Abstract
Instant noodle manufacturing waste was used as feedstock to convert it into two products, bioethanol and biodiesel. The raw material was pretreated to separate it into two potential feedstocks, starch residues and palm oil, for conversion to bioethanol and biodiesel, respectively. For the production of bioethanol, starch residues were converted into glucose by α-amylase and glucoamylase. To investigate the saccharification process of the pretreated starch residues, the optimal pretreatment conditions were determined. The bioethanol conversion reached 98.5 % of the theoretical maximum by Saccharomyces cerevisiae K35 fermentation after saccharification under optimized pretreatment conditions. Moreover, palm oil, isolated from the instant noodle waste, was converted into valuable biodiesel by use of immobilized lipase (Novozym 435). The effects of four categories of alcohol, oil-to-methanol ratio, reaction time, lipase concentration and water content on the conversion process were investigated. The maximum biodiesel conversion was 95.4 %.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ever increasing global economic activities have led to a worldwide increase in oil consumption. According to some reports by international agencies, there will be a 53 % increase in energy demand by the year 2030 [1]. Hence, the inevitable shortage of the world’s traditional energy supplies has generated huge interest in alternative energy sources. In addition, the rising cost and reserves depletion of fossil fuels are and will be serious problems to humanity, making the discovery of new energy resources an extremely urgent mission [2, 3].
Considering the cost of conversion processes, waste biomass can be used to obtain alternative feedstock for biofuels. With the increasing demand for sustainable fuels and feedstock, waste biomass from industrial food manufacturing and daily life has become an obviously better choice as a source for alternative feedstock than waste biomass from agriculture and most lignocelluloses for biofuel conversion, because it is can provide a high yield of biofuel without any compromise to food supplies and other natural sources [4]. Apparently, using food waste as a raw material for bioenergy conversion has a higher value than using it as animal feed. In addition, food manufacturing waste is more attractive than wastes from other sources because of its low cost and constant consumption [5]. So far, starch and oil, which are common components of food, are the most attractive feedstock, widely used for conversion into bioenergy [6].
Noodle is one of most popular daily food, worldwide. The instant noodle manufacturing industry, one of the most important industries, depends completely on the supply of starch. Instant noodle manufacturing waste has been rarely used as a feedback source for bioenergy conversion. Moreover, there have been reports on the use of one food product for conversion into multiple bioenergy products. During the instant noodle manufacturing process, a huge quantity of residue stream is drained as a naturally abundant substrate of fermentation sugar with constant components. Although instant noodle waste has been recognized as being harmful as animal feed because of the adsorptive palm oil on the residual fragments, it can be a potential feedstock for the more valuable production of biodiesel. In addition, the Korean instant noodle production capacity has been ranked in the top ten worldwide in past decades. According to the statistics of Food Circulation Almanac of Korea (2011), around 3 billion packages of instant noodle were consumed in Korea in 2011, and more than 2,106 tons of instant noodle residues were treated as waste during the manufacturing process; these figures suggest that instant noodle manufacturing residue can be a sustainable resource for biofuel conversion [7]. In this study, we chose instant noodle, a very popular fast food for all age groups, which comprises the largest proportion of the food market in Korea, as a raw material for bioenergy conversion based on the results of industrial and economic feasibility [8, 9].
This study investigated the use of instant noodle (food waste) for bioethanol and biodiesel production, and determined the optimal saccharification conditions of starch and biodiesel conversion by lipase.
Materials and methods
Materials
The raw material evaluated in this study was instant noodle from a commercial company.
In the bioethanol conversion study, the commercial enzyme in powder form was obtained from Sigma (St. Louis. MO, USA). Two kinds of enzyme used, i.e., α-amylase from Aspergillus oryzae and glucoamylase from Aspergillus niger, had activities of 30 U/mg and 30–60 U/mg, respectively. The activity units of the enzymes were provided by the manufacturer. For α-amylase, 1 U corresponds to the amount of enzyme which liberates 1 μmol maltose per minute at pH 6.0 and 25 °C. For glucoamylase, 1 U will liberate 1.0 mg of glucose from starch in 3 min at pH 4.5 at 55 °C. S. cerevisiae K35 was used for ethanol production [10].
In the biodiesel conversion study, another commercial lipase, Novozym 435 (Candida antarctica lipase B immobilized on acrylic resin), was used, which was from the Novozymes company (in Netherlands). All other chemicals used in this study were of reagent grade.
Pretreatment of instant noodle
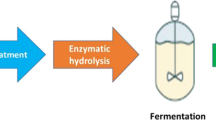
Figure 1 shows the schematic diagram of bioethanol and biodiesel production from instant noodle. Physical and chemical pretreatments isolated the substrate, starch and palm oil for two target conversions, from instant noodle waste. To obtain starch and palm oil from instant noodle, instant noodle residue (100 g) was boiled in distilled water (1 l), cooled to room temperature within 20 min, and then washed using hexane (500 ml). After filtration, starch residue (solid phase) and hexane solution (clean liquid phase) were obtained. For bioethanol production, starch residue was dried in the oven to remove moisture and ground to powder by a homogenizer. Then, it was used for saccharification as described below. For biodiesel production, the hexane solution was evaporated (above 100 °C, 30 min), and the palm oil was employed as substrate for conversion.
Bioethanol production
Enzymatic saccharification of starch residue
From basic experiments, the optimization for enzymatic saccharification was evaluated under the following conditions: 250 ml flask, 200 rpm, sodium acetate buffer (pH 5), temperature 37 °C, reaction time (3–24 h), dried starch residue (5–35 %, w/v), α-amylase (75–2,400 U/ml), glucoamylase (0.6–60.0 U/ml) and loading time of glucoamylase (0–12 h). Reaction mixtures were adjusted to pH 1.5 with 2 M HCl to stop the reaction and stored at 4 °C for subsequent high-performance liquid chromatography analysis (HPLC) [11].
Fermentation
The seed culturing of S. cerevisiae K35 was carried out in a 250-ml flask containing 100 ml YM medium at 30 °C and 200 rpm for 24 h. The seed medium was composed of 3 g/l yeast extract and 3 g/l malt extract. The culture broth was centrifuged at 12,000 rpm for 10 min [12, 13]. The cell pellet was washed using saline and suspended in 5 ml of saline. This suspension (5 %, v/v) was then inoculated into the main culture. The main culture, which was obtained from the broth of enzyme saccharification of instant noodle waste, was centrifuged to obtain the supernatant liquor for ethanol fermentation. The composition of the main medium was as follows: 5 g/l yeast extract, 1 g/l K2HPO4, 1 g/l MgSO4, 5 g/l peptone and diluted glucose solution from the instant noodle waste saccharification. The main culture for ethanol production was performed in a 250 ml flask containing 100 ml medium at 30 °C and 200 rpm for 72 h. Cell growth was monitored by measuring the optical density (OD) of the samples at 600 nm with a UV–Vis spectrophotometer (UVmini-1240, Shimadzu, Japan) [14].
HPLC analysis
The concentrations of sugar, ethanol and by-products were analyzed by HPLC (Shimadzu, Japan) with a refraction index detector using an Aminex HPX-87H column (300 mm × 7.8 mm, Bio-Rad, USA). The temperature of the column was set to 50 °C, and the injection volume was 20 μL at a flow rate of 0.8 ml/min. The mobile phase was 5 μM sulfuric acid [14]. The ethanol yield was calculated as a percentage of a maximum theoretical ethanol yield of 0.51 g ethanol per gram of glucose, which is a typical calculation method used in previous researches [15].
Biodiesel production
Enzymatic biodiesel conversion
Biodiesel was produced in a 100-ml flask, and stirred steadily at 300 rpm [16–18]. The reactants included 3 ml feedstock, i.e., palm oil isolated from instant noodle waste residue, methanol and catalysts. The optimized conditions for biodiesel conversion by immobilized lipase were determined in a stepwise manner, during which the effects of multiple alcohols (methanol, ethanol, butanol or isopropyl alcohol), methanol-to-oil molar ratios (0.3–45), methanol loading method (all loading at initial or 3-step loading at 0, 4 and 16 h reaction), lipase concentration (2.5–35 %, w/v) and distilled water concentration (0–4 %, v/v) were evaluated. The compositions of the products were analyzed using a gas chromatograph (GC) [17].
GC analysis
Biodiesel was analyzed using the GC M600D (Younglin Co. Ltd., Korea) with an HP-innowax 1909IN-133 column (30 m × 25 μm, Agilent, USA). The column temperature was raised from 140 to 245 °C at a rate of 5 °C/min, and then maintained at 245 °C for 10 min. The injector and detector temperatures were set to 250 °C [19]. Biodiesel conversion is based on weight determination. Conversion yield is calculated by the following equation:
ΣA: area of all peaks in result
A EI: area of methyl heptadecanoate
C EI: concentration of methyl heptadecanoate (mg/ml)
V EI: volume of methyl heptadecanoate (ml)
m: sample weight (mg)
Results and discussion
Effect of pretreatment
Depending on the instant noodle composition, the surface of the instant noodle residue absorbs fried palm oil during the manufacturing process. Therefore, the instant noodle residue contains both starch and fried palm oil, which represent at least two kinds of potential bioenergy substrates. Therefore, this experiment has two objectives: to convert the pretreated starch to bioethanol by enzymatic hydrolysis and yeast fermentation, and to convert the palm oil to biodiesel by transesterification. Pretreatment of instant noodle successfully separates it into two kinds of feedstocks for production of two kinds of bioenergies, respectively. The starch residue used for ethanol conversion was purified. The average yield of starch residue from instant noodle waste (100 g) was 83.3 g. In addition, 5 ml palm oil was isolated from 100 g of instant noodle waste.
Many studies indicated that modification of substrate properties before hydrolysis can efficiently increase the saccharification reaction [20]. In this study, pretreatment increased saccharification by 18.7 % by removing the hydrophobic composition, i.e., oil, as a result, the enzyme ability increased in the hydrolysis reaction. Due to boiling pretreatment, the physicochemical properties of starch were modified and improved in gelatinization. Size reduction increased the accessibility of enzyme in the hydrolysis reaction [21]. Furthermore, physical treatment (drying and grinding) can increase the surface area of residual starch granules (in case of decreased crystallinity) to increase the contact between enzymes and substrate and consequently, enhance the saccharification efficacy [4]. Accordingly, it is important to pretreat instant noodle for high saccharification efficiency.
Meanwhile, the oil isolated from instant noodle was used as the substrate for biodiesel conversion. Therefore, instant noodle waste has been utilized completely.
Starch hydrolysis and bioethanol production
Since saccharification is a prerequisite for fermentation of starch to produce ethanol, optimization of this step has necessarily been investigated individually for fermentation at high glucose concentration. Therefore, separate hydrolysis and fermentation (SHF) process was selected in this study. The optimal saccharification conditions of starch residue by α-amylase and glucoamylase were investigated in a stepwise manner.
The optimal saccharification conditions are shown in Fig. 2. The glucose yield increased from 37.3 to 48.4 % (w/w) with increasing α-amylase concentration (75–300 U/ml) (Fig. 2a). Higher concentrations of α-amylase (over 300 U/ml) did not enhance saccharification significantly, which means that the saccharification by α-amylase was saturated at the enzyme concentration of 300 U/ml. Similar results were obtained for glucoamylase (Fig. 2b). With increasing glucoamylase concentration (0.6–60.0 U/ml), the glucose yield continued to increase. A significant increase in glucose yield (69.4 %, w/w) was obtained at glucoamylase concentration of 12 U/ml. As shown in Fig. 2a, b, the optimal concentrations of α-amylase and glucoamylase were 300 and 12 U/ml, respectively. In addition, the effect of the glucoamylase on saccharification efficacy was much stronger than α-amylase, even though glucoamylase concentration was much lower than α-amylase concentration.
Effects of α-amylase concentration (a), glucoamylase concentration (b), glucoamylase loading time (c) and substrate concentration (d) on enzymatic saccharification of starch residue. a 75–2,400 U/ml α-Amylase; 6 U/ml glucoamylase; 30 % (w/v) starch; 12 h; 37 °C; 200 rpm. b 300 U/ml α-Amylase; 0.6–60 U/ml glucoamylase 30 % (w/v) starch; 12 h; 37 °C; 200 rpm. c 300 U/ml α-Amylase; 12 U/ml glucoamylase; 30 % (w/v) starch; 24 h; 37 °C; 200 rpm. d 300 U/ml α-Amylase; 12 U/ml glucoamylase; 5–30 % (w/v) starch; 37 °C, 200 rpm
The hydrolysis effect of each enzyme was investigated with two enzymes loading in a stepwise manner. Figure 2c shows the results of saccharification when glucoamylase was loaded at 0, 3, 6, 9 and 12 h after α-amylase loading, respectively. The saccharification rate increased after glucoamylase loading. The initial 3 h reaction rates after glucoamylase loading were 14.5, 37.8, 26.5 and 20.1 g/l h when glucoamylase was loaded at 0, 3, 6, and 9 h later, respectively. In addition, the most efficient hydrolysis was achieved at glucose concentration of 182.1 g/l when glucoamylase was loaded 3 h after α-amylase loading. These two analysis results showed that saccharification was improved when glucoamylase was loaded 3 h after α-amylase loading. This improvement may be due to the different functions and reaction orders of α-amylase and glucoamylase in starch digestion kinetics. First, α-amylase breaks down starch polymers into soluble dextrin by hydrolyzing α-1,4 bonds; then, it is hydrolyzed by glucoamylase and converted into glucose [5]. Therefore, the hydrolysis by α-amylase prepares the substrate for glucoamylase. Because of these chain reactions that increase the substrate for the glucoamylase, glucoamylase loading after α-amylase loading can increase the glucose yield.
In addition to the enzymes, substrate concentration was another critical factor of starch residue saccharification. Figure 2d shows the effect of substrate concentration on saccharification. The curves show similar trends. The initial glucose concentration was almost 0, it increased until 6 h, and it maintained around the maximum until 9 or 12 h. There was almost no insoluble substance after enzymatic hydrolysis at substrate concentrations under 5 % (w/v). And when the substrate concentration was above 35 % (w/v), the viscosity of the mixture of substrate and buffer was too high to make the mixture fluid. Therefore, the proper range, 5–35 % (w/v), of substrate concentration was investigated. Figure 2d shows that the substrate concentration increased from 5 to 25 % (w/v) regularly, but sharply decreased from 25 to 35 % (w/v). This result indicated that 25 % (w/v) was the saturation substrate concentration for this enzymatic hydrolysis reaction. When the substrate concentration was over 25 % (w/v), the low fluidity of the reaction system obstructed the activities of the enzymes and substrate in the reaction. The decreased accessibility of enzymes was probably one of the most important inhibition factors here. Moreover, the low solubility of the substrate was probably another negative influence of this hydrolysis reaction. Considering the subsequent process, fermentation, high saccharification was required in the hydrolysis process. Figure 2d also indicates that the glucose concentration remained between 150 and 200 g/l, when the substrate concentration and reaction time were over 20 % (w/v) and 9 h, respectively. The highest glucose concentration was obtained with 25 % (w/v) substrate. The hydrolysis reaction finished within 9 h, unaffected by the increasing substrate concentration.
In conclusion, both α-amylase and glucoamylase were used for the saccharification of the starch residue, and the optimal conditions were: 300 U/ml α-amylase, 12 U/ml glucoamylase, 25 % (w/v) instant noodle starch residue at 37 °C, and 9 h reaction time. The results also indicated that the rate of saccharification by the two enzymes increased when glucoamylase was added to the reaction 3 h after the α-amylase loading.
After saccharification, ethanol was produced by S. cerevisiae K35. The starch residue was fermented within 24 h at 30 °C, 250 rpm, with an initial glucose concentration around 84 g/l under the optimum saccharification conditions. The result of the fermentation is shown in Fig. 3. Many researches showed that ethanol fermentation using microorganism can be influenced by impure substrate [10, 22]. They also indicated that initial fermentation performance can be improved by an additional process of fermentable sugar purification. Cell growth was slower in the initial 6 h compared to its growth with reagent level pure glucose as a carbon source (not shown in this paper but in [22]), probably because of the negative influence of the impure substrate, which was crude glucose, extracted and degraded from biomass without purification. The cell grew and ethanol production sharply increased within the next 6 h, but a noticeable increase in ethanol production was not observed after 12 h due to complete glucose consumption. The maximum concentration of ethanol was obtained after 24 h (approximately 41.3 g/l), and the ethanol conversion reached 98.5 % after 24 h of fermentation.
By-product level is one of the important factors affecting the production of target products in the bioindustry, due to its influence on microorganism metabolism. Therefore, it is necessary to investigate the performance of by-products conversion to understand and control fermentation process. In this study, glycerol and acetate were the main by-products of the ethanol fermentation process, which are common by-products of S. cerevisiae K35 in aerobic condition ethanol fermentation [23, 24]. By-product conversion performance is demonstrated in Fig. 3. The concentrations of glycerol and acetate increased from 0 to 3.4 g/l and 3.5 g/l, respectively, within the initial 9 h of fermentation. After then, the glycerol concentration was maintained at approximately 3.1 g/l. The acetate concentration was increased with the same trend as the ethanol concentration in the following 9 h, and then finally maintained at approximately 6.9 g/l.
Biodiesel production
Palm oil isolated from instant noodle was employed as a feedstock for biodiesel production. The most significant factors for biodiesel production by lipase were investigated in a stepwise manner, i.e., alcohol effect, ratio of alcohol-to-oil, alcohol loading method, lipase and water concentration [25–27].
The results of biodiesel production by use of lipase are shown in Fig. 4. The performances of four kinds of alcohols are shown in Fig. 4a. The highest conversion was 85.7 % when methanol was employed. Some researches mentioned that fatty alcohols with the carbon lengths above three could completely dissolve in vegetable oils, which is an advantage in biodiesel conversion [28]. But, our result indicates that the shortest chain alcohol has the highest performance in enzymatic conversion of used palm oil isolated from instant noodle wastes. The accessibility of lipase, alcohol and substrate oil is an important factor in biodiesel conversion. It is reasonable hypothesize that methanol as the smallest molecular alcohol in this study, advantaged to increase accessibility of lipase, alcohol and substrate oil. The boiling temperature of methanol is 64.5–64.7 °C, which is the lowest one of these four kinds of alcohol. This low boiling temperature can be a physical performance advantage of downstream separation process in industrial application.
Effects of alcohol (a), Methanol-to-oil molar ratio (b), Methanol 3-step loading and reaction time (c), lipase concentration (d), and water content (e). a 6 Alcohol-to-oil molar ratio; 48 h; 20 % (w/v) lipase; 3 % (v/v) water; 40 °C. b 1–18 Methanol-to-oil molar ratio; 48 h; 20 % (w/v) lipase; 3 % (v/v) water; 40 °C. c 9 Methanol-to-oil molar ratio; 72 h; 20 % (w/v) lipase; 3 % (v/v) water; 40 °C. d 9 Methanol-to-oil molar ratio; 36 h; 2.5–35 % (w/v) lipase; 3 % (v/v) water; 40 °C. e 9 Methanol-to-oil molar ratio; 36 h; 15 % (w/v) lipase; 0.5–4 % (v/v) water; 40 °C
Figure 4b represents the effect of methanol-to-oil molar ratio on biodiesel production. The conversion increased rapidly at a molar ratio <6, and dropped after reaching the molar ratio of 12. The maximum conversion (88.8 %) was obtained at a methanol-to-oil molar ratio of 9, which was much higher than the stoichiometric amount of methanol (methanol-to-oil ratio 3:1) required for complete conversion. Talukder et al. [29] research claimed that Novozym 435 was deactivated because of its low solubility of methanol in refined oil. On the contrary, the molar ratio of methanol-to-oil in this study is reasonably over 3, for the adequately higher stoichiometric amount of methanol. This is probably due to half of the stoichiometric amount of methanol being completely dissolved in palm oil [30]. In addition, the overdose of methanol could have enhanced the solubility of the by-product glycerol of biodiesel conversion to reduce the product inhibition and viscosity of this reaction system. However, the conversion decreased when the ratio was over 12, indicating that excessive amounts of methanol (over loading) would be a barrier to enzyme and substrate contact.
The traditional method of methanol loading is to add the total amount at initiation. Recent research showed that conversion could be increased by adding methanol in three steps to the reaction, i.e., 1/3 of the stoichiometric amount at each time—at initiation, 4 and 16 h [29]. The result in Fig. 4c shows that three successive additions of 1/3 of methanol dosage resulted in conversion of above 85.1 and 88.7 % after 36 and 48 h, respectively. Figure 4c also shows three plateaus in the biodiesel conversion curve, i.e., at 3–6 h, 12–15 h and after 24 h, respectively. The conversion increased rapidly after each methanol loading, which means that methanol loading in steps decreased the inhibition of the enzyme reaction. Therefore, this enzymatic biodiesel conversion process was optimized in a stepwise manner. According to a reference survey and fundamental experiments, biodiesel conversion by Novozym 435 took approximately 48 h [25, 28–33]. In a previous study, 48 h has been used as reaction time condition in the investigation of the two factors, which are the effect of the alcohol categories and methanol-to-oil ratio. Figure 4c also represents the biodiesel conversion in 72 h at 3 h intervals. Considering the reaction cost and productivity, the optimal reaction time was 36 h, which was employed in the subsequent investigation.
The biodiesel conversion increased with the increase in lipase concentration up to 15 % (w/v), after which it remained at almost the same concentration of 30 % (w/v) (Fig. 4d). The highest conversion (88.0 %) was reached within 36 h. The results indicated that the conversion reaction was saturated when lipase concentration reached 15 % (w/v). Figure 4e shows the effect of the water concentration. The optimum biodiesel conversion was 95.4 % when water content was 2.0 % (v/v). The influence of water was significant when its concentration was greater than 0.5 % (v/v), and inhibitory effect of water on the conversion appeared when its concentration was over 2 % (v/v). Since both water and methanol act as hydroxyl donors and react with the acyl-enzyme complex, hydrolysis decreased at a lower water activity, while methanolysis was favored [31]. Appropriate methanol and water loadings can increase the diffusion and decrease the inhibition of the by-product glycerol, which was adsorbed onto Novozym 435. In addition, the highest biodiesel conversion of 95.4 % was achieved in 12 h, a shorter reaction time (36 h) than the average optimum reaction time (48 h) in current studies [16].
The palm oil isolated from instant noodle residue was the feedstock for biodiesel production. The optimum conversion conditions of immobilized lipase (Novozym 435) were as follows: methanol-to-oil molar ratio of 9; three optimal methanol loading times at 0, 4, and 16 h; enzyme concentration of 15 % (w/v); water concentration of 2 % (w/v); reaction temperature of 40 °C; and reaction time of 36 h.
Generally, chemical conversion of biodiesel is more efficient and easier to control than enzymatic one [32, 33]. However, an immobilized lipase is environmentally friendly, and its reuse can decrease the process cost. Moreover, the negative effect of carotenoids, the common components of used palm oil, on the activity of Novozym 435 was difficult to avoid [25].To optimize the enzymatic biodiesel production, the avoidance of enzyme inhibition, decrease of reaction time and lowering of cost should be investigated in future works. Meanwhile, the choice of the catalyst also depends on the components of the feedstock oil. Although some lipases, like Novozym 435, catalyze both the esterification of free fatty acids and the transesterification of triglycerides, where both reactions produce biodiesel, a pretreatment to enhance the effect of esterification cannot be avoid before alkali catalysis. An acid catalyst is more effective than an alkali one when the free fatty acid content of feedstock oil is >0.5 % (w/w) [34–36].
Conclusions
This study successfully established a conversion process for instant noodle waste as an economical and environmentally friendly source to produce two kinds of biofuels—bioethanol and biodiesel—after pretreatment. The conversion conditions were optimized to yield high conversions. In this study, instant noodle waste was completely isolated as starch residue and palm oil by a pretreatment process and converted to bioethanol and biodiesel at conversion 98.5 and 95.4 %, respectively. The high yield of instant noodle manufacturing would support the industrial-scale use of instant noodle waste as an attractive substrate of bioenergy.
References
Miranda JR, Passarinho PC, Gouveia L (2012) Pre-treatment optimization of Scenedesmus obliquus microalgae for bioethanol production. Bioresour Technol 104:342–348
Kim KH, Eom IY, Lee SM, Cho ST, Choi IG, Choi JW (2012) Applicability of sub- and supercritical water hydrolysis of woody biomass to produce monomeric sugars for cellulosic bioethanol fermentation. J Ind Eng Chem 16:918–922
Zhu MJ, Zhu ZS, Liang L, Xu WX (2012) Production of bioethanol from sugarcane biomass using NH4OH–H2O2 pretreatment and simultaneous saccharification and co-fermentation. Biotechnol Bioprocess Eng 17:316–325
Hashem M, Darwish SMI (2010) Production of bioethanol and associated by-products from potato starch residue stream by Saccharomyces cerevisiae. Biomass Bioenergy 34:953–959
Chen H, Qiu W (2010) Key technologies for bioethanol production from lignocellulose. Biotechnol Adv 28:556–562
Shanavas S, Padmaja G, Moorthy SN, Sajeev MS, Sheriff JT (2011) Process optimization for bioethanol production from cassava starch using novel eco-friendly enzymes. Biomass Bioenergy 35:901–909
Food Journal (2011) Food distribution yearbook of Korea. Food Journal Co. Ltd, South Korea
Yan SB, Li J, Chen XS, Wu JY, Wang PC, Ye JF, Yao JM (2011) Enzymatical hydrolysis of food waste and ethanol production from the hydrolysis. Renew Energy 36:1259–1265
Rojan PJ, Anisha GS, Nampoothiri KM, Pandey A (2011) Micro and macro algal biomass: a renewable source for bioethanol. Bioresour Technol 102:186–193
Lee H, Cho DH, Kim YH, Shin SJ, Kim SB, Han SO, Lee J, Kim SW, Park C (2011) Tolerance of Saccharomyces cerevisiae K35 to lignocellulose-derived inhibitory compounds. Biotechnol Bioprocess Eng 16:755–760
Iliana BM, Axayácatl RGG, Edgar SM, Juan SAB (2011) A Simple metabolic flux balance analysis of biomass and bioethanol production in Saccharomyces cerevisiae fed-batch cultures. Biotechnol Bioprocess Eng 16:13–22
Talebnia F, Karakashev D, Angelidaki I (2010) Production of bioethanol from wheat straw: an overview on pretreatment, hydrolysis and fermentation. Bioresour Technol 101:4744–4753
Won KY, Kim YS, Oh KK (2012) Comparison of bioethanol production of simultaneous saccharification & fermentation and separation hydrolysis & fermentation from cellulose-rich barley straw. Korean J Chem Eng 29(10):1341–1346
Lee SJ, Kim SB, Kang SW, Han SO, Park C, Kim SW (2012) Effect of crude glycerol-derived inhibitors on ethanol production by Enterobacter aerogenes. Bioprocess Biosyst Eng 35:85–92
Itsuki W, Naonori M, Akira A, Riki S, Ken T, Toshihide N (2012) Ethanol production by repeated-batch simultaneous saccharification and fermentation (SSF) of alkali-treated rice straw using immobilized Saccharomyces cerevisiae cells. Bioresour Technol 123:695–698
Seong PJ, Jeon BW, Lee M, Cho DH, Kim DK, Jung KS, Kim SW, Han SO, Kim YH, Park C (2011) Enzymatic coproduction of biodiesel and glycerol carbonate from soybean oil and dimethyl carbonate. Enzyme Microb Technol 48:505–509
Lee JH, Kim SB, Park C, Kim SW (2010) Effect of a buffer mixture system on the activity of lipases during immobilization process. Bioresour Technol 101:566–570
Lee JH, Kim SB, Kang SW, Song YS, Park C, Han SO, Kim SW (2011) Biodiesel production by a mixture of Candida rugosa and Rhizopus oryzae lipases using a supercritical carbon dioxide process. Bioresour Technol 102:2105–2108
Lee JH, Kwon CH, Kang JW, Park C, Tea B, Kim SW (2009) Biodiesel production from various oil under supercritical fluid conditions by Candida antartica lipase B using a stepwise reaction method. Appl Biochem Biotechnol 156:454–464
Simone B, Charles EW (2010) Review: continuous hydrolysis and fermentation for cellulosic ethanol production. Bioresour Technol 101:4862–4874
Han M, Kim Y, Chung B, Kim Y, Choi GW (2011) Bioethanol production from optimized pretreatment of cassava stream. Korean J Chem Eng 28(1):119–125
Kim SB, Lee SJ, Lee JH, Jung YR, Thapa LP, Kim JS, Um Y, Park C, Kim SW (2013) Pretreatment of rice straw with combined process using dilute sulfuric acid and aqueous ammonia. Biotechnol Biofuels 6:109
Yu KO, Kim SW, Han SO (2010) Engineering of glycerol utilization pathway for ethanol production by Saccharomyces cerevisiae. Bioresour Technol 101:4157–4161
Frick O, Wittmann C (2005) Characterization of the metabolic shift between oxidative and fermentative growth in Saccharomyces cerevisiae by comparative 13C flux analysis. Microb Cell Fact 4:30
Gog A, Roman M, Tosa M, Paizs C, Irimie FD (2012) Biodiesel production using enzymatic transesterification—current state and perspectives. Renew Energy 39:10–16
Jang MG, Kim DK, Soon C, Park SC, Lee JS, Kim SW (2012) Biodiesel production from crude canola oil by two-step enzymatic processes. Renew Energy 42:99–104
Jung H, Lee Y, Kim D, Han SO, Kim WK, Lee J, Kim YH, Park C (2012) Enzymatic production of glycerol carbonate from by-product after biodiesel manufacturing process. Enzyme Microb Technol 51:143–147
Talukder MR, Wu JC, Nguyen TBV, Ng MF, Melissa YLS (2009) Novozym 435 for production of biodiesel from unrefined palm oil: comparison of methanolysis methods. J Mol Catal B Enzym 60:106–122
Talukder MR, Das P, Fang TS, Wu JC (2011) Enhanced enzymatic transesterification of palm oil to biodiesel. Biochem Eng J 55:119–122
Park JY, Lee JS, Wang ZM, Kim DK (2010) Production and characterization of biodiesel from trap grease. Korean J Chem Eng 27(6):1791–1795
Egues I, Alriols MG, Herseczki Z, Marton G, Labidi J (2010) Hemicelluloses obtaining from rapeseed cake residue generated in the biodiesel production process. J Ind Eng Chem 16:293–298
Go A, Ko JK, Lee SJ, Kim SW, Han SO, Lee J, Woo HM, Um Y, Nam J, Park C (2012) Process design and evaluation of value-added chemicals production from biomass. Biotechnol Bioprocess Eng 17:1055–1061
Yan J, Li A, Xu Y, Ngo TPN, Phua S, Li Z (2012) Efficient production of biodiesel from waste grease: one pot esterification and transesterification with tandem lipases. Bioresour Technol 123:332–337
Saqib M, Mumtaz MW, Mahmood A, Abdullah MI (2012) Optimized biodiesel production and environmental assessment of produced biodiesel. Biotechnol Bioprocess Eng 17:617–623
Saifuddin N, Raziah AZ, Farah HN (2009) Production of biodiesel from high acid value waste cooking oil using an optimized lipase enzyme/acid-catalyzed hybrid process. J Chem 6(S1):S485–S495
Tan KT, Lee KT, Mohamed AR (2011) Potential of waste palm cooking oil for catalyst-free biodiesel production. Energy 36:2085–2088
Acknowledgments
This work was supported by the Advanced Biomass R&D Center (ABC-2011-0031360) of Global Frontier Project funded by the Ministry of Science, ICT and Future Planning of Korea and Advanced Production Technology Development Program (309016-5) of the Ministry of Agriculture, Food and Rural Affairs, Republic of Korea and Creative Allied Project (CAP) of the Korea Research Council of Fundamental Science and Technology (KRCF)/Korea Institute of Science and Technology (KIST) (2E24832).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yang, X., Lee, J.H., Yoo, H.Y. et al. Production of bioethanol and biodiesel using instant noodle waste. Bioprocess Biosyst Eng 37, 1627–1635 (2014). https://doi.org/10.1007/s00449-014-1135-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-014-1135-3