Abstract
Background
We investigated the potential beneficial effect of the antioxidant 2-mercaptoethane-sulfonate (mesna) against oxidative stress induced by pneumoperitoneum in splanchnic organs.
Methods
Wistar rats were subjected to either (a) CO2 pneumoperitoneum (15 mmHg for 60 min) (group P), (b) pretreatment with mesna (400 mg/kg, p.o.) followed by pneumoperitoneum with a 180 min interval (group MP), (c) sham operation (group S), or (d) administration of mesna only (group M). Forty-five minutes after desufflation (groups P and MP), 60 + 45 min after the induction of anesthesia (group S), or 180 min after mesna administration (group M), tissue specimens were excised from liver, kidneys, jejunum and stomach. Tissue oxidative state was assessed on the basis of glutathione-to-glutathione disulfide ratio, malondialdehyde concentration , and superoxide dismutase activity.
Results
Pneumoperitoneum deteriorated all the oxidative stress markers in the organs studied. Mesna prevented the occurrence of oxidative stress following pneumoperitoneum in all the organs studied. In the absence of pneumoperitoneum, the administration of mesna caused mild enhancement of the oxidative state of liver, stomach, and kidneys compared to sham controls.
Conclusions
Prophylaxis with mesna prevents oxidative stress induced by pneumoperitoneum in splanchnic organs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Pneumoperitoneum facilitates the visualization of abdominal organs during laparoscopy or laparoscopic surgery. Despite the numerous advantages offered by this technique, there is growing evidence both from experimental and clinical studies that the increase in intra-abdominal pressure causes oxidative damage of splanchnic and even remote organs. In animal studies, increases in oxidative stress markers were observed in blood plasma, intestine, liver, spleen, kidney, peritoneum, and even lung following CO2 pneumoperitoneum [1–4]. In the clinical setting, laparoscopically performed operations, such as cholecystectomy [5] and hernia repair [6], induce oxidative stress in contrast to open surgery.
Insufflation of gas results in increased intra-abdominal pressure with subsequent alterations in systemic, splanchnic, and renal circulation. These changes include a decrease in cardiac output and mean arterial pressure, transient elevations of aortic pressure and carotid arterial blood flow, and a reduction in blood flow in portal vein, superior mesenteric artery, liver, spleen, pancreas, intestine and kidneys [1, 7–10]. These changes are considered as a main causative factor of ischemia–reperfusion (I/R) in the splanchnic organs with subsequent oxidative damage.
Various strategies are under study for the prevention of pneumoperitoneum-induced oxidative stress. Ischemic preconditioning [11, 12], establishment of low intra-abdominal pressure [13], insufflation with the inert gas helium [4], and pretreatment with erythropoietin [14] or the antioxidant melatonin [15] have yielded promising results.
2-Mercaptoethane-sulfonate (mesna) is a thiol used in chemotherapy regimens for the prevention of hemorrhagic cystitis induced by the oxazaphosphorines cyclophoshamide and ifosfamide [16–18]. Apart from its uroprotective action, an antioxidative effect against I/R injury in kidneys [19], liver [20], and intestine [21, 22] has also been shown. On these grounds, we sought to investigate a potential beneficial effect of mesna’s prophylactic administration against pneumoperitoneum-induced oxidative stress in splanchnic organs.
Materials and methods
Animals
Thirty-two Wistar rats, weighing 300–350 g, provided from our laboratory’s rat colony, were used. They were housed in macrolon cages, three rats per cage, at 20–22°C room temperature, in a 12:12 hour dark cycle and provided with a commercial pelleted diet (certified rat chow, #510/ EL.VI.Z, Xanthi, Greece) and tap water ad libitum. The facilities were in accordance with Directive 86/609/EEC.
Experimental protocol
Animals were randomly assigned to four groups of eight animals each as follows: group P, 60 min pneumoperitoneum; group MP, mesna administration (400 mg/kg, p.o.) followed by 60 min pneumoperitoneum with a 180 min interval; group S, sham operation (anesthesia for 60 min and a catheter inserted into the abdomen); group M, mesna only administration (400 mg/kg, p.o.).
Forty-five minutes after desufflation (groups P, MP), 60 + 45 min after the induction of anesthesia (group S), or 180 min after mesna administration (group M), tissue specimens were excised under general anesthesia from liver, kidney, the mid-part of the jejunum, and stomach and processed for oxidative state assessment on the basis of glutathione-to-glutathione disulfide ratio (GSH/GSSG ratio), malondialdehyde (MDA) concentration, and superoxide dismutase (SOD) activity. The study design is schematically represented in Fig. 1.
Subsequently, all the animals were euthanized by intracardiac injection of thiopental. The experimental protocol was approved by the Animal Care and Use committee of the local Veterinary Service since it was in compliance with Directive 86/609/EEC.
Pneumoperitoneum
The animals were anesthetized by intraperitoneal administration of ketamine (90 mg/kg) and placed at a supine position. After proper preparation of the surgical field, a 18 G Abbocath catheter was inserted into the abdominal cavity through the umbilicus and connected to an insufflator tube. Then, CO2 was insufflated up to a 15 mmHg pressure for 60 min, using an abdominal CO2 insufflator (Olympus-Walz Electronik GmbH, D-7271 Rohrdorf, Germany), followed by immediate desufflation.
Mesna administration
Mesna tablets (Uromitexan 600 mg/tab./ Baxter Oncology GmbH, Westfalen, Germany) were powdered. The appropriate dose was preweighed and dissolved in 2 ml normal saline. Appropriate concentrations of the drug were then administered orally using an intragastric catheter.
GSH/GSSG ratio assessment
The concentrations (μΜ) of GSH and GSSG were measured in the jejunal epithelium using a commercially available kit (GSH/GSSG Ratio Assay Kit, CALBIOCHEM, San Diego, CA) according to the manufacturer’s instructions. The assay utilizes glutathione reductase and allows quantitative assessment of GSSG by the use of the thiol-scavenging reagent 1-methyl-2-vinylpyridinium trifluoromethane sulfonate (M2VP, US Patent 5,543,298) that rapidly scavenges GSH without interfering with the glutathione reductase assay. Thus, jejunal segments, approximately 10 cm long, were excised and opened longitudinally to expose the mucosal epithelium which was rinsed with ice-cold normal saline (0.9% NaCl). The mucosal layer was then harvested by gentle scraping of the epithelium with a glass slide. Tissue specimens were also harvested from liver, kidneys, and stomach. Homologous pieces of each tissue (weighing approximately 10 mg) were collected in pairs of preweighed Eppendorf tubes. One tube was empty while the other contained the scavenger M2VP. The samples were homogenized therein before being snap frozen in liquid nitrogen and stored at −75°C until later used for GSH/GSSG ratio assessment. The tube which did not contain the scavenger was meant for quantification of GSH concentration, while the other was meant for quantification of GSSG concentration. Consequently, the assay was performed measuring the rate of optical density change of 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB) used as a chromophore at 412 nm (ΔOD412/min). For each pair of homologous samples, the levels of GSH and GSSG were determined by reference to standard corresponding concentrations of GSH (supplied by the manufacturer) and were recalculated per mg of protein content of the corresponding samples. Finally, the GSH/GSSG ratio was calculated according to the formula: ratio = [(GSH)−2(GSSG)]/(GSSG).
MDA concentration assessment
The concentration of MDA was measured by a colorimetric method using a commercially available kit (Lipid Peroxidation Assay Kit/Calbiochem, San Diego, CA, USA). The jejunal segments, approximately 10 cm long, were excised and opened longitudinally to expose the mucosal epithelium which was rinsed with ice cold 0.9% NaCl containing ethylenediaminetetraacetic acid (EDTA) and harvested by gentle scraping of the epithelium with a glass slide. The rest of the tissues (liver, kidney, stomach) were perfused with 0.9% NaCl containing EDTA. Tissue samples were snap frozen in liquid nitrogen and kept at −75°C until later assayed for MDA concentration assessment. Each tissue was washed, disrupted and then homogenized in 20 mM phosphate buffered saline (PBS) using a rotor–stator homogenizer. The homogenates were centrifuged at 3000 g for 10 min at 4°C. The concentration of MDA was measured in the supernatant according to the manufacturer’s instructions. MDA concentration was then divided by the total protein and the results were expressed as μmol/g protein. Total protein was measured using the Bradford method [23] as modified by Bearden [24].
SOD activity assessment
The activity of SOD was measured by a colorimetric method using a commercially available kit (Superoxide Dismutase Assay Kit / Cayman Chemical Company, Michigan, USA). The tissues were treated as described in MDA concentration assessment, snap frozen in liquid nitrogen, and kept at −75°C until later assayed for SOD activity assessment. Each tissue was perfused with PBS containing heparin and homogenized in 20 mM HEPES buffer, pH 7.2, 1 mM ethyleneglycoltetraacetic acid (EGTA), 210 mM mannitol, and 70 mM sucrose per gram of tissue. The homogenates were centrifuged at 3000 g for 10 min at 4°C. SOD activity was measured in the supernatant according to the manufacturer’s instructions. SOD activity was then divided by the total protein and the results were expressed as SOD units/mg protein. Total protein was measured using the Bradford method [23] as modified by Bearden [24].
Statistical analysis
The data were expressed as means ± standard deviation and were subjected to one-way analysis of variance. The Bonferroni test was used for multiple comparisons within means. A probability of less than 5% (p < 0.05) was considered to be statistically significant.
Results
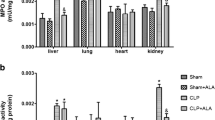
Figures 2–5 depict the measurements of the oxidative stress markers for all the organs studied. Pneumoperitoneum induced oxidative stress in all the organs studied. This was suggested by a deterioration in all the oxidative stress markers, a decrease in GSH/GSSG ratio, and an increase in the MDA concentration and SOD activity in group P compared to group S. On the basis of GSH/GSSG, ratio, the most sensitive index, liver was the least affected organ (36.1%, 39.8%, 43.0%, and 67.0% decrease for liver, stomach, jejunum, and kidneys, respectively).
In the mesna pretreated plus pneumoperitoneum group of animals, oxidative stress was prevented in all the organs studied since all oxidative stress markers remained at sham levels (p > 0.05, group MP versus group S).
In the absence of pneumoperitoneum, mesna administration led to mild enhancement of the oxidative state of liver, kidneys, and stomach compared to sham controls. This was shown by an increase in GSH/GSSG ratio in liver (p = 0.02, group M versus group S) and stomach (p = 0.016, group M versus group S) and a decrease in MDA concentration in the kidneys (p < 0.001, group M versus group S). No change was observed in jejunum.
Discussion
Animal and clinical studies have demonstrated the induction of oxidative stress following pneumoperitoneum [1–6]. In the present study we investigated the antioxidant effect of mesna administration on the oxidative state of liver, kidneys, jejunum, and stomach following pneumoperitoneum in the rat. According to our results, oral pretreatment with mesna, 3 hours before the establishment of CO2 pneumoperitoneum for 60 min at 15 mmHg, prevented the occurrence of oxidative stress in these organs. In addition, when mesna was administered in the absence of pneumoperitoneum, there was a mild enhancement of the oxidative state of liver, stomach, and kidneys, but not of intestine compared to sham controls.
Various markers of oxidative stress have been measured in the blood and the tissues to evaluate this phenomenon in relation to pneumoperitoneum. In our study we determined the tissue GSH/GSSG ratio, the concentration of MDA, and the activity of SOD. The main nonenzymatic intracellular antioxidant GSH acts as a redox buffering system by scavenging reactive oxygen species thereby helping to maintain protein sulfhydryl groups in the reduced state and the heme iron in the ferrous (Fe2+) state. Reactive oxygen species, overproduced at oxidative stress, are potent oxidizing and reducing agents that can directly damage cellular membranes by lipid peroxidation [25]. Peroxidation of endogenous lipids leads to conversion of reduced glutathione to glutathione disulfide [26]. Therefore, the determination of GSH/GSSG ratio is a convenient and sensitive marker of oxidative stress. MDA is an end product derived from the peroxidation of polyunsaturated fatty acids and related esters. Thus, MDA concentration is a true reporter of lipid peroxidation. Another line of cellular defense against free radicals is a system of two enzymes, SOD and catalase. SOD catalyzes the conversion of superoxides to hydrogen peroxide (H2O2), which is subsequently converted to water and oxygen by catalase or glutathione peroxidase. Because SOD plays such a key role in cellular defense against free radicals, it is also an important indicator of the oxidative state [27].
It has been demonstrated that even a small increase in intra-abdominal pressure (4 mmHg) by insufflation of CO2 results in deterioration of systemic and splanchnic circulation attributable mainly to mechanical compression of vessels and hypercapnia [10]. During pneumoperitoneum, blood flow in splanchnic organs as well as in the portal vein, the renal vein, and the superior mesenteric artery is decreased, returning to normal after desufflation [7, 9]. This pattern of changes in the blood supply in splanchnic organs, constitutes a model of I/R injury which is manifested by induction of oxidative stress.
It is well known that ischemia causes considerable tissue damage which is exacerbated by reperfusion with oxygenated blood [28]. For this reason, in our study, the 60 min pneumoperitoneum was followed by a 45 min resting period before tissue biopsies were taken for evaluation of the oxidative state, according to the rat model described by Yilmaz et al. [11, 12].
In our study, an increase in intra-abdominal pressure at 15 mmHg led to substantial oxidative stress in kidneys, jejunum, and stomach, while liver sustained less oxidative stress. A deterioration in the oxidative state of these organs has also been reported in previous studies [1, 11, 12, 14, 15]. Although one would expect the degree of oxidative damage to be proportional to the reduction in blood flow, this is not supported by previous studies; the deterioration of blood flow during pneumoperitoneum was more prominent in solid organs, such as liver, pancreas, spleen, and kidneys compared to that in hollow viscus organs such as intestine, while nonsignificant in stomach [10]. This discrepancy suggests a potentially variable degree of sensitivity to ischemic insult among different tissues. Intestine is one of the most sensitive organs with respect to I/R injury [29]. On the other hand, in the present study we observed that the liver afforded a higher degree of tolerance to I/R, compared to the other organs tested, although it has been reported that it is subjected to substantial blood flow reduction during pneumoperitoneum [7, 10, 30]. The oxidative state of stomach was severely compromised in our 15 mmHg pneumoperitoneum model. Although Schafer et al. [10] did not report any significant change in the blood flow of the stomach during 4 or 10 mmHg CO2 pneumoperitoneum, there are no data available on the effects of a 15 mmHg intra-abdominal pressure. Finally, the kidneys, although retroperitoneally located, were also severely affected, in agreement with reports of reduced renal blood flow during pneumoperitoneum [9, 10].
Different strategies, including prophylactic administration of antioxidants, are under investigation for the prevention of pneumoperitoneum-induced oxidative damage of splanchnic organs. In a recent experimental study, the administration of melatonin 5 min before the insufflation of the abdominal cavity with CO2 for 60 min at 15 mmHg and just before the desufflation prevented oxidative damage in ileum, liver, and kidneys on the basis of tissue MDA levels and the histopathological findings in the ileal mucosa and submucosa [15]. In our study, pretreatment with mesna prevented the occurrence of oxidative stress in liver, kidneys, jejunum, and stomach as evidenced by the maintenance of tissue oxidative stress markers at sham-operated levels.
Mesna is used in current clinical practice as a uroprotectant for the prevention of hemorrhagic cystitis in oxazaphosphorine chemotherapy protocols [16–18]. However, recent animal studies demonstrate its antioxidant properties in the amelioration or even prevention of oxidative damage induced by I/R injury in kidneys [19], liver [20], and intestine [21, 22]. In our previous study, oral pretreatment with mesna at 400 mg/kg, 3 hours before the induction of 60 min ischemia in the rat jejunum, prevented the induction of oxidative stress, keeping the intestinal mucosa literally intact [22]. The oral route of administration proved to be more effective than the intravenous, offering the advantage of delayed absorption and long residence time [31] that should provide prolonged protection against the overproduction of reactive oxygen species during the ischemic and the reperfusion period. Since this administration schedule was effective in the protection of the intestine, known for its sensitivity to I/R injury, we decided to use it in the present study. Our results confirm the protective antioxidant effect of mesna reported in studies using an I/R model affecting liver, kidneys, and intestine by transient occlusion of their blood supplying artery and also broaden its application to the relevant pneumoperitoneum-induced I/R model in these organs. In addition, to our knowledge, this is the first report of antioxidative protection of mesna on the stomach.
The administration of mesna in the absence of pneumoperitoneum, led to mild enhancement of the antioxidant capacity of liver, stomach, and kidneys. However, no change was observed in jejunal mucosa, in agreement with our previous work [22]. The mechanism of mesna’s protective action against oxidative damage is still under investigation. It has been shown in vitro that the drug has the ability to scavenge reactive oxygen species by virtue of its sulfhydryl group [32]. Recently, the protective effect of mesna on the intestinal mucosa was related to inhibition of the activation of nuclear factor-κB (NF-κB) [22]. The NF-κB proteins are a family of transcription factors controlling the expression of genes involved in many critical physiological responses, including immune and acute-phase inflammatory responses, cell adhesion, differentiation, oxidative stress responses, and apoptosis [33]. A multitude of signaling factors, including viruses, oxidants, inflammatory cytokines, and immune stimuli, can activate transcription factor NF-κB. Reactive oxygen species and especially H2O2, are considered as effective inducers of NF-κB activation [34]. Among other antioxidant compounds used as NF-κB inhibitors [35–37], mesna has been shown to inhibit the activation of NF-κB following I/R insult in rat intestine [22]. This effect was related to the protective action of mesna against I/R-induced oxidative injury.
In conclusion, prophylactic oral administration of the antioxidant mesna prevented the occurrence of oxidative stress in liver, kidneys, intestine, and stomach induced by CO2 pneumoperitoneum in rat. The protective effect of mesna was related to mild enhancement of the antioxidant capacity of liver, stomach, and kidney. The results of our experimental study provide evidence for the potentially effective application of mesna pretreatment in the clinical setting as a prophylactic strategy against pneumoperitoneum-induced oxidative damage of splanchnic organs.
References
Eleftheriadis E, Kotzampassi K, Papanotas K, Heliadis N, Sarris K (1996) Gut ischemia, oxidative stress, and bacterial translocation in elevated abdominal pressure in rats. World J Surg 20:11–16
Pross M, Schulz HU, Flechsig A, Manger T, Halangk W, Augustin W, Lippert H, Reinheckel T (2000) Oxidative stress in lung tissue induced by CO(2) pneumoperitoneum in the rat. Surg Endosc 14:1180–1184
de Souza AM, Wang CC, Chu CY, Lam PM, Rogers MS (2003) The effect of intra-abdominal pressure on the generation of 8-iso prostaglandin F2alpha during laparoscopy in rabbits. Hum Reprod 18:2181–2188
Yilmaz S, Polat C, Kahraman A, Koken T, Arikan Y, Dilek ON, Gokce O (2004) The comparison of the oxidative stress effects of different gases and intra-abdominal pressures in an experimental rat model. J Laparoendosc Adv Surg Tech A 14:165–168
Glantzounis GK, Tselepis AD, Tambaki AP, Trikalinos TA, Manataki AD, Galaris DA, Tsimoyiannis EC, Kappas AM (2001) Laparoscopic surgery-induced changes in oxidative stress markers in human plasma. Surg Endosc 15:1315–1319
Polat C, Kahraman A, Yilmaz S, Koken T, Serteser M, Akbulut G, Arikan Y, Dilek ON, Gokce O (2003) A comparison of the oxidative stress response and antioxidant capacity of open and laparoscopic hernia repairs. J Laparoendosc Adv Surg Tech A 13:167–173
Ishizaki Y, Bandai Y, Shimomura K, Abe H, Ohtomo Y, Idezuki Y (1993) Changes in splanchnic blood flow and cardiovascular effects following peritoneal insufflation of carbon dioxide. Surg Endosc 7:420–423
Chiu AW, Chang LS, Birkett DH, Babayan RK (1995) The impact of pneumoperitoneum, pneumoretroperitoneum, and gasless laparoscopy on the systemic and renal hemodynamics. J Am Coll Surg 181:397–406
McDougall EM, Bennett HF, Monk TG, Siegel CL, Li D, McFarland EG, Clayman RV, Sharp T, Rayala HJ, Miller SB, Haacke EM (1997) Functional MR imaging of the porcine kidney: physiologic changes of prolonged pneumoperitoneum. JSLS 1:29–35
Schafer M, Sagesser H, Reichen J, Krahenbuhl L (2001) Alterations in hemodynamics and hepatic and splachnic circulation during laparoscopy in rats. Surg Endosc 15:1197–1201
Yilmaz S, Koken T, Tokyol C, Kahraman A, Akbulut G, Serteser M, Polat C, Gokce C, Gokce O (2003) Can preconditioning reduce laparoscopy-induced tissue injury? Surg Endosc 17:819–824
Yilmaz S, Ates E, Polat C, Koken T, Tokyol C, Akbulut G, Gokce O (2003) Ischemic preconditioning decreases laparoscopy-induced oxidative stress in small intestine. Hepatogastroenterology 50:979–982
Cevrioglu AS, Yilmaz S, Koken T, Tokyol C, Yilmazer M, Fenkci IV (2004) Comparison of the effects of low intra-abdominal pressure and ischaemic preconditioning on the generation of oxidative stress markers and inflammatory cytokines during laparoscopy in rats. Hum Reprod 19:2144–2151
Ates E, Yilmaz S, Ihtiyar E, Yasar B, Karahuseyinoglu E (2006) Preconditioning-like amelioration of erythropoietin against laparoscopy-induced oxidative injury. Surg Endosc 20:815–819
Cay A, Imamoglu M, Unsal MA, Aydin S, Alver A, Akyol A, Sarihan H (2006) Does anti-oxidant prophylaxis with melatonin prevent adverse outcomes related to increased oxidative stress caused by laparoscopy in experimental rat model? J Surg Res 135:2–8
Dechant KL, Brogden RN, Pilkington T, Faulds D, Ifosfamide/mesna (1991) A review of its antineoplastic activity, pharmacokinetic properties and therapeutic efficacy in cancer. Drugs 42:428–467
Haselberger MB, Schwinghammer TL (1995) Efficacy of mesna for prevention of hemorrhagic cystitis after high-dose cyclophosphamide therapy. Ann Pharmacother 29:918–921
Siu LL, Moore MJ (1998) Use of mesna to prevent ifosfamide-induced urotoxicity. Support Care Cancer 6:144–154
Kabasakal L, Sehirli AO, Cetinel S, Cikler E, Gedik N, Sener G (2004) Mesna (2-mercaptoethane sulfonate) prevents ischemia/reperfusion induced renal oxidative damage in rats. Life Sci 75:2329–2340
Sener G, Sehirli O, Ercan F, Sirvanci S, Gedik N, Kacmaz A (2005) Protective effect of MESNA (2-mercaptoethane sulfonate) against hepatic ischemia/reperfusion injury in rats. Surg Today 35:575–580
Ypsilantis P, Lambropoulou M, Tentes I, Kortsaris A, Papadopoulos N, Simopoulos C (2006) Mesna protects intestinal mucosa from ischemia-reperfusion injury. J Surg Res 134:278–284
Ypsilantis P, Tentes I, Lambropoulou M, Anagnostopoulos K, Papadopoulos N, Kortsaris A, Simopoulos C (2008) Prophylaxis with mesna prevents oxidative stress induced by ischemia-reperfusion in the intestine via inhibition of nuclear factor-κB activation. J Gastroen Hepatol 23:328–335
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bearden JC Jr (1978) Quantitation of submicrogram quantities of protein by an improved protein-dye binding assay. Biochim Biophys Acta 533:525–529
Toyokuni S (1999) Reactive oxygen species-induced molecular damage and its application in pathology. Pathol Int 49:91–102
Brivida K, Sies H (1994) Non enzymatic antioxidant defense system. In: Frei B (ed) Natural Antioxidants in Human Health and Disease. Academic Press, San Diego, pp 107–128
de Zwart LL, Meerman JH, Commandeur JN, Vermeulen NP (1999) Biomarkers of free radical damage applications in experimental animals and in humans. Free Radic Biol Med 26:202–226
Carden DL, Granger DN (2000) Pathophysiology of ischaemia-reperfusion injury. J Pathol 190:255–266
Granger DN, Korthuis RJ (1995) Physiologic mechanisms of postischemic tissue injury. Annu Rev Physiol 57:311–332
Windberger UB, Auer R, Keplinger F, Längle F, Heinze G, Schindl M, Losert UM (1999) The role of intra-abdominal pressure on splanchnic and pulmonary hemodynamic and metabolic changes during carbon dioxide pneumoperitoneum. Gastrointest Endosc 49:84–91
Stofer-Vogel B, Cerny T, Borner M, Lauterburg BH (1993) Oral bioavailability of mesna tablets. Cancer Chemother. Pharmacol 32:78–81
Gressier B, Cabanis A, Lebegue S, Brunet C, Dine T, Luyckx M, Cazin M, Cazin JC (1994) Decrease of hypochlorous acid and hydroxyl radical generated by stimulated human neutrophils: comparison in vitro of some thiol-containing drugs. Methods Find Exp Clin Pharmacol 16:9–13
Pahl HL (1999) Activators and target genes of Rel/NF-κB transcription factors. Oncogene 18:6853–6866
Gloire G, Legrand-Poels S, Piette J (2006) NF-kappa B activation by reactive oxygen species: fifteen years later. Biochem Pharmacol 72:1493–1505
Schulze-Osthoff K, Beyaert R, Vandevoorde V, Haegeman G, Fiers W (1993) Depletion of the mitochondrial electron transport abrogates the cytotoxic and gene-inductive effects of TNF. EMBO J 12:3095–3104
Sen CK, Roy S, Packer L (1996) Involvement of intracellular Ca2+ in oxidant-induced NF-kappa B activation. FEBS Lett 385:58–62
Manna SK, Kuo MT, Aggarwal BB (1999) Overexpression of gamma-glutamylcysteine synthetase suppresses tumor necrosis factor-induced apoptosis and activation of nuclear transcription factor-kappa B and activator protein-1. Oncogene 18:4371–4382
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ypsilantis, P., Tentes, I., Anagnostopoulos, K. et al. Mesna protects splanchnic organs from oxidative stress induced by pneumoperitoneum. Surg Endosc 23, 583–589 (2009). https://doi.org/10.1007/s00464-008-9887-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-008-9887-y