Abstract
This study examined the effects of climate on tree-ring widths and maximum latewood densities of Abies veitchii and Abies mariesii at the upper and lower distribution limits in central Japan. A. veitchii and A. mariesii dominated at the lower and upper parts of the subalpine zone, respectively. Residual chronologies of tree-ring width and maximum latewood density were developed for the two Abies species at the upper and lower distribution limits, and were compared with monthly mean temperatures and monthly sums of precipitation. Tree-ring widths of the two Abies species at the upper and lower distribution limits positively correlated with temperatures during the beginning of the dormant season and during the growing season of the current year, except for A. veitchii at the lower distribution limit, which showed no positive correlation with temperature. Maximum latewood densities of the two Abies species at the upper and lower distribution limits positively and negatively correlated with temperatures and precipitation, respectively, during the growing season of the current year. Therefore, tree-ring widths and maximum latewood densities of the two Abies species were sensitive to low temperature, except for the tree-ring width of A. veitchii at the lower distribution limit with the warmest thermal conditions along the altitude. Global warming is suggested to affect maximum latewood densities and tree-ring widths of the two Abies species along the altitude.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The growth period of plants decreases with increasing altitude, in association with changes in air temperature and precipitation. Such altitudinal environmental changes affect plant growth along altitudes. Many studies investigated the effects of climate on radial growth of trees by using dendrochronological techniques (Cullen et al. 2001; Nöjd and Hari 2001; Peterson and Peterson 2001; Hopton and Pederson 2005; Fan et al. 2009). In a study of the change in growth–climate relationships along an altitudinal gradient, the tree-ring width of Picea abies (L.) H. Karst negatively and positively correlated with early summer temperature and precipitation, respectively, at a low altitude, but inversely correlated at a high altitude (Wilson and Hopfmueller 2001). Similar altitudinal changes in growth–climate relationships were also observed in other studies (Adams and Kolb 2005; Wang et al. 2005; Massaccesi et al. 2008; Peng et al. 2008), although growth–climate relationships vary at the same altitude according to topographical aspects (Liang et al. 2006). Therefore, the results of previous studies suggest that the factors limiting tree growth are drought stress at low altitudes and low temperatures at high altitudes on a meso scale.
Within a genus, plant species are often distributed at different altitudes. For example, Betula platyphylla var. japonica Hara is distributed in the upper part of the montane zone (~1,600 m a.s.l.) in central Japan, but this species is replaced by Betula ermanii Cham. in the subalpine zone above 1,600 m a.s.l. In this case, the altitude of 1,600 m a.s.l. is the altitudinal ecotone between the two species. The climatic conditions of an altitudinal ecotone are cold and moist for species of the upper distribution limit within its altitudinal distribution range, but in the same ecotone are warm and dry for species of the lower distribution limit within its altitudinal distribution range. Thus, species at the upper distribution limit may respond to climatic conditions differently from species at the lower distribution limit in an altitudinal ecotone. For example, growth of B. ermanii at the lower distribution limit is more reduced by drought stress compared with B. platyphylla var. japonica at the upper distribution limit in the altitudinal ecotone (Takahashi et al. 2003). Examination of growth responses to climatic conditions for both species at the upper and lower distribution limits of an altitudinal ecotone is important to understand the effects of global warming on altitudinal plant distributions. However, a few studies have compared growth responses to climatic conditions between tree species at their upper and lower distribution limits in an altitudinal ecotone.
In the subalpine zone on Mount Norikura, central Japan, Abies veitchii Lindl. and Abies mariesii Mast. are dominant conifers at the lower (1,600–2,000 m a.s.l.) and upper parts (2,000–2,500 m a.s.l.), respectively (Miyajima et al. 2007). The altitude around 2,000 m a.s.l. is the transitional vegetation ecotone of the two Abies species along the altitude. In the case of conifers, climatic factors affect not only tree-ring width but also maximum latewood density (Hughes et al. 1984; Briffa et al. 1998; Barber et al. 2004). Wide tracheids with a thin secondary wall are formed in the early growing season (earlywood), and then narrow tracheids are formed with a thick secondary wall (latewood). Wood density is wood mass per unit volume, which is usually greater in tracheids formed in the later growing season (Yasue et al. 2000).
In this study, we investigated the two Abies species at their upper and lower distribution limits on Mount Norikura by attempting to answer the following questions:
-
1.
Are tree-ring width and maximum latewood density of the two Abies species more sensitive to low temperature at the upper distribution limit than at the lower distribution limit?
-
2.
Are tree-ring width and maximum latewood density of A. veitchii at the upper distribution limit reduced by low temperature more than those of A. mariesii at the lower distribution limit of an altitudinal ecotone?
Materials and methods
Study site
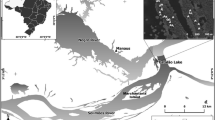
This study was done in the subalpine forest zone on the east slope of Mount Norikura (36°06′N, 137°33′E, 3,026 m above sea level) in central Japan. The mean annual temperature recorded at Nagawa Weather Station (1,068 m a.s.l., approximately 12 km in horizontal distance from the summit) was 8.2°C between 1979 and 2005. The mean monthly temperatures in January and in August were −3.6 and 20.4°C, respectively. The mean annual precipitation was 1,968 mm.
Three major vegetation zones can be recognized along the altitude on Mount Norikura: a montane deciduous broad-leaved forest zone between 800 m and 1,600 m a.s.l., a subalpine coniferous forest zone between 1,600 m and 2,500 m a.s.l. and an alpine dwarf pine (Pinus pumila Regel) scrub zone between 2,500 m and 3,000 m a.s.l. (Miyajima et al. 2007; Miyajima and Takahashi 2007). The timberline is at about 2,500 m a.s.l. on the east slope of Mount Norikura (Takahashi 2003).
Dominant tree species are A. veitchii and Tsuga diversifolia Mast. between 1,600 m and 2,000 m a.s.l., and A. mariesii, B. ermanii, Sorbus commixta Hedl. and Sorbus matsumurana Koehne between 2,200 m and 2,500 m a.s.l. Anthropogenic effects on vegetation were negligible from 1,600 m a.s.l. to the summit.
Sampling and measurements
This study was done at three altitudes corresponding to the lower distribution limit of A. veitchii (1,600 m a.s.l.), the upper distribution limit of A. mariesii (2,400 m a.s.l.) and the altitudinal ecotone from A. veitchii to A. mariesii distributions (1,900 m a.s.l.). To include as long a common time period as possible, wood cores (5 mm in diameter) were taken from trunks of canopy trees (about 20 m in height) at breast height (1.3 m) at 2,400 m a.s.l. in 2003 and at 1,600 m and 1,900 m a.s.l. in 2006. The number of cores was between 36 and 44 for each species at each altitude (Table 1). The trunk diameter at breast height (DBH) measured for each tree was 27–61 cm (Table 1).
Increment cores were cut transversely into 1.5 mm thick strips by using a twin-bladed saw. The strips were oven-dried and then they underwent soft X-ray analysis at 20 kV and 14 mA for 210 s from a distance of 2.2 m; a calibration wedge was also used in the soft X ray analysis. The resultant radiographs were scanned at resolution 2,400 dpi. The tree-ring widths and maximum latewood densities were measured by using a WinDENDRO (Regent Instruments Inc., Quebec City, Canada).
Chronology development
All cores were cross-dated visually by matching characteristic wide and narrow rings that were synchronous within sample trees. Visual cross-dating was statistically verified by using the COFECHA program (Holmes 1983, 1994) that tests each individual series against a master dating series (mean of all series) from correlation coefficients.
Growth of trees is affected not only by climatic factors but also by age, disturbance and competition between neighboring trees. To reduce the variations caused by such non-climatic factors, all raw ring-width series were standardized by fitting smoothing splines (Cook and Peters 1981) with a 50% frequency–response cutoff of 40 years by using the ARSTAN program (Cook 1985; Holmes 1994). After standardizing each individual series, standard chronologies of the tree-ring width and maximum latewood density were obtained by averaging the standardized individual series in each year. We used at least five cores for each chronology in each year.
A standardized series usually shows autocorrelation that negates the assumption of independence necessary for most statistical analyses (Fritts 1976; Monserud 1986). To remove the effects of autocorrelation, we transformed each standardized series to a residual series through pooled autoregressive modeling by using the ARSTAN program.
Five statistics were calculated for residual chronologies: (1) mean correlation between trees represents the chronology signal strength; (2) mean sensitivity and (3) standard deviation are measures of interannual variation; (4) signal-to-noise ratio expresses the strength of the observed common signal among trees; (5) expressed population signal quantifies the degree to which a particular sample chronology portrays the hypothetically perfect chronology, which may in turn be regarded as the potential climatic signal. A threshold of 0.85 was empirically suggested as an acceptable statistical quality (Wigley et al. 1984).
Statistical analysis
Relationships of residual chronologies of tree-ring width and maximum latewood density with monthly climatic data were analyzed by using a bootstrapped correlation function for the common interval (1944–2001, Table 1) between the two Abies species at the upper and lower distribution limits by using the DENDROCLIM2002 program (Biondi and Waikul 2004). DENDROCLIM2002 uses 1,000 bootstrapped samples to compute correlation coefficients, and to test their significance at the 0.05 level.
Monthly mean temperature and the sum of precipitation data were used for the analysis. The nearest weather station to the study site was Nagawa (1,068 m a.s.l., approximately 11 km from the study site at 2,400 m a.s.l.). However, the available meteorological data at Nagawa started in 1979, whereas long-term records from 1898 were available at Matsumoto (610 m a.s.l., approximately 40 km from the study site at 2,400 m a.s.l.). Correlations of climatic data between Matsumoto and Nagawa stations were highly significant, i.e., correlation coefficients ranged between 0.89 (August) and 0.98 (April) for monthly mean temperature and between 0.59 (May) and 0.94 (November) for monthly precipitation. Thus, the general temporal trends of temperature and precipitation were consistent between Matsumoto and Nagawa, indicating that the climatic trend of Matsumoto represents that of the study region. Thus, we used long-term climatic data recorded at Matsumoto in this study for the bootstrapped correlation function.
The bootstrapped correlation function was done by using climatic data from the start of the previous growing season to the end of the current growing season because the growth of many tree species is affected not only by climatic conditions of the current year, but also by those of the previous year (cf. Eshete and Ståhl 1999; Takahashi et al. 2001; Speer et al. 2004). The approximate growing season at 1,600 m and 1,900 m a.s.l. was estimated as May to October, because the mean monthly temperatures exceeded 5°C, the effective heat for plant growth (Kira 1948), during this period, and so the correlation analysis was done using climatic data from May of the previous year to October of the current year (18 months total). The growing season at 2,400 m a.s.l. was estimated as June–September, so the correlation analysis was done using climatic data from June of the previous year to September of the current year (16 months total).
Results
Basic statistics
Wood cores were taken from the similar DBH range (ca. 30–60 cm) of the two Abies species at the upper and lower distribution limits (Table 1). However, the chronology range was considerably shorter for A. veitchii at 1,600 m a.s.l. than the other chronology ranges because they were young and the growth rate was high (Table 1, Fig. 1). Mean correlation coefficients between trees were 0.273–0.353 for the two Abies species at the upper and lower distribution limits. The mean sensitivity and standard deviation were lower in the four chronologies of the maximum latewood densities than those of the tree-ring widths (ex., 0.037–0.049 vs. 0.132–0.206 for mean sensitivity, 0.034–0.046 vs. 0.126–0.186 for standard deviation). The signal-to-noise ratio and expressed population signal were lowest for A. mariesii at 2,400 m a.s.l. among the four chronologies for both tree-ring width and maximum latewood density (Table 1). The expressed population signal was greater than the empirical threshold of 0.85, indicating a good estimation of the population value for all chronologies, except for the maximum latewood density of A. mariesii at 2,400 m a.s.l.
Correlations between climatic factors and tree-ring widths
The tree-ring width chronology of A. veitchii at 1,600 m a.s.l. negatively correlated with July and August temperatures of the previous year and positively correlated with March and August precipitation of the previous year (Fig. 2). Responses of the tree-ring width chronology of A. veitchii at 1,900 m a.s.l. considerably differed from those at 1,600 m a.s.l. The tree-ring width of A. veitchii at 1,900 m a.s.l. positively correlated with May precipitation of the previous year and October precipitation of the current year and negatively correlated with July precipitation of the current year. Tree-ring width of A. veitchii at 1,900 m a.s.l. positively correlated with temperatures of 8 months between November of the previous year and October of the current year. Thus, tree-ring width of A. veitchii positively responded to temperatures more at 1,900 m a.s.l. than at 1,600 m a.s.l.
Correlation coefficients between tree-ring width chronologies and monthly climate data (temperature [left] and precipitation [right]) for Abies mariesii at 2,400 m a.s.l., A. mariesii at 1,900 m a.s.l., A. veitchii at 1,900 m a.s.l., and A. veitchii at 1,600 m a.s.l. on Mount Norikura, central Japan. Shaded bars indicate significant correlation (P < 0.05). PGS, DS and CGS mean previous-year growing season, dormant season and current-year growing season, respectively
The responses of the tree-ring width chronology of A. mariesii at 1,900 m and 2,400 m a.s.l. were similar to those of A. veitchii at 1,900 m a.s.l. (Fig. 2), although the number of months with significant correlations was less in A. mariesii at 1,900 m and 2,400 m a.s.l. than A. veitchii at 1,900 m a.s.l. Especially, precipitation did not correlate with the tree-ring width chronology of A. mariesii at 2,400 m a.s.l. The tree-ring widths of A. mariesii at 1,900 m and 2,400 m a.s.l. positively correlated with temperatures of the beginning of the dormant season and ad summer temperatures.
Correlations between climatic factors and maximum latewood densities
The maximum latewood densities of the two Abies species at the upper and lower distribution limits negatively correlated with July temperature of the previous year, and positively correlated with temperatures between late spring and early autumn in the current year (Fig. 3). Correlation patterns of precipitation were the reverse of those of temperature for the two Abies species at the upper and lower distribution limits. The maximum latewood densities were positively correlated with summer precipitation in the previous year and negatively correlated with precipitation between late spring and summer in the current year. Thus, high temperatures and little precipitation between late spring and early autumn in the current year increased the maximum latewood densities of the two Abies species irrespective of altitude.
Correlation coefficients between maximum latewood density chronologies and monthly climate data (temperature [left] and precipitation [right]) for Abies mariesii at 2,400 m a.s.l., A. mariesii at 1,900 m a.s.l., A. veitchii at 1,900 m a.s.l., and A. veitchii at 1,600 m a.s.l. on Mount Norikura, central Japan. Shaded bars indicate significant correlation (P < 0.05). PGS, DS and CGS mean previous-year growing season, dormant season and current-year growing season, respectively
Discussion
Responses of tree-ring widths
The responses of tree-ring width chronology of A. veitchii at 1,600 m a.s.l. differed from those of the three other tree-ring width chronologies at 1,900 m and 2,400 m a.s.l. Although A. veitchii sampled at 1,600 m a.s.l. was younger than A. veitchii at 1,900 m a.s.l. and A. mariesii at 1,900 m and 2,400 m a.s.l., this study developed tree-ring width chronologies assuming an age-invariant growth–climate relationship. However, some studies reported that such an assumption is invalid because growth–climate relationships change with tree age (Szeicz and MacDonald 1994; Rozas 2005; Rossi et al. 2008; Vieira et al. 2009). For example, the tree-ring width of young Juniperus thurifera is prone to respond to precipitation in a continental Mediterranean region, probably because of undeveloped roots (Rozas et al. 2009). In this study, positive correlation with summer precipitation was recognized for only A. veitchii at 1,600 m a.s.l. However, whether the root of A. veitchii at 1,600 m was less developed compared with the other trees examined at 1,900 m and 2,400 m a.s.l. is difficult to consider, because trunk height and the range of DBH of trees examined in this study were similar among the two Abies species at the upper and lower distribution limits. Carrer and Urbinati (2004) found that sensitivity of tree-ring width of Larix deciduas to climatic conditions was greater in old trees than young trees, and suggested that the high sensitivity of old trees was ascribed to the strong hydraulic resistance due to the tall trunk height. Thus, the effect of tree age on the growth–climate relationship among the four chronologies would be small in this study because of the similar trunk height among the two Abies species at the upper and lower distribution limits.
Probably, the different growth–climate relationship of A. veitchii at 1,600 m a.s.l. was mainly caused by altitudinal change in climatic conditions. Generally, precipitation is less and temperature is higher at lower altitudes. Especially, the precipitation of August (104 mm at Matsumoto) is less than the other summer months (July 130 mm, September 153 mm), which causes drought stress in August at 1,600 m a.s.l. (Takahashi et al. 2003). By contrast, the tree-ring width of A. veitchii at 1,900 m a.s.l. was positively correlated with temperatures between late spring and early autumn. Such altitudinal changes in growth–climate relationships were also observed in other studies (Adams and Kolb 2005; Wang et al. 2005). Thus, the different growth–climate relationship of A. veitchii is suggested to be ascribed to altitudinal change in climatic conditions.
The tree-ring width of A. mariesii positively correlated with July and August temperatures at 2,400 m a.s.l. and June and July temperatures at 1,900 m a.s.l. in the current year, like A. veitchii at 1,900 m a.s.l. Fujiwara et al. (1999) also observed that the tree-ring width of A. mariesii positively correlated with summer temperatures of the current year at about 2,000 m and 2,200 m a.s.l. on Mount Norikura. Cambial activity (cell division) is generally slow under cold conditions (Gricar et al. 2007). Therefore, tree-ring width is often positively correlated with summer temperatures at high altitudes (Buckley et al. 1997; Oberhuber 2004; Savva et al. 2006).
Tree-ring widths of A. veitchii (1,900 m a.s.l.) and A. mariesii at (1,900 m and 2,400 m a.s.l.) showed positive correlations with temperatures of the beginning of the dormant season (October to December) of the previous year. Although radial growth already finishes by October, high temperature is beneficial for photosynthetic production in this period. Thus, much photosynthetic production due to warm temperatures between October and December is suggested to increase tree growth in the next year.
Responses of maximum latewood densities
The maximum latewood densities of the two Abies species at the upper and lower distribution limits showed significant positive correlations with temperatures during the growing season of the current year. Maximum latewood densities also negatively correlated with precipitation during the growing season in the current year. However, this would not mean that much precipitation directly reduced maximum latewood densities because of negative correlation between temperature and precipitation during the growing season (Takahashi et al. 2005).
Cell division of the cambium of the two Abies species starts from early June to early July at 1,600 m to 2,400 m a.s.l. on Mount Norikura, and thickening of the secondary wall continues until mid-September (Koike 2009). Yasue et al. (2000) reported that interannual variation in maximum latewood density of Picea glehnii Mast. is due to interannual variation in cell wall thickness of the last-formed cells, which varies depending on the climatic conditions in summer. In this study, positive correlations of maximum latewood density with current-year April and May temperatures before the start of cell division were also found. It is well known that warm spring temperature cause earlier onset of shoot growth (Gostev et al. 1996), which might lead to higher photosynthetic production due to earlier development of current-year needles. Hence, more carbon might be available for cell wall growth and lignification of latewood cells. Therefore, maximum latewood density would respond to climatic conditions in late spring and during the growing season.
In this study, July temperature of the previous year was negatively correlated with maximum latewood densities of the two Abies species at the upper and lower distribution limits. Unfortunately, we do not have enough ecophysiological information to explain the relationships. Possibly, July temperature is not a causal factor if the temperature correlates with other climatic factors that correlate with maximum latewood densities. However, July temperature of the previous year did not correlate with other climatic factors, except for the positive correlations with temperatures of the other months during the growing season of the same year. However, these temperatures did not correlate with the maximum latewood densities of the two Abies species at the upper and lower distribution limits. Thus, the negative correlation of July temperature of the previous year was, at least, not indirectly caused by correlation with other climatic factors.
Conclusion
This study concludes that (1) tree-ring widths of the two Abies species above 1,900 m a.s.l. are increased by high temperatures of the previous-year late autumn and current-year summer, and maximum latewood densities of the two Abies species at the upper and lower distribution limits are increased by high temperatures between spring and summer and redued by high summer precipitation in the current year, (2) responses of tree-ring width and maximum latewood density to climate were similar between A. veitchii at the upper distribution limit and A. mariesii at the lower distribution limit of the altitudinal ecotone (1,900 m a.s.l.). Therefore, global warming is suggested to increase maximum latewood densities and tree-ring widths of the two Abies species, except for the tree-ring width of A. veitchii at the lower distribution limit. The issue whether global warming will alter chronologies of the two Abies species in old-growth stands is useful to predict effects of global warming on stand dynamics along the altitude.
References
Adams HD, Kolb TE (2005) Tree growth response to drought and temperature in a mountain landscape in northern Arizona, USA. J Biogeogr 32:1629–1640
Barber VA, Juday GP, Finney BP, Wilmking M (2004) Reconstruction of summer temperatures in interior Alaska from tree-ring proxies: evidence for changing synoptic climate regimes. Clim Chang 63:91–120
Biondi F, Waikul K (2004) DENDROCLIM2002: a C++ program for statistical calibration of climate signals in tree-ring chronologies. Comput Geosci 30:303–311
Briffa KR, Schweingruber FH, Jones PD, Osborn TJ, Shiyatov SG, Vaganov EA (1998) Reduced sensitivity of recent tree-growth to temperature and high northern latitudes. Nature 391:678–682
Buckley BM, Cook ER, Peterson MJ, Barbetti M (1997) A changing temperature response with elevation for Lagarostrobos franklinii in Tasmania, Australia. Clim Chang 36:477–498
Carrer M, Urbinati C (2004) Age-dependent tree-ring growth responses to climate in Larix decidua and Pinus cembra. Ecology 85:730–740
Cook ER (1985) A time series analysis approach to tree ring standardization. PhD thesis, Univ Arizona, Tucson
Cook ER, Peters K (1981) The smoothing spline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree-Ring Bull 41:45–53
Cullen LE, Palmer JG, Duncan RP, Stewart GH (2001) Climate change and tree-ring relationships of Nothofagus menziesii tree-line forests. Can J For Res 31:1981–1991
Eshete G, Ståhl G (1999) Tree rings as indicators of growth periodicity of acacias in the Rift Valley of Ethiopia. For Ecol Manage 116:107–117
Fan ZX, Brauning A, Cao KF, Zhu SD (2009) Growth-climate responses of high-elevation conifers in the central Hengduan Mountains, southwestern China. For Ecol Manage 258:306–313
Fritts HC (1976) Tree rings and climate. The Blackburn Press, New Jersey
Fujiwara T, Okada N, Yamashita K (1999) Comparison of growth response of Abies and Picea species to climate in Mt. Norikura, central Japan. J Wood Sci 45:2–97
Gostev M, Wiles G, D’Arrigo R, Jacoby G, Khomentovsky P (1996) Early summer temperatures since 1670 A.D. for Central Kamchatka reconstructed based on a Siberian larch tree-ring width chronology. Can J For Res 26:2048–2052
Gricar J, Zupancic M, Cufar K, Oven P (2007) Regular cambial activity and xylem and phloem formation in locally heated and cooled stem portions of Norway spruce. Wood Sci Technol 41:463–475
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-ring Bull 43:69–78
Holmes RL (1994) Dendrochronology program library version 1994. Laboratory of Tree-Ring Research, Univ Arizona, Tucson
Hopton HM, Pederson N (2005) Climate sensitivity of Atlantic white cedar at its northern range limit. General Technical Report SRS-91. USDA Forest Service, Millersville, pp 22–30
Hughes MK, Schweingruber FH, Cartwright D, Kelly PM (1984) July–August temperature at Edinburgh between 1721 and 1975 from tree-ring density and width data. Nature 308:341–344
Kira T (1948) On the altitudinal arrangement of climatic zones in Japan. Kanti Nogaku 2:143–173 (in Japanese)
Koike S (2009) Seasonal changes of radial growth of trees along an altitudinal gradient and effects of daily changes of climate on radial growth. MS thesis. Shinshu Univ, Nagano (in Japanese)
Liang E, Shao X, Eckstein D, Huang L, Liu X (2006) Topography- and species-dependent growth responses of Sabina przewalskii and Picea crassifolia to climate on the northeast Tibetan Plateau. For Ecol Manage 236:268–277
Massaccesi G, Roig FA, Pastur GJM, Barrera MD (2008) Growth patterns of Nothofagus pumilio trees along altitudinal gradients in Tierra del Fuego, Argentina. Trees 22:245–255
Miyajima Y, Takahashi K (2007) Changes with altitude of the stand structure of temperate forests on Mount Norikura, central Japan. J For Res 12:187–192
Miyajima Y, Sato T, Takahashi K (2007) Altitudinal changes in vegetation of tree, herb and fern species on Mount Norikura, central Japan. Veg Sci 24:29–40
Monserud RA (1986) Time-series analyses of tree-ring chronologies. For Sci 32:349–372
Nöjd P, Hari P (2001) The effect of temperature on the radial growth of Scots pine in northernmost Fennoscandia. For Ecol Manage 142:65–77
Oberhuber W (2004) Influence of climate on radial growth of Picea cembra within the alpine timberline ecotone. Tree Physiol 24:291–301
Peng J, Gou X, Chen F, Li J, Liu P, Zhang Y (2008) Altitudinal variability of climate-tree growth relationships along a consistent slope of Anyemaqen Mountains, northeastern Tibetan Plateau. Dendrochronologia 26:87–96
Peterson DW, Peterson DL (2001) Mountain hemlock growth responds to climatic variability at annual and decadal time scales. Ecology 82:3330–3345
Rossi S, Deslauriers A, Anfodillo T, Carrer M (2008) Age-dependent xylogenesis in timberline conifers. New Phytol 177:199–208
Rozas V (2005) Dendrochronology of pedunculate oak (Quercus robur L.) in an old-growth pollarded woodland in northern Spain: tree-ring growth responses to climate. Ann For Sci 62:209–218
Rozas V, DeSoto L, Olano JM (2009) Sex-specific, age-dependent sensitivity of tree-ring growth to climate in the dioecious tree Juniperus thurifera. New Phytol 182:687–697
Savva Y, Oleksyn J, Reich PB, Tjoelker MG, Vaganov EA, Modrzynski J (2006) Interannual growth response of Norway spruce to climate along an altitudinal gradient in the Tatra Mountains, Poland. Trees 20:735–746
Speer JH, Orvis KH, Grissino-Mayer HD, Kennedy LM, Horn SP (2004) Assessing the dendrochronological potential of Pinus occidentalis Swartz in the Cordillera Central of the Dominican Republic. The Holocene 14:563–569
Szeicz JM, MacDonald GM (1994) Age-dependent tree-ring growth responses of subarctic white spruce to climate. Can J For Res 24:120–132
Takahashi K (2003) Effects of climatic conditions on shoot elongation of alpine dwarf pine (Pinus pumila) at its upper and lower altitudinal limits in central Japan. Arct Antarct Alp Res 35:1–7
Takahashi K, Homma K, Shiraiwa T, Vetrova VP, Hara T (2001) Climatic factors affecting the growth of Larix cajanderi in the Kamchatka Peninsula, Russia. Eurasian J For Res 3:1–9
Takahashi K, Azuma H, Yasue K (2003) Effects of climate on the radial growth of tree species in the upper and lower distribution limits of an altitudinal ecotone on Mt. Norikura, central Japan. Ecol Res 18:549–558
Takahashi K, Tokumitsu Y, Yasue K (2005) Climatic factors affecting the tree-ring width of Betula ermanii at the timberline on Mount Norikura, central Japan. Ecol Res 20:445–451
Vieira J, Campelo F, Nabais C (2009) Age-dependent responses of tree-ring growth and intra-annual density fluctuations of Pinus pinaster to Mediterranean climate. Trees 23:257–265
Wang T, Ren H, Ma K (2005) Climatic signals in tree ring of Picea schrenkiana along an altitudinal gradient in the central Tianshan Mountains, northwestern China. Trees 19:735–741
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendrochronology and hydrometeorology. J Clim Appl Meteorol 23:201–213
Wilson RJS, Hopfmueller M (2001) Dendrochronological investigations of Norway spruce along an elevational transect in the Bavarian Forest, Germany. Dendrochronologia 19:67–79
Yasue K, Funada R, Kobayashi O, Ohtani J (2000) The effects of tracheid dimensions on variations in maximum density of Picea glehnii and relationships to climatic factors. Trees 14:223–229
Acknowledgments
We thank Dr. Takeshi Fujiwara and his colleagues of Forestry and Forest Products Research Institute for giving facilities for X-ray. This study was partially supported by grants from the Ministry of Education, Culture, Sports, Science and Technology, Japan (Nos. 15710007, 19580168).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Wieser.
Rights and permissions
About this article
Cite this article
Takahashi, K., Okuhara, I., Tokumitsu, Y. et al. Responses to climate by tree-ring widths and maximum latewood densities of two Abies species at upper and lower altitudinal distribution limits in central Japan. Trees 25, 745–753 (2011). https://doi.org/10.1007/s00468-011-0552-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-011-0552-z