Abstract
Background
The long-term outcomes after laparoscopic surgery for colon cancer remain debatable, as randomized trials have reported similar outcomes for open and laparoscopic surgery but population-based data are scarce. Thus, it is unclear whether, outside of clinical trials, laparoscopic surgery that is performed as a standard clinical treatment has detrimental effects on patients’ long-term survival.
Methods
This study examined a unified database of 30 German regional cancer registries for patients with colorectal cancer who were diagnosed between 2003 and 2011. Among 216,682 patients with colorectal carcinoma, we identified 37,068 patients with Union for International Cancer Control stage I–III colon carcinoma (>12 cm from the anal verge), including 3825 patients (10.38 %) who underwent laparoscopic surgery. Multivariate Cox regression analyses were also used to evaluate factors that influenced the likelihood of a patient undergoing laparoscopic surgery. Kaplan–Meier analysis with the log-rank test was used to analyse differences in short- and long-term survival outcomes after open or laparoscopic surgery.
Results
Younger age, lower T-stage, and left-sided surgery were independent predictors of the patient undergoing laparoscopic surgery (all, p < 0001). The 30-day mortality rate was significantly lower for patients who underwent laparoscopic surgery for left-sided tumours (odds ratio [OR] 0.49; 95 % confidence interval [CI] 0.33–0.77). Compared to open surgery, laparoscopic surgery was a significant and independent predictor of prolonged long-term survival for right- and left-sided surgeries (right-side, OR 0.67; 95 % CI 0.56–0.82; left-sided, OR 0.70; 95 % CI 0.62–0.78).
Conclusion
Our results indicate that laparoscopic surgery provides favourable outcomes even when used outside controlled trials and should be considered as a standard treatment for patients with colon cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Laparoscopic surgery for colon cancer has slowly been adopted during the last decade in Germany, despite large randomised trials reporting similar long-term oncologic outcomes for laparoscopic and open surgery [1–7], and better short-term results for laparoscopic surgery [8]. The reason(s) for the limited adoption of laparoscopic surgery remains unclear, although it seems likely that the absence of any clear advantage in the long-term oncologic outcomes has not motivated surgeons to switch from open surgery, which is considered an established, safe, and cost-effective procedure. However, several recent population-based analyses have reported a clear benefit in the short-term results for laparoscopic surgery, compared to open surgery, in patients with colon cancer [9–11]. Nevertheless, only two studies have reported stage dependent long-term results from population-based registries of laparoscopic colon cancer surgery [12, 13].
Colorectal cancer cases in Germany have historically been tracked in 30 regional registries. Each regional registry collects data from all hospitals in a specified area. The overall coverage of these 30 registries is approximately 28 % of the German population. The German Society of Clinical Cancer registries combine the individual data from these registries every 2 years for a nationwide quality conference. For the current analysis, we used these data to compare the short- and long-term outcomes after laparoscopic and open surgery for German patients with colon cancer.
Materials and methods
The preliminary data were collected in 30 regional registries covering approximately 28 % of the German population. Thus, all levels of hospitals were included. The area that is covered by these registries is mainly the east of Germany including Bavaria and parts of Baden-Württemberg. However, these registries used different data sets for their primary data collection. Therefore, a unified “transfer data set” was created to merge all patient data into a single unified SPSS database. All patient data were anonymized before this transfer. In addition, identifying information about single hospitals was removed. This study’s design was reviewed and approved by the Ethical Review Board of University of Regensburg, Germany (approval no. 15-170-0000).
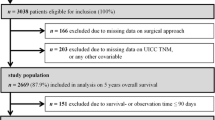
Using the unified database, we identified 216,682 patients who were diagnosed with colorectal carcinoma (ICD codes: C18–20) between 2002 and 2011. Among these patients, 83,934 patients fulfilled the criteria for stage I–III colon adenocarcinoma, which included the upper-third of the rectum (12–16 cm from the anal verge), and were treated via standard oncologic resection (right or left hemicolectomy). Patients with adenocarcinoma in the upper-third of the rectum were included because of variations in the international definitions of the upper end of the rectum, and because the operation essentially involves a left hemicolectomy with a mid-rectum anastomosis. Cases with a tumour in the transverse colon were included if they were treated using right or left hemicolectomy (n = 1160) and were excluded if another procedure was used (n = 9997). After excluding patients with incomplete information regarding the procedure type, short-term outcomes, and long-term oncologic outcomes, we identified 37,068 patients with complete records for analysis (Table 1).
In the database, information regarding adjuvant chemotherapy was recorded as “yes,” “no,” or “no information.” As only 4.1 % of the patients’ records indicated “no” for adjuvant chemotherapy, we combined the “no” and “no information” records under the assumption that these patients did not undergo adjuvant chemotherapy. Thus, for our analyses, 47.6 % of patients had undergone adjuvant chemotherapy, and 52.3 % of patients were assumed to have not undergone adjuvant chemotherapy. For all other analysis items, the groups with missing data were reported separately. The primary study outcomes were short-term (30-day mortality) and long-term (5-year overall survival) survival outcomes.
Statistical analyses
All data were analysed using SPSS software (version 23.0; SPSS Inc., Chicago, IL), and a p value of <0.05 was considered statistically significant. Categorical data were analysed using the Chi-square test. The median follow-up was calculated using an inverse Kaplan–Meier analysis. Univariate survival analyses for the different groups were performed using the Kaplan–Meier method and the log-rank test. Stratification was carried out according to UICC stage, type of surgery (laparoscopic vs. open) and localization of the tumour (right vs. left). Survival curves were generated using R software (version 3.2.2) and the KMWin Interface (version 15.2). Multivariate survival analyses were performed using a Cox proportional hazard model (with a forward selection strategy using likelihood ratio statistics), and the results were reported as hazard ratios (HRs) and 95 % confidence intervals (95 % CIs). The patients’ demographic and disease-related characteristics were classified as indicated in Table 2. The multivariate linear regression model was adjusted for the significant factors in the univariate analyses, and the results were reported as odds ratios (ORs) and 95 % CIs for 30-day mortality or the likelihood of undergoing laparoscopic surgery. Factors included in the multivariate analysis for short-term results were: type of surgery, T-stage, N-stage, age, sex, grading, R-classification and lymph node retrieval. For long-term results adjuvant chemotherapy was included additionally.
Results
Demographic characteristics
Among the 37,068 patients who were included, 33,243 (89.3 %) patients underwent open surgery and 3825 (10.7 %) patients underwent laparoscopic surgery. During the study period, the proportion of laparoscopic surgery increased from 4.3 % in 2002 to 15.4 % in 2011, although the proportions of laparoscopic surgery varied from 1.2 to 35.4 % between the different registries. The median estimated follow-up was 53.5 months (95 % CI 53.1–53.8 months). As shown in Table 2a, b, the laparoscopic surgery group included a significantly larger proportion of men (55.8 vs. 52.4 %, p < 0.001), and the patients who underwent laparoscopic surgery were approximately 2.68 years younger than the patients who underwent open surgery (p < 0.001). Differences in the specimens’ pathological characteristics were also observed, with the laparoscopic surgery group exhibiting a trend towards lower tumour stages (T, N, and Union for International Cancer Control staging) and better differentiation. The open surgery group exhibited a higher number of retrieved lymph nodes, although the proportion of R1/2 resections was also larger in the open surgery group. Multivariate logistic regression was used to evaluate the pre-surgery factors’ effects on the likelihood of undergoing laparoscopic surgery (Table 3), and we found that T-stage (T2: OR 0.467, T3: OR 0.295, T4: OR 0.193; all p < 0.001) exhibited a strong influence and age exhibited a lesser influence (OR 0.98/10 years, p < 0001), on the use of laparoscopic surgery.
Perioperative mortality
In the univariate analysis (Table 4), the 30-day mortality rate was significantly lower in the laparoscopic surgery group (0.9 vs. 3.3 %, p < 0.001). Even when we grouped the conversions (30-day mortality 3.68 %) with laparoscopic surgeries (intent-to-treat) in the multivariate analysis (Table 5), we observed a significantly reduced OR for 30-day mortality in the laparoscopic left-sided surgery group (0.48, p < 0.001). The right-side group exhibited a similar trend (OR 0.6), although this trend was not statistically significant (p = 0.062). Age, male sex, higher T-stage, and non-R0 resections were independently associated with an increased risk of post-operative mortality. A high lymph node count was not associated with post-operative mortality, although a low lymph node count was an independent risk factor for post-operative mortality (OR 1.49, p < 0.0001).
Long-term survival
In the univariate intent-to-treat analysis, the laparoscopic surgery groups (both right- and left-sided) exhibited a prolonged long-term survival, with an especially prominent increase for stage III patients (Table 6). This association remained even after we excluded cases that experienced a survival of ≤30 days (Fig. 1A–D). Open and laparoscopic right hemicolectomy provided worse outcomes in stage III patients, compared to left colic resection, and right laparoscopic resection was superior to all other treatments in stage II patients. In the multivariate intent-to-treat analysis, laparoscopic resection remained a highly significant predictor of prolonged long-term survival, regardless of the tumour site. Furthermore, adverse tumour-related factors (T-stage, N-stage, and R-classification) were independent predictors of a poor prognosis (Table 7).
Discussion
This population-based study revealed that, compared to open surgery, laparoscopic surgery for colon cancer also provided favourable long-term outcomes. Furthermore, our results indicate that laparoscopic left-sided surgery was associated with a reduced post-operative mortality rate. However, both findings are not entirely congruent with those of previous meta-analyses, [14] which reported that laparoscopic surgery was associated with a reduction in post-operative morbidity, but that there were no significant differences in the short- or long-term mortality outcomes.
It may be speculated that these discrepancies may be related to the exclusion a large proportion of the patients from our analyses for various reasons. However, inclusion of 37,068 patients with colon cancer from 30 registries over a 9-year period likely provides a representative picture of the standard care in Germany during the study period. In addition, our findings are similar to the results from British, French, and American population-based registry studies, which reported a marked reduction in the rates of 30-day or in-hospital mortality among patients who underwent laparoscopic surgery [9–11].
In contrast to these population-based studies’ findings, meta-analyses of large randomised trials have reported that laparoscopic surgery provided a reduction in the total complication rate, earlier resumption of gastrointestinal function, and a reduction in the average hospital stay, but there was no corresponding reduction in post-operative mortality rates [7, 8]. However, it is important to note that the mortality rate in the randomised trials was only 1.1 % [7], which is considerably lower than the mortality rates in the population-based studies (France: 4.4 %, UK: 3.3 %, US: 4.1 %)0.[8,9,13] including our analysis.
Meta-analyses of the prospective randomised trials that have evaluated the long-term outcomes after laparoscopic colon and colorectal cancer surgery have not revealed any significant differences in overall survival for stages I–III [6–8]. One population-based study assessing the very early phase (1998–2002) of laparoscopic colon cancer surgery reported a significant benefit for stages I and II but not for stage III. A recent Norwegian population-based study showed better results for the laparoscopic approach for the first 2 years after surgery in stages I–III, which disappeared after correction for emergency operations [13]. Two other population-based studies have investigated long-term survival of which two are of limited value because of either low numbers of laparoscopic cases [15] or a lack of analysis of stage dependent survival [16]. Two additional studies of non-population-based registries have reported no beneficial effects for laparoscopic resection [17, 18].
Unlike the previous trials, we observed better outcomes for stages I–III, regardless of the tumour site. Furthermore, this relationship remained even after we excluded patients who died within 30 days, which prevents any bias related to the better short-term results in the laparoscopic surgery group. The discrepancy between our findings and those of the randomised trials may partially be related to patient selection, as it is conceivable that patients who were especially suitable would have undergone laparoscopic surgery, and that these patients would likely have been young, had a low body mass index, had low tumour stages, had few comorbidities and had a scheduled operation. Indeed, we observed that the patients in the laparoscopic surgery group were significantly younger and had lower tumour stages, which is consistent with this explanation. Therefore, it is clear that patient selection has taken place in clinical practice; however, this selection likely does not fully explain our findings, as the beneficial effect of laparoscopic surgery was independent of tumour stage in our multivariate analyses, which included large numbers of patients in each group. Unfortunately, we could not control for obesity, as the related data were not recorded in our registry. Obesity is a well-known risk factor for anastomotic leakage and poor short-term outcomes. However, a recent large cohort analysis revealed that the long-term survival outcomes were better among obese patients, compared to lean patients [19]. Also, Makino et al. found no effect of obesity on long-term survival in their single-centre series of laparoscopic colorectal resections [20].Therefore, although it is likely that patient selection affects the use of laparoscopic surgery, it is unlikely that this selection fully explains our findings.
Comorbidity may also be an important selection factor that may affect long-term outcomes in different ways. It increases the risk of all-cause long-term mortality and also is a reason for performing limited resections, thus causing an increase in tumour-related mortality. This effect cannot entirely be ruled out by our data because we were only able to include age but not comorbidity itself into multivariate analysis. Also, we could not control for emergency operations as it was possible in the Norwegian study [13]. However, it is noteworthy that in that study without correction for emergency status higher survival rates in the laparoscopy group were only noted in the first 2 years after surgery. This difference disappeared after multivariable analysis including the emergency status. Conversely, in our study the difference tends to increase with time suggesting that the emergency status may only explain differences in the middle but not in long-term survival that we have observed.
In addition to patient-related factors, technical issues may be important to explain the higher survival rate after laparoscopic surgery. Long-term outcomes vary for different surgeons presumably according to the degree of compliance with the principles of complete mesocolic excision (CME) [21–23]. In this context, the integrity of the specimen’s mesocolon and the degree of central vascular ligation are considered key factors for ensuring positive outcomes. Both, laparoscopic surgery and compliance with CME principles are technically more demanding than standard open surgery and are typically performed by dedicated, well-trained surgeons who are more likely to be specialized and more familiar with current standards and advances in oncologic surgery. Therefore, we hypothesize that a higher rate of high-quality specimens in the laparoscopic surgery group may explain the long-term benefit that we have observed. Thus, selection as an explanation for the beneficial effect for laparoscopic surgery observed in this study would be more a selection of surgeons and to a lesser extend a selection of patients. This idea is supported by the finding of West et al. [24], who reported that the influence of dissection in the right–mesocolic-plane increased from stage I to stage III, which provided an approximate difference of 15 % in the 5-year overall survival rate. This pattern is very similar to what we observed in our study and is thought to be a main contribution to the beneficial effect of CME. The concept of CME was published in 2009; however, many German surgeons have performed CME type surgery long before this date especially for left-sided tumours. This may explain the difference especially in stage III in our observation compared to the early analysis of the US data [12] and also to the Norwegian analysis [13].
As in randomized trials [25], we observed a lower lymph node count after laparoscopic surgery which seems to contradict the pervious statement. However, in all groups still in a large proportion of more than 12 lymph nodes were retrieved as required for adequate staging. In addition, current evidence suggests that preservation of the integrity of the mesocolon [24] and the degree of central dissection [26] may be more important than the overall size of the specimen.
The strength of the study is the population-based setting with a high number of patients from 30 different regional registries. The data appear to be representative and comparable to other large cohorts, which is demonstrated by similar post-operative mortality. Moreover, specific findings that have previously been described in other population-based investigations could be found in our data as well such as the lower post-operative mortality in the laparoscopy groups and the difference in survival for right- and left-sided tumours [27].
The shortcomings of our analysis are that we excluded a large proportion of patients due to incomplete data. In addition, the registry did not contain more detailed data that would have been helpful for the multivariate analyses (e.g., body mass index, emergency surgery status, and comorbidities).
In conclusion, our population-based analyses revealed that, compared to open surgery, laparoscopic surgery was associated with favourable short- and long-term outcomes for patients with colon cancer. Given the previous randomized reports that laparoscopic surgery was associated with better short-term outcomes and equal long-term outcomes, our data indicate that laparoscopic surgery for patients with colon cancer performed by well-trained surgeons can be encouraged as a first choice.
References
Clinical Outcomes of Surgical Therapy Study G (2004) A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med 350:2050–2059
Abraham NS, Young JM, Solomon MJ (2004) Meta-analysis of short-term outcomes after laparoscopic resection for colorectal cancer. Br J Surg 91:1111–1124
Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AM, Heath RM, Brown JM, Group UMCT (2007) Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol Off J Am Soc Clin Oncol 25:3061–3068
Colon Cancer Laparoscopic or Open Resection Study G, Buunen M, Veldkamp R, Hop WC, Kuhry E, Jeekel J, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy A, Bonjer HJ (2009) Survival after laparoscopic surgery versus open surgery for colon cancer: long-term outcome of a randomised clinical trial. Lancet Oncol 10:44–52
Bagshaw PF, Allardyce RA, Frampton CM, Frizelle FA, Hewett PJ, McMurrick PJ, Rieger NA, Smith JS, Solomon MJ, Stevenson AR, Australasian Laparoscopic Colon Cancer Study G (2012) Long-term outcomes of the australasian randomized clinical trial comparing laparoscopic and conventional open surgical treatments for colon cancer: the Australasian Laparoscopic Colon Cancer Study trial. Ann Surg 256:915–919
Theophilus M, Platell C, Spilsbury K (2014) Long-term survival following laparoscopic and open colectomy for colon cancer: a meta-analysis of randomized controlled trials. Colorectal Dis Off J Assoc Coloproctol Great Br Ireland 16:O75–O81
Schwenk W, Neudecker J, Haase O (2014) Current evidence for laparoscopic surgery of colonic cancer. Der Chirurg; Zeitschrift fur alle Gebiete der operativen Medizen 85:570–577
Ohtani H, Tamamori Y, Arimoto Y, Nishiguchi Y, Maeda K, Hirakawa K (2012) A meta-analysis of the short- and long-term results of randomized controlled trials that compared laparoscopy-assisted and open colectomy for colon cancer. J Cancer 3:49–57
Panis Y, Maggiori L, Caranhac G, Bretagnol F, Vicaut E (2011) Mortality after colorectal cancer surgery: a French survey of more than 84,000 patients. Ann Surg 254:738–743 (discussion 743-734)
Mamidanna R, Burns EM, Bottle A, Aylin P, Stonell C, Hanna GB, Faiz O (2012) Reduced risk of medical morbidity and mortality in patients selected for laparoscopic colorectal resection in England: a population-based study. Arch Surg 147:219–227
Juo YY, Hyder O, Haider AH, Camp M, Lidor A, Ahuja N (2014) Is minimally invasive colon resection better than traditional approaches?: first comprehensive national examination with propensity score matching. JAMA Surg 149:177–184
Bilimoria KY, Bentrem DJ, Nelson H, Stryker SJ, Stewart AK, Soper NJ, Russell TR, Ko CY (2008) Use and outcomes of laparoscopic-assisted colectomy for cancer in the United States. Arch Surg 143:832–839 (discussion 839-840)
Stormark K, Søreide K, Søreide JA, Kvaløy JT, Pfeffer F, Eriksen MT, Nedrebø BS, Kørner H (2016) Nationwide implementation of laparoscopic surgery for colon cancer: short-term outcomes and long-term survival in a population-based cohort. Surg Endosc. doi:10.1007/s00464-016-4819-8
Bonjer HJ, Hop WC, Nelson H, Sargent DJ, Lacy AM, Castells A, Guillou PJ, Thorpe H, Brown J, Delgado S, Haglind E, Pahlman L, Transatlantic Laparoscopically Assisted vs Open Colectomy Trials Study G (2007) Laparoscopically assisted vs open colectomy for colon cancer: a meta-analysis. Arch Surg 142:298–303
Cummings LC, Delaney CP, Cooper GS (2012) Laparoscopic versus open colectomy for colon cancer in an older population: a cohort study. World J surg Oncol 10:31
Dobbins TA, Young JM, Solomon MJ (2014) Uptake and outcomes of laparoscopically assisted resection for colon and rectal cancer in Australia: a population-based study. Dis Colon Rectum 57:415–422
Kube R, Gastinger I, Mroczkowski P, Ptok H, Wolff S, Lippert H (2011) The care of patients with colon cancer: current treatment, and evaluation of new surgical approaches. Deutsches Arzteblatt Int 108:41–46
Sammour T, Jones IT, Gibbs P, Chandra R, Steel MC, Shedda SM, Croxford M, Faragher I, Hayes IP, Hastie IA (2015) Comparing oncological outcomes of laparoscopic versus open surgery for colon cancer: analysis of a large prospective clinical database. J Surg Oncol 111:891–898
Renfro LA, Loupakis F, Adams RA, Seymour MT, Schmoll HJ, Douillard JY, Hurwitz H, Fuchs CS, Diaz-Rubio E, Porschen R, Tournigand C, Chibaudel B, Falcone A, Tebbutt NC, Punt CJ, Hecht JR, Bokemeyer C, Van Cutsem E, Goldberg RM, Saltz LB, de Gramont A, Sargent DJ, Lenz HJ (2015) Body mass index is prognostic in metastatic colorectal cancer: pooled analysis of patients from first-line clinical trials in the ARCAD database. J Clin Oncol 34:144–150
Makino T, Trencheva K, Shukla PJ, Rubino F, Zhuo C, Pavoor RS, Milsom JW (2014) The influence of obesity on short- and long-term outcomes after laparoscopic surgery for colon cancer: a case-matched study of 152 patients. Surgery 156:661–668
Bertelsen CA, Neuenschwander AU, Jansen JE, Wilhelmsen M, Kirkegaard-Klitbo A, Tenma JR, Bols B, Ingeholm P, Rasmussen LA, Jepsen LV, Iversen ER, Kristensen B, Gogenur I, Danish Colorectal Cancer G (2015) Disease-free survival after complete mesocolic excision compared with conventional colon cancer surgery: a retrospective, population-based study. Lancet Oncol 16:161–168
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S (2009) Standardized surgery for colonic cancer: complete mesocolic excision and central ligation–technical notes and outcome. Colorectal Dis Off J Ass Coloproctol Great Br Ireland 11:354–364 (discussion 364-355)
Bokey EL, Chapuis PH, Dent OF, Mander BJ, Bissett IP, Newland RC (2003) Surgical technique and survival in patients having a curative resection for colon cancer. Dis Colon Rectum 46:860–866
West NP, Morris EJ, Rotimi O, Cairns A, Finan PJ, Quirke P (2008) Pathology grading of colon cancer surgical resection and its association with survival: a retrospective observational study. Lancet Oncol 9:857–865
Kuhry E, Schwenk WF, Gaupset R, Romild U, Bonjer HJ (2008) Long-term results of laparoscopic colorectal cancer resection. The Cochrane database of systematic reviews:CD003432
West NP, Kobayashi H, Takahashi K, Perrakis A, Weber K, Hohenberger W, Sugihara K, Quirke P (2012) Understanding optimal colonic cancer surgery: comparison of Japanese D3 resection and European complete mesocolic excision with central vascular ligation. J Clin Oncol Official J Am Soc Clin Oncol 30:1763–1769
Benedix F, Kube R, Meyer F, Schmidt U, Gastinger I, Lippert H, Colon/Rectum Carcinomas Study G (2010) Comparison of 17,641 patients with right- and left-sided colon cancer: differences in epidemiology, perioperative course, histology, and survival. Dis Colon Rectum 53:57–64
Acknowledgments
The authors thank all participating registries for their cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Drs. Stefan Benz, Hagen Barlag, Michael Gerken, Alois Fuerst and Monika Klinkhammer-Schalke have no conflicts of interest or financial ties to disclose.
Rights and permissions
About this article
Cite this article
Benz, S., Barlag, H., Gerken, M. et al. Laparoscopic surgery in patients with colon cancer: a population-based analysis. Surg Endosc 31, 2586–2595 (2017). https://doi.org/10.1007/s00464-016-5266-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-016-5266-2