Abstract
The aim of this study was to report on the clinical characteristics and outcomes of Belgian children with chronic kidney disease (CKD). Between 2001 and 2005, we followed 143 new successive patients younger than 20 years of age with a glomerular filtration rate of <60 ml/min/1.73 m2 prospectively in a Belgian department of pediatric nephrology. The incidence of diagnosed CKD was 11.9 per million child population (pmcp), and the incidence of renal replacement therapy was 6.2 pmcp. There were 67% patients in CKD stage 3, 19% in CKD stage 4 and 14% in CKD stage 5. Patients with congenital anomalies of the kidney and urinary tract (CAKUTs), hereditary diseases and glomerular diseases were diagnosed at a median age of 1, 2 and 10 years, respectively. CAKUTs were the main causes of CKD, accounting for 59% of all cases. After 3, 4 and 5 years of follow-up, 27, 31 and 38% of patients treated conservatively, respectively, reached end-stage renal failure (ESRF). The progression rate to ESRF was eightfold higher in patients with CKD stage 4 than in those with CKD stage 3. Among our patient group, hereditary diseases progressed more rapidly to ESRF than CAKUTs. Transplantation was performed preemptively in 22% of these children. Infections and cardiovascular diseases were the main causes of death.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most epidemiological information on chronic kidney disease (CKD) originates from data available on terminal stages of CKD. Data on end-stage renal failure (ESRF) among the Belgian pediatric population collected from 1985 to 2000 were published by the European Renal Association–European Dialysis and Transplantation Association (ERA–EDTA) in 2004 [1]. Due to the scarcity of available information on early stages of CKD, a new Belgian Pediatric Nephrology Registry (BPNR) was set up in 2001 to include all children diagnosed with CKD, even those with the mildest stages.
The aims of study reported here were to provide up-to-date reliable information on the clinical characteristics and nature of primary renal diseases, to analyze the modalities of treatment, and to evaluate the outcomes, such as co-morbidities, progression to ESRF and causes of death, of Belgian children newly diagnosed with CKD and entered into the national registry between 2001 and 2005.
Methods
Data collection
Between 1 January 2001 and 31 December 2005, 143 young patients were newly entered in the Belgian registry. All of these patients were younger than 20 years of age and had an estimated glomerular filtration rate (eGFR) of <60 ml/min/1.73 m2 [2]. Serum creatinine was measured using a Roche/Hitachi Modular P analytical system with a blanked modification of the Jaffe reaction. The method is based on the Jaffe reaction as modified by Bartels et al. [3]. Demographic data (date of birth, gender) and primary renal disease were recorded for each patient.
The annual patient’s evaluation report contained the following information: stage of CKD, treatment modality, date of start of renal replacement therapy (RRT), date and type of renal transplantation (preemptive, living-related donor, cadaveric donor), co-morbidities (hypertension, anemia, mineral and bone disorders) and causes of death.
Data were entered by all Belgian departments of pediatric nephrology in Brussels, Liege, Antwerp, Ghent and Leuven through a web-based system. At least one local investigator in each department was responsible for encoding all of the cases that met the inclusion criteria at his/her center and for providing the relevant follow-up information.
A systematic audit was undertaken by the principal investigator (Tran TM Hiep) to check the completeness of all data relating to new pediatric CKD cases in Belgium. There was also an internal data validation of the registry by an annual questionnaire. Centers were asked to enter their new patients with CKD or ESRF and to mention the adaptations/changes in treatment modalities on a real-time basis. General evaluations, co-morbidities and handicaps were up-dated on a yearly basis.
Definitions used
Stages of CKD were defined according to the Kidney Disease Outcomes Quality Initiative (K/DOQI) [4], with stage 3 defined as moderately decreased GFR (30–59 ml/min/1.73 m2), stage 4 as severely decreased GFR (15–29 ml/min/1.73 m2) and stage 5 as kidney failure (GFR <15 ml/min/1.73 m2).
The date of onset of ESRF was defined as the date of start of RRT (preemptive renal transplantation or dialysis).
The waiting time for transplantation was defined as the time from the first dialysis to the first transplantation.
Anemia was defined according to The European Pediatric Peritoneal Dialysis Working Group [5].
Hypertension was determined according to the task force criteria [6].
Data analysis
The incidence of diagnosed CKD and RRT was defined as the number of new cases per year and per million children of the Belgian child population (pmcp) younger than 20 years of age, respectively. A new case of diagnosed CKD was defined as a child with CKD stages 3 to 5 who was followed in a Belgian pediatric nephrology department.
For clarity, we have classified the primary renal diseases into five groups: congenital anomalies of the kidney and urinary tract (CAKUT), hereditary renal disorders, glomerular diseases, miscellaneous causes and unknown origin.
Survival was computed till death or censored until the 21 July 2007 if alive.
Statistical analyses of progression to ESRF were estimated using the Kaplan–Meier method. Risk for progression to ESRF was assessed using multiple Cox proportional hazards regression. A two-sided p value <0.05 was considered to be statistically significant. Statistical analyses were performed using SPSS ver. 15.0 for Windows (SPSS, Chicago, IL).
Results
Demographic characteristics
From January 2001 to December 2005, 143 new patients with CKD stages 3 to 5 (82 boys, 61 girls, male/female ratio 1.3) were registered and followed prospectively. The median age at registration was 3 years (interquartile range 1–10). Patients with CAKUT, hereditary and glomerular diseases were diagnosed at a median age of 1 (interquartile range 1–5), 2 (interquartile range 1–7), and 10 years (interquartile range 4–12), respectively. Among these 143 patients, 132 (92%) were of Caucasian ethnicity, eight (6%) were African and three (2%) were Asian.
Twenty-nine and 15 new patients per year either diagnosed with CKD or as needing RRT, respectively. The mean annual Belgian population under 20 years of age from 2001 to 2005 was 2 410 206 children [7], the incidence of diagnosed CKD was at least 11.9 pmcp and the incidence of RRT was 6.2 pmcp.
Children’s characteristics at entry
Among the 143 patients, 136 (95%) were treated conservatively, and seven (5%) children required RRT immediately.
These children were classified at entry according to the K/DOQI criteria [4]. There were 96 (67%) patients with CKD stage 3, 27 (19%) with CKD stage 4 and 20 (14%) with CKD stage 5. Among the 20 children with stage 5, 13/20 (65%) were treated conservatively at entry, and the remaining 7/20 (35%) children needed immediate RRT.
Primary renal diseases
The primary renal diseases identified are listed in Table 1. CAKUTs were the main cause of CKD and accounted for 84/143 (59%) of the cases. Hereditary diseases accounted for 27/143 (19%) of the cases, nephronophthisis being the most frequent hereditary pathology (7/27, 26%). Glomerular diseases were found in 19/143 (13%) of the cases, with sequelae of hemolytic uremic syndrome being the most frequent cause of glomerular disease (9/19 children, 47%).
Primary renal diseases by stage and age are shown in Table 2. Glomerular diseases were more frequently found in patients with CKD stages 4 and 5 and in teenagers (10–19 years of age).
Co-morbidities at entry
Antihypertensive drugs were needed at entry in 20/143 (14%) patients. Among these children, 9/96 (9%) were in CKD stage 3, 6/27 (22%) in CKD stage 4 and 5/20 (25%) in CKD stage 5.
Anemia needing erythropoietin therapy at entry was found in 11/143 (8%) patients, among whom 5/27 (18%) were in CKD stage 4 and 6/20 (30%) were in CKD stage 5. Treatment was tailored in order to reach a hemoglobin concentration of at least 11 g/dl.
Oral vitamin D analogs capable of effective parathyroid suppression (mostly alfacalcidol) were needed at entry in 45/143 (32%), among whom 20/96 (21%), 12/27 (44%) and 13/20 (65%) were in CKD stages 3, 4 and 5, respectively.
CKD evolution during follow-up
The median follow-up time for the 143 patients was 3.9 years (interquartile range 2.7–4.9).
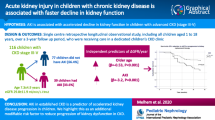
Figure 1 shows the cumulative incidence of ESRF among the 136 children treated conservatively at entry. After 3, 4 and 5 years of follow-up, 27.0 ± 3.9, 30.7 ± 4.4 and 37.6 ± 5.1% of these children reached ESRF, respectively.
Using Cox regression (Fig. 2) in order to adjust for age and gender, we found that progression rate to ESRF was 8.47-fold higher in children with CKD stage 4 than those with CKD stage 3. Hereditary diseases progressed threefold more rapidly to ESRF than CAKUTs (P = 0.003).
Renal replacement therapy
Among the 143 patients followed, 39/143 (27%) started dialysis during follow-up. Hemodialysis (HD) was performed in 19/39 (49%) children and peritoneal dialysis (PD) in 20/39 (51%). The median age at the start of dialysis was 4 years (interquartile range 1–11).
In the same period, 36/143 (25%) patients were transplanted, among whom 8/36 (22%) had a preemptive transplantation (one with a living related donor allograft). The median age at first transplantation was 5 years (interquartile range 3–11). Among all transplanted children, 31/36 (86%) received a graft from cadaveric origin and 5/36 (14%) received a graft from a living-related donor. The median waiting time for the first transplantation was 1.0 year (interquartile range: 0.6–2.1).
Mortality
Among the 143 patients, four patients (3%) died during follow-up. The first patient died because of septicemia after combined renal and liver transplantation at the age of 3 years. The second patient died while on PD with acute pneumonia at the age of 4 years. The two remaining patients, both of whom were in stage 4 CKD at entry, were on conservative treatment and had a severe cardiovascular disease; they died at the age of 1 and 13 years, respectively.
Discussion
Worldwide, the number of patients with CKD is rising markedly, especially among the adult population [8]. In contrast, pediatric CKD patients constitute a very small proportion of the total CKD population, but they pose unique challenges to pediatricians and to the health care system, which must address the primary renal disorder and many extra-renal manifestations that affect clinical and social development [8].
We carried out this nationwide study of newly diagnosed Belgian young patients with stage 3–5 CKD at the beginning of 2001. The Belgian health system allows patients free access to all medical specialties, including pediatric nephrology. Therefore, all Belgian departments of pediatric nephrology participated to the study. Whether the whole Belgian pediatric population is covered in the study, however, is uncertain, especially for adolescents, as some of these patients may be cared for and followed by adult caregivers. Consequently, we believe that the data presented here provide only a reliable evaluation of the incidence of diagnosed CKD in Belgian children and newly diagnosed adolescents who were followed by a pediatric nephrology department.
Previous studies attempting to describe the epidemiology of renal insufficiency in children have provided important estimates, but they only included severe degrees of renal insufficiency, thereby providing an incomplete picture. As CKD is often asymptomatic in its early stages, it is both misdiagnosed and underreported. Many children with renal impairment (particularly congenital abnormalities) may reach ESRF in adulthood before the disease is detected as a result of the pediatrician missing the diagnosis during the patient’s childhood. This may also explain why the proportion of CKD stages 4 and 5 represents 33% of newly diagnosed cases.
The eGFR as calculated by the Schwartz formula significantly overestimates the GFR [9]. In our study, serum creatinine was measured using a modification of the Jaffe reaction. The Schwartz formula is, however, fairly reliable. Although glomerular function needs to be measured as accurately as possible, this is difficult in children. For all new patients, we have therefore decided to measure creatinine clearance in 24-h urine collection in continent children and chromium ethylenediaminetetraacetic acid in the youngest age group.
With an incidence of diagnosed CKD of at least 11.9 pmcp of children found in this study, Belgium falls in the same category as Italy (12.1 pmcp) [10]. The incidence of RRT in this study (6.2 pmcp) was also comparable to those reported by other West European countries, such as the Netherlands (5.8 pmcp) [11] and France (7.5 pmcp) [12]. However—and unfortunately—methodological differences in case definitions and disease classifications continue to make it difficult to compare studies of the epidemiology of CKD in young populations in different geographical areas, even in Europe [13].
CAKUTs account for the greatest percentage of all cases of CKD seen in children [8]. However, whereas CAKUTs are the most common reported etiology in developed countries where CKD is diagnosed in its earlier stages, infectious or acquired causes predominate in developing countries, where patients are referred in the later stages of CKD [14]. In North America, almost one-half of all CKD cases are accounted for by patients with the diagnoses of obstructive uropathies (22%), aplasia/hypoplasia/dysplasia (18%) and reflux nephropathy (8%) [15]. The same figures have been found in Belgium and other European countries [10].
These generalizations apart, certain disorders giving rise to CKD are, indeed, more common in some countries than in others. For reasons that are as yet unexplained, hemolytic uremic syndrome represents the largest subgroup of systemic diseases responsible for CKD found in our study (6.3%). The same finding was reported in the Netherlands (5.4%) [11]. Other countries, such as Sweden and the USA, reported comparatively lower numbers of such cases [15, 16].
It has recently been recognized that children and young adults with CKD are also at a significantly increased risk of cardiovascular morbidity and even mortality compared to the general pediatric population [17]. In the USA, hypertension and anemia requiring erythropoietin therapy were found in 39 and in 18%, respectively, of all CKD patients at entry [15]. In comparison, our data show a lower number of cases receiving antihypertensive drugs and erythropoietin. This discrepancy is probably due to the higher proportion of CKD stage 3 patients reported in our study.
Although the stages of CKD are now reasonably well defined, the natural history of the early stages is variable and often unpredictable. Our data have shown, however, that the progression rate to ESRF was higher in patients in a severe stage of CKD at entry. Irrespective of the underlying kidney disease, it is clear that the risk of progression to ESRF in childhood is inversely proportional to the baseline creatinine clearance [18].
In developed countries, the distribution of dialysis modalities varies widely. As an example, PD in children is highest in Finland and Scotland, accounting for 73 and 67%, respectively of prevalent dialysis patients [1]. In our study, PD and HD were performed in the same proportion. It is possible that the choice of dialysis modalities in Belgium depends mainly on medical skills and preference of the different centers, such as trained nursing staff and comprehensive supportive services.
In Europe, preemptive transplantation was performed in 13–25% of patients during the period 1995–2000 [1]. Norway has had the highest preemptive transplant rate. Considering these figures, it is encouraging, in view of its advantages over dialysis, to note that preemptive transplantations were performed in 22% of all newly diagnosed CKD children between 2001 and 2005 in Belgium.
In general, the pattern of the causes of death in Belgium was comparable to that in other European countries, where infectious and cardiac diseases were the leading causes of death [1].
Conclusion
This study provides detailed information on the incidence, causes and overall outcomes of mild to severe CKD among Belgian children. The incidences of diagnosed CKD and RRT among our patient population are comparable to those in other European countries. CAKUTs were the main causes of CKD. Risk for rapid progression to ESRF was found to be 8.5-fold higher among patients with stage 4 CKD than those with stage 3. Hereditary diseases have a higher risk of rapidly progressing to ESRF than CAKUTs.
Abbreviations
- CKD:
-
Chronic kidney disease
- ESRF:
-
End-stage renal failure
- HD:
-
Hemodialysis
- PD:
-
Peritoneal dialysis
- pmcp:
-
Per million child population
- RRT:
-
Renal replacement therapy
References
Van der Heijden BJ, van Dijk PC, Verrier-Jones K, Jager KJ, Briggs JD (2004) Renal replacement therapy in children: data from 12 registries in Europe. Pediatr Nephrol 19:213–221
Schwartz GJ, Brion LP, Spitzer A (1987) The use of plasma creatinine concentration for estimating glomerular filtration rate in infants, children and adolescents. Pediatr Clin North Am 34:571–590
Bartels H, Bohmer M, Heierli C (1972) Serum creatinine determination without protein precipitation. Clin Chim Acta 37:193–197
Levey AS, Coresh J, Balk E, Kausz AT, Levin A, Steffes MW, Hogg RJ, Perrone RD, Lau J, Eknoyan G (2003) National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Intern Med 139:137–147
Schröder CH, European Pediatric Peritoneal Dialysis Working Group (2003) The management of anemia in pediatric peritoneal dialysis patients. Guidelines by an ad hoc European Committee. Pediatr Nephrol 18:805–809
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents (2004) The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 114[Suppl 2]:555–576
Institut national de Statistique. Population par sexe et par groupe d’âge-Belgique (2000-2007). Available at: http://www.statbel.fgov.be/homefr.asp. Accessed 26 Nov 2007
Warady BA, Chadha V (2007) Chronic kidney disease in children: the global perspective. Pediatr Nephrol 22:1999–2009
Skinner R, Cole M, Pearson AD, Keir MJ, Price L, Wyllie RA, Coulthard MG, Craft AW (1994) Inaccuracy of glomerular filtration rate estimation from height/plasma creatinine ratio. Arch Dis Child 70:387–390
Ardissino G, Daccò V, Testa S, Bonaudo R, Appiani A, Taioli E, Giuseppina M, Edefonti A, Sereni F (2003) Epidemiology of chronic kidney disease in children: Data from the Italkid Project. Pediatrics 111:382–387
Miklovicova D, Cornelissen M, Cransberg K, Groothoff DL, Schroder CH (2005) Etiology and epidemiology of end-stage renal failure in Dutch children 1987-2001. Pediatr Nephrol 20:1136–1142
Deleau J, Andre JL, Briancon S, Musse JP (1994) Chronic kidney disease in children: an epidemiological survey in Lorraine (France). Pediatr Nephrol 8:472–476
Jager KJ, Zoccali C (2008) Quality of care in end-stage renal disease: the importance of comparing ‘apples with apples’. Nephrol Dial Transplant 23:1116
Tran TMH, Janssen F, Ismaili K, Khai Minh D, Vuong Kiet D, Robert A (2008) Etiology and outcome of chronic renal failure in hospitalized children in Ho Chi Minh City, Vietnam. Pediatr Nephrol 23:965–970
North American Pediatric Renal Transplant and Collaborative Studies (2006) NAPRTCS annual report. Available at https://web.emmes.com/study/ped/annlrept/annlrept2006.pdf. Accessed 24 Aug 2006
Esbjorner E, Berg U, Hansson S (1997) Epidemiology of chronic kidney disease in children: a report from Sweden 1986-1994. Pediatr Nephrol 11:438–442
Mitsnefes MM (2002) Pediatric end-stage renal disease: heart as a target. J Pediatr 141:162–164
Ismaili K, Schurmans T, Wissing M, Hall M, Van Aelst C, Janssen F (2001) Early prognostic factors of infants with chronic renal failure caused by renal dysplasia. Pediatr Nephrol 16:260–264
Acknowledgments
Presented in part at the French-speaking Society of Pediatric Nephrology (SNP), 13–15 November 2008, Nice, France. This study was supported by a grant from the Commission Universitaire pour le Développement, Projet Inter Universitaire Ciblé (CUD-PIC 2004), Belgian Ministry of Foreign Affairs, Brussels, Belgium (to Dr. Tran Thi Mong Hiep).
Author information
Authors and Affiliations
Corresponding author
Additional information
Other participants to the study were as follows: Michelle Hall, Brigitte Adams, Ksenija Lolin, Thierry Schurmans, Laure Collard, Jean-Marin Desgrottes, Pernille Hansen, An Bael, Dominique Trouet, Johan Van de Walle, Mieke Van Dyck, Jean Herman, Elena Levtchenko, Laurence Habimana, Julie Francart and Severine Henrard.
Françoise Janssen and Annie Robert share senior co-authorship.
Rights and permissions
About this article
Cite this article
Mong Hiep, T.T., Ismaili, K., Collart, F. et al. Clinical characteristics and outcomes of children with stage 3–5 chronic kidney disease. Pediatr Nephrol 25, 935–940 (2010). https://doi.org/10.1007/s00467-009-1424-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-009-1424-2