Abstract
Background
Although it has been 25 years since the introduction of laparoscopy to cholecystectomy, outcomes remain largely unchanged, with rates of bile duct injury higher in the modern age than in the era of open surgery. The SAGES Safe Cholecystectomy Task Force (SCTF) initiative seeks to encourage a culture of safety in laparoscopic cholecystectomy (LC) and reduce biliary injury. An expert consensus study was conducted to identify interventions thought to be most effective in pursuit of this goal.
Methods
An initial list of items for safer practice in LC was identified by the SCTF through a nominal group technique (NGT) process. These were put forward to 407 SAGES committee members in two-stage electronically distributed Delphi surveys. Consensus was achieved if at least 80 % of respondents ranked an item as 4 or 5 on a Likert scale of importance (1–5). Additionally, respondents ranked five top areas of importance for the following domains: training, assessment, and research.
Results
Thirty-nine initial items were identified through NGT. Response rates for each Delphi round were 40.2 and 34 %, respectively. Final consensus was achieved on 15 items, the majority of which related to non-technical factors in LC. Key domains for training, assessment, and research were identified. Critical view of safety was deemed most important for overall safety, as well as training and assessment of LC. Intraoperative cholangiography was identified as an additional priority area for future research.
Conclusions
Consensus items to progress surgical practice, training, assessment, and research have been identified, to promote safe practice and improve patient outcomes in LC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduced in the late 1980s [1, 2], the laparoscopic approach to cholecystectomy has revolutionised the surgeon’s approach to the procedure. Laparoscopy has been shown to offer improved cosmesis, reduced length of inhospital stay, and more rapid convalescence compared with open cholecystectomy [3, 4]. Over 700,000 cholecystectomies are now performed annually in the USA alone, the vast majority laparoscopically [5].
The rapid uptake of laparoscopy, however, has not been without cost. Despite studies demonstrating equivalent morbidity and mortality rates for laparoscopic and open approaches [3, 6], early studies suggested that this was tempered by the fact that a reduction in minor complications such as wound infection was, in fact, offset by an increase in major bile duct injury (BDI) complications [7]. The Southern Surgeons’ Club’s 1991 analysis of 1518 laparoscopic cholecystectomies (LCs) reported a BDI rate of 0.5 % [7], up to five times higher than the previously cited rates of 0.1–0.2 % for open cholecystectomy [8, 9].
Regrettably, this higher rate of BDI has persisted in the modern era of LC. Recent studies drawing upon large-scale or national database cohorts have reported rates of BDI ranging from 0.2 to 1.1 % [10–13]. The severe nature of BDI following LC is reflected in the greatly increased risk of serious morbidity, mortality, and length of stay [14], in addition to reduced quality of life [15] and long-term survival [16, 17]. Furthermore, the economic impact of BDI must also be considered. Analyses of litigation claims demonstrate BDI to be the cause of the majority of lawsuits following LC, resulting in average settlements of up to $500,000 USD [18–20]. One estimate places the annual cost of litigation related to BDI in excess of $1 billion USD in the USA alone [21].
Efforts to improve safety in LC have greatly increased the body of knowledge across nearly all domains of care relevant to cholecystectomy. These include timing of the procedure and patient selection [22], to training and assessment of surgeons performing LC [23]. Intraoperative processes to ensure safety of the procedure have been championed, such as photographic documentation [24] of the “critical view of safety” first described by Strasberg and colleagues almost 20 years ago [25], and the use of intraoperative cholangiography [26].
Despite this, many of the above-described interventions have failed to achieve significant uptake, and outcomes have remained largely unchanged. BDI rates remain significantly higher now than a quarter of a century ago in the era of open surgery [27]. It is likely that a new, consensus-driven approach is needed, if change is to be achieved.
The Society of American Gastrointestinal Endoscopic Surgeons (SAGES) has launched a new initiative in this context, to improve safety in laparoscopic cholecystectomy (LC), spearheaded by the Safe Cholecystectomy Task Force (SCTF). This group’s objective is to encourage and catalyze a universal culture of safety for cholecystectomy with the goal that its adoption would result in a decrease in the incidence of BDI. This study, representing the first output from the Task Force, sought to identify the future directions for process improvement, training and research with the greatest potential to improve outcomes in LC, through expert consensus.
Methods
Initiation of the SAGES Safe Cholecystectomy Task Force
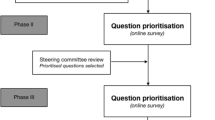
The current leadership of SAGES has placed efforts to enhance patient safety and improve surgical outcomes at the center of the society’s strategy. The improvement of safety in LC has been given a central role, directed in part by a group of practicing laparoscopic surgeons with an interest in patient safety, the SCTF. To ensure that the interests of the broader surgical community were represented and to create a “road map” for this initiative, a two-phase consensus process, consisting of nominal group and Delphi techniques, was used.
Nominal group technique
In April 2014, the SAGES SCTF, a group of expert gastrointestinal surgeons with an interest in surgical education and patient safety, convened a meeting to generate an initial list of factors to improve safe practice in LC. The nominal group technique (NGT) was applied. NGT is a highly structured process of idea generation, with resulting ideas aggregated according to importance and relevance [28]. It allows ideas to be generated by individuals without the risk of decision being biased by choices or opinions of other group members, before combining ideas with the rest of the group to be agreed on by a majority.
Participants were asked to suggest critical safety factors for LC across three domains: operative (technical), perioperative, and non-technical factors. Ideas across all participants were collated, with participants then asked to each select and rank factors in order of their perceived importance. A rank-ordered list was presented and finalized through the combining of similar or redundant items, and exclusion of irrelevant ones, as agreed by the group majority.
Delphi consensus
The list of items generated by NGT was then subjected to a Delphi process to achieve consensus. The Delphi process is an iterative consensus technique which provides anonymous feedback to participants in each round about the choices made by their peers in previous rounds. In this manner, individuals are free to change their opinion based on the group’s influence, but without the risk of disproportionate bias from any particularly senior or dominant individuals, as might otherwise result from live discussion [29].
All SAGES committee members were invited to participate in the two-round Delphi consensus via an e-mailed online survey, with explicit instructions to participate only if they performed LC as part of their current independent surgical practice. Participants completed a demographic questionnaire, before ranking each NGT item on a Likert scale of 1 (not at all important) to 5 (very important) for their importance to safe practice in LC. Free-text space was given to suggest additional items, if necessary. In addition, participants were asked to name the five most important NGT-determined items in rank order of perceived relevance to (1) resident education, (2) performance assessment, and (3) future scientific research for safe practice in LC. A reminder was sent at 2 weeks, and the first-round survey closed at 4 weeks following the initial invitation.
Results (mean ± SD of each item) were circulated as part of the second round survey 2 weeks later, in which participants re-ranked items in a similar manner. Again, an electronic reminder was circulated at 2 weeks, and the Delphi process closed at 4 weeks following initial invitation.
Data analysis
Analysis of the second round Delphi results was conducted in Microsoft Excel 2011 (Microsoft Corp, Redmond, WA). For the primary question—determining factors critical to safe practice of LC—consensus was reached if at least 80 % of respondents ranked an item as 4 or higher (on a Likert scale of 1–5). Each item was designated as relating to a technical, non-technical, or perioperative domain of care as determined during the initial NGT process. For responses to the secondary questions—naming the top five factors for education, assessment, and research, items were ranked in terms of number of times they were named as a top-five item; if two items achieved the same number of nominations, the final order was determined by higher mean ranking.
Results
Nominal group technique
Eleven surgeons (members of the SAGES SCTF) took part in the NGT. All were board-certified general surgeons and practiced primarily in academic centers (8/11), community hospital (1/11), private practice (1/11), no reply given (1/11), with a median 22 years of independent practice (range 6–60 years). Most indicated an annual volume of 50–100 LCs [5/11; also 100–200 LC/year (3/11), 20–50 LC/year (2/11), 10–20/year (1/11)], 8 of 11 had completed over 1,000 previous LCs 2 of 11 500–1,000 LCs, and 1 of 11 300–500 LCs.
Through the NGT process, a total of 39 factors were identified and deemed relevant to safe practice in LC (Table 1). These were included in the subsequent Delphi process.
Delphi consensus
Demographic data
Four hundred and seven SAGES committee members received an invitation to complete each round of the Delphi consensus survey (SurveyMonkey, Palo Alto, CA).
The first-round survey was completed by 164 respondents (40.2 % response rate), with the second round completed by 139 respondents (34 %). Respondents referred to themselves as general surgeons (78/164, 47.6 %), upper gastrointestinal surgeons (35/164, 21.3 %), bariatric surgeons (31/164, 18.9 %), colorectal surgeons (7/164, 4.3 %), hepatobiliary surgeons (7/164, 4.3 %), pediatric surgeons (5/164, 3.1 %), and endocrine surgeons (1/164, 0.6 %). Approximately 90 % (148/164) possessed relevant board certification and had 12.4 ± 9.4 (mean ± SD) years of independent practice. One hundred and thirteen of 164 (68.9 %) respondents conducted a majority of their practice in a university-affiliated center, 34 of 164 (20.7 %) in a community teaching hospital, and 17 of 164 (10.4 %) in a community non-teaching hospital. Most had access to specialist services such as endoscopy (162/164, 98.8 %), endoscopic retrograde cholangiopancreatography (156/164, 95.1 %), endoscopic ultrasound (145/162, 88.4 %), and interventional radiology (151/164, 92.1 %). Only two respondents (1.2 %) reported access to none of the above.
Respondents were asked to indicate their surgical experience. The mode was 21–50 annual LCs (55/164, 33.5 %), with a total of 201–300 previously completed LCs (55/164, 33.5 %). One hundred and sixty of 164 (97.6 %) were comfortable performing and interpreting IOC. While 29 of 164 (17.7 %) performed IOC routinely (>90 % of cases), the mode (85/164, 51.8 %) performed IOC less than 10 % of the time. One hundred and ten of 164 (67.1 %) were comfortable performing transcystic laparoscopic common bile duct exploration (CBDE), and 59 of 164 (36.0 %) reported the ability to perform transcholedochal CBDE; 25 of 164 (27.4 %) did not perform CBDE. The mode annual volume was less than five CBDEs (92/164, 56.1 %).
Delphi consensus results
Twenty-six factors were identified by 80 % or greater of respondents as being “important” or “very important” through the first Delphi round (Appendix 1). Following the second Delphi round, final consensus was achieved on fifteen items (Table 2).
Across both rounds of the Delphi process, the same items were ranked at the top as the five most important factors to safe practice, with two major themes emerging. The first related to safe identification of biliary anatomy: (rank 1) establishing the critical view, (2) understanding of relevant anatomy, and (3) appropriate retraction/exposure. The second theme focused on surgical decision making: (4) knowing when to call for help and (5) recognizing the need for conversion or an alternate procedure (such as subtotal cholecystectomy).
Asked to identify factors relevant specifically to resident training for LC (Table 3, full results Appendix 2), experts identified the top five items as being (1) establishing the critical view, (2) understanding of relevant anatomy, (3) appropriate retraction and exposure, (4) correct approach to dissection of Calot’s triangle from high on the gallbladder, and (5) knowing when to call for senior assistance.
For assessment (Table 3, full results Appendix 3), experts again included (1) establishing the critical view, and (2) understanding of anatomy, as well as (3) appropriate retraction and exposure, in addition to (4) surgeon ability to perform intraoperative cholangiography and (5) appropriate use of energy devices.
Finally, the most commonly identified topics for research (Table 3, full results Appendix 4) identified were as follows: (1) surgeon ability to perform intraoperative cholangiography, (2) establishing the critical view, (3) optimal timing of LC with reference to the disease history, (4) appropriate use of energy devices, and (5) how to recognize the need for conversion or alternate procedure.
Discussion
Twenty-five following the introduction of LC, bile duct injury remains a significant risk of this procedure. That such a serious complication may result from a very common procedure undertaken for benign disease, underlines the need for renewed initiative to improve safety in LC. It is estimated that 1,400–7,700 patients a year will suffer a major bile duct injury during LC [27]. The number is staggering if one considers that the vast majority of these patients come for an outpatient procedure and are expected to be back to their normal activities within 2 weeks or less.
This study presents the results of a structured Delphi consensus study, with participants drawn from the leading members of the world’s largest society for laparoscopic surgery, SAGES. The resulting consensus list forms the cornerstone for what we hope will catalyze a renewed push to objectively define and standardize measures to ensure safe practice in LC. Consensus was reached on 15 final items, with a “top five” list of priorities additionally identified for the specific domains of residency training, assessment, and research.
Of the 15 consensus items, half of the top 10 consensus items related to non-technical skills; of the remainder, four related to technical items and one item to perioperative care. Thus, the consensus has reflected the evolution of surgery over the past few decades, which has seen a shift in the historical emphasis on technical ability, to incorporate a growing understanding of the importance of non-technical performance [30] and perioperative processes [31] for surgical outcomes.
The continued need to improve knowledge in this area is highlighted by the 2003 analysis of 252 bile duct injury cases [32] reported by Way and colleagues. In this study, the authors reported that in 97 % of cases, non-technical errors—predominantly relating to perceptual errors, in which the operator was led to make false assumptions regarding the position of the bile duct in relation to the anatomy at hand—were at the root cause of the injury. Many of their recommendations to avoid BDI are mirrored in the leading items identified by this consensus study, with the establishment of the critical view (Delphi consensus rank 1) and understanding the relevant anatomy (2), aided by adequate retraction and visualization (3), all crucial to correct identification of the cystic and common bile ducts.
Considering the areas of priority for implementing change for the next generation of laparoscopic surgeons, the results were similar for both training and assessment domains. While some items are already the subject of active research and educational interventions, such as the SAGES Fundamental Use of Surgical Energy (FUSE) program [33] to improve and assess the appropriate use of energy devices, many other areas remain unaddressed.
On the basis of this consensus study, future efforts must prioritize trainees’ understanding of the critical view and it is formalized assessment. In addition to setting training goals, the exploration and development of valid and unobtrusive assessment tools is required. Routine photographic or video-based documentation of the CVS has been championed in some countries such as the Netherlands, where it has been endorsed by the Dutch Surgical Society [34] and is utilized there by the large majority of surgeons [35]. However, resource limitations, such as a lack of necessary equipment to facilitate image or video recording and linkage to medical records, may mean that such practice is more difficult to achieve on a broader international scale. Future efforts will require a pragmatic approach to introducing feasible and universally implementable interventions.
In addition to the items prioritized for research through the Delphi consensus as presented in this study, future research must also consider appropriate outcome measures. Commonly cited outcomes include morbidity, bile duct injury rates, time taken, or rates of conversion. However, these represent an outcomes-based, surgical perspective. It does not take into account what should perhaps be a more patient-focused approach in what is, after all, an operation performed for benign disease. Future studies should thus place greater emphasis on patient-oriented outcomes such as resolution of symptoms, or quality of life, in addition to traditionally reported surgical measures.
The strengths of the Delphi process are highlighted by the gradual refinement of items throughout the stages of this study. From 39 original NGT items, the staged Delphi process reduced these first to 26 and then to 15 items, with the Delphi process ensuring unbiased opinion sharing and achieving consensus across a larger group. Though the participants were drawn from a select group representing a small proportion of the greater community of laparoscopic gastrointestinal surgeons with a potential overrepresentation of academic centers, as SAGES committee members, the participating surgeons also represent those nominated to represent their peers on a national and international level. A suitable response rate (mean response rate 37 %) for the Delphi survey will have minimized any potential non-response bias [36].
The results of this consensus process form a preliminary “road map” toward the ultimate goal—improved outcomes and reduced injuries in LC. They also, however, illustrate some of the difficulties to be encountered along the way. Despite 39 items being put forward by the original Task Force group, consensus could only be reached on little more than a third of them (15/39, 38 %). Participant “buy-in” is critical to any initiative which takes on the difficult task of introducing change in established practice. Seeking to also instill changes in organizational safety culture is even more difficult. Recent studies of the WHO Safer Surgery Checklist, for example, have demonstrated the potential for failure which looms if initiatives are introduced without proper training, buy-in, and leadership—resulting in completed “tick-box exercises” without actual change, negative responses among staff, and stagnant patient outcomes [37, 38]. Such lessons highlight the importance of achieving broad consensus, as presented in this study, to identify items of perceived greatest importance, which received the greatest support, and, therefore, have the greatest chance of succeeding in future.
Conclusion
In summary, this study reports the results of a Delphi consensus process intended to identify key items to ensuring safe practice in LC, as well as priorities for future training, assessment, and research. Across all domains, emphasis was placed upon safe establishment of the critical view of safety, in addition to appropriate surgical decision making, and intraoperative judgement. Priorities for educators, researchers, and surgeons alike have been identified, with which to structure a campaign to reduce bile duct injury rates and improve patient safety in cholecystectomy.
References
Litynski GS (1999) Profiles in laparoscopy: Mouret, Dubois, and Perissat—the laparoscopic breakthrough in Europe (1987–1988). JSLS 3:163–167
Dubois F, Icard P, Berthelot G, Levard H (1990) Coelioscopic cholecystectomy. Preliminary report of 36 cases. Ann Surg 211:60–62
Keus F, de Jong JA, Gooszen HG, van Laarhoven CJ (2006) Laparoscopic versus open cholecystectomy for patients with symptomatic cholecystolithiasis. Cochrane Database Syst Rev 18(4):CD006231
Unger SW, Rosenbaum G, Unger HM, Edelman DS (1993) A comparison of laparoscopic and open treatment of acute cholecystitis. Surg Endosc 7:408–411
Shaffer EA (2006) Gallstone disease: epidemiology of gallbladder stone disease. Best Pract Res Clin Gastroenterol 20:981–996
McMahon AJ, Russell IT, Baxter JN, Ross S, Anderson JR, Morran CG, Sunderland G, Galloway D, Ramsay G, O’Dwyer PJ (1994) Laparoscopic versus minilaparotomy cholecystectomy: a randomised trial. Lancet 343:135–138
The Southern Surgeons Club (1991) A prospective analysis of 1518 laparoscopic cholecystectomies. N Engl J Med 324:1073–1078
Raute M, Schaupp W (1988) Iatrogenic damage of the bile ducts caused by cholecystectomy. Treatment and results. Langenbecks Arch Chir 373:345–354
Morgenstern L, Wong L, Berci G (1992) Twelve hundred open cholecystectomies before the laparoscopic era. A standard for comparison. Arch Surg 127:400–403
Harboe KM, Bardram L (2011) The quality of cholecystectomy in Denmark: outcome and risk factors for 20,307 patients from the national database. Surg Endosc 25:1630–1641
Waage A, Nilsson M (2006) Iatrogenic bile duct injury: a population-based study of 152 776 cholecystectomies in the Swedish Inpatient Registry. Arch Surg 141:1207–1213
Flum DR, Dellinger EP, Cheadle A, Chan L, Koepsell T (2003) Intraoperative cholangiography and risk of common bile duct injury during cholecystectomy. JAMA 289:1639–1644
Navez B, Ungureanu F, Michiels M, Claeys D, Muysoms F, Hubert C, Vanderveken M, Detry O, Detroz B, Closset J, Devos B, Kint M, Navez J, Zech F, Gigot JF, Belgian Group for Endoscopic S, the H, Pancreatic Section of the Royal Belgian Society of S (2012) Surgical management of acute cholecystitis: results of a 2-year prospective multicenter survey in Belgium. Surg Endosc 26:2436–2445
Stewart L, Way LW (1995) Bile duct injuries during laparoscopic cholecystectomy. Factors that influence the results of treatment. Arch Surg 130:1123–1128
Bouras G, Burns EM, Howell AM, Bagnall NM, Lee H, Athanasiou T, Darzi A (2014) Systematic review of the impact of surgical harm on quality of life after general and gastrointestinal surgery. Ann Surg 260(6):975–983
Tornqvist B, Zheng Z, Ye W, Waage A, Nilsson M (2009) Long-term effects of iatrogenic bile duct injury during cholecystectomy. Clin. Gastroenterol Hepatol 7:1013–1018
Pucher PH, Aggarwal R, Qurashi M, Darzi A (2014) Meta-analysis of the effect of postoperative in-hospital morbidity on long-term patient survival. Br J Surg 101(12):1499–1508
Kern KA (1997) Malpractice litigation involving laparoscopic cholecystectomy. Cost, cause, and consequences. Arch Surg 132:392–397
McLean TR (2006) Risk management observations from litigation involving laparoscopic cholecystectomy. Arch Surg 141:643–648
Alkhaffaf B, Decadt B (2010) 15 years of litigation following laparoscopic cholecystectomy in England. Ann Surg 251:682–685
Berci G, Hunter J, Morgenstern L, Arregui M, Brunt M, Carroll B, Edye M, Fermelia D, Ferzli G, Greene F, Petelin J, Phillips E, Ponsky J, Sax H, Schwaitzberg S, Soper N, Swanstrom L, Traverso W (2013) Laparoscopic cholecystectomy: first, do no harm; second, take care of bile duct stones. Surg Endosc 27:1051–1054
de Mestral C, Rotstein OD, Laupacis A, Hoch JS, Zagorski B, Alali AS, Nathens AB (2014) Comparative operative outcomes of early and delayed cholecystectomy for acute cholecystitis: a population-based propensity score analysis. Ann Surg 259:10–15
Aggarwal R, Crochet P, Dias A, Misra A, Ziprin P, Darzi A (2009) Development of a virtual reality training curriculum for laparoscopic cholecystectomy. Br J Surg 96:1086–1093
Sanford DE, Strasberg SM (2014) A simple effective method for generation of a permanent record of the critical view of safety during laparoscopic cholecystectomy by intraoperative “doublet” photography. J Am Coll Surg 218:170–178
Strasberg SM, Hertl M, Soper NJ (1995) An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 180:101–125
Fletcher DR, Hobbs MS, Tan P, Valinsky LJ, Hockey RL, Pikora TJ, Knuiman MW, Sheiner HJ, Edis A (1999) Complications of cholecystectomy: risks of the laparoscopic approach and protective effects of operative cholangiography—a population-based study. Ann Surg 229:449–457
Strasberg SM, Brunt LM (2010) Rationale and use of the critical view of safety in laparoscopic cholecystectomy. J Am Coll Surg 211:132–138
Gallagher M, Hares T, Spencer J, Bradshaw C, Webb I (1993) The nominal group technique: a research tool for general practice? Fam Pract 10:76–81
Dalkey NC (1967) Delphi. RAND Corporation, Santa Monica
Mishra A, Catchpole K, Dale T, McCulloch P (2008) The influence of non-technical performance on technical outcome in laparoscopic cholecystectomy. Surg Endosc 22:68–73
Pucher PH, Aggarwal R, Darzi A (2014) Surgical ward round quality and impact on variable patient outcomes. Ann Surg 259:222–226
Way LW, Stewart L, Gantert W, Liu K, Lee CM, Whang K, Hunter JG (2003) Causes and prevention of laparoscopic bile duct injuries: analysis of 252 cases from a human factors and cognitive psychology perspective. Ann Surg 237:460–469
Feldman LS, Brunt LM, Fuchshuber P, Jones DB, Jones SB, Mischna J, Munro MG, Rozner MA, Schwaitzberg SD, Committee SF (2013) Rationale for the fundamental use of surgical Energy (FUSE) curriculum assessment: focus on safety. Surg Endosc 27:4054–4059
Wauben LS, Goossens RH, van Eijk DJ, Lange JF (2008) Evaluation of protocol uniformity concerning laparoscopic cholecystectomy in the Netherlands. World J Surg 32:613–620
Buddingh KT, Hofker HS, ten Cate Hoedemaker HO, van Dam GM, Ploeg RJ, Nieuwenhuijs VB (2011) Safety measures during cholecystectomy: results of a nationwide survey. World J Surg 35:1235–1241
Mealing NM, Banks E, Jorm LR, Steel DG, Clements MS, Rogers KD (2010) Investigation of relative risk estimates from studies of the same population with contrasting response rates and designs. BMC Med Res Methodol 10:26
Urbach DR, Govindarajan A, Saskin R, Wilton AS, Baxter NN (2014) Introduction of surgical safety checklists in Ontario, Canada. N Engl J Med 370:1029–1038
Borchard A, Schwappach DL, Barbir A, Bezzola P (2012) A systematic review of the effectiveness, compliance, and critical factors for implementation of safety checklists in surgery. Ann Surg 256:925–933
Disclosures
Rajesh Aggarwal is a consultant for Applied Medical. Robert D. Fanelli is a consultant for EndoGastric Solutions and Cook Surgical, Inc. All other authors declare no financial ties or conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the SAGES Safe Cholecystectomy Task Force.
Appendices
Appendix 1
See Table 4
Appendix 2
See Table 5
Appendix 3
See Table 6
Appendix 4
See Table 7
Rights and permissions
About this article
Cite this article
Pucher, P.H., Brunt, L.M., Fanelli, R.D. et al. SAGES expert Delphi consensus: critical factors for safe surgical practice in laparoscopic cholecystectomy. Surg Endosc 29, 3074–3085 (2015). https://doi.org/10.1007/s00464-015-4079-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-015-4079-z