Abstract
Background
Since only a few extensive reports are available on the less invasive nature of laparoscopic gastrectomy, we compared postoperative changes over time in vital signs and hematological parameters between this surgery and laparotomic gastrectomy.
Methods
Of 188 patients who underwent distal gastrectomy for preoperatively diagnosed early gastric cancer between January 2004 and September 2006, 87 underwent laparoscopy-assisted distal gastrectomy (LADG) and 101 underwent laparotomic distal gastrectomy (DG). The invasiveness of the two procedures was evaluated in 164 patients with no postoperative complications (82 cases of LADG and 82 cases of DG by measuing vital signs daily and performing hematological examination on postoperative days (POD) 1, 4, 7, and 10.
Results
For body temperature, heart rate, and blood pressure, significantly lower values were obtained with LADG on 3 and 4 POD, 4 POD, and 3 and 4 POD, respectively. For white blood cell counts (WBC) and C-reactive protein (CRP), significantly lower values were obtained with LADG on 7 and 10 POD, and 10 POD, respectively. For serum protein levels and lymphocyte counts, significantly higher values were obtained with LADG on 1, 4, 7, and 10 POD, and 4 and 10 POD, respectively. Body temperature, WBC, and CRP showed no significant difference immediately after surgery but earlier recovery occurred with LADG. For protein levels and lymphocyte counts, higher values were obtained immediately after surgery. There seemed to be two patterns of less invasiveness in the parameters: the early recovery found for body temperature, WBC and CRP, and the smaller shift immediately after surgery in protein level and lymphocyte count, and probably, heart rate and blood pressure. The complication rate was 18.8% for DG and 5.7% for LADG.
Conclusions
LADG is a less-invasive surgical procedure as it produces early normalization or smaller shifts in various parameters and exhibits a low prevalence of complications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Although laparoscopy-assisted gastrectomy (LAG) is recognized as a less-invasive surgery that produces small wounds, only a few extensive reports are available on its less-invasive nature [1–4]. Some authors are suspicious about the safety of LAG because it is technically more sophisticated than laparotomic gastrectomy [5]. Against this background, we extensively investigated the less invasive nature of LAG, compared to laparotomic gastrectomy, by comparing changes over time in vital signs and hematological parameters. We also examined the status of postoperative complications.
Materials and methods
We chose distal gastrectomy, a commonly performed procedure for gastrectomy, as the subject surgical technique. The study population consisted of 188 patients who underwent distal gastrectomy for preoperatively diagnosed early gastric cancer between January 2004 and September 2006. More specifically, 87 underwent laparoscopy-assisted distal gastrectomy (LADG) and 101 underwent open distal gastrectomy (DG). Postoperative changes over time in various parameters related to invasiveness were evaluated in 164 cases with no postoperative complications (82 for LADG and 82 for DG). Vital signs were analyzed using daily maximum values. Hematological examination was performed on 1, 4, 7, and 10 postoperative days (POD). The extent of lymph node dissection and degree of disease progression were described in accordance with the Japanese Classification of Gastric Carcinoma, second English edition [6].
Statistical analysis
Data obtained were statistically analyzed by the t-test and chi-square test using SPSS 11.0 for Windows (SPSS Inc., Chicago, USA). A P-value of less than 0.05 was considered to indicate a significant difference.
Results
Patient background
No significant difference was observed in age, sex, American Society of Anesthesiology (ASA) classification, or body mass index (BMI) between the DG and LADG groups (Table 1).
Surgical findings
No significant difference was observed on operation. A significantly lower bleeding volume was obtained with LADG (108.2 ± 129.4 ml) than with DG (240.4 ± 193.4 ml). There was no significant difference in the extent of lymph node dissection, number of dissected lymph nodes, or degree of pathological stage (Table 2).
Evaluation of invasiveness
Changeovers in vital signs
No significant difference in body temperature was observed between the two groups immediately after surgery. However, significantly lower values were obtained with LADG on 3 and 4 POD, indicating earlier normalization. Regarding changes over time in heart rate and systolic blood pressure, lower values were obtained with LADG immediately after surgery; however, a significant difference was observed on 4 POD, and 3 and 4 POD, respectively (Figure 1).
Changes in hematological parameter values
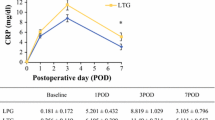
For white blood cell count (WBC) and C-reactive protein (CRP), there was no significant difference immediately after surgery; however, significantly lower values were obtained with LADG for WBC on 7 and 10 POD, and for CRP with a slight delay on 10 POD. Although hematological examination did not take place thereafter, it is anticipated that the significant differences will disappear eventually. It can be said that WBC and CRP show early reductions with LADG (Figure 2). Generally, nutritional parameters such as serum total protein levels and lymphocyte counts decrease transiently after surgery. In the present study, both serum total protein levels and lymphocyte counts showed generally higher values with LADG from immediately after surgery; a significant difference was observed in serum total protein levels on 1, 4, 7, and 10 POD, and in lymphocyte counts on 4 and 10 POD (Figure 3).
Safety assessment
Complications developed in 18.8% of the subjects with DG. With LADG, the complication rate was lower (5.7%) but the difference was not statistically significant. Regarding the breakdown of complications in cases of LADG, duodenal cut-end leakage in Roux-en-Y reconstruction was observed in one case (1.1%), pancreatic fistula in one case (1.1%), and stenotic symptoms at the anastomosis necessitating fasting in two cases (2.3%) (Table 3). In both groups, there was no operative death or death during hospitalization.
Recurrence
Since duration of observation was short at 20.4 ± 9.3 months (range 1.6–34.8 months) for DG and 16.0 ± 9.7 months (range 1.1–34.8 months) for LADG, further follow-up will be necessary to describe long-term outcome. No recurrences have occurred to date.
Discussion
Traditionally, medical intervention for gastric cancer has emphasized treatment for advanced cancer; there has been remarkable progress in extended operation and chemotherapy. On the other hand, recently there has been an opposite shift, from typical surgery to contracted treatment, for early cancer with a good prognosis. In gastroenterological medicine, endoscopic treatments such as endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD), which enables more-reliable resection of lesions than conventional EMR, have become therapeutic options for cases of early gastric cancer thought to have no lymph node metastasis [7]. In cases of early cancer with possible lymph node metastasis, surgery is indicated. Even in such cases, however, contracted operation involving a narrower range of lymph node dissection and less-invasive laparoscopic surgery of low invasion are now available [2, 4]. Laparoscopic gastrectomy for gastric cancer was performed for the first time in Japan in 1991 [8]. Since then, there has been a rapid increase in the number of patients undergoing this surgery.
Due to its less invasive nature, laparoscopic gastrectomy is characterized by small bleeding volume, little pain, early restoration of normal intestinal peristalsis, and early hospital discharge [1, 3, 4, 8, 9]. However, only a few extensive studies have been performed to date on, for example, time-related changes in various parameters in laparoscopic gastrectomy compared with laparotomic gastrectomy [1]. Additionally, since laparoscopic surgery is technically more sophisticated than laparotomic surgery, some problems remain to be fully resolved, including the assurance of its safety and the radical treatment of cancer [3–5, 11].
In this study, we evaluated the less-invasive nature of LADG by comparing changes over time in various parameters with those obtained by DG. All the parameters examined demonstrated the advantage of LADG, but their changes over time revealed two patterns. One pattern was found for inflammation-related parameters, such as body temperature, WBC, and CRP, which showed values similar to those for DG immediately after surgery but achieved early recovery. The other pattern was found for nutritional parameters, such as protein levels and lymphocyte counts, which showed values significantly different from those with DG immediately after surgery. For dynamic circulatory parameters, such as heart rate and blood pressure, there seemed to be an apparent difference from immediately after surgery. However, a significant difference was observed later on 4 POD for heart rate and on 3 and 4 POD for blood pressure; which pattern applies to these parameters remains unknown. This period also corresponds to the end of epidural anesthesia, and intensifying pain may have an influence, though further investigation in a larger study population would clarify this. In addition to the traditionally recognized advantages of laparoscopic surgery, such as minor surgical wounds and low bleeding volume, the less-invasive nature of LADG is attributable to the retention of a wet environment in the abdominal cavity and the absence of manual sustaining and traction of non-extirpated organs during surgery.
Laparoscopic gastrectomy is technically more sophisticated than laparotomic gastrectomy. So, does it actually produce more complications? On the contrary, we noted that the prevalence of complications is lower with laparoscopic gastrectomy. According to the data from the eighth questionnaire survey conducted by the Japanese Society for Endoscopic Surgery in Japan, 4,799 patients underwent LAG in 2004 and 2005 and 8.71% of them experienced complications [12]. Four patients (0.083%) died during hospitalization [12]. In our department, the complication rate was 4.6% for LADG and none died during hospitalization. Therefore, LADG can be considered a safer than laparotomic gastrectomy if performed with due basic skills. The low prevalence of complications is also deemed characteristic of the less-invasive nature of laparoscopic gastrectomy.
In the context of cancer treatment, top priority should be given to radically treating the cancer lesion. Although only a few studies are available on long-term prognosis in laparoscopic gastrectomy because it is a relatively new surgical technique, this technique has not been reported to be inferior to laparotomic gastrectomy in terms of radical treatment [3, 5, 11]. In our department, we introduced LAG in 2001 with the belief that less-invasive treatment should be performed for cases of early gastric cancer with a good prognosis. Since then, we have encountered no recurrences. At least for early gastric cancer, there seems to be no problem with radical treatment. It should be noted, however, that there are a significant number of cases where the degree of progression differs between preoperative and postoperative diagnoses, and that some institutions have begun to apply LAG to the treatment of advanced cancer [5, 8, 13]. With these facts in mind, we conclude that a randomized study of LAG should be conducted not only for early but also for advanced gastric cancer.
References
Adachi Y, Shiraishi N, Shiromizu A, Bavdoh T, Aramaki M, Kitano S (2000) Laparoscopy-assisted Billroth I gastrectomy compared with conventional open gastrectomy. Arch Surg 135: 806–810
Kitagawa Y, Kitano S, Kubota T, Kumai K, Otani Y, Saikawa Y, Yoshida M, Kitajima M (2005) Minimally invasive surgery for gastric cancer – toward a confluence of two major streams: a review. Gastric Cancer 8: 103–110
Tanimura S, Higashino M, Fukumaga Y, Kishida S, Nishikawa M, Ogata A, Osugi H (2005) Laparoscopic distal gastrectomy with regional lymph node dissection for gastric cancer. Surg Endosc 19: 1177–1181
Kitano S, Yasuda K, Shiraishi N (2006) Laparoscopic surgical resection for early gastric cancer. Eur J Gastroenterol Hepatol 18: 855–861
Huscher CGS, Mingoli A, Sgarzini G, Sansonetti A, Paola MD, Recher A, Ponzano C (2005) Laparoscopic versus open subtotal gastrectomy for distal gastric cancer. Five-year results of a randomized prospective trial. Ann Surg 241: 232–37
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma −2nd English edition. Gastric Cancer 1: 10–24
Ono H, Kondo H, Gotoda T, Shirao K, Yamaguchi H, Saito D, Hosokawa K, Shimoda T, Yoshida S (2001) Endoscopic mucosal resection for treatment of early gastric cancer. Gut 48: 225–229
Oda I, Saito D, Tada M, Iishi H, Tanabe S, Oyama T, Doi T, Otani Y, Fujisaki J, Ajioka Y, Hamada T, Inoue H, Gotoda T, Yoshida S (2006) A multicenter retrospective study of endoscopic resection for early gastric cancer. Gastric Cancer 9: 262–270
Kitano S, Iso Y, Moriyama M, Sugimachi K (1994) Laparoscopy-assisted Billroth I gastrectomy. Surg Laparosc Endosc 4: 146–148
Tanimura S, Higashino M, Fukumaga Y, Kishida S, Ogata, Fujiwara Y, Osugi H (2006) Respiratory function after laparoscopic distal gastrectomy – an index of minimally invasive surgery. World J Surg 30: 1211–1215
Shiraishi N, Yasuda K, Kitano S (2006) Laparoscopic gastrectomy with lymph node dissection for gastric cancer. Gastric Cancer 9: 167–176
Japanese Society for Endoscopic Surgery (2006) The 8th questionnaire survey of endoscopic surgery. J Jpn Soc Endosc Surg 5:528–628
Asao T, Hosouchi Y, Nakabayashi T, Haga N, Mochiki E, Kuwano H (2001) Laparoscopically assisted total or distal gastrectomy with lymph node dissection for early gastric cancer. Br J Surg 88: 128–132
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawamura, H., Okada, K., Isizu, H. et al. Laparoscopic gastrectomy for early gastric cancer targeting as a less invasive procedure. Surg Endosc 22, 81–85 (2008). https://doi.org/10.1007/s00464-007-9373-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-007-9373-y