Abstract
Key message
Leaf shape in European beech provenances varies geographically, with narrower and longer lamina observed in southern provenances, indicating a direct selection favoring leaf shape that likely safeguards trees performances under less favorable growing conditions.
Abstract
Spatial and environmental patterns of European beech (Fagus sylvatica L.) leaf size and shape variations were studied using landmark-based geometric morphometrics. The study involved eight provenances originating from three biogeographic regions (i.e., Alpine, Continental, and Pannonian), spanning across six European countries (Bosnia and Herzegovina, Croatia, Germany, Hungary, Romania and Serbia). All specimens were cultivated in a common garden experiment. The symmetric component of leaf shape variation was analyzed by Procrustes ANOVA and multi‐variate analyses [principal component analysis and canonical variate analysis (CVA)], whereas MANOVA was used to examine asymmetry. Partial least square (PLS) analysis was used to assess the covariation between leaf shape and size, and geographical position and environmental variables at the sites of provenance origin, respectively. A highly observed phenotypic variation for the shape and size of leaf both within and among provenances, indicates a strong local adaptation of provenances within the species natural range. CVA revealed the existence of two clusters of provenances based on the leaf shape, i.e., the first group included provenances originating from Balkan Peninsula and Central Europe, while the second group consisted of two Romanian provenances and the northernmost provenance from Germany. Likewise, PLS evidenced that leaf shape was spatially structured along latitudinal (− 0.64) and longitudinal (0.60) gradients, with southern provenances having longer and narrower (ovate) lamina compared to the northern ones. In contrast, no correlation was found between centroid size and spatial and environmental variables. Results suggest the presence of direct selection favoring a leaf shape that likely safeguards trees from heat and water loses under less favorable growing conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The European beech (Fagus sylvatica L.) is one of the major broadleaved tree species in Europe, covering approximately 14 million hectares of forested land (Gavranović et al. 2018). According to its wide ecological amplitude, beech successfully adapted to divergent selective pressures, which, in turn, favored selection of ecotypes capable to survive and reproduce across a range of environmental conditions (Wortemann et al. 2011; Funk et al. 2017). As a consequence of evolutionary adaptation, an intraspecific phenotypic traits variation has occurred in different provenances, having a central role in shaping plant functioning (Hajek et al. 2016; Kurjak et al. 2019). Hence, great attention has been paid to the natural variations in plant phenotypic traits and their ecological and evolutionary relevance (Wellstein et al. 2013; Adler et al. 2014; Duruflé et al. 2019).
Leaves are trees’ primary interface with the atmosphere and affect a range of ecological processes (Bussotti and Pollastrini 2015). As the result of local adaptations, trees have evolved various physiological and morphological adaptation mechanisms, primarily related to light interception and radiative exchange (Scartazza et al. 2016; Pšidová et al. 2018), as well as temperature regulation and heat dissipation (Tognetti et al. 1995; Guerin et al. 2012; Albrecht et al. 2020). For example, Robson et al. (2012) reported that beech provenances originating from southern Europe exhibited higher carbon assimilation rate under drought conditions in comparison to provenances from central and northern parts, which showed severely reduced photosynthetic capacity. In addition, leaf morphology has been recognized as one of the key components of plant species adaptation to environmental conditions, influencing plant growth and productivity (Wang and Zhang 2012; Ren et al. 2020). Several studies have shown that spatial variation of leaf traits, including leaf size and shape are significantly related to climate (Peppe et al. 2011; Miljković et al. 2019; Jahdi et al. 2020), suggesting strong local adaptation.
Provenance trials may provide useful information about stress tolerance and adaptation potentials of tree species to variation in climate (Pauls et al. 2013), thereby presenting a valuable decision tool in the programs aimed at conservation of genetic resources, species genetic improvement and selection of suitable reproductive material. Indeed, a number of researchers reported that different European beech provenances have shown variable performances when subjected to stress factors in manipulative experiments, such as water shortage (e.g., Thiel et al. 2014; Cocozza et al. 2016), freezing temperature (e.g., Borghetti et al. 1993), or drought stress at different irradiances (e.g., Tognetti et al. 1994; Wang et al. 2021).
Although the variability of leaf morphology has been extensively studied in European beech (Šijačić-Nikolić et al. 2013; Uhl, 2014; Stojnić et al. 2016; Kempf et al. 2018), to the best of our knowledge, this paper is the first to study beech provenances leaf form (i.e., size and shape) using geometric morphometric (GM) methods. Namely, previous studies have relied on standard (so called “traditional”) linear measurements of leaf character lengths, widths and ratios, thus containing limited information about the geometry of the object (Breno et al. 2011). Another disadvantage of “traditional” morphometrics lies in the fact that although measured traits are considered independent of each other, certain measurements frequently originate from a single point, thus showing multi co-linearity (Zelditch et al. 2004; Cooke and Terhune 2015). Finally, the influence of leaf size on the studied attributes, makes it difficult to distinguish environmental effects from genotypic differences, especially in the case of in situ populations. For these reasons, a number of authors believe that “traditional” measurements do not provide enough power to discriminate closely related entities (Maderbacher et al. 2008; Abdel-Rahman et al. 2009; Schmieder et al. 2015). In contrast to linear measurements, GM uses the landmark configuration to describe and visualize the shape of the object, while retaining its original geometry (Watanabe 2018). Landmarks have been seen as anatomically recognizable loci which are the same for all specimens in the study (Zelditch et al. 2004). In general, three main types of landmarks have been recognized depending on whether they clearly define the biological structure (Type I), describe maxima (or minima) of curvature (Type II) or correspond to the geometric constructions (e.g., endpoints of the longest diameter) and the point furthest away from another landmark (Type III) (see Savriama 2018; Palci and Lee 2019). On the other side, semi-landmarks represent the points arrayed along an outline with the aim to capture the information on geometry of curves and surfaces (Bookstein 1997). Thus, using sliding semi-landmarks enables quantification of curved outlines, enhancing overall estimation of organism shape (Gunz and Mitteroecker 2013). For example, combining landmark and semi-landmark configurations, Stefanović et al. (2017) investigated differences among Taxus baccata L. populations concerning the shape of needles, and presence of sexual dimorphism in each of them. Lastly, as the object shape, which is defined by landmark configuration, is “size-free”, leaf size and shape variations can be analyzed separately, as well as the factors influencing this variation along environmental or spatial gradients (Maestri et al. 2018). Therefore, GM appeared highly effective in analyzing intra- and interspecific leaf shape variability and discrimination of tree species and populations (Viscosi et al. 2009; Viscosi 2015; Miljković and Čortan 2020), as well as in evaluation of subtle shape changes on the intraspecific level influenced by environmental factors (Kubínová et al. 2018).
The GM techniques have been used to assess the patterns of leaf size and shape variations in eight European beech provenances occurring across different biogeographic regions, as well as to evaluate the association between the pattern of leaf size and shape variation and a set of spatial and environmental variables. It was specifically hypothesized that: (1) significant differences in leaf shape and size will be observed both at intra- and inter-provenance levels, supporting previous findings reporting a high genetic variability in European beech provenances (Stojnić et al. 2015; Müller et al. 2018; Vastag et al. 2019), (2) morphological adjustments in leaf shape and size will occur along spatial and environmental gradient as a result of provenances local adaptation to different selective pressures.
Materials and methods
Experimental setup and European beech provenances
European beech provenance trial in Croatia was established in 2007 at the Medvednica Mountain (N 45° 52′, E 15° 55′; 450 m a.s.l.), as the part of pan-European network of beech provenances trials that are founded in the framework of the COST Action E52—Evaluation of the Genetic Resources of Beech for Sustainable Forestry. The climate of the Medvednica Mountain is temperate continental. Mean annual temperature is approximately 8.9 °C, while total annual precipitation amounts to 1100 mm. The active growth period (April–September), has a mean air temperature of 14.8 °C, and a precipitation amount of 615 mm.
The provenance trial was established of 2- and 3-year-old seedlings produced in the nursery of Thünen-Institute for Forest Genetics (Grosshansdorf, Germany), distributed to several project partner institutions. The experiment is arranged in a randomized complete block design, with three replications. Each block consists of 50 seedlings planted in 1 × 2 m spacing. To avoid edge effects, two border rows with local provenances were planted around the trial (Bogunović et al. 2020).
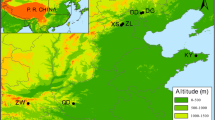
The study involved eight provenances from six European countries (Bosnia and Herzegovina, Croatia, Germany, Hungary, Romania and Serbia), with distribution in the Alpine, Continental, and Pannonian biogeographic regions (Table 1). The provenances were selected to cover a gradient of climate and environmental conditions from the northern region of the beech distribution range (i.e., North Germany), across its central parts (i.e., Central Europe) to the Balkan Peninsula and Romania at the south-eastern region of the species natural area (Fig. 1). The information about climate at the sites of provenances origin and the trial locality were obtained from WorldClim 2 database for the period between 1970 and 2000 (Fick and Hijmans 2017), whereas altitudes were derived from Shuttle Radar Topography Mission (SRTM) digital elevation dataset (Van Zyl 2001).
Distribution of European beech provenances involved in the study. The provenances are abbreviated (filled circle) and the trial site ME, (concentric circles) as shown in Table 1. The blue area represents natural distribution range of beech (after von Wuehlisch 2008). ME—provenance trial site Medvednica (Croatia); HR25—Vrani Kamen (Croatia); BA30—Tajan (Bosnia and Herzegovina); HU42—Valkonya (Hungary); DE48—Höllerbach (Germany); DE49—Hasbruch (Germany); RO63—Alesd (Romania); RO64—Alba-Iulia (Romania); RS66—Avala (Serbia)
To evaluate suitability of provenances origin and provenance trial sites for beech production, the Forest Aridity Index (FAI) was calculated according to the formulae:
where TVII and TVIII represent mean temperature in July and August, respectively, and PV, PVI, PVII and PVIII the sum of precipitation for May, June, July and August, respectively (Gavrilov et al. 2019). The FAI was also used to examine the covariation between leaf size and shape and environmental conditions of the provenance’s origins.
Leaf measurements
Leaf material was collected at the end of July 2016. Twenty leaves were collected from ten trees within each provenance. Fully sun exposed leaves were sampled from the upper third part of the crown using telescope scissors. Only fully developed and healthy leaves were considered for sampling. After sampling, the leaves were herbarized (i.e., the leaves were arranged, labeled and spread flat between thin sheets of paper and dried).
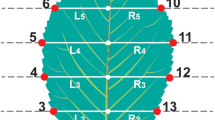
A total of 1600 leaves were scanned with a Mustek A3 2400S scanner at a resolution of 600 dpi to obtain high-quality images. Scanned images were used to digitize 2 landmarks (LM) and 20 semi-landmarks (SML) for each leaf (specimen), using TpsDig 2.17 software (Rohlf 2013). The first LM represented the intersection of the petiole and the leaf blade, while the second LM defined the apex of the leaf lamina. The sliding semi-landmarks method was used to describe the outline shape of the leaf blade. Two curves were manually traced along the external edges of the right blade (SLM 3–12) and left blade (SLM 13–22) sides, going from the last nerve to the beginning one. The curves were resampled as 10 equally spaced points, each (Fig. 2).
The specimens were superimposed by translating them to the origin, scaling to unit centroid size, and rotating to align with the reference shape by employing the generalized Procrustes analysis (GPA) (Rohlf and Slice 1990; Rohlf 2002) in TpsRelw 1.69 software (Rohlf 2017). The centroid size (CS) of each specimen was saved for further analyses of size variation across provenances.
Statistical analyses
A full Procrustes fit was performed from the aligned coordinates to extract the symmetric component of leaf shape (Klingenberg et al. 2012). Analyses of leaf shape were performed on the covariance matrix of the symmetric component averaged by tree, produced in MorphoJ software (Klingenberg 2011). Procrustes ANOVA was run on the shape coordinates to test for the effect of variation within leaves (i.e., directional asymmetry), and within and among trees of the same provenance. The pattern of leaf shape variation among trees was then explored with principal component analysis (PCA), while variation among provenances was summarized with canonical variate analysis (CVA). Leaf shape differences among provenance pairs were tested through Mahalanobis and Procrustes distances extracted from the tangent Euclidean (i.e., CVA scores) and the Kendall shape space (i.e., Procrustes distances), respectively. The covariation between leaf shape and geographic and environmental characteristics at the sites of provenances origin was investigated with Two-block Partial Least Square (PLS) analysis. The first block consisted of the symmetric component of shape variation and the second block of studied provenances latitude (Lat), longitude (Long) and altitude (Alt) and three climate variables: mean annual temperature (Tann), annual sum of precipitation (Pann) and Forest Aridity Index (FAI) (Table 1). All variables were standardized prior to the analysis, according to the equation:
where z is standardized value, x is specific value for which the standardized value is calculated, \(\overline{x}\) is mean of the given distribution, and SD is standard deviation of the given distribution. All individuals belonging to the same provenance had the same value for the climate variables. The full Procrustes fit, PCA, CVA and PLS analysis were carried out in MorphoJ software (Klingenberg 2011).
Differences in centroid size (CS) within and among provenances were assessed by applying nested analysis of variance (ANOVA) in Statistica 13 software (TIBCO Software Inc 2017). “Provenance” was considered to be a fixed factor as it was clearly defined, whereas individual “tree” (nested in provenance) was set as a random factor due to the trees within provenances being randomly selected. Tukey's honestly significant difference test (Tukey's HSD) was applied to test differences among studied provenances of European beech. Significant differences were determined at p < 0.05 throughout.
Results
Leaf shape variation
The two-factor Procrustes ANOVA showed that, in each provenance, there was more variation among leaves of different trees than among leaves within the same tree (Table 2), which justified data averaging per tree for further analyses. The effect of side was significant for all beech provenances (Pillai’s trace), indicating directional asymmetry.
The PCA of the symmetric component revealed that highest proportion of shape variation (73.3%) was associated with PC1 (Fig. 3a). On the plot of the first two PCs, no differentiation of provenances could be observed (except for a few individuals belonging to the German provenances), indicating high natural variability of leaf shape both at intra- and inter-provenance level (Fig. 3b). The leaf shapes along PC1 varied from longer and obovate blade with narrow-cuneate base at the negative end, to shorter and ovate blade with wide-cuneate base at the positive end of PC1. PC2 accounted for 16.2% of total symmetric variation and was associated with widening of the lamina in the upper part and its overall shortening, at the negative end, to longer and narrower lamina at the positive end.
a Percentage of variability explained by main principal components (PCs). Extreme leaf shapes (in dark blue) were presented for − 0.1 and + 0.1 of PC1, and − 0.05 and + 0.05 of PC2. Consensus configuration was presented in gray color. b Scatterplot of the first two PCs from PCA of symmetric covariance matrix generated on the averaged data for 80 trees. Each symbol represents average leaf shape per individual (i.e., tree). ME—provenance trial site Medvednica (Croatia); HR25—Vrani Kamen (Croatia); BA30—Tajan (Bosnia and Herzegovina); HU42—Valkonya (Hungary); DE48—Höllerbach (Germany); DE49—Hasbruch (Germany); RO63—Alesd (Romania); RO64—Alba-Iulia (Romania); RS66—Avala (Serbia)
The three canonical variates (CV1–CV3) explained 82.3% of the total variation among provenances, with first two CVs accounting for approximately 61.7% of the variation. The results of the first two CVs scores are shown in the scatter plot of Fig. 4. Provenances originating from Southern (BA30, HR25, RS66) and Central Europe (HU42) were differentiated from the others on the negative side of CV1. The lower scores on CV1 corresponded with obovate and longer lamina and narrow-cuneate bases (HR25), while trees on positive end had ovate and shorter leaf lamina and wide-cuneate base (DE49) (Fig. 4). Trees on negative end of CV2 were characterized with ovate lamina and acuminate apex (DE48), while positive end was associated with obovate lamina and acute apex (BA30) (Fig. 4). Along CV1, which accounted for 37.6% of variation, provenances HR25 and DE49 were the most discriminated (Fig. 5a), whereas the second CV accounted for 24.1% and mainly separated provenance DE48 and BA30 (Fig. 5b).
a Plot of the first two CVs scores of symmetric component of shape, generated on averaged data for 80 trees. Ellipses represent 90% confidence interval for provenance means. b Extreme leaf shapes (in dark blue) are shown for − 5.0 and + 5.0 along CV1, and − 5.0 and + 5.0 along CV2. The gray line represents the average leaf shape configuration. ME—provenance trial site Medvednica (Croatia); HR25—Vrani Kamen (Croatia); BA30—Tajan (Bosnia and Herzegovina); HU42—Valkonya (Hungary); DE48—Höllerbach (Germany); DE49—Hasbruch (Germany); RO63—Alesd (Romania); RO64—Alba-Iulia (Romania); RS66—Avala (Serbia)
Differences in the leaf shape among European beech provenances according to the canonical scores for the first two CVs. The boxplot colors correspond to the colors of provenances as presented in Figs. 3 and 4. ME—provenance trial site Medvednica (Croatia); HR25—Vrani Kamen (Croatia); BA30—Tajan (Bosnia and Herzegovina); HU42—Valkonya (Hungary); DE48—Höllerbach (Germany); DE49—Hasbruch (Germany); RO63—Alesd (Romania); RO64—Alba-Iulia (Romania); RS66—Avala (Serbia)
Mahalanobis distances among studied provenances were significant for all pairwise comparisons except for geographically close provenances originating from Romania (provenances RO63 and RO64) and Serbia (RS66) and Hungary (HU42), respectively. The inter-provenance distances showed that provenances DE49 (sum of Mahalanobis distances—26.852 and sum of Procrustes distances—0.281), HR25 (sum of Mahalanobis distances—25.112 and sum of Procrustes distances—0.166) and BA30 (sum of Mahalanobis distances—23.892 and sum of Procrustes distances—0.159) were the most differentiated (Table 3). Considering Mahalanobis distances, the highest differentiation was observed between provenance DE49 on one side, and provenances HR25, BA30, RS66 and HU42 on the other side, as well as between provenances DE48 and BA30. Similarly, significant Procrustes distances were found between provenance DE49, and provenances BA30, HU42 and RS66, as well as another German provenance, DE48, and BA30 and HU42. Highly significant differentiation was also recorded between easternmost provenance RO64, and both German provenances (DE48 and DE49), as well as RO64 and HR25 (Table 3). Mahalanobis distances are transformed Euclidean distances, so that variation within groups become circular. Significantly different distances appear only in Mahalanobis distances (but not Prucrustes distances) between certain provenances (e.g., DE49 and HR25) meaning that leaf shape in these provenances is characterized by non-isotropic variation (Klingenberg and Monteiro 2005).
Leaf size variation
The studied provenances differed in leaf CS (Table 4; Fig. 6). The results showed that variation in CS among provenances, as well as variation within provenances was highly significant (p < 0.001). The largest leaves were recorded in the provenance originating from Serbia (RS66), and the smallest in the provenance originating from Hungary (HU42).
Differences in CS among beech provenances. Homogenous groups are obtained by post hoc Tukey pairwise comparisons with 95% confidence. The boxplot colors correspond to the colors presented in Figs. 3 and 4. ME—provenance trial site Medvednica (Croatia); HR25—Vrani Kamen (Croatia); BA30—Tajan (Bosnia and Herzegovina); HU42—Valkonya (Hungary); DE48—Höllerbach (Germany); DE49—Hasbruch (Germany); RO63—Alesd (Romania); RO64—Alba-Iulia (Romania); RS66—Avala (Serbia)
Relationship between leaf shape and size and environmental factors
The PLS analysis examined the association between leaf shape (block 1) and some environmental characteristics of provenance origin sites (block 2). The overall strength of association between blocks was significant (p < 0.05) and the first pair of PLS axes explained 97% of total variation. The strength of correlation between blocks for PLS1 was moderate (r = 0.42, p = 0.026). In block 2, the highest loadings on PLS1 had latitude (− 0.64) and longitude (0.60) (Fig. 7, Table 5). We found that leaf shape patterns along latitudinal/longitudinal gradients were influenced by local climate at the sites of provenance origin. However, the association between climate and leaf shape variation was complex, since there was no single climate factor supported as a predictor for leaf shape variation. PLS between centroid size and environmental variables showed non-significant correlation (r = 0.11; p = 0.828).
Discussion
We observed a significant differentiation of provenances for both leaf size and shape, indicating the presence of local adaptation within the species natural range. The ability of plants to adjust to given environmental conditions is determined by long-term local adaptation and short-term acclimation (i.e., phenotypic plasticity), which jointly influence phenotypic differentiation of populations (Hamrick 2004; Nicotra et al. 2010). However, despite the relevance of this issue to overall understanding of evolutionary and ecological history of species, there remains the debate about relative contribution of genetic variation and phenotypic plasticity in the plant phenotypic response to climate (Voltas et al. 2018). Although certain studies identified phenotypic plasticity as the main driver of phenotypic variation in beech provenances (Gárate‐Escamilla et al. 2019; Müller et al. 2020), our results are more in line with the findings that demonstrated strong genetic control of different leaf functional traits in this species (Bolte et al. 2016; Kurjak et al. 2019; Petrík et al. 2020). As the local adaptation is shaped under the influence of the environment, spatial structuring of environmental conditions in nature may also lead to a hierarchical structuring of phenotypic trait variation, which could be either continuous (clinal) or discontinuous (ecotypic) (Alves et al. 2016). Previous studies showed contrasting results concerning variation patterns in functional traits of beech. Several studies on leaf anatomical and morphological traits variability revealed the presence of ecotypic (random) pattern of genetic variation (Stojnić et al. 2015; Vastag et al. 2019). On the other side, numerous research, mostly linked to beech phenology and growth rate, showed clinal (gradual) variation over geographic distance (Nielsen and Jørgensen 2003; Robson et al. 2013; Harter et al. 2015). Moreover, different studies showed geographical variation in physiological response of beech provenances to drought induced stress (Robson et al. 2012; Cocozza et al. 2016; Kurjak et al. 2019).
Our results confirmed presence of clinal pattern of leaf shape variation in beech provenances in a direction northwest—southeast. For example, Robson et al. (2013) reported that early-flushing provenances from the southeast and parts of central Europe were among the tallest, while late-flushing provenances from northern and western Europe were among the shortest in the field trial established in Spain. Similar results have been reported by von Wuehlisch et al. (1995) for time of flushing and growth cessation of beech provenances, i.e., more eastern provenances tended to flush and cease growth earlier, whereas northern provenances flushed and ended growing season later. Although all the provenances had average leaf shapes that differed significantly from one another, the CVA showed that provenances were aligned along CV1 relative to their geographic origin. In general, the first group, situated on the negative side of CV1, included provenances originating from Balkan Peninsula (HR25, BA30 and RS66) and Central Europe (HU42; geographically close to HR25), while the second group, located on the positive side of CV1, consisted of two Romanian provenances (RO63 and RO64) and the northernmost provenance DE49, which was also the most differentiated from other provenances. The pattern of leaf shape geographical variation largely corresponds to past migration history of the species, supporting previous assumptions about beech populations expansion during post-glacial period. According to Hazler et al. (1997), beech survived the last glacial period in numerous protected areas (refugia) from which the species re-colonized Europe. Based on palaeobotanical records and genetic data (i.e., nuclear and chloroplast markers), Magri et al. (2006) concluded that eastern Alps–Slovenia–Istria, and southern Moravia-southern Bohemia were the main refugium from which beech colonized central and northern Europe (including Germany). Another opinion by the same authors, they believe that Balkan Peninsula was colonized from several glacial refugia which spread locally (e.g., Balkan populations did not spread northward), and was clearly separated from the Romanian populations. Using a species distribution model and relaying the research on data of Magri et al. (2006), Saltré et al. (2013) additionally explained post-glacial colonization of beech in Europe. The authors indicated that Northern Alps was the main contributor of the present-day distribution of beech in Germany (e.g., the region from which provenances DE48 and DE49 originate), whereas large parts of Balkan and Eastern Europe (e.g., the region from which other provenances came from) were colonized from refugium situated in Slovenia and Czech Republic.
Yet, PLS showed that latitude and longitude were the best environmental predictors of leaf shape. Interestingly, no significant correlation was found between leaf shape and individual climatic variables, indicating rather complex effect of the climate on this trait. Likewise, it is very possible that other factors that are not quantified by this research, such as soil conditions and frequency of extreme climate events, affected the tree’s structure and function. For example, studying microbial soil biodiversity in beech forests, Dinca et al. (2021) found that microbial communities play significant role in increasing tree population’s resilience toward drought stress. In addition, several studies showed that local soil conditions may determine variation among beech provenances, even in geographically close ones (Robson et al. 2013; Stojnić et al. 2015). Moreover, certain authors evidenced that variation in leaf traits could be the result of trees adaptation to the physiological risk of drought stress (Bussotti et al. 2005).
Our results showed that provenances originating from higher latitudes and lower longitudes (i.e., Germany) were characterized with shorter and wider (ovate) lamina, which was less expanded in the apical part. In contrast, southern provenances tended to have longer and narrower (ovate) lamina, which might reflect direct selection aimed at improvement of leaf thermal regulation and hydraulic efficiency under more arid climatic conditions (and more frequent drought events), prevailing in this region (Nicotra et al. 2011). According to Yates et al. (2010), leaf shape and size determine the balance between photosynthesis, transpiration and thermoregulation, affecting the rates of gas and heat exchange between the leaf and the surrounding air. In this respect, small effective leaf width and leaf size have been seen as principal adaptive mechanisms of plants evolved in drier environments (Leigh et al. 2017), although an increase in leaf length has been reported to occur in response to warmer climate, as well (Gurevitch 1992). For example, Guerin et al. (2012) found that leaf width in Dodonaea viscosa subsp. angustissima was negatively correlated with latitude regionally, concluding that lamina narrowing was related to maximum temperature increase. Similar patterns of leaf width variation have been observed in other plant species associated with hot environments (Fonseca et al. 2000; Yates et al. 2010). Broader leaves have a thicker boundary layer of still air that reduce convective heat loss, tending to increase leaf-to-air temperature differences during sunny conditions (Fonseca et al. 2000). Indeed, studying the relationship between leaf morphology and leaf thermal regulation across 68 Proteaceae species, Leigh et al. (2017) demonstrated that leaf cooling time and leaf-to-air temperature differences significantly increased with leaf width. This may be particularly critical during hot spells, where thicker boundary layer tends to reduce leaf transpiration cooling, exposing the plant to a higher risk of overheating (Wright et al. 2017). In contrast, as noted by Yates et al. (2010), narrow leaves allow plants to shed heat through sensible heat exchange with surrounding air, without the need for cooling by transpiration water loss.
Besides heat transfer, leaf shape may also reflect direct selection for increased leaf lamina hydraulic conductance, which, in turn, depends on leaf venation architecture and extravascular pathways (Sack et al. 2003). For example, study of Sack et al. (2004) showed that the highest resistance to water flow through Acer saccharum and Quercus rubra leaves was in minor venation, followed by pathways outside the venation through mesophyll, which accounted for 68% and 75% of total leaf hydraulic resistance, respectively. Plants may increase leaf hydraulic efficiency through either deeply lobed leaves or entirely narrow leaves (Nicotra et al. 2011). Namely, narrow leaves have a lower ratio of minor veins and mesophyll tissue to lower-order veins, having a reduced hydraulic resistance relative to broad leaves. In contrast, broad leaves characterize with large areas of mesophyll far from highly conductive veins, which are supplied by large number of minor veins, therefore contributing to lower leaf hydraulic conductance (Sack and Tyree 2005).
Contrary to leaf shape, we found no correlation between leaf size and spatial and environmental variables, supporting previous findings obtained in common garden experiments, which showed that leaf area in beech provenances may vary independently of geographical and ecological gradients (Stojnić et al. 2016). Although certain studies have shown that leaf area in beech provenances was spatially structured along geographical gradients (Kempf et al. 2018), Leigh et al. (2017) stated that leaf thermal regulation is not a simple function of total leaf size, but largely depend on distances across the lamina, which may differ even in the case of a given leaf area. The same authors, thereby, believe that leaf width may have a greater influence on leaf thermal dynamics than leaf size (Leigh et al. 2017), indicating that leaf shape adjustments could be an efficient alternative to decreasing leaf area toward more arid environments.
Conclusion
High leaf size and shape variations occur in European beech across its native distribution range. Our results demonstrated that leaf shape, but not leaf size, varied geographically. The observed shape-geography covariation could be, therefore, interpreted in adaptive terms. Leaf shape relationships between European beech provenances and the longitude and latitude of their origin sites, suggest that species genetic differentiation occurred as a consequence of long-term selection by local environmental conditions. Narrower and longer lamina observed in southern provenances suggests a direct selection favoring leaf shape that likely safeguards trees performances under drier climatic conditions. Bearing in mind the predictions related to climate change impact on beech, our findings emphasize that assisted migration should be considered in future reforestation programs, where the provenances from Southern Europe might be potentially used to complement local seed sources at higher latitudes to enhance their resilience toward altered environmental conditions.
Author contribution statement
SS, RT, and AL conceived the ideas and designed the study. MI collected the leaf samples and herbarized the leaves. AL and VVi set up methodological approach of data processing. VVa scanned the leaves and prepared the leaf photos for analyses. SO recorded landmarks and semi-landmarks for each leaf (a total of 1600 leaves were processed). MM conducted the statistical analysis. SS and VVi wrote the first version of the manuscript, and all of the authors contributed critically to the drafts and gave final approval for publication.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Abdel-Rahman EH, Taylor PJ, Contrafatto G, Lamb JM, Bloomer P, Chimimba CT (2009) Geometric craniometric analysis of sexual dimorphism and ontogenetic variation: a case study based on two geographically disparate species, Aethomys ineptus from southern Africa and Arvicanthis niloticus from Sudan (Rodentia: Muridae). Mamm Biol 74:361–373. https://doi.org/10.1016/j.mambio.2008.06.002
Adler PB, Salguero-Gómez R, Compagnoni A, Hsu JS, Ray-Mukherjee J, Mbeau-Ache C, Franco M (2014) Functional traits explain variation in plant life history strategies. Proc Natl Acad Sci USA 111:740–745. https://doi.org/10.1073/pnas.1315179111
Albrecht H, Fiorani F, Pieruschka R, Müller-Linow M, Jedmowski C, Schreiber L, Schurr U, Rascher U (2020) Quantitative estimation of leaf heat transfer coefficients by active thermography at varying boundary layer conditions. Front Plant Sci 10:1684. https://doi.org/10.3389/fpls.2019.01684
Alves VM, Moura MO, de Carvalho CJB (2016) Wing shape is influenced by environmental variability in Polietina orbitalis (Stein) (Diptera: Muscidae). Rev Bras Entomol 60:150–156. https://doi.org/10.1016/j.rbe.2016.02.003
Bogunović S, Bogdan S, Lanšćak M, Ćelepirović N, Ivanković M (2020) Use of a common garden experiment in selecting adapted beech provenances for artificial stand restoration. South East Eur for 11:1–10. https://doi.org/10.15177/seefor.20-07
Bolte A, Czajkowski T, Cocozza C, Tognetti R, de Miguel M, Pšidová E, Ditmarová Ĺ, Dinca L, Delzon S, Cochard H, Ræbild A, de Luis M, Cvjetkovic B, Heiri C, Müller J (2016) Desiccation and mortality dynamics in seedlings of different European beech (Fagus sylvatica L.) populations under extreme drought conditions. Front Plant Sci 7:751. https://doi.org/10.3389/fpls.2016.00751
Bookstein FL (1997) Landmark methods for forms without landmarks: morphometrics of group differences in outline shape. Med Image Anal 1:225–243. https://doi.org/10.1016/S1361-8415(97)85012-8
Borghetti M, Leonardi S, Raschi A, Snyderman D, Tognetti R (1993) Ecotypic variation of xylem embolism, phenological traits, growth parameters and allozyme characteristics in Fagus sylvatica. Funct Ecol 7:713–720. https://doi.org/10.2307/2390193
Breno M, Leirs H, van Dongen S (2011) Traditional and geometric morphometrics for studying skull morphology during growth in Mastomys natalensis (Rodentia: Muridae). J Mamm 92:1395–1406. https://doi.org/10.1644/10-MAMM-A-331.1
Bussotti F, Pollastrini M (2015) Evaluation of leaf features in forest trees: methods, techniques, obtainable information and limits. Ecol Indic 52:219–230. https://doi.org/10.1016/j.ecolind.2014.12.010
Bussotti F, Pancrazi M, Matteucci G, Gerosa G (2005) Leaf morphology and chemistry in Fagus sylvatica (beech) trees as affected by site factors and ozone: results from CONECOFOR permanent monitoring plots in Italy. Tree Physiol 25:211–219. https://doi.org/10.1093/treephys/25.2.211
Cocozza C, de Miguel M, Pšidová E, Ditmarová L, Marino S, Maiuro L, Alvino A, Czajkowski T, Bolte A, Tognetti R (2016) Variation in ecophysiological traits and drought tolerance of beech (Fagus sylvatica L.) seedlings from different populations. Front Plant Sci 7:886. https://doi.org/10.3389/fpls.2016.00886
Cooke SB, Terhune CE (2015) Form, function, and geometric morphometrics. Anat Rec 298:5–28. https://doi.org/10.1002/ar.23065
Dinca L, Onet A, Samuel AD, Tognetti R, Uhl E, Bosela M, Gömöryová E, Bielak K, Skrzyszewski J, Hukić E, Zlatanov T, de Dios-Garcia J, Tonon G, Giammarchi F, Svoboda M, Dobor L, Rolando L, Rauseo J, Pescatore T, Garbini GL, Visca A, Patrolecco L, Caracciolo AB, Grenni P (2021) Microbial soil biodiversity in beech forests of European mountains. Can J for Res 51:1–13. https://doi.org/10.1139/cjfr-2020-0139
Duruflé H, Ranocha P, Mbadinga Mbadinga DL, Déjean S, Bonhomme M, San Clemente H, Viudes S, Eljebbawi A, Delorme-Hinoux V, Sáez-Vásquez J, Reichheld J-P, Escaravage N, Burrus M, Dunand C (2019) Phenotypic trait variation as a response to altitude-related constraints in Arabidopsis populations. Front Plant Sci 10:430. https://doi.org/10.3389/fpls.2019.00430
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Fonseca CR, Overton JM, Collins B, Westoby M (2000) Shifts in trait-combinations along rainfall and phosphorus gradients. J Ecol 88:964–977. https://doi.org/10.1046/j.1365-2745.2000.00506.x
Funk JL, Larson JE, Ames GM, Butterfield BJ, Cavender-Bares J, Firn J et al (2017) Revisiting the Holy Grail: using plant functional traits to understand ecological processes. Biol Rev 92:1156–1173. https://doi.org/10.1111/brv.12275
Gárate-Escamilla H, Hampe A, Vizcaíno-Palomar N, Robson TM, Benito Garzón M (2019) Range-wide variation in local adaptation and phenotypic plasticity of fitness-related traits in Fagus sylvatica and their implications under climate change. Glob Ecol Biogeogr 28:1336–1350. https://doi.org/10.1111/geb.12936
Gavranović A, Bogdan S, Lanšćak M, Čehulić I, Ivankovic M (2018) Seed yield and morphological variations of beechnuts in four European beech (Fagus sylvatica L.) populations in Croatia. South East Eur for 9:17–27. https://doi.org/10.15177/seefor.18-06
Gavrilov MB, Lukić T, Janc N, Basarin B, Marković SB (2019) Forestry aridity index in Vojvodina, North Serbia. Open Geosci 11:367–377. https://doi.org/10.1515/geo-2019-0029
Guerin GR, Wen H, Lowe AJ (2012) Leaf morphology shift linked to climate change. Biol Lett 8:882–886. https://doi.org/10.1098/rsbl.2012.0458
Gunz P, Mitteroecker P (2013) Semilandmarks: a method for quantifying curves and surfaces. HYSTRIX 24:103–109. https://doi.org/10.4404/hystrix-24.1-6292
Gurevitch J (1992) Sources of variation in leaf shape among two populations of Achillea lanulosa. Genetics 130:385–394
Hajek P, Kurjak D, von Wühlisch G, Delzon S, Schuldt B (2016) Intraspecific variation in wood anatomical, hydraulic, and foliar traits in ten European beech provenances differing in growth yield. Front Plant Sci 7:791. https://doi.org/10.3389/fpls.2016.00791
Hamrick JK (2004) Response of forest trees to global environmental changes. For Ecol Manag 197:323–335. https://doi.org/10.1016/j.foreco.2004.05.023
Harter DEV, Nagy L, Backhaus S, Beierkuhnlein C, Fussi B, Huber G, Jentsch A, Konnert M, Thiel D, Kreyling J (2015) A comparison of genetic diversity and phenotypic plasticity among european beech (Fagus sylvatica L.) populations from Bulgaria and Germany under drought and temperature manipulation. Int J Plant Sci 176:232–244. https://doi.org/10.1086/679349
Hazler K, Comps B, Šugar I, Melovski L, Tashev A, Gračan J (1997) Genetic structure of Fagus sylvatica L. populations in southeastern Europe. Silvae Genet 46:229–236
Jahdi R, Arabi M, Bussotti F (2020) Effect of environmental gradients on leaf morphological traits in the Fandoghlo forest region (NW Iran). iForest 13:523–530. https://doi.org/10.3832/ifor3391-013
Kempf M, Banach J, Skrzyszewska K (2018) Morphological variability of beech leaves from early and late flushing provenances. Balt for 24:210–217
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Resour 11:353–357. https://doi.org/10.1111/j.1755-0998.2010.02924.x
Klingenberg CP, Monteiro LR (2005) Distances and directions in multidimensional shape spaces: implications for morphometric applications. Syst Biol 54(4):678–688. https://doi.org/10.1080/10635150590947258
Klingenberg CP, Duttke S, Whelan S, Kim M (2012) Developmental plasticity, morphological variation and evolvability: a multilevel analysis of morphometric integration in the shape of compound leaves. J Evol Biol 25:115–129. https://doi.org/10.1111/j.1420-9101.2011.02410.x
Kubínová Z, Janáček J, Lhotáková Z, Šprtová M, Kubínová L, Albrechtová J (2018) Norway spruce needle size and cross section shape variability induced by irradiance on a macro- and microscale and CO2 concentration. Trees (berl West) 32:231–244. https://doi.org/10.1007/s00468-017-1626-3
Kurjak D, Konôpková A, Kmeť J, Mackov M, Frýdl J, Živčák M, Palmroth S, Ditmarová Ľ, Gömöry D (2019) Variation in the performance and thermostability of photosystem II in European beech (Fagus sylvatica L.) provenances is influenced more by acclimation than by adaptation. Eur J for Res 138:79–92. https://doi.org/10.1007/s10342-018-1155-7
Leigh A, Sevanto S, Close JD, Nicotra AB (2017) The influence of leaf size and shape on leaf thermal dynamics: does theory hold up under natural conditions? Plant Cell Environ 40:237–248. https://doi.org/10.1111/pce.12857
Maderbacher M, Bauer C, Herler J, Postl L, Makasa L, Sturmbauer C (2008) Assessment of traditional versus geometric morphometrics for discriminating populations of the Tropheus moorii species complex (Teleostei: Cichlidae), a Lake Tanganyika model for allopatric speciation. J Zool Syst Evol Res 46:153–161. https://doi.org/10.1111/j.1439-0469.2007.00447.x
Maestri R, Monteiro LR, Fornel R, Freitas TRO, Patterson BD (2018) Geometric morphometrics meets metacommunity ecology: environment and lineage distribution affects spatial variation in shape. Ecography 41:90–100. https://doi.org/10.1111/ecog.03001
Magri D, Vendramin GG, Comps B, Dupanloup I, Geburek T, Gömöry D, Latalowa M, Litt T, Paule L, Roure JM, Tantau I, van der Knaap WO, Petit RJ, de Beaulieu JL (2006) A new scenario for the Quaternary history of European beech populations: palaeobotanical evidence and genetic consequences. New Phytol 171:199–221. https://doi.org/10.1111/j.1469-8137.2006.01740.x
Miljković D, Čortan D (2020) Morphometric and morphological analysis of Populus nigra L. leaves in flooded regions. Šumar List 3–4:139–147. https://doi.org/10.31298/sl.144.3-4.3
Miljković D, Stefanović M, Orlović S, Stanković-Neđić M, Kesić L, Stojnić S (2019) Wild cherry (Prunus avium (L.) L.) leaf shape and size variations in natural populations at different elevations. Alp Bot 129:163–174. https://doi.org/10.1007/s00035-019-00227-1
Müller M, Cuervo-Alarcon L, Gailing O, Rajendra KC, Chhetri MS, Seifert S, Arend M, Krutovsky KV, Finkeldey R (2018) Genetic variation of European beech populations and their progeny from Northeast Germany to Southwest Switzerland. Forests 9:469. https://doi.org/10.3390/f9080469
Müller M, Kempen T, Finkeldey R, Gailing O (2020) Low population differentiation but high phenotypic plasticity of European beech in Germany. Forests 11:1354. https://doi.org/10.3390/f11121354
Nicotra AB, Atkin OK, Bonser SP, Davidson AM, Finnegan EJ, Mathesius U, Poot P, Purugganan MD, Richards CL, Valladares F, van Kleunen M (2010) Plant phenotypic plasticity in a changing climate. Trends Plant Sci 15:684–992. https://doi.org/10.1016/j.tplants.2010.09.008
Nicotra AB, Leigh A, Boyce CK, Jones CS, Niklas KJ, Royer DL, Tsukaya H (2011) The evolution and functional significance of leaf shape in the angiosperms. Funct Plant Biol 38:535–552. https://doi.org/10.1071/fp11057
Nielsen CN, Jørgensen FV (2003) Phenology and diameter increment in seedlings of European beech (Fagus sylvatica L.) as affected by different soil water contents: variation between and within provenances. For Ecol Manag 174:233–249. https://doi.org/10.1016/S0378-1127(02)00042-7
Palci A, Lee MSY (2019) Geometric morphometrics, homology and cladistics: review and recommendations. Cladistics 35:230–242. https://doi.org/10.1111/cla.12340
Pauls SU, Nowak C, Bálint M, Pfenninger M (2013) The impact of global climate change on genetic diversity within populations and species. Mol Ecol 22:925–946. https://doi.org/10.1111/mec.12152
Peppe DJ, Royer DL, Cariglino B, Oliver SY, Newman S, Leight E, Enikolopov G, Fernandez-Burgos M, Herrera F, Adams JM, Correa E, Currano ED, Erickson JM, Hinojosa LF, Hoganson JW, Iglesias A, Jaramillo CA, Johnson KR, Jordan GJ, Kraft NJ, Lovelock EC, Lusk CH, Niinemets U, Peñuelas J, Rapson G, Wing SL, Wright IJ (2011) Sensitivity of leaf size and shape to climate: global patterns and paleoclimatic applications. New Phytol 190:724–739. https://doi.org/10.1111/j.1469-8137.2010.03615.x
Petrík P, Petek A, Konôpková A, Bosela M, Fleischer P, Frýdl J, Kurjak D (2020) Stomatal and leaf morphology response of european beech (Fagus sylvatica L.) provenances transferred to contrasting climatic conditions. Forests 11:1359. https://doi.org/10.3390/f11121359
Pšidová E, Živčák M, Stojnić S, Orlović S, Gömöry D, Kučerová J, Ditmarová L, Střelcová K, Brestič M, Kalaji HM (2018) Altitude of origin influences the responses of PSII photochemistry to heat waves in European beech (Fagus sylvatica L.). Environ Exp Bot 152:97–106. https://doi.org/10.1016/j.envexpbot.2017.12.001
Ren J, Ji X, Wang C, Hu J, Nervo G, Li J (2020) Variation and genetic parameters of leaf morphological traits of eight families from Populus simonii × P. nigra. Forests 11(12):1319. https://doi.org/10.3390/f11121319
Robson TM, Sánchez-Gómez D, Cano FJ, Aranda I (2012) Variation in functional leaf traits among beech provenances during a Spanish summer reflects the differences in their origin. Tree Genet Genomes 8:1111–1121. https://doi.org/10.1007/s11295-012-0496-5
Robson TM, Rasztovits E, Aphalo PJ, Alia R, Aranda I (2013) Flushing phenology and fitness of European beech (Fagus sylvatica L.) provenances from a trial in La Rioja, Spain, segregate according to their climate of origin. Agric for Meteorol 280:76–85. https://doi.org/10.1016/j.agrformet.2013.05.008
Rohlf FJ (2002) Geometric morphometrics and phylogeny. In: MacLeod N, Forey PL (eds) Morphology, shape and phylogeny. CRC Press, New York, pp 175–193
Rohlf FJ (2013) TpsDig2 ver 2.1.7, ecology & evolution. SUNY, Stony Brook
Rohlf FJ (2017) Relative warps (Version 1.69) ecology & evolution and anthropology. Stony Brook University, New York
Rohlf FJ, Slice DE (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Zool 39:40–59. https://doi.org/10.2307/2992207
Sack L, Tyree MT (2005) Leaf hydraulics and its implications in plant structure and function. In: Holbrook NM, Zwieniecki MA (eds) Vascular transport in plants. Academic Press, New York, pp 93–114
Sack L, Cowan PD, Jaikumar N, Holbrook NM (2003) The ‘hydrology’ of leaves: co-ordination of structure and function in temperate woody species. Plant Cell Environ 26:1343–1356. https://doi.org/10.1046/j.0016-8025.2003.01058.x
Sack L, Streeter CM, Holbrook NM (2004) Hydraulic analysis of water flow through leaves of sugar maple and red oak. Plant Physiol 134:1824–1833. https://doi.org/10.1104/pp.103.031203
Saltré F, Saint-Amant R, Gritti ES, Brewer S, Gaucherel C, Davis BAS, Chuine I (2013) Climate or migration: what limited European beech post-glacial colonization? Glob Ecol Biogeogr 22:1217–1227. https://doi.org/10.1111/geb.12085
Savriama Y (2018) A step-by-step guide for geometric morphometrics of floral symmetry. Front Plant Sci 9:1433. https://doi.org/10.3389/fpls.2018.01433
Scartazza A, Di Baccio D, Bertolotto P, Gavrichkova O, Matteucci G (2016) Investigating the European beech (Fagus sylvatica L.) leaf characteristics along the vertical canopy profile: leaf structure, photosynthetic capacity, light energy dissipation and photoprotection mechanisms. Tree Physiol 36(9):1060–1076. https://doi.org/10.1093/treephys/tpw038
Schmieder DA, Benítez HA, Borissov IM, Fruciano C (2015) Bat species comparisons based on external morphology: a test of traditional versus geometric morphometric approaches. PLoS One 10:e0127043. https://doi.org/10.1371/journal.pone.0127043
Šijačić-Nikolić M, Milovanović J, Nonić M, Knežević R, Stanković D (2013) Leaf morphometric characteristics variability of different beech provenances in juvenile development stage. Genetika 45:369–380. https://doi.org/10.2298/GENSR1302369S
Stefanović M, Nikolić B, Matić B, Popović Z, Vidaković V, Bojović S (2017) Exploration of sexual dimorphism of Taxus baccata L. needles in natural populations. Trees (berl West) 31:1697–1710. https://doi.org/10.1007/s00468-017-1579-6
Stojnić S, Orlović S, Miljković D, Galić Z, Kebert M, von Wuehlisch G (2015) Provenance plasticity of European beech leaf traits under differing environmental conditions at two Serbian common garden sites. Eur J for Res 134:1109–1125. https://doi.org/10.1007/s10342-015-0914-y
Stojnić S, Orlović S, Miljković D, von Wuehlisch G (2016) Intra- and interprovenance variation of leaf morphometric traits in European beech (Fagus sylvatica L.) provenances. Arch Biol Sci 68:781–788. https://doi.org/10.2298/ABS151008064S
Thiel D, Kreyling J, Backhaus S, Beierkuhnlein C, Buhk C, Egen K, Huber G, Konnert M, Nagy L, Jentsch A (2014) Different reactions of central and marginal provenances of Fagus sylvatica to experimental drought. Eur J for Res 133:247–260. https://doi.org/10.1007/s10342-013-0750-x
TIBCO Software Inc (2017) Statistica (data analysis software system), version 13. http://statistica.io
Tognetti R, Michelozzi M, Borghetti M (1994) Response to light of shade-grown beech seedlings subjected to different watering regimes. Tree Physiol 14:751–758. https://doi.org/10.1093/treephys/14.7-8-9.751
Tognetti R, Johnson JD, Michelozzi M (1995) The response of European beech (Fagus sylvatica L.) seedlings from two Italian populations to drought and recovery. Trees (berl West) 9:348–354. https://doi.org/10.1007/BF00202499
Uhl D (2014) Variability of selected leaf traits in European beech (Fagus sylvatica) in relation to climatic factors—some implications for palaeoenvironmental studies. Phytol Balc 20:145–153
Van Zyl JJ (2001) The shuttle radar topography mission (SRTM): a breakthrough in remote sensing of topography. Acta Astronaut 48:559–565. https://doi.org/10.1016/S0094-5765(01)00020-0
Vastag E, Kovačević B, Orlović S, Kesić L, Bojović M, Stojnić S (2019) Leaf stomatal traits variation within and among fourteen European beech (Fagus sylvatica L.) provenances. Genetika 51:937–959. https://doi.org/10.2298/GENSR1903937V
Viscosi V (2015) Geometric morphometrics and leaf phenotypic plasticity: assessing fluctuating asymmetry and allometry in European white oaks (Quercus). Bot J Linn Soc 179:335–348. https://doi.org/10.1111/boj.12323
Viscosi V, Fortini P, Slice DE, Loy A, Blasi C (2009) Geometric morphometric analyses of leaf variation in four oak species of the subgenus Quercus (Fagaceae). Plant Biosyst 143:575–587. https://doi.org/10.1080/11263500902775277
Voltas J, Shestakova TA, Patsiou T, di Matteo G, Klein T (2018) Ecotypic variation and stability in growth performance of the thermophilic conifer Pinus halepensis across the Mediterranean basin. Forest Ecol Manag 424:205–215. https://doi.org/10.1016/j.foreco.2018.04.058
Von Wuehlisch G, Krusche D, Muhs HJ (1995) Variation in temperature sum requirement for flushing of beech provenances. Silvae Genet 44:343–346
von Wuehlisch G (2008) EUFORGEN technical guidelines for genetic conservation and use for European beech (Fagus sylvatica). Bioversity International, Rome
Wang Z, Zhang L (2012) Leaf shape alters the coefficients of leaf area estimation models for Saussurea stoliczkai in central Tibet. Photosynthetica 50:337–342. https://doi.org/10.1007/s11099-012-0039-1
Wang F, Israel D, Ramírez-Valiente JA, Sánchez-Gómez D, Aranda I, Aphalo PJ, Robson MT (2021) Seedlings from marginal and core populations of European beech (Fagus sylvatica L.) respond differently to imposed drought and shade. Trees (berl West) 35:53–67. https://doi.org/10.1007/s00468-020-02011-9
Watanabe A (2018) How many landmarks are enough to characterize shape and size variation? PLoS One 13:e0198341. https://doi.org/10.1371/journal.pone.0198341
Wellstein C, Chelli S, Campetella G, Bartha S, Galié M, Spada F, Canullo R (2013) Intraspecific phenotypic variability of plant functional traits in contrasting mountain grasslands habitats. Biodivers Conserv 22:2353–2374. https://doi.org/10.1007/s10531-013-0484-6
Wortemann R, Herbette S, Barigah TS, Fumanal B, Alia R, Ducousso A, Gömöry D, Roeckel-Drevet P, Cochard H (2011) Genotypic variability and phenotypic plasticity of cavitation resistance in Fagus sylvatica L. across Europe. Tree Physiol 31:1175–1182. https://doi.org/10.1093/treephys/tpr101
Wright IJ, Dong N, Maire V, Prentice C, Westoby M, Díaz S, Gallagher RV, Jacobs BF, Kooyman R, Law EA, Leishman MR, Niinemets Ü, Reich PB, Sack L, Villar R, Wang H, Wilf P (2017) Global climatic drivers of leaf size. Science 357:917–921. https://doi.org/10.1126/science.aal4760
Yates MJ, Verboom GA, Rebelo AG, Cramer MD (2010) Ecophysiological significance of leaf size variation in Proteaceae from the Cape Floristic Region. Funct Ecol 24:485–492. https://doi.org/10.1111/j.1365-2435.2009.01678.x
Zelditch ML, Swiderski DL, Sheets HD, Fink WL (2004) Geometric morphometric for biologissts: a primer. Elsevier Academic Press, London
Acknowledgements
This study was performed within COST (European Cooperation in Science and Technology) Action CLIMO (Climate-Smart Forestry in Mountain Regions—CA15226), supported by the EU Framework Programme for Research and Innovation HORIZON 2020. The research was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (contract number: 451-03-9/2021-14/200197). The authors greatly appreciate Mr. Ed Bauer from USDA Forest Service Northern Research Station, Rhinelander, WI, for editing the manuscript for English language and style.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by J. Major.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Stojnić, S., Viscosi, V., Marković, M. et al. Spatial patterns of leaf shape variation in European beech (Fagus sylvatica L.) provenances. Trees 36, 497–511 (2022). https://doi.org/10.1007/s00468-021-02224-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-021-02224-6