Abstract
Background
A multicentric study was performed to evaluate the clinical results after laparoscopic treatment of pancreatic pseudocysts (PP).
Methods
We collected the data of 17 patients presenting with PP and operated on by laparoscopy between 1996 and 2001. There were nine men and eight women with a median age of 42 years (range 30–72). In 15 patients the PP developed after acute pancreatitis and the median delay between the acute onset and surgery was 7 months (range: 2–24). In two patients the PP was associated with chronic pancreatitis. All the patients had a single PP with a median diameter of 9 cm (range: 5–20).
Results
According to the location of the PP, a cystogastrostomy was performed in 10 patients and a cystojejunostomy in seven patients. The median operative time was 100 min (range: 80–300). Laparoscopic PP surgery was completed successfully in 16 patients and the median size of the cystoenterostomy was 3 cm (range: 2–5). Necrotic debris was present within the PP in 11 patients. The median postoperative hospital stay was 6 days (range: 4–24). No mortality and no immediate morbidity were recorded.
However, two patients were readmitted within the first 3 postoperative weeks because of secondary PP infection. The first patient had an early closure of cystogastrostomy and was treated by endoscopic placement of a stent. The second represented with a right retrocolic abscess after cystojejunostomy and was treated by percutaneous drainage. One patient was lost for follow-up 2 months after surgery. The others had regular clinical and radiological controls. With a median follow-up of 12 months (range: 6–36), no recurrence of PP was observed.
Conclusions
The laparoscopic treatment of PP was associated with a low postoperative complication rate and an effective permanent result. That approach avoided some difficulties, particularly bleeding that is classically linked with endoscopic internal drainage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Pancreatic pseudocyst (PP) is a collection arising in or adjacent to the pancreas but lacking a true epithelial lining [4, 20]. It is widely accepted that PP may have different origins according to the underlying disease. PP is an inflammatory process in nature and may be caused by acute and chronic pancreatitis or by pancreatic trauma. Depending on the series quoted, PP occurs in roughly 10% to 20% of all pancreatitis cases [19, 25]. PP associated with acute necrotizing pancreatitis results from autodigestion of the pancreatic gland and extravasation of pancreatic juice inducing necrosis of the tissues with which it comes in contact [12]. Up to 85% of those cysts resolve spontaneously within 6 weeks and seldom require intervention [32]. Beyond that period, cysts >6 cm are associated with a poor chance of resolution and with a significant risk for the development of complications including infection, bleeding, compression, or rupture [6]. On the other hand, it is hypothesized that PP associated with chronic pancreatitis results from duct outflow obstruction due to stone or stricture causing distension and rupture of smaller ducts. Therefore, a persistent communication between the pancreatic duct and the PP is often present and spontaneous cyst resolution is very uncommon [30]. Internal drainage is the treatment of choice for uncomplicated symptomatic cysts. The most recent treatment option to be developed is laparoscopy, but its role among the armamentarium of available therapeutic procedures remains to be established. The aim of that study is to determine if laparoscopic internal drainage is a safe and efficient therapeutic option for the treatment of PP.
Materials and methods
Patients
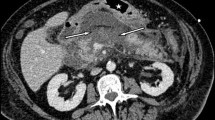
In a multicentric study performed among the members of the Club Coelio and the BGES, we analyzed data for 17 patients who were operated by laparoscopy between 1996 and 2001 for a symptomatic PP. There were nine men and eight women with a median age of 42 years (range 30–72). In 15 patients the PP developed after acute pancreatitis: 10 from alcoholism and five from gallstones. In those patients, the median delay between the acute onset and surgery was 7 months (range: 2–24). In two patients the PP was associated with chronic pancreatitis. Indication for internal drainage was pain or dyspepsia and abdominal discomfort. CT scan was systematically performed before surgery to establish the number, size, and location of the cysts and to define the relationship with the surrounding viscera. All the patients had a single PP: the median diameter was 9 cm (range: 5–20) and the cyst wall was always <1 cm thick. No significant dilatation of the Wirsung canal was evidenced and no patient had a preoperative ERCP.
Surgical techniques
The first step of the operation consisted in establishing a CO2 pneumoperitoneum and placing a standard port and laparoscope at the umbilical level. The number and position of the additional trocars were dictated by the chosen procedure. A cystogastrostomy was performed in 10 patients. Six had an endoluminal gastric procedure and four a totally intraperitoneal transgastric procedure. Whatever the approach, the position of the cyst was first verified by puncture and afterward the posterior wall of the stomach and the cyst wall were incised utilizing electrocautery or, more recently, ultrasonic dissection. The communication was extended to the desired length and the cyst contents were thoroughly debrided. In four patients, the edges of the cystogastrostomy were not sutured. The cyst and the gastric wall were secured with an endo-GIA stapler in four and with sutures in two patients. A nasogastric tube was used postoperatively in all of the patients. Most of the time, it was removed on the first postoperative day, but in three patients the tube, which had been pushed into the cyst cavity, was systematically left in place for a period of 4 days in order to obtain a constant suctioning of PP contents. At the end of the procedure, the defects in the gastric wall or the anterior gastrotomy were closed with sutures. A cystojejunostomy was performed in seven patients. The omentum and transverse colon were rolled upward. The usual site of entry to the PP was just superior and lateral to the ligament of Treitz. Aspiration with a laparoscopic needle was performed to ensure the cyst location. The PP was then opened through the transverse mesocolon and the cyst contents were thoroughly debrided. The cystojejunostomy was performed with an endo-GIA stapler or with sutures. Three patients had a direct latero-lateral anastomosis without jejuno-jejunal anastomosis and four patients had drainage with a 60-cm isolated Roux-en-Y loop of jejunum.
Results
Laparoscopic PP drainage was completed successfully in 16 patients. In one patient, who had an endoluminal procedure, the PP could not be correctly localized and a conversion to open surgery was required to perform the cystogastrostomy. The median size of the cystoenterostomy was 3 cm (range: 2–5). In 11 patients, necrotic material was present within the PP. The median operative time was 100 min (range: 80–300) and the median postoperative hospital stay was 6 days (range: 4–24). Perioperative data are summarized in Table 1 for each of the techniques used in this study. Among the patients treated by cystogastrostomy, one had to stay 24 days postoperatively for psychiatric reasons. It was also noted that the three patients who had a prolonged drainage of the cyst cavity via a nasogastric tube had a longer median postoperative stay: 8 days versus 4 days. No mortality and no immediate morbidity were recorded. However, two patients were readmitted within the first 3 postoperative weeks because of secondary PP infection. The first patient had a 2-cm large cystogastrostomy and a prolonged cyst aspiration. He presented with an infected PP due to early closure of cystogastrostomy that was treated by endoscopic placement of a stent. The second one presented with a right retrocolic abscess after cystojejunostomy on a Roux-en-Y loop and was treated by percutaneous drainage. The patient, who required conversion to open surgery, was lost for follow-up 2 months after surgery. The others had regular clinical and radiological controls. The mean follow-up after PP drainage was 12 months (range: 6–36). No recurrent PP was observed but relapse of acute pancreatitis was noted in two patients due to persistence of alcohol intake.
Discussion
The heterogeneity of PP origins is now widely acknowledged and outcome data of cysts associated with acute or chronic pancreatitis must be distinguished. In case of PP complicating acute pancreatitis, it is commonly suggested that pseudocysts ≥6 cm in size and which persist longer than 6 weeks need therapy to prevent potential complications [6, 12]. At that time, the cyst wall is mature enough to allow a cystoenterostomy. However, other studies have shown that the majority of those patients can be safely observed, treatment being reserved for those developing symptoms or those with enlarging PP over time [29, 30]. In cases of PP associated with chronic pancreatitis, it is generally accepted that treatment must be proposed without delay because of the high risk of complications such as haemorrhage and obstruction [16, 32], Only asymptomatic cysts of <4 cm can be managed expectantly [30].
When treatment is required, internal drainage is recognized as the treatment of choice. Pancreatic resection is only advised for PP of the tail of the pancreas with no connection to nearby organs, and external drainage must be reserved for septic patients with infected PP because that approach is marred by a high risk of pancreaticocutaneous fistula, cyst infection, and recurrence [10, 12, 20]. Surgery is the conventional and most proven therapy for internal drainage, which can be performed into the stomach, the duodenum, or the jejunum depending on the location of the PP [12, 18, 22]. But nowadays, there are other therapeutic options currently available for the management of PP. Several authors proposed percutaneous cystogastrostomy with stenting as an alternative nonsurgical treatment [1, 9, 16]. But during recent decades, the endoscopic drainage procedures have greatly improved and have become increasingly popular and prevalent [5, 10, 25]. Endoscopically, PP can be drained via three different routes: the stomach and the duodenum if the PP is bulging into the gastrodudodenal lumen, or the papilla if the PP communicates with the pancreatic duct. Some remarks must be made about the efficiency of those endoscopic techniques. First, literature data indicate that the initial technical success, morbidity, mortality and recurrence rate are, respectively; 94%, 20%, 1%, and 16% after endoscopic drainage and 100%, 25%, 5%, and 10 % after surgical drainage [4, 20]. Despite the fact that no randomized study has been performed to compare endoscopy and surgery, it appears that the results recorded after both techniques are globally equivalent. Nevertheless, compared to conventional surgery, endoscopy offers undoubtedly a minimally invasive approach that is very attractive for patients and clinicians [10, 20]. Second, unlike surgery, endoscopic drainage is only feasible when a favorable anatomical situation is present. A number of essential prerequisites are necessary before transmural drainage is undertaken: the distance between the PP and the gastric or duodenal wall must be < 1cm and there must be a clear compression of the PP into the gut wall [5, 19, 20, 25]. These criteria are met in 55% of PP complicating chronic pancreatitis, and only in 27% of PP complicating acute pancreatitis [4]. Transpapillary drainage is possible only when there is a communication between the pancreatic ductal system and the PP. Such a situation is also more common in case of PP associated with chronic pancreatitis (49%) than with acute pancreatitis (20%) [4]. Third, the highest rate of complication (22%) and recurrence (18%) is observed after transgastric drainage, bleeding (8%) and cyst infection (8%) being the most frequent complications [4, 5, 10, 11, 27]. The risk of bleeding increases when the size of the anastomosis is enlarged, but a too-narrow incision leads to inadequate drainage or early closure and subsequently to PP infection or recurrence [5, 20, 27]. To avoid those complications, stent placement has been utilized, but common stent problems such as dislodgement, kinking, perforation, and clogging with necrotic debris are frequently observed [9, 16, 25, 27].
There are few studies about the laparoscopic management of PP, and most of them concern case reports [2, 3, 15, 17, 24, 28, 31] or a limited number of patients [7, 8, 13, 26]. More recently, two series including >10 patients have been published [21, 23]. The results recorded in our study confirm that laparoscopic techniques for PP drainage are feasible and practical and that the results are reproducible. Undoubtedly, cystogastrostomy is the most popular laparoscopic procedure. When cumulating the results noted in our patients (n = 10) with those noted in the patients of Mori et al. (n = 14) [21] and Park and Heniford (n = 25) [23], we observe a conversion rate of 10% (5/49), a morbidity rate of 8% (4/49), and no late recurrence. Those results compare favorably with those reported after endoscopic cystogastrostomy. It must be emphasized that laparoscopy has two main potential advantages that should avoid some difficulties linked with the endoscopic drainage techniques: Hemostasis is excellent, as the entire procedure is performed under complete visual control and magnification, and a wide cystoenterostomy minimizes the risk of early closure or recurrence. However, there are no precise criteria to determine what can be considered a wide cystostomy. According to our experience and the literature data, we advise performing an opening of ≥3 cm. Indeed, early closure and PP infection [21] or late recurrence [2] is only reported after laparoscopic cystogastrostomy of 2 cm or less on size. Laparoscopic cystojejunostomy is a more challenging technique, but it may be the only way to drain a symptomatic PP that is not adjacent to the gastroduodenal lumen. Our series show that the procedure can be simplified and shortened by performing a direct cystoenterostomy instead of a Roux-en-Y drainage with equivalent results.
Some authors advocate that endoscopic [10] or percutaneous [16] drainage therapies should be the procedure of choice for the treatment of PP, with surgery reserved for failed attempts. Their arguments are particularly based on the minimally invasive character of those procedures. Nowadays, laparoscopy bridges the wide gap between endoscopy and conventional surgery. Yet, the literature data do not allow a clear statement of which patient will best benefit from that new therapeutic option. The choice of the technique of PP drainage should primarily be a collaborative decision involving interested endoscopists, radiologists, and surgeons [19]. The procedure of drainage also depends on case specifics. For instance, chronic pain, a hallmark of patients with chronic pancreatitises, is difficult to manage. Most of the time, that pain cannot be solely treated by PP drainage, and multiple endoscopic therapies such as sphincterotomy, stone extraction, and pancreatic or biliary stenting are usually performed concurrently [27]. On the other hand, it is suggested that large cysts are better managed surgically [14] and that the presence of significant debris or cloudy material in the PP might lead to stent failure and should discourage an endoscopic attempt [16].
Finally, our series confirm that long-term results are influenced by the quality of the drainage of the PP as well as by the persistence or cessation of alcohol intake, continued drinking being a frequent problem [17] and the main cause of late death in those patients [18].
References
R Anderson W Cwikiel (2002) ArticleTitlePercutaneous cystogastrostomy in patients with pancreatic pseudocyst Eur J Surg 168 345–348 Occurrence Handle10.1080/11024150260284851 Occurrence Handle12428872
U Atabek D Mayer A Amin RC Camishion (1993) ArticleTitlePancreatic cystogastrostomy combined upper endoscopy and percutaneous transgastric instrumentation J Laparoendosc Surg 3 501–504 Occurrence Handle8251667
I Baca I Klempa V Gotzen (1994) ArticleTitleLaparoscopic pancreatiocojejunostomy without entero-enteral anastomosis Chirurg 65 378–381 Occurrence Handle8020361
U Beckingham JEJ Krige PC Bornman J Terblanche (1997) ArticleTitleEndoscopic management of pancreatic pseudocyst Br J Surg 84 1638–45 Occurrence Handle10.1046/j.1365-2168.1997.00561.x Occurrence Handle9448608
H Bejanin C Ligoury O Ink J Fritsch AD Choury JF Lefebvre V Vilgrain JP Etienne (1993) ArticleTitleDrainage endoscopique des pseudo-kystes du pancreas Gastroenterol Clin Biol 17 804–810 Occurrence Handle8143945
EL Bradley JL Clements AC Gonzalez (1979) ArticleTitleThe natural history of pancreatic pseudocysts: a unified concept of management Am J Surg 137 135–141 Occurrence Handle10.1016/0002-9610(79)90024-2 Occurrence Handle758840
G Champault N Rizk E Lebhar JM Catheline C Barrat F Cazacu (1998) ArticleTitleTraitement laparoscopique des faux kystes du pancreas Ann Chir 52 41–44 Occurrence Handle9752407
PK Chowbey V Soni A Sharma S Khullar M Baijal A Vashita (2001) ArticleTitleLaparoscopic intragastric stapled cystogastrostomy for pancreatic pseudocysts J Laparoendosc Adv Surg Tech 11 10–14 Occurrence Handle10.1089/109264201750539709
MR Cox RP Davies RC Bowyer J Toouli (1993) ArticleTitlePercutaneous cystogastrostomy for treatment of pancreatic peudocysts Aust N-Z J Surg 63 693–698 Occurrence Handle8363478
M Cremer J Deviere L Engelholm (1989) ArticleTitleEndoscopic management of cysts and pseudocysts in chronic pancreatitis: long-term follow-up after 7 years of experience Gastrointest Endosc 35 1–9 Occurrence Handle2920879
KP Guzman ParticleDe WH Holdermen A Abu-Hamour (1995) ArticleTitleEndoscopic drainage of pancreatic pseudocyst: patient selection and evaluation of the outcome by endoscopic ultrasonography Endoscopy 27 329–333 Occurrence Handle7555940
M Eeftinck Schattenkerk JE Vries ParticleDe HA Braining WF Eggink H Obertop (1982) ArticleTitleSurgical treatment of pancreatic pseudocysts Br J Surg 69 593–594 Occurrence Handle7127039
L Fernandez-Cruz A Saenz E Astudillo JP Pantoja E Uzcategui S Navarro (2002) ArticleTitleLaparoscopic surgery in patients with chronic pancreatitis Surg Endosc 16 996–1003 Occurrence Handle10.1007/s00464-001-9065-y Occurrence Handle12163971
G Froeschle U Meyer-Pannwitt M Brueckner (1993) ArticleTitleA comparison between surgical, endoscopic and percutaneous management of pancreatic pseudocysts—long term results Acta Belg Chir 93 102–106
EJ Hagopian JA Teixeira M Smith FM Steichen (2001) ArticleTitlePancreatic pseudocyst treated by laparoscopic Roux-en-Y cystojejunostomy Surg Endosc 14 967–970
FW Henriksen S Hancke (1994) ArticleTitlePercutaneous cystogastrostomy for chronic pancreatic pseudocyst Br J Surg 81 1525–1528 Occurrence Handle7820494
P Holeczy J Danis (1998) ArticleTitleLaparoscopic transgastric pancreatic pseudocystogastrostomy—first experience with extraluminal approach Hepato Gastroenterol 45 2215–2218
H Kohler A Schafmayer FE Ludtke G Lepsien H-J Peiper (1987) ArticleTitleSurgical treatment of pancreatic pseudocysts Br J Surg 74 813–815 Occurrence Handle3664248
RA Kozarek (1997) ArticleTitleEndoscopic treatment of pancreatic pseudocysts Gastrointest Endosc Clin North Am 7 271–283
SK Lo A Rowe (1997) ArticleTitleEndoscopic management of pancreatic pseudocysts Gastroenterogist 5 10–25
T Mori N Abe M Sugiyama Y Atomi LW Way (2000) ArticleTitleLaparoscopic pancreatic cystgastrostomy J Hepatobiliary Pancreat Surg 7 28–34 Occurrence Handle10.1007/s005340050150 Occurrence Handle10982588
KA Newell T Liu GV Aranha RA Prinz (1990) ArticleTitleAre cystogastrostomy and cystojejunostomy equivalent operations for pancreatic pseudocysts? Surgery 108 635–640 Occurrence Handle2218873
A.E. Park B.T Heniford (2002) ArticleTitleTherapeutic laparoscopy of the pancreas Ann Surg 236 149–158 Occurrence Handle10.1097/00000658-200208000-00002 Occurrence Handle12170019
S Pekmezci K Saribey T Karahasanoglu H Tasci (2002) ArticleTitleTotal laparoscopic cystogastrostomy for the treatment of pancreatic pseudocyst J Laparoendosc Adv Surg Tech 12 119–123 Occurrence Handle10.1089/10926420252939655
SS Sharma N Bhargawa A Govil (2002) ArticleTitleEndoscopic management of pancreatic pseudocyst: a long term follow-up Endoscopy 34 203–207 Occurrence Handle11870570
C Smadja A Badawy C Vons V Giraud D Franco (1999) ArticleTitleLaparoscopic cystogastrostomy for pancreatic pseudocyst is safe and effective J Laparoendosc Adv Surg Tech 9 16–18
M Smits E Rauws G Tytgat (1995) ArticleTitleThe efficacy of endoscopic treatment of pancreatic pseudocysts Gastrointest Endosc 42 202–207 Occurrence Handle10.1016/S0016-5107(95)70051-X Occurrence Handle7498683
M Trias EM Targarona C Balague A Cifuentes P Taura (1995) ArticleTitleIntraluminal stapled laparoscopic cystogastrostomy for treatment of pancreatic pseudocyst Br J Surg 82 403 Occurrence Handle7796025
GJ Vitas MG Sarr (1992) ArticleTitleSelected management of pancreatic pseudocysts: operative versus expectant management Surgery 116 123
CJ Yeo JA Bastidas A Lynch-nyhan (1990) ArticleTitleThe natural history of pancreatic pseudocysts documented by computed tomography Surg Gynecol Obstet 170 411–417 Occurrence Handle2326721
Y Yunoki H Takeuchio Y Yasui K Tanakaya E Konaga K Hamazaki (1999) ArticleTitleUse of disposable stapler in operative cystogastrostomy for pancreatic pseudocyst Hepato–Gastroenterol 46 3271–3273
AL Warshaw DW Rattner (1985) ArticleTitleTiming of surgical drainage for pancreatic pseudocyst—Clinical and chemical criteria Ann Surg 202 720–724 Occurrence Handle4073984
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hauters, P., Weerts, J., Navez, B. et al. Laparoscopic treatment of pancreatic pseudocysts. Surg Endosc 18, 1645–1648 (2004). https://doi.org/10.1007/s00464-003-9280-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-003-9280-9