Abstract
Background
Endoscopic management of full-thickness gastrointestinal tract defects (FTGID) has become an attractive management strategy, as it avoids the morbidity of surgery. We have previously described the short-term outcomes of over-the-scope clip management of 22 patients with non-acute FTGID. This study updates our prior findings with a larger sample size and longer follow-up period.
Methods
A retrospective analysis of prospectively collected data was conducted. All patients undergoing over-the-scope clip management of FTGID between 2013 and 2019 were identified. Acute perforations immediately managed and FTGID requiring endoscopic suturing were excluded. Patient demographics, endoscopic adjunct therapies, number of endoscopic interventions, and need for operative management were evaluated. Success was strictly defined as complete FTGID closure.
Results
We identified 92 patients with 117 FTGID (65 fistulae and 52 leaks); 27.2% had more than one FTGID managed simultaneously. The OTSC device (Ovesco Endoscopy, Tubingen, Germany) was utilized in all cases. Additional closure attempts were required in 22.2% of defects. With a median follow-up period of 5.5 months, overall defect closure success rate was 66.1% (55.0% fistulae vs. 79.6% leaks, p = 0.007). There were four mortalities from causes unrelated to the FTGID. Only 14.9% of patients with FTGID underwent operative management. There were no complications related to endoscopic intervention and no patients required urgent surgical intervention.
Conclusions
Over-the-scope clip management of FTGID represents a safe alternative to potentially morbid operative intervention. When strictly defining success as complete closure of all FTGID, endoscopy was successful in 64.4% of patients with only a small minority of patients ultimately requiring surgery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Full-thickness gastrointestinal defects (FTGID) are serious, potentially life threatening processes that present in a variety of fashions. Acute FTGID (perforations) are often the result of endoscopic or surgical intervention and are often identified and managed immediately [1]. Non-acute FTGID (leaks and fistulae) are typically more difficult to manage due to a delayed diagnosis, recent surgical interventions, or chronicity. These conditions can be associated with poor nutrition, skin irritation and breakdown, infection, sepsis, and even death [2, 3]. Traditional methods to address non-acute FTGID include treating underlying infection, optimizing nutrition, removing foreign bodies, relieving downstream obstructions, and collecting gastrointestinal (GI) effluent. Surgical intervention, which is often performed open, can be challenging. These procedures can require washout and drainage of infection, access to challenging anatomical locations, takedown or recreation of surgical anastomoses or staple lines, ostomy formation, and parenteral nutrition. The morbidity of these operations is substantial [4,5,6].
As a direct consequence of operative morbidity, there has been ongoing interest in endoscopic methods to manage FTGID [7]. Therapeutic endoscopic methods include endoluminal stents, cardiac septal occluders, endoscopic clips, fibrin sealant, endoluminal suturing devices, and over-the-scope clips [8,9,10,11,12,13,14]. Of these, only endoluminal suturing devices and over-the-scope clips are designed, and Food and Drug Administration approved, to permit full-thickness closure of the GI tract wall. Small case series demonstrate the safety and potential success of endoscopy to manage FTGID, but the current standard for managing GI fistulae and non-acute leaks remains surgical repair [4, 15]. A small minority of patients with these conditions are referred for endoscopic management to close the FTGID and avoid surgical morbidity. The majority of endoscopically managed FTGID reported in the literature are acute defects that were iatrogenic perforations created (and ultimately closed) during the course of an endoscopic intervention [16, 17].
The endoscopic management of non-acute, FTGID has been less well studied. Since the first case report in 2007, over-the-scope clip management of FTGIDs has been increasingly described in multiple case series and is a promising alternative to surgical management [9, 18,19,20,21,22,23]. Our institution has previously reported the outcomes of 22 patients managed with over-the-scope clip closure of non-acute FTGID [24]. In that series, we found that over-the-scope clip application was successful in the management of leaks in 100% of cases and in the management of chronic fistulae in 76% of cases with a median follow-up of 4.7 months. This study updates our previously reported outcomes with a larger sample size and with a longer duration of post-endoscopy observation. We hypothesized that endoscopic closure of FTGID would be a safe and effective alternative to surgical intervention.
Materials and methods
Data sources
Data were evaluated retrospectively from a prospectively maintained, institutional review board (IRB) approved database. Following IRB approval for the study, the database was augmented using the institutional electronic medical record (EMR).
Population
All patients undergoing attempted closure of FTGID utilizing over-the-scope clips between 2013 and 2019 were included. All procedures were performed by two fellowship-trained endoscopic surgeons (EMP, VVA) using the same procedure protocol at a single tertiary-care institution. Exclusion criteria included patients with acute perforations managed immediately at the time of perforation (acute FTGID) or FTGID managed with endoscopic suturing (including suturing combined with over-the-scope clip use) [25]. Patients with previous attempts at endoscopic FTGID management (including the use of over-the-scope clips, stents, endoscopic suturing and surgical interventions) prior to referral to our endoscopy service were included in the study cohort.
Procedure protocol
Prior to over-the-scope clip placement, each patient underwent a diagnostic endoscopy under carbon dioxide insufflation with fluoroscopy and contrast injection to identify the number of defects and defect location. All foreign bodies were removed (Fig. 1A). If an associated abscess or communication with the peritoneal cavity was noted, the cavity would be thoroughly irrigated and debrided (Fig. 2). Existing percutaneous drain position relative to the cavity would be evaluated and repositioned endoscopically as necessary (Fig. 1B). If existing percutaneous drains were inadequate to drain the cavity, additional percutaneous drains would be placed under fluoroscopic and endoscopic guidance (Video 1). Downstream obstructions/strictures would be treated with botulinum toxin (to lower esophageal sphincter or pylorus as indicated), endoluminal stenting, or balloon dilatation. Epithelial ablation with argon plasma coagulation would be attempted in all cases were the FTGID was not in close proximity to structures (e.g., loops of small bowel or blood vessels) that could be adversely affected by thermal spread (Fig. 1C). The OTSC clip (Ovesco Endoscopy, Tubingen, Germany) was utilized in all cases (Fig. 3). In cases with thickened and friable mucosa, an accessory device (OTSC Anchor or OTSC Twin Grasper, Ovesco Endoscopy) would be used as an adjunct to aggressive suctioning to ensure adequate purchase of the defect edges for closure (Fig. 1D). After OTSC deployment, aggressive insufflation and contrast injection with fluoroscopy would be used to ensure successful FTGID closure (Video 2). Select patients with upper GI defects would have downstream enteral access placed if necessary.
Endoscopic adjuncts utilized with the over-the-scope clip system. A Foreign body removal (pill fragments) with endoscopic grasper; B Adjustment of existing percutaneous drain into abscess cavity (including representative fluoroscopic image); C Tissue ablation of defect os using argon plasma coagulation; D Tissue acquisition device (anchor grasper) to pull defect edges into endoscope cap
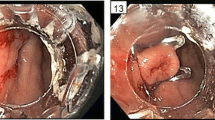
Patient with a free intraperitoneal rectal stump staple line leak after initiation of chemotherapy for management of colon cancer. A Endoscopic view of fistula opening at rectal stump staple line; B Endoscopic view of grossly contaminated peritoneal cavity; C Endoscopic view of peritoneum after washout and placement of new drain and endoscopic adjustment of existing drain; D Computed tomography image of abscess cavity following procedure prior to final drain removal (Arrow: rectal stump staple line with clip; Box: percutaneous drain)
Outcomes
Patients undergoing over-the-scope clip closure of GI defects were identified from the prospectively collected dataset. This dataset was augmented through retrospective review of the EMR to define patient demographics, patient comorbidities, GI defect characteristics (location, number, size, chronicity, etiology), number and type of procedural interventions, endoscopic adjuncts used to help manage the defects, and follow-up time. The primary outcome was long-term closure rate with success defined as clinical or radiographic evidence of complete closure of the full-thickness defect at the conclusion of follow-up. For patients without additional imaging at follow-up, clinical notes were reviewed to identify patient complaints and physical examination findings indicative of long-term failure.
A fistula was defined as a non-anatomical connection between two luminal structures or between one luminal structure and the skin. A leak was defined as the extraluminal drainage of GI contents into a contained cavity or abscess with no other connection to the GI tract.
Statistical analysis
Continuous outcome variables were compared using two-sample t-tests and categorical outcome variables were compared using Chi squared tests. SAS (version 9.4, SAS Institute Inc., Cary, NC, USA) statistical software was used for all statistical analysis. Statistical significance level was predefined as p value < 0.05.
Results
A total of 92 patients underwent 150 procedures to endoscopically close 117 FTGID (52 leaks, 65 fistulae). Mean age was 54.6 ± 15.4 years and patients were 87.0% Caucasian and 64.1% female with a mean body mass index of 30.7 ± 10.4 kg/m2 (Table 1). Most patients (72.8%) had a single FTGID. There were no complications secondary to the endoscopic intervention and no patients required urgent or emergent surgery.
Comparison was undertaken between the patients with leaks and those with fistulae (Table 2). Patients with a leak were less likely to have a prior history of radiation treatment at the defect site than those with fistulae (0.0% vs. 7.7%, p = 0.0409). Patients with a leak were more likely to have multiple FTGID than those with fistulae (1.85 vs. 1.38, p = 0.0047). There were no other statistically significant differences between the two groups. Among the leaks, there were three FTGID (5.8%) without follow-up after the last attempt at over-the-scope clip closure and among the fistulae there were five FTGID (7.7%) lost to follow-up.
The GI defect characteristics were subsequently compared (Table 3). The majority of defects were located in the upper GI tract (64.1%). The median age of the defect(s) was lower with leaks than with fistulae (9 vs. 90 days, p = 0.0179). Estimated defect diameter was smaller for leaks than for fistulae (mean 4.6 vs. 6.8 mm, p = 0.0109). Leaks were more likely to have an abscess associated with the defect (65.4% vs. 18.5%, p < 0.0001) and were more likely to have had a percutaneously placed drain present at time of endoscopic FTGID closure (67.3% vs. 10.8%, p < 0.0001). Groups were equally likely to have undergone prior procedural intervention (e.g., percutaneous drainage, endoscopic or surgical repair) for the FTGID, with overall prior intervention rate at 40.2% with a mean of 0.56 prior interventions. Of those with prior intervention, four (all in the fistula group) had undergone a previous attempt at over-the-scope clip closure prior to referral to our endoscopy service.
The median follow-up time was 5.5 months (interquartile range 2.2–14.8). The 87 patients with documented follow-up were stratified by long-term success and compared (Table 4). Patients with multiple FTGID were only counted as successfully managed if all FTGID were closed at the end of follow-up. There were 31 patients that failed over-the-scope clip management of their FTGID and 56 with successful management leading to an overall success rate of 64.4%. Long-term success was significantly higher for leaks than for fistulae (79.6% vs. 55.0%, p = 0.007) and more fistulae patients ultimately underwent definitive operative management (16.9% vs. 3.9%, p = 0.0253, Table 3). Patients who failed FTGID closure were more likely to have had a history of radiation treatment at the defect site compared to those successfully managed (12.9% vs. 1.8%, p = 0.0329). A history of previous smoking was also more common in failed patients than successfully closed patients (67.7% vs. 32.1%, p = 0.0014). No other statistically significant differences in patient characteristics were noted between the two groups.
Defect characteristics were then compared by long-term success (Table 5). Fistula defects were disproportionately represented in the failure group compared to the successful management group (73.0% vs. 27.0%, p = 0.0070). All defects which had previously undergone attempted over-the-scope clip closure prior to referral to our endoscopy service failed subsequent attempts at closure (comprising 10.8% of all failures vs. 0.0% of all successes, p = 0.0045). More attempts at defect closure were undertaken in the failure group compared to the successful management group (mean total attempts 2.78 vs. 2.07, p = 0.0083). The median age of FTGID did not differ between the success and failure groups (31 vs. 49.5 day, p = 0.1630, Fig. 4).
The 150 over-the-scope clip interventions were then stratified by GI defect type and compared (Table 6). Endoscopic adjuncts were utilized in 90.0% of procedures (Fig. 5). Tissue ablation was less common in leaks than in fistulae (39.7% vs. 69.0%, p = 0.0004). Adjustment of existing percutaneous drains was attempted more frequently with leaks (19.1% vs. 5.8%, p = 0.0112) as was establishment of distal enteral feeding access (30.2% vs. 10.3%, p = 0.0021). Successful long-term closure, with no subsequent endoscopic attempt at closure, was more common in procedures for leaks than for fistulae (63.3% vs. 43.9%, p = 0.022). Finally, interventions were stratified by long-term success and compared (Table 7). Only the adjustment of existing percutaneous drains was more common among successful over-the-scope clip interventions compared to failures (14.9% vs. 4.4%, p = 0.0369).
Discussion
This study demonstrates the efficacy and safety of over-the-scope clip management of FTGID. With 5.5 months median follow-up, we successfully closed 64.4% of all non-acute FTGID endoscopically utilizing the OTSC device. Consistent with other literature, long-term closure success differed significantly between leaks (79.6%) and fistulae (55.0%). Notably, no patients experienced complications from the endoscopic closure procedures and no patients required urgent or emergent surgical intervention. Only a small subset of patients (14.9%) ultimately underwent a surgical intervention for their FTGID.
Our endoscopic methodology to approach FTGID emphasizes the implementation of surgical principles via a flexible endoscopic platform. This includes treating underlying infection, optimizing nutrition, removing foreign bodies, de-epithelizing/ablating mucosa, relieving downstream obstructions, and collecting GI effluent. We believe that the high closure rate identified in this series (particularly with difficult to manage fistulae) is a reflection of adherence to these techniques. The liberal application of endoscopic adjuncts to FTGID closure may be as important to successful closure as the actual over-the-scope clip application itself. This study further demonstrates that the successful endoscopic management of a FTGID frequently requires multiple endoscopic procedures. We believe that despite the repeated procedures, the ultimate avoidance of the morbidity of surgical intervention is well warranted. The concept of multimodal therapy with repeat interventions as indicated is akin to the endoscopic management of GI tract bleeding.
Our management algorithm prioritizes closure of internal defects with external drainage over internal drainage whenever possible. As internal drainage of these defects has increasingly become an accepted standard of care in the management of these defects, we continue to believe that internal defect closure is preferable with appropriate defect characteristics. We do believe that small internal defects with large extraluminal collections are better served by internal drainage via double pigtail stents or endoluminal vac therapy instead of the algorithm detailed in this manuscript. However, we believe that other defects are still best suited with internal defect closure. Our preference for internal closure is evidenced by the inclusion in this series of defects with small extraluminal collections without internal or external drainage.
In our management algorithm, endoscopic stenting had a limited role as evidenced by none of the patients included in this series having a stent placed at the time of attempted endoscopic closure. Endoscopic stenting for management of FTGID is currently an off-label application of stents. Stenting in these defects has fallen out of favor in our group given experiences with stent migration and concern for the radial tension placed on the GI lumen. We favor OTSC management of FTGID except in cases of near-complete anastomotic disruption.
This study represents the largest single center study on over-the-scope clip management of non-acute FTGID. With 5.5 months of median follow-up, this study also represents one of the longest post-endoscopy follow-up periods after over-the-scope clip management of FTGID.
The single center nature of this study permits several benefits despite the retrospective data collection. First, the same technical steps were followed during every procedure. Second, given that our study includes cases performed by two fellowship-trained endoscopic surgeons, the inter-provider variability is likely limited. We believe that uniformity is a strength of this study.
The study has several limitations. We included only patients in whom an over-the-scope clip closure was attempted and did not include patients with defects repaired with other methods, or in whom over-the-scope clip therapy was determined to be not the best option for management. The results of this study may therefore be more reflective of our group’s previously described treatment algorithm than the efficacy of the OTSC system alone [7, 26, 27]. Additionally, the single center, two surgeon nature of the study also means that the results may not be reproducible if the techniques are more broadly adopted.
Most studies of over-the-scope clip management of non-acute FTGID to date have included 30 or less patients except for studies by Haito-Chavez et al., Law et al. and Honegger et al. [18, 19, 22, 23, 28,29,30,31,32]. The present study, therefore, represents one of the largest case series evaluating the success of over-the-scope clip management of non-acute FTGID with extensive follow-up. Additionally, when focusing on the subset of patients with non-acute post-operative leaks that were managed with OTSC, it is the largest case series of those particular defects.
Overall, 64.4% of all patients and 66.1% of all defects were managed successfully using the over-the-scope clip system. As noted in previous case series, the long-term successful closure of fistulae at 55.0% in our study was lower than that of leaks at 79.6%. These respective success rates are consistent with the rates in previous studies with fistulae ranging from 43–53% and leaks from 67–83% [18, 19, 22, 23, 28, 30]. A systematic review from 2018 with a total of 388 fistulae and 97 cases of anastomotic dehiscence from 30 articles reported an overall fistula closure success rate of 51.5% and an overall anastomotic dehiscence success rate of 66% [33]. We believe that our results, and those of Haito-Chavez et al. (fistulae 43%, leaks 73%), are the best estimates of true procedural success rates given that the two studies reflect the largest case series to date with over-the-scope clip management of non-acute FTGID.
We failed to identify specific patient characteristics associated with failed over-the-scope clip management of FTGID. Although history of prior radiation treatment at the defect site was more common among patients that failed over-the-scope clip management, radiation history was also more common among fistula defects and this study demonstrates a lower success rate among fistulae. Care must be taken in interpreting these results as some findings may be more indicative of the underlying disease process instead of a predictor of failure or success.
Multiple endoscopic adjuncts were utilized in conjunction with over-the-scope clip management. We believe endoscopic surgical management requires utilization of conventional surgical principles such as treatment of the underlying infection, nutritional optimization, foreign body removal, relieving downstream obstructions, and washout and drainage of any infection. We believe our data support the use of endoscopic washout and adjustment of existing percutaneous drains as these were employed more commonly in successful over-the-scope clip interventions. Care must be taken to identify all existing defects; our data demonstrated that multiple defects were present in approximately one quarter of all patients (Fig. 6).
The patients in our study underwent multiple procedural interventions aimed at managing the GI defects. This study was not designed to evaluate the cost-effectiveness of attempted over-the-scope clip closure of GI defects. Further study is necessary to determine the cost-effectiveness of over-the-scope clip given the multiple procedural interventions. Additionally, this study was not designed to evaluate the optimal timing of deciding over-the-scope clip failure and undergoing attempted definitive operative management of the FTGID.
Surprisingly, defect age did not vary significantly as expected between successfully managed FTGID and failures indicating that defect age is not a predictor of long-term success. This suggests that successful management of these defects can be undertaken regardless of defect age and that endoscopic closure attempts can be offered to all patients regardless of defect age as it appears that other defect characteristics are more predictive of long-term success.
There were four mortalities in this series that were all unrelated to the FTGID. No patients required urgent surgical intervention to address the FTGID and there were no complications (e.g., bleeding, perforation, aspiration) related to endoscopic intervention. It has previously been demonstrated that prior attempts at endoscopic closure of fistulas do not increase complications if the patient subsequently undergoes surgical revision [34]. We believe that attempts at over-the-scope clip management of FTGID are warranted despite the relatively high rates of failure of 45.0% in fistulae and 19.4% in leaks. Over-the-scope closure of FTGID represents a low-risk, high-reward procedure. The potential to avoid the morbidity of surgical revision of these defects represents a significant benefit to patients when these endoscopic interventions are successful.
The 14.9% rate of definitive operative management of FTGID is low and argues in favor of attempting endoscopic closure prior to surgical management. Even when definitive endoscopic closure failed, the endoscopic adjuncts rendered (such as drain repositioning, and placement of downstream enteral access) have the potential to improve patient management. Further, endoscopic therapy failure did not necessitate urgent surgery and did not appear to negatively affect or complicate the subsequent surgical repair. It is worth noting that many of the patients who underwent over-the-scope FTGID management were not candidates for surgical intervention. Only 41.9% of patients who failed our endoscopic closure attempts were ultimately offered a surgical intervention, suggesting that endoscopic therapy was being offered as a salvage therapy when all other options have been exhausted.
Closure of FTGID with the OTSC system falls within a spectrum of endoscopic management options. In addition to OTSC, these options include endoclips, endoscopic stents, endoscopic suturing, internal drainage with double pigtail stents, endoscopic vacuum therapy, fistula plugs, and tissue sealants [27]. The result from this series represents our use of the OTSC system within a broader spectrum of management options for FTGID closure. Future study is necessary to develop treatment algorithms that appropriately match patients to the best treatment modality for management of FTGID.
In conclusion, over-the-scope clip management of FTGID represents a safe alternative to potentially morbid operative intervention. This study underscores the utility of attempted endoscopic management of FTGID. Further study is needed to identify predictors of long-term closure and to improve success rates.
References
Verlaan T, Voermans RP, van Berge Henegouwen MI, Bemelman WA, Fockens P (2015) Endoscopic closure of acute perforations of the GI tract: a systematic review of the literature. Gastrointest Endosc 82:618–628.e615
Almahmeed T, Gonzalez R, Nelson LG, Haines K, Gallagher SF, Murr MM (2007) Morbidity of anastomotic leaks in patients undergoing Roux-en-Y gastric bypass. Arch Surg 142:954–957
Alanezi K, Urschel JD (2004) Mortality secondary to esophageal anastomotic leak. Ann Thorac Cardiovasc Surg 10:71–75
Luning TH, Keemers-Gels ME, Barendregt WB, Tan AC, Rosman C (2007) Colonoscopic perforations: a review of 30,366 patients. Surg Endosc 21:994–997
Jentschura D, Raute M, Winter J, Henkel T, Kraus M, Manegold BC (1994) Complications in endoscopy of the lower gastrointestinal tract therapy and prognosis. Surg Endosc 8:672–676
Garbay JR, Suc B, Rotman N, Fourtanier G, Escat J (1996) Multicentre study of surgical complications of colonoscopy. Br J Surg 83:42–44
Winder JS, Pauli EM (2015) Comprehensive management of full-thickness luminal defects: the next frontier of gastrointestinal endoscopy. World J Gastrointest Endosc 7:758–768
van Boeckel PG, Dua KS, Weusten BL, Schmits RJ, Surapaneni N, Timmer R, Vleggaar FP, Siersema PD (2012) Fully covered self-expandable metal stents (SEMS), partially covered SEMS and self-expandable plastic stents for the treatment of benign esophageal ruptures and anastomotic leaks. BMC Gastroenterol 12:19
Kim JS, Kim BW, Kim JI, Kim JH, Kim SW, Ji JS, Lee BI, Choi H (2013) Endoscopic clip closure versus surgery for the treatment of iatrogenic colon perforations developed during diagnostic colonoscopy: a review of 115,285 patients. Surg Endosc 27:501–504
Juza RM, Haluck RS, Pauli EM, Rogers AM, Won EJ, LynSue JR (2015) Gastric sleeve leak: a single institution’s experience with early combined laparoendoscopic management. Surg Obes Relat Dis 11:60–64
Fernandez-Esparrach G, Lautz DB, Thompson CC (2010) Endoscopic repair of gastrogastric fistula after Roux-en-Y gastric bypass: a less-invasive approach. Surg Obes Relat Dis 6:282–288
Bhardwaj A, Cooney RN, Wehrman A, Rogers AM, Mathew A (2010) Endoscopic repair of small symptomatic gastrogastric fistulas after gastric bypass surgery: a single center experience. Obes Surg 20:1090–1095
Raithel M, Albrecht H, Scheppach W, Farnbacher M, Haupt W, Hagel AF, Schellerer V, Vitali F, Neurath MF, Schneider HT (2017) Outcome, comorbidity, hospitalization and 30-day mortality after closure of acute perforations and postoperative anastomotic leaks by the over-the-scope clip (OTSC) in an unselected cohort of patients. Surg Endosc 31:2411–2425
Hourneaux de Moura DT, Jirapinyo P, Hathorn KE, Thompson CC (2019) Use of a cardiac septal occluder in the treatment of a chronic GI fistula: what should we know before off-label use in the GI tract? VideoGIE 4:114–117
Corcelles R, Jamal MH, Daigle CR, Rogula T, Brethauer SA, Schauer PR (2015) Surgical management of gastrogastric fistula. Surg Obes Relat Dis 11:1227–1232
Baron TH, Wong Kee Song LM, Zielinski MD, Emura F, Fotoohi M, Kozarek RA (2012) A comprehensive approach to the management of acute endoscopic perforations (with videos). Gastrointest Endosc 76:838–859
Bemelman WA, Baron TH (2018) Endoscopic management of transmural defects, including leaks, perforations, and fistulae. Gastroenterology 154:1938–1946.e1931
Lee HL, Cho JY, Cho JH, Park JJ, Kim CG, Kim SH, Han JH (2018) Efficacy of the over-the-scope clip system for treatment of gastrointestinal fistulas, leaks, and perforations: a korean multi-center study. Clin Endosc 51:61–65
Mennigen R, Colombo-Benkmann M, Senninger N, Laukoetter M (2013) Endoscopic closure of postoperative gastrointestinal leakages and fistulas with the over-the-scope clip (OTSC). J Gastrointest Surg 17:1058–1065
Niland B, Brock A (2017) Over-the-scope clip for endoscopic closure of gastrogastric fistulae. Surg Obes Relat Dis 13:15–20
Kirschniak A, Traub F, Kueper MA, Stuker D, Konigsrainer A, Kratt T (2007) Endoscopic treatment of gastric perforation caused by acute necrotizing pancreatitis using over-the-scope clips: a case report. Endoscopy 39:1100–1102
Haito-Chavez Y, Law JK, Kratt T, Arezzo A, Verra M, Morino M, Sharaiha RZ, Poley JW, Kahaleh M, Thompson CC, Ryan MB, Choksi N, Elmunzer BJ, Gosain S, Goldberg EM, Modayil RJ, Stavropoulos SN, Schembre DB, DiMaio CJ, Chandrasekhara V, Hasan MK, Varadarajulu S, Hawes R, Gomez V, Woodward TA, Rubel-Cohen S, Fluxa F, Vleggaar FP, Akshintala VS, Raju GS, Khashab MA (2014) International multicenter experience with an over-the-scope clipping device for endoscopic management of GI defects (with video). Gastrointest Endosc 80:610–622
Mercky P, Gonzalez JM, Aimore Bonin E, Emungania O, Brunet J, Grimaud JC, Barthet M (2015) Usefulness of over-the-scope clipping system for closing digestive fistulas. Dig Endosc 27:18–24
Winder JS, Kulaylat AN, Schubart JR, Hal HM, Pauli EM (2016) Management of non-acute gastrointestinal defects using the over-the-scope clips (OTSCs): a retrospective single-institution experience. Surg Endosc 30:2251–2258
Alli V, Doble J, Pauli E (2017) Multimodal endoscopic therapy for closure of gastro-gastric fistula: The “Triple O” Method., abstr society of American Gastrointestinal and Endoscopic Surgeons 2017 Annual Meeting, Houston, TX
Witte SR, Pauli EM (2018) Endoscopic management of gastrointestinal complications. Dig Dis Interv 2:346–358
Winder JS, Pauli EM (2019) Novel endoscopic modalities for closure of perforations, leaks and fistula in the gastrointestinal tract. Tech Gastrointest Endosc 21:109–114
Arezzo A, Verra M, Reddavid R, Cravero F, Bonino MA, Morino M (2012) Efficacy of the over-the-scope clip (OTSC) for treatment of colorectal postsurgical leaks and fistulas. Surg Endosc 26:3330–3333
Monkemuller K, Peter S, Toshniwal J, Popa D, Zabielski M, Stahl RD, Ramesh J, Wilcox CM (2014) Multipurpose use of the ‘bear claw’ (over-the-scope-clip system) to treat endoluminal gastrointestinal disorders. Dig Endosc 26:350–357
Sulz MC, Bertolini R, Frei R, Semadeni GM, Borovicka J, Meyenberger C (2014) Multipurpose use of the over-the-scope-clip system (“Bear claw”) in the gastrointestinal tract: swiss experience in a tertiary center. World J Gastroenterol 20:16287–16292
Law R, Wong Kee Song LM, Irani S, Baron TH (2015) Immediate technical and delayed clinical outcome of fistula closure using an over-the-scope clip device. Surg Endosc 29:1781–1786
Honegger C, Valli PV, Wiegand N, Bauerfeind P, Gubler C (2017) Establishment of over-the-scope-clips (OTSC®) in daily endoscopic routine. United Eur Gastroenterol J 5:247–254
Kobara H, Mori H, Nishiyama N, Fujihara S, Okano K, Suzuki Y, Masaki T (2019) Over-the-scope clip system: a review of 1517 cases over 9 years. J Gastroenterol Hepatol 34:22–30
Flicker MS, Lautz DB, Thompson CC (2011) Endoscopic management of gastrogastric fistulae does not increase complications at bariatric revision surgery. J Gastrointest Surg 15:1736–1742
Acknowledgements
The authors have no acknowledgements to make.
Funding
No internal or external financial support was used for this report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Eric Pauli receives honoraria and research support from Cook Biotech and C. R. Bard, consultant fees from Boston Scientific and Actuated Medical, and royalties from UpToDate. Salvatore Docimo receives consultant fees from Boston Scientific. David Morrell, Josh Winder, Ansh Johri, Ryan Juza, Samantha Witte, and Vamsi Alli have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MP4 141726 kb) Video 1 Endoscopic foreign body removal and irrigation of contaminated peritoneal cavity from staple line leak with endoscopic and fluoroscopic guided placement of percutaneous drain and adjustment of existing percutaneous drain
Supplementary material 2 (MP4 343045 kb) Video 2 Endoscopic closure of FTGID using OTSC clip and OTSC Anchor with endoscopic and fluoroscopic confirmation of successful closure
Rights and permissions
About this article
Cite this article
Morrell, D.J., Winder, J.S., Johri, A. et al. Over-the-scope clip management of non-acute, full-thickness gastrointestinal defects. Surg Endosc 34, 2690–2702 (2020). https://doi.org/10.1007/s00464-019-07030-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-019-07030-3