, with no time lag, of 0.71 and 0.27 in adults and children, respectively (Kim et al. 1996). In the case of GAS pharyngitis, there are no previous data about the influence of meteorologic factors on the development of the disease. In our study, we have tested most of the meteorologic factors but only temperature and solar radiation correlated with GAS activity. On the contrary, atmospheric pressure, humidity, rainfall, and wind speed did not show any correlation. Some of these meteorologic factors seem to play a role in the transmission of certain viruses like RSV and influenza (Hervás et al. 2012; Shaman et al. 2009), but their impact on the transmission of bacterial infections has not been demonstrated (Tang et al. 2009). Our results suggest that the prediction rates of the models with bacterial infections are lower than with viral infections. Bacterial transmission most probably requires a closer contact from person to person, and the persistence of bacteria in the environment as bioaerosols may be less durable or determinant than for viruses.
Studying the linkage between climate and disease may yield insights into the factors that drive the emergence and seasonal variations in epidemic diseases. However, we are aware of the limited value of this kind of approach, which cannot fully account for the complex web of causation that underlies disease dynamics. Infectious diseases are affected by many factors other than climate, and the relationship between climatic variations and disease outbreaks is often mediated by ecological, biological, or societal changes. One of the biases of our study was the patient’s age since we only studied the pediatric population and cannot exclude different patterns in adults. Another limitation would be the reliability of the number of GAS phayngitis cases. Although we isolated GAS from throat cultures of all children diagnosed with acute pharyngitis, this isolation did not confirm causality and this GAS count may include both infections and colonizations. To establish causality, it is theoretically necessary to demonstrate the host’s response to the infection in the form of antibody production (serologic evidence) that takes 2 or 3 weeks.
In conclusion, the combination of low mean temperature with high solar radiation seems to play a role in the dynamics and seasonality of GAS pharyngitis. In order to confirm the importance of these factors, we need other studies from different geographic areas that evaluate the effects of meteorologic factors on the development of this infection. In addition, further experimental studies of the effects of temperature and solar radiation on the bacteria and human innate immunity would be useful to understand the involvement of these meteorologic factors in the development of GAS pharyngitis.
In this study, we have also seen that school vacation decreased the number of GAS sore throat infections, demonstrating the role of schooling in the dynamics of the disease. Overall, the present study contributes to a better understanding of weather-related factors and schooling in the development of GAS pharyngitis in children, with the help of multiple regression and time series methods.
References
Barros AJD, Ross DA, Fonseca WVC, Williams LA, Moreira-Filho DC (1999) Preventing acute respiratory infections and diarrhoea in child care centres. Acta Paediatr 88:1113–1118
Carapetis JR, Steer AC, Mulholland EK, Weber M (2005) The global burden of group A streptococcal diseases. Lancet Infect Dis 5:685–694
Danchin MH, Rogers S, Selvaraj G, Kelpie L, Rankin P, Vorich R, Howson M, Carlin JB, Curtis N, Nolan TM, Carapetis JR (2004) The burden of group A streptococcal pharyngitis in Melbourne families. Indian J Med Res 119(Suppl):144–147
Danchin MH, Rogers S, Kelpie L, Selvaraj G, Curtis N, Carlin JB, Nolan TM, Carapetis JR (2007) Burden of acute sore throat and Group A streptococcal pharyngitis in school-aged children and their families in Australia. Pediatrics 120:950–957
Diggle PJ (1990) Time series: a biostatistical introduction (Oxford Statistical Science Series-5). Clarendon Press, Oxford
Falck G, Kjellander J (1992) Outbreak of Group A streptococcal infection in a day-care center. Pediatr Infect Dis J 11:914–919
Giraldez-García C, Rubio B, Gallegos-Braun JF, Imaz I, González-Enriquez J, Sarría-Santamera A (2011) Diagnosis and management of acute pharyngitis in a paediatric population: a cost-effectiveness analysis. Eur J Pediatr 170:1059–1067
Givner LB, Abramson JS, Wasilauskas B (1991) Apparent increase in the incidence of invasive Group A beta-hemolytic streptococcal disease in children. J Pediatr 118:341–346
Gombart AF (2009) The vitamin D-antimicrobial peptide pathway and its role in protection against infection. Future Microbiol 4:1151–1165
Hervás D, Reina J, Hervás JA (2012) Meteorologic conditions and respiratory syncitial virus activity. Pediatr Infect Dis 31(10):e176–81. doi:10.1097/INF.0b013e31825cef14
Hervás D, Hervás-Masip J, Nicolau A, Reina J, Hervás JA (2015) Solar radiation and water vapor pressure to forecast chickenpox epidemics. Eur J Clin Microbiol Infect Dis 34(3):439–46. doi:10.1007/s10096-014-2243-3
Heymann A, Chodick G, Reichman B, Kokia E, Laufer J (2004) Influence of school closure on the incidence of viral respiratory diseases among children and on health care utilization. Pediatr Infect Dis J 23:675–677
Kang SO, Wright JO, Tesorero RA, Lee H, Beall B, Cho KH (2012) Thermoregulation of capsule production by Streptococcus pyogenes. PLoS One 7(5), e37367. doi:10.1371/journal.pone.0037367
Kaplan EL, Wotton JT, Johnson DR (2001) Dynamic epidemiology of group A streptococcal serotypes associated with pharyngitis. Lancet 358:1334–1337
Kim PE, Musher DM, Glezen WP, Rodriguez-Barradas MC, Nahm WK, Wright CE (1996) Association of invasive pneumococcal disease with season, atmospheric conditions, air pollution, and the isolation of respiratory viruses. Clin infect Dis 22:100–106
Krause RM, Rammelkamp CH, Denny FW, Wannamaker LW (1962) Studies of the carrier state following infection with group A streptococci. I. Effect of climate. J Clin Invest 41:575–578
Lamagni TL, Neal S, Keshishian C, Alhaddad N, George R, Duckworth G, Vuopio-Varkila J, Efstratiou A (2008) Severe Streptococcus pyogenes infections, United Kingdom, 2003-2004. Emerg Infect Dis 14:201–209
Love JF, Tran-Winkler HJ, Wessels MR (2012) Vitamin D and the human antimicrobial peptide LL-37 enhance group A streptococcus resistance to killing by human cells. M Bio. 23; 3(5). doi: 10.1128/mBio.00394-12
Martin JM (2010) Pharyngitis and streptococcal throat infections. Pediatr Ann 39:22–27
Martin JM, Green M, Barbadora KA, Wald ER (2004) Group A streptococci among school-aged children: clinical characteristics and the carrier state. Pediatrics 114:1212–1219
Nandi S, Kumar R, Ray P, Vohra H, Ganguly NK (2001) Group A streptococcal sore throat in a periurban population of northern India: a one-year prospective study. Bull World Health Organ 79:528–533
Norval M (2006) The effect of ultraviolet radiation on human viral infections. Photochem Photobiol 82:1495–1504
Norval M, Garssen J, Loveren HV, El-Ghorr AA (1999) UV-induced changes in the immune response to microbial infections in human subjects and animal models. J Epidemiol 9:S84–S92
Nseir W, Mograbi J, Abu-Rahmeh Z, Mahamid M, Abu-Elheja O, Shalata A (2012) The association between vitamin D levels and recurrent group A streptococcal tonsillopharyngitis in adults. Int J Infect Dis 16(10):e735–8. doi:10.1016/j.ijid.2012.05.1036
Ralph AP, Lucas RM, Norval M (2013) Vitamin D and solar ultraviolet radiation in the risk and treatment of tuberculosis. Lancet Infect Dis 13:77–88
Ross PW, Chisty SMK, Knox JDE (1971) Sore throat in children: its causation and incidence. BMJ 2:624–626
Rubinstein G, Bavdaz B, De Bunder S, Blazquez N (2005) Incidencia de faringitis por Streptococcus pyogenes en Bariloche, Argentina. Rev Argent Microbiol 37:84–86
Shaman J, Kohn M (2009) Absolute humidity modulates influenza survival, transmission and seasonality. PNAS 106:3243–3248
Smoot LM, Smoot JC, Graham MR, Somerville GA, Sturdevant DE, Lux-Migliaccio CA, Sylva GL, Musser JM (2001) Global differential gene expression in response to growth temperature alteration in group A Streptococcus. PNAS 98:10416–10421
Tang JW (2009) The effect of environmental parameters on the survival of airborne infectious agents. J R Soc Interface 6(6):737–46. doi:10.1098/rsif.2009.0227.focus
Wessels MR, Bronze MS (1994) Critical role of the group A streptococcal capsule in pharyngeal colonization and infection in mice. Proc Natl Acad Sci USA 91:12238–12242
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hervás, D., Hervás-Masip, J., Ferrés, L. et al. Effects of meteorologic factors and schooling on the seasonality of group A streptococcal pharyngitis. Int J Biometeorol 60, 763–769 (2016). https://doi.org/10.1007/s00484-015-1072-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-015-1072-0