Abstract
The aims of this research were to screen and characterize a new microbial source of γ-PGA, to optimize aspects of culture conditions and medium composition using central composite design and response surface methodologies. The influence of bioreactor stirring rates on the production of γ-PGA was also investigated and the oxygen volumetric mass transfer coefficients (k La) were established. The most productive strain was identified by 16S rDNA analysis as Bacillus subtilis, and its γ-PGA production in rotatory shaker was threefold increased under optimized conditions (37 °C, pH 6.9, and 1.22 mM Zn2+), compared to conventional medium. In bioreactor, the γ-PGA production was further increased, reaching 17 g l−1, 70 % higher than shaker cultures. γ-PGA production showed high dependency on oxygen transfer. At k La of 210 h−1, the cultivation time could be reduced to 48 h, about 50 % of the time required for operations at k La 55 h−1.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Poly-gamma-glutamic acid (γ-PGA) is a homo-polyamide of d- and l-glutamic acid units [1]. This polymer presents γ-amide linkages between α-amino and γ-carboxylic groups as opposed to α-amino linkages in proteins. Consequently, this polyamide is resistant to proteases [2]. γ-PGA is water soluble, biodegradable, edible, biocompatible, non-toxic, and non-immunogenic. Applications of this biopolymer and its derivatives have been widely reported for food, chemical, medical, and environmental-related industries, as flocculants in water and wastewater treatment, thickener, cryoprotectant and functional ingredient in food processing, and as an anticancer drug-release agent in medicine [1, 3–8], but these applications are still very limited due to the high cost of γ-PGA.
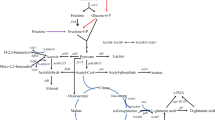
γ-PGA was first identified 70 years ago as a component of the Bacillus anthracis capsule [1, 9]. Further studies have also shown its presence in extracellular viscous material of other non-pathogenic Bacillus species such as B. subtilis, B. licheniformis, B. megaterium, and B. halodurans [1, 9–11]. A few Gram (+) bacteria, archeobacteria, and some eukaryotes can also synthesize γ-PGA [2, 3]. γ-PGA biosynthesis is a membrane-associated process, which requires ATP and glutamic acid as a substrate and cations as cofactors. The enzymatic complex PgsBCA (polyglutamate synthetase) is responsible for the polymerization and transport of γ-PGA through the cellular membrane [2, 3]. The role of PgsBCA complex was described in detail by Ashiuchi and co-workers [11–13]. Initially, glutamic acid binds to the ATP molecule, which is converted to ADP providing energy for the polymerization. PgsBCA acts as an amide-ligase, the subunits PgsB and PgsC forming a catalytic site, while the subunit PgsA removes the elongated chain from the active site allowing for the addition of a new monomer. According to Ashiuchi et al. [13], Mg2+ is essential, and Zn2+ acts as a booster to PgsBCA activity in vitro.
In recent years, the demand for environmentally safe materials, especially those obtained from renewable resources, have increased the interest for γ-PGA biosynthesis and its applications [4, 8, 14]. Bacillus γ-PGA producers have been isolated from soil and traditional foods, mainly in Asian countries [12, 15–20]. Some researchers have investigated the influence of nutritional and process parameters on γ-PGA production in order to seek strategies to increase yield of production to allow cost-effective industrial production of this polymer.
The media formulation for the production of γ-PGA is still one of the major industrial problems, mainly due to cost. The glutamic acid monomers incorporated in γ-PGA can be obtained from culture medium or can be synthesized from precursors derived from tricarboxylic acid (TCA) cycle by microorganisms [1, 3]. Medium “E”, developed by Leonard and co-workers [21], is frequently used to grow Bacillus for γ-PGA production in submerged cultivation [22–25]. Glycerol, citric acid and l-glutamic acid as medium components are reported as necessary for biomass and γ-PGA production [22–28]. Citric and l-glutamic acids act as precursors of polymer [1, 3, 26], while glycerol acts as a co-substrate [27].
The analysis of the influence of oxygen transfer mechanism is of fundamental importance in aerobic bioprocesses, especially those involving the production of biopolymers that change the rheological properties of culture broth over time, and is regarded as the key factor for understanding and optimizing these processes. However, relatively few studies have investigated the influence of the oxygen mass transfer on the production of γ-PGA [26, 29, 30], and the optimal oxygen volumetric transfer rates for γ-PGA production have never been reported.
In this context, the aims of this research were to identify new microbial sources of γ-PGA, characterize them, and to apply the central composite design and response surface methodology in order to optimize culture conditions in terms of pH, temperature, and Zn2+ concentration to enhance γ-PGA production. Furthermore, we scaled up the γ-PGA cultivation in bioreactors, evaluated the influence of stirring rate over the oxygen uptake rate, oxygen volumetric mass transfer coefficient, cell viability and production and productivity of γ-PGA.
Materials and methods
Microorganisms
A total of 87 different bacterial strains of our own collection composed of isolates from the aquatic and dumped soils of the Amazonian environment in Brazil were pre-tested for biopolymer production. Six of them, named as BL15, BL16, BL32, BL53, BL62, and BL74, showed highly viscous culture broths and were chosen for γ-PGA production screening. These strains were kept as lyophilized reference stocks in the Culture Collection of the Microbiology Department of The Federal University of Rio Grande do Sul State, Brazil. For working purposes, isolates were kept in 20 % glycerol at −20 °C. Bacteria isolates were reactivated twice in Luria–Bertani (LB) broth, streaked onto LB agar plates and subcultured onto LB slants. Strain slants were kept under refrigeration until usage.
Cultivation medium
Microorganisms were cultivated in medium E (ME), which was composed of (in g l−1) l-glutamic acid, 20; citric acid, 12; glycerol, 80; NH4Cl, 7; MgSO4·7H2O, 0.5; FeCl3·6H2O, 0.04; K2HPO4, 0.5; CaCl2·2H2O, 0.15, and MnSO4·H2O, 0.04 [21].
Inoculum preparation
The inocula preparation for cultivations were prepared by transferring a loopful of cells from LB slants into 50 ml LB broth in 250 ml Erlenmeyer flasks and incubated at 37 °C in a rotatory shaker at 180 rpm. The inocula were standardized to 1.0 ± 0.1 OD at 600 nm and added to cultivation medium at 4 % (volume fraction).
Screening γ-PGA-producing strains
Screening cultivations were carried out on medium E, with the final pH adjusted to 6.5 using either 1 M HCl or NaOH. Each strain was inoculated into 25 ml by cultivation on medium E in 125-ml Erlenmeyer flasks and incubated at 37 °C for 96 h in a rotatory shaker at 180 rpm. Concentration of γ-PGA in culture medium was measured and the results were subjected to analyses of variance (ANOVA) followed by Fisher LSD (p < 0.01). The ANOVA and LSD were performed with Statistica 7.0 (Statsoft Inc., Tulsa, USA). All shake experiments were run in triplicates.
Identification and characterization of γ-PGA producer strain
The best producer of γ-PGA was characterized and identified by 16S rDNA analysis and by morphological and physiological characteristics. DNA from a bacterial suspension equivalent to a McFarland 0.5 (200 μl) was extracted using the QIAamp DNA minikit (Quiagen®) following the manufacturer instructions. PCR reaction was performed using the Platinun Taq Supermix (Invitrogen) and 10 μl of the extracted DNA in a final volume of 50 μl. The 16S rDNA gene was amplified using primers 285 and 261 under cycling conditions previously described [31]. PCR products were separated by agarose gel electrophoresis and the ethidium bromide-stained bands were visualized under UV light. After confirmation, the remaining PCR mix was purified using the AccuPrep(R) PCR Purification Kit (Bioneer) and submitted to direct sequencing using the BigDye Terminator kit (Applied Biosystems), as directed by the manufacturer instructions. Following sequencing, the nucleotide sequence was subjected to BLAST sequence analysis using the NCBI (National Center for Biotechnology Information) website, and the nucleotide sequence was deposited in GenBank database.
Morphological and biochemical characterization was performed according to the Manual of Bergey [32] and MacFadin [33], and also included additional references.
Optimization of culture parameters for γ-PGA production by response surface methodology (RSM)
The influence of temperature, initial culture pH, and concentration of Zn2+ supplemented to medium E were evaluated for the best producer of γ-PGA strain in experiments conducted with 25 ml of medium in 125-ml Erlenmeyer flasks in rotary shaker at 180 rpm. A 23 central composite design (k = 3, α = 1.68), including 6 axial points and 3 replicates at the central point was designed, resulting in 17 runs, and the three culture parameters were optimized for γ-PGA production by response surface methodology (RSM). A second-order polynomial regression model was obtained from experimental design according to the following equation:
where Y is the response variable (γ-PGA production); β 0 is the constant; β i , β ii , and β ij are the coefficients for the linear, quadratic, and interaction effects, respectively; X i and X j are the coded independent variables or factors.
Analysis of variance (ANOVA) was employed in order to evaluate the significance of the regression model. Significance of regression coefficients was determined by Student’s t test and the variance explained by the model is given by the multiple determination coefficient, R 2. Experimental design and analysis of results were carried out using Statistica 7.0 (Statsoft Inc., Tulsa, USA).
The time course of γ-PGA production was evaluated for 96 h using the optimal parameters obtained. Submerged cultures were conducted with 25 ml of medium in 125-ml Erlenmeyer flasks, incubated in rotatory shaker at 180 rpm, and samples were taken for quantification of γ-PGA, and count of viable cells.
Submerged cultivations in bioreactors
Submerged batch cultures were performed in a fully instrumented 5-l stirred tank bioreactor (Biostat B bioreactor, B. Braun Biotech International, Germany), under the optimized conditions using an aeration rate of 2.0 vvm (2 l of air/liters of medium/min) and stirring agitation of 500, 750, and 1,000 rpm. The cultivations were carried out without pH control. Assays were carried out with 2.5 l of culture medium including 4 % of inoculum in LB broth standardized to 1.0 ± 0.1 OD at 600 nm. A small amount of 0.2 % (volume fraction) of Antifoam 204 (organic non-silicone Emulsion, Sigma, USA) was added for foaming control. During the time course of cultivation, samples were taken for quantification of γ-PGA, dry biomass, and for count of viable cells. All bioreactor experiments were run in duplicates.
Determination of growth kinetic parameters
Curves of viable cell growth were fitted to the Logistic Model shown in Eq. 2 [34] by using the OriginPro 8.0 software (OriginLab, USA).
where, X is the total count of viable cells (CFU ml−1) and t is time (h). The parameters X max, μ max and t c are obtained from the experimental data fit. X max represents the maximum amplitude of X in the stationary phase plateau, μ max is the maximum specific growth rate (exponential phase) and t c is the time in which X = X max/2.
Determination of OUR, Q O2 and kLa
Dissolved oxygen concentration (DOC) in the culture broth was measured using a polarographic electrode (Mettler-Toledo, Germany), and was expressed as percentage of oxygen saturation. An acquisition board connected to a computer running MATLAB (Mathworks, USA) was used to acquire data of dissolved oxygen. Oxygen uptake rate (OUR), specific rate of oxygen consumption (Q O2), and volumetric oxygen transfer coefficient (k La) were determined following the dynamic gassing-out method [35]. The parameters OUR, Q O2, and k La were calculated within the exponential growth phase (at 3, 6 and 8 h), except for 500 rpm, which was calculated only at 3 h of run because dissolved oxygen was very low after this time.
Analytical methods
The amount of γ-PGA was determined by spectrophotometric method based on the complexation reaction of γ-PGA with cetylmethylammonium bromide (CTAB) [36]. In this method CTAB binds very specifically to γ-PGA, and form a water-insoluble, highly dispersed micelle-like complex, resulting in an increase in turbidity, which is measured at 400 nm [36, 37]. γ-PGA (MM of 70–100 kDa, Sigma-Aldrich, USA) was used as spectrophotometric standard. Procedure for polymer extraction from culture medium was as follows. Trichloroacetic acid solution was added to samples until pH was reduced to 3.0 in order to separate cells and proteins [22, 36]. The mixture was kept at 50 °C for 30 min, and centrifuged at 15,000g, 15 °C for 30 min. The supernatant was collected, the pH adjusted to 7.0 ± 0.1 and five volumes of ethanol were added to samples. A second centrifugation was conducted at 15,000g, 4 °C and 30 min. The pellet was dissolved in buffer solution pH 7.0 ± 0.1 and the complexation reaction was carried out.
Biomass was determined as dry cell weight by centrifugation of the culture medium at 3,000g for 20 min at 4 °C; cell pellets were twice washed and dried at 75 °C until constant weight. The viable cells were determined by direct plate count procedure using the spread plate technique. After successive decimal dilutions of the cultivation broth, the adequate dilutions were spread on to Agar Plate Count Medium and incubated at 37 °C for 18 h.
Results and discussion
Screening and identification of γ-PGA-producing strains
Table 1 shows the γ-PGA production by BL15, BL16, BL32, BL53, BL62, and BL74 strains in ME after 24, 72 and 96 h. It can be observed that all strains had the ability to produce γ-PGA, except for BL32. The highest productions were obtained for BL53 and BL74 strains. BL53 produced approximately 4 g l−1 γ-PGA in 72 h of cultivation. As a comparison, for a well-studied strain B. licheniformis CCRC 12826, Shih and co-workers [24] obtained 5.27 g l−1 of γ-PGA, but only after 96 h of cultivation in medium E under similar cultivation conditions. After 96 h all strains showed a decline on γ-PGA production, probably due to PGA depolymerase enzymes, which could decrease the yield of γ-PGA during stationary phase [3]. Results also showed that BL53 produced significantly more γ-PGA at early growth stages when compared to BL74 strain. The strain identification showed that BL74 could be morphologically and physiologically identified as B. anthracis. Consequentially, BL74 was discharged and only BL53 strain was selected for further experiments.
The 16S rDNA sequence obtained from BL53 strain was deposited in NCBI GenBank database under the accession number JQ359757. This nucleotide sequence was compared to available sequences at NCBI GenBank database, and revealed 100 % of similarity with several B. subtilis and Bacillus amyloliquefaciens strains, and also to some Bacillus polyfermenticus and Bacillus siamensis. Taxonomic studies have indicated that Bacillus species are very similar to each other with respect to 16S rDNA, and B. amyloliquefaciens is indeed closely related to B. subtilis on the basis of 16S–23S ITS nucleotide sequences [38–40]. Nevertheless, some physiological characteristics allowed distinguishing among these species [32, 40–44]. The morphological and physiological characterization of BL53 strain showed that it is aerobic, rod-shaped, spore-forming, catalase-positive, Gram(+) bacterium, capable of producing acid from glucose, as a typical Bacillus sp. [32]. The positive citrate reaction, negative oxidase test, growth at 50 °C, no acid formation from lactose and sorbitol, and growth in NaCl 7 % but not in NaCl 10 % allowed for differentiating from other Bacillus species and for the identification of BL53 as B. subtilis. All morphological and physiological characteristics for BL 53 strain are shown in Table 2.
Optimization of cultivation using the CCD and RSM
Although previously described for the production of γ-PGA, ME has not been statistically optimized for cultivation conditions such as temperature, pH, and the influence of Zn2+ concentration. The Zn2+ cation was pointed out by Ashiuchi and co-workers [13] as an important factor in the enzymatic in vitro synthesis of γ-PGA, however, this cation is not present in the composition of the ME. Moreover, since B. subtilis BL53 is a new strain for bioprocess, it is important to assess its physiological characteristics to define optimal operating conditions. Therefore, the influence of temperature, initial pH of cultivation, and Zn2+ concentration on γ-PGA production by B. subtilis BL53 was investigated using a 23 central composite design (CCD). Table 3 shows the coded and actual values of 3 factors and the observed γ-PGA production for each run at 72 h of cultivation, time of the highest production. The effects estimation showed that the negative quadratic effect of initial pH was the most significant (p = 0.067) on γ-PGA production, followed by the positive linear effect (p = 0.115) and negative quadratic effect (p = 0.118) of Zn2+ concentration. The strong influence of pH was reported for some other Bacillus strains. Richards and Margaritis [45] reported the highest production of γ-PGA for pH between 6.5 and 7.5, for B. subtilis IFO 3335 strain, growing at 37 °C. Cromwick and co-workers [26] observed the strong influence of pH on the production of γ-PGA by B. licheniformis ATCC 9945A, correlating it to cell viability and to the preferential use of citrate via TCA cycle. According to these authors, the consumption of glycerol and glutamic acid remained approximately constant under any pH range, but the consumption of citrate was increased under pH 6.5. Wu and co-workers [46] showed that the glutamic acid uptake by cells was also dependent on the medium pH, and reported the best production of γPGA at pH 6.5–7.0, suggesting that the influence of pH might be due to the participation of H+ in the transport driving force of glutamate throughout membrane.
In order to determine the optimal values for the 3 studied variables, a second-order model was obtained from regression of CCD data. All coefficients were considered in the fitted model presented in Eq. (1), except for X 1 X 2 (Zn2+ concentration–initial pH interaction), and X 2 X 3 (temperature–initial pH interaction), which were the least significant effects. Equation 1 can now be rewritten as:
where Y is the response variable (γ-PGA production, g l−1), X 1 , X 2 and X 3 are the coded independent variables temperature, Zn2+ concentration and initial pH, respectively. The analysis of variance demonstrated that the model is highly significant, as evidenced by Fisher’s F test with very low p value (p = 0.0024), and presenting insignificant lack of fit (p = 0.84). The determination coefficient R 2 was 0.87, indicating that 87 % of variation was explained by the model. These results suggest that the proposed model satisfactorily represents the phenomenon under analysis. The fitted response for the regression model is plotted in Fig. 1, showing the contour plots for each two variables. The maximal predicted γ-PGA production was determined by the fitted model as 12.3 g l−1 when the temperature, initial pH, and Zn2+ concentration were 37 °C, 6.9, and 1.22 mM, respectively. The experimental validation of the model in shaker flasks produced 10.4 g l−1 of γ-PGA.
Ashiuchi et al. [12] demonstrated that the optimal pH for the in vitro synthesis of γ-PGA by PgsBCA complex is around 7.0, using enzyme-associated membrane from B. subtilis subspecies chungkookjang. Richards and Margaritis [29] showed that optimal pH for γ-PGA production by B. subtilis IFO 3335 was also 7.0. Finally, for B. licheniformis ATCC 9945, optimum pH was defined as 6.5 [26]. These studies performed using the traditional ‘one-factor-at-a-time’ technique without verifying the interaction among factors, which is only possible in factorial designs. Shih, Van, and Chang [24] used CCD to evaluate medium composition and initial medium pH. Nevertheless, the chosen range of pH was very narrow (6.0–7.0), resulting in the exclusion of pH from the design due to its low significance.
Although initial pH played an important role in the γ-PGA production by B. subtilis BL53 and other Bacillus strains, the experiments in this study indicated that strict control of pH was not necessary, since pH tends to buffer around 7.0–7.5 (data not shown), which are optimal values for γ-PGA production after 24 h of cultivation. Therefore, B. subtilis BL53 cultures contrasted with that of other strains, as reported for B. licheniformis CCRC 12826 [23], where the initial pH of 6.5 dropped to 5.6 after 36 h of cultivation at 37 °C. Cromwick and co-workers [26] reported that in the cultivation of B. licheniformis ATCC 9945, the pH was acidified from 7.4 to 5.6 between 48 and 96 h, at 37 °C.
As shown on the analysis of the effects (Table 3), Zn2+ concentration was a relevant factor in the production of γ-PGA by B. subtilis BL53. Ashiuchi et al. [13] observed that the Zn2+ was able to boost the action of Mg2+ on activation of complex PgsBCA responsible for γ-PGA enzymatic synthesis. Soliman and co-workers [17] using Plackett–Burman analysis to investigate γ-PGA production by a new strain of B. licheniformis, also identified Zn2+ concentration to have a positive effect on γ-PGA production, but it was negative for biomass formation. Negative effects Zn2+ concentration on biomass was not evidenced for B. subtilis BL 53 (data not shown).
Kinetics of B. subtilis BL53 cultivation
In order to evaluate the kinetics of B. subtilis BL53 cultivation and the effects of small scaling-up of the process, cultivations were performed in Erlenmeyer flasks (125 ml) and 5 l stirred tank bioreactors, with results shown in Fig. 2. For the bioreactor, aeration rate of 2 vvm and stirred agitation of 500 rpm were used to improve aeration of the system and prevent the oxygen depletion, while in the rotatory shaker, 180 rpm was adopted. The number of viable cells rapidly increased after 6 h in bioreactors. At this time, γ-PGA began to be excreted to the medium broth. Kinetics of both shaker and bioreactor suggests that γ-PGA produced by B. subtilis BL53 was partially associated with cell growth, similar to what is reported for other Bacillus strains [18, 47, 48].
γ-PGA production by Bacillus subtilis BL53 under optimized CCD conditions in rotatory shaker at 180 rpm (wingdings), and in bioreactor at 500 rpm and 2 vvm (filled circle). Viable cells in rotatory shaker (square), and in bioreactor (filled square). Results are the mean of triplicates for shaker experiments and duplicates for bioreactor experiments
Shaker cultivations produced 10.4 g l−1 of γ-PGA in 72 h, while in the bioreactor a 70 % higher production (17 g l−1) was obtained, strongly suggesting that aerobic metabolism is essential for this biopolymer production. Since γ-PGA is an extracellular polymer, the culture broth becomes viscous as cultivation time progresses, which may lead to oxygen limitation.
Effects of dissolved oxygen concentration, stirring rate, OUR, Q O2, and k La on cultivation for γ-PGA production
Dissolved oxygen concentration of medium was monitored during cultivation of B. subtilis BL53 for production of γ-PGA and results are shown in Fig. 3. The DOC declined sharply before 12 h of cultivation indicating high oxygen demand by cells during the exponential phase, exceeding oxygen transfer capacity in most conditions, except for 1,000 rpm, when DOC was around 10 %. The results indicate the difficulty in cultivations with high γ-PGA concentrations, which directly affects oxygen mass transfer capacity of the system. Data in Table 4 show that there was an increase in oxygen demand (OUR) from 3 to 6 h in cultures performed at 750 and 1,000 rpm, with a subsequent reduction at 8 h. Similar results were demonstrated by Richards and Margaritis [29] in the first hours of cultivation of B. subtilis IFO 3335 in a bioreactor operating at 2 vvm and 700 rpm, however, the maximal OUR was 20 mmol O2 l−1 h−1 and occurred later, after 10 h of cultivation. Normally, OUR of bacteria increases during the exponential growth phase because, at this stage, there is a high substrate consumption, decreasing thereafter due to reduced metabolic activity of cells as it approaches stationary phase [50]. The highest value of Q O2 for B. subtilis BL53 occurred at the beginning of exponential phase around 11–12 mmol O2 g−1 h−1 for all conditions (Table 4). These values are within the range of Q O2 values for other biopolymers produced by microorganisms as reviewed by Garcia-Ochoa et al. [50], which are in the range between 0.2 and 31.2 mmol O2 g−1 h−1. In the production of xanthan gum by Xanthomonas campestris NRRL 1775, for example, Q O2 values ranged from 2 to 15 mmol O2 g−1 h−1, and the maximal value was observed after 5 h of cultivation [51]. Richard and Margaritis [29] also showed a peak of Q O2 at the beginning of cultivation of B. subtilis IFO 3335 during production of γ-PGA. Considering the cultivation time of 3 h, which corresponded to early-exponential growth phase, the increase in stirring rate from 500 to 1,000 rpm caused an increase of approximately four times on the value of k La (Table 4). Bandaiphet and Prasertsan [52] observed the same effect in the production of an exopolysaccharide by Enterobacter cloacae, when the increase from 500 to 1,200 rpm caused an increase of approximately nine times in the value of k La, while the increase in the aeration rate was far less effective.
Figure 4 shows the growth kinetics for B. subtilis BL53 at 37 °C in the bioreactor operating using aeration rate of 2 vvm at different stirring rates. For the three conditions, a concentration of viable cells around 109 CFU ml−1 was reached after 36 h, with no loss of cell viability until the end of cultivation (96 h), even at high stirring rates. Similar results were reported by Cromwick and co-workers [26] for B. licheniformis ATCC 9945A cultures under milder aeration conditions (0.5 vvm and 250 rpm). In many cases, the oxygen transfer controls microbial growth and may affect the biological process [50]. As data in Table 5 show, the increase in the stirring rate produced a small increase in the specific growth rate of the logarithmic phase (μ max), but cell viability was not affected, probably due to the shear protective effect of γ-PGA, also reported for mammalian cells [53]. The correlation between oxygen availability and microbial growth in the exponential phase can be perceived from data on Fig. 3 and the kinetic parameters in Table 5. As more abrupt was the decline in DOC in the first hours of cultivation, the lower was the specific growth rate, extending the exponential phase.
Viable cells during γ-PGA production by Bacillus subtilis BL53, cultivation conducted at 2 vvm and stirring rates of 500 rpm (filled circle), 750 rpm (square), and 1,000 rpm (filled triangle). Results are the mean of duplicates and curves are obtained by fit to the Logistic Model in Eq. (2)
Figure 5 depicts the production of γ-PGA as a function of stirring rate. Results showed a positive effect of increased stirring rates on the productivity of the biopolymer, but did not affect its final concentration, which was kept around 17 g l−1 until 96 h. Cromwick and co-workers [26] were the first to investigate the influence of different conditions of aeration/agitation on the production of γ-PGA. These authors tested two different conditions (0.5 vvm/250 rpm and 1 vvm/800 rpm) for the cultivation of B. licheniformis ATCC 9945A and showed that the production of the polymer was twice as high at the higher aeration. Bajaj and Singhal [49] evaluated the effects of aeration and agitation on cultivations of B. licheniformis NCIM 2324, reporting higher concentrations of γ-PGA with increased agitation and, to a lesser extent, by increased aeration. However, k La was not calculated in those studies, so it is not possible to make further considerations regarding the transfer of oxygen. Comparing the γ-PGA production by B. subtilis BL53 (Fig. 5) to their respective k La values for each stirring rate (Table 4), it can be observed that a higher k La during exponential phase leads to a higher γ-PGA production. The k La decreases along time cultivation, as a direct influence of γ-PGA concentration, an effect already observed for other biopolymers systems, such as xanthan gum [54].
Production of γ-PGA by Bacillus subtilis BL53 for cultivation conducted at 2 vvm and stirring rates of 500 rpm (filled circle), 750 rpm (square), and 1,000 rpm (filled triangle). Results are the mean of duplicates and curves are obtained by fit to the Logistic Model in Eq. (2)
The maximal productivity of γ-PGA, which was obtained at 1,000 rpm (kLa = 170 h−1, middle exponential phase), was about 0.8 g l−1 h−1 (Fig. 6), exceeding the highest productivity obtained by Cromwick and co-workers [26] for B. licheniformis ATCC 9945A, and Bajaj and Singhal [49] for B. licheniformis NCIM 2324; it was similar to that reported by Richards and Margaritis [29] for B. subtilis IFO 3335. However, the highest productivity of γ-PGA from B. subtilis BL53 occurred before 24 h of cultivation, earlier than reported by the other authors. Using the strategy of increasing oxygen transfer through stirring rate, it was possible to reduce the time of process to <48 h at kLa of 210 h−1, about 50 % of the time required to produce de same amount of γ-PGA operating at kLa of 55 h−1. This result is especially relevant for future industrial applications because the enhancement of productivity contributes to reducing process costs.
Productivity γ-PGA by Bacillus subtilis BL53 for cultures conducted at 2 vvm and stirring rates of 500 rpm (filled circle), 750 rpm (square), and 1,000 rpm (filled triangle). Results are the mean of duplicates and curves are obtained by fit to the Logistic Model in Eq. (2)
The difficulty in producing γ-PGA along the time course of cultivation is related to the increase in the medium viscosity that negatively affects oxygen transfer. The sharp decline in the γ-PGA concentration shown in Fig. 6 for 1,000 rpm may be due the high shear rate, which could breakdown the biopolymer molecule. This effect has already been reported for the production of other biopolymers such as xanthan gum [55], gellan gum [56], and an exopolysaccharide produced by E. cloacae [52]. However, the influence of depolymerase enzymes, in the specific case of γ-PGA, cannot be dismissed, since their activities are induced under conditions of carbon source limitations or increased concentration of γ-PGA [26].
Conclusions
Exploring biodiversity is a key step in the development of bioprocess for new or alternative sources of industrially important products. In this study, five strains isolated from Amazonian environment showed the ability to produce γ-PGA when cultured in medium E. The best producer strain was characterized and identified as B subtilis. γ-PGA production could be optimized concerning temperature, pH, and Zn2+, which is an important inducer of the biopolymer. The scaling-up of γ-PGA production in bioreactors, where agitation strategies were evaluated and the best kLa determined, increased its production in more than 70 % compared to shaker cultivations. The production of γ-PGA by B. subtilis BL53 showed to be highly dependent on oxygen transfer, and productivity could be increased, while cultivation time could be reduced to half the time compared to conditions where k La was not optimal, thereby contributing to reducing process costs.
References
Shih IL, Van YT (2001) The production of poly-(gamma-glutamic acid) from microorganisms and its various applications. Bioresour Technol 79:207–225
Candela T, Fouet A (2006) Poly-gamma-glutamate in bacteria. Mol Microbiol 60:1091–1098
Buescher JM, Margaritis A (2007) Microbial biosynthesis of polyglutamic acid biopolymer and applications in the biopharmaceutical, biomedical and food industries. Crit Rev Biotechnol 27:1–19
Manocha B, Margaritis A (2008) Production and characterization of gamma-polyglutamic acid nanoparticles for controlled anticancer drug release. Crit Rev Biotechnol 28:83–99
Sung MH, Park C, Kim CJ, Poo H, Soda K, Ashiuchi M (2005) Natural and edible biopolymer poly-gamma-glutamic acid: synthesis, production, and applications. Chem Rec 5:352–366
Taniguchi M, Kato K, Shimauchi A, Xu P, Nakayama H, Fujita KI, Tanaka T, Tarui Y, Hirasawa E (2005) Proposals for wastewater treatment by applying flocculating activity of cross-linked poly-gamma-glutamic acid. J Biosci Bioeng 99:245–251
Carvajal-Zarrabal O, Nolasco-Hipólito C, Barradas-Dermitz DM, Hayward-Jones PM, Aguilar-Uscanga MG, Bujang K (2012) Treatment of vinasse from tequila production using polyglutamic acid. J Environ Manag 95:S66–S70
Bajaj I, Singhal R (2011) Poly (glutamic acid)–an emerging biopolymer of commercial interest. Bioresour Technol 102:5551–5561
Schallmey M, Singh A, Ward OP (2004) Developments in the use of Bacillus species for industrial production. Can J Microbiol 50:1–17
Oppermann-Sanio FB, Steinbuchel A (2002) Occurrence, functions and biosynthesis of polyamides in microorganisms and biotechnological production. Naturwissenschaften 89:11–22
Ashiuchi M, Misono H (2002) Biochemistry and molecular genetics of poly-gamma-glutamate synthesis. Appl Microbiol Biotechnol 59:9–14
Ashiuchi M, Kamei T, Baek DH, Shin SY, Sung MH, Soda K, Yagi T, Misono H (2001) Isolation of Bacillus subtilis (chungkookjang), a poly-gamma-glutamate producer with high genetic competence. Appl Microbiol Biotechnol 57:764–769
Ashiuchi M, Shimanouchi K, Nakamura H, Kamei T, Soda K, Park C, Sung M-H, Misono H (2004) Enzymatic synthesis of high-molecular-mass poly-{gamma}-glutamate and regulation of its stereochemistry. Appl Environ Microbiol 70:4249–4255
Kunioka M (2004) Biodegradable Water Absorbent Synthesized from Bacterial Poly(amino acid)s. Macromol Biosci 4:324–329
Hoppensack A, Oppermann-Sanio FB, Steinbüchel A (2003) Conversion of the nitrogen content in liquid manure into biomass and polyglutamic acid by a newly isolated strain of Bacillus licheniformis. FEMS Microbiol Lett 218:39–45
Mahmoud DAR (2006) Isolation of polyglutamic acid flocculant producing bacteria from extreme Egyptian environments. J Appl Sci Res 2:608–612
Soliman N, Berekaa M, Abdel-Fattah Y (2005) Polyglutamic acid (PGA) production by Bacillus sp. SAB-26: application of Plackett–Burman experimental design to evaluate culture requirements. Appl Microbiol Biotechnol 69:259–267
Xu H, Jiang M, Li H, Lu D, Ouyang P (2005) Efficient production of poly([gamma]-glutamic acid) by newly isolated Bacillus subtilis NX-2. Process Biochem 40:519–523
Cao M, Geng W, Liu L, Song C, Xie H, Guo W, Jin Y, Wang S (2011) Glutamic acid independent production of poly-γ-glutamic acid by Bacillus amyloliquefaciens LL3 and cloning of pgsBCA genes. Bioresour Technol 102:4251–4257
Zhang H, Zhu J, Zhu X, Cai J, Zhang A, Hong Y, Huang J, Huang L, Xu Z (2012) High-level exogenous glutamic acid-independent production of poly-(γ-glutamic acid) with organic acid addition in a new isolated Bacillus subtilis C10. Bioresour Technol
Leonard CG, Housewright RD, Thorne CB (1958) Effects of some metallic ions on glutamyl polypeptide synthesis by Bacillus subtilis. J Bacteriol 76:499–503
Do JH, Chang HN, Lee SY (2001) Efficient recovery of gamma-poly (glutamic acid) from highly viscous culture broth. Biotechnol Bioeng 76:219–223
Shih IL, Van YT, Yeh LC, Lin HG, Chang YN (2001) Production of a biopolymer flocculant from Bacillus licheniformis and its flocculation properties. Bioresour Technol 78:267–272
Shih IL, Van YT, Chang YN (2002) Application of statistical experimental methods to optimize production of poly(gamma-glutamic acid) by Bacillus licheniformis CCRC 12826. Enzyme Microb Technol 31:213–220
Shih I-L, Yu Y-T (2005) Simultaneous and selective production of levan and poly(γ-glutamic acid) by Bacillus subtilis. Biotechnol Lett 27:103–106
Cromwick AM, Birrer GA, Gross RA (1996) Effects of pH and aeration on gamma-poly(glutamic acid) formation by Bacillus licheniformis in controlled batch fermenter cultures. Biotechnol Bioeng 50:222–227
Du G, Yang G, Qu Y, Chen J, Lun S (2005) Effects of glycerol on the production of poly([gamma]-glutamic acid) by Bacillus licheniformis. Process Biochem 40:2143–2147
Yoon SH, Hwan Do J, Yup Lee S, Nam Chang H (2000) Production of poly-γ-glutamic acid by fed-batch culture of Bacillus licheniformis. Biotechnol Lett 22:585–588
Richard A, Margaritis A (2003) Rheology, oxygen transfer, and molecular weight characteristics of poly(glutamic acid) fermentation by Bacillus subtilis. Biotechnol Bioeng 82:299–305
Bajaj IB, Singhal RS (2009) Enhanced production of poly (gamma-glutamic acid) from Bacillus licheniformis NCIM 2324 by using metabolic precursors. Appl Biochem Biotechnol 159:133–141
Kirschner P, Meier A, Böttger EC (1993) Genotypic identification and detection of mycobacteria: facing novel and uncultured pathogens. In: Persing DH, Smith TF, Tenover FC, White TJ (eds) Diagnostic molecular microbiology: principles and applications. American Society for Microbiology, Washington (DC)
Sneath PHA (1986) Endospore-forming gram-positive rods and cocci. In: Sneath PHA, Mair NS, Sharpe ME, Holt JG (eds) Bergey’s Manual of Systematic Bacteriology. Williams & Wilkins, Baltimore
MacFadin JF (ed) (1980) Biochemical test for identification of medical bacteria, 2nd edn. Williams & Wilkins, Baltimore
Zwietering MH, Jongenburger I, Rombouts FM, Vantriet K (1990) Modeling of the Bacterial-Growth Curve. Appl Environ Microbiol 56:1875–1881
Doran PM (1995) Bioprocess engineering principles. Academic Press, London
Kanno A, Takamatsu H (1995) Determination of polyglutamic acid in “Natto” using cetyltrimethylammonium bromide (Studies on “Natto” part V). Nippon Shokuhin Kagaku Kogaku Kaishi 42:878–886
Ashiuchi M (2011) Analytical approaches to poly-γ-glutamate: quantification, molecular size determination, and stereochemistry investigation. J Chromatogr B 879:3096–3101
Porwal S, Lal S, Cheema S, Kalia VC (2009) Phylogeny in aid of the present and novel microbial lineages: diversity in Bacillus. PLoS One 4:e4438
Xu D, Cote JC (2003) Phylogenetic relationships between Bacillus species and related genera inferred from comparison of 3′ end 16S rDNA and 5′ end 16S-23S ITS nucleotide sequences. Int J Syst Evol Microbiol 53:695–704
Sumpavapol P, Tongyonk L, Tanasupawat S, Chokesajjawatee N, Luxanani P, Visessanguan W (2010) Bacillus siamensis sp. nov., isolated from salted crab (poo-khem) in Thailand. Int J Syst Evol Microbiol 60:2364–2370
Priest FG, Goodfellow M, Todd C (1988) A Numerical Classification of the Genus Bacillus. J Gen Microbiol 134:1847–1882
Reva ON, Sorokulova IB, Smirnov VV (2001) Simplified technique for identification of the aerobic spore-forming bacteria by phenotype. Int J Syst Evol Microbiol 51:1361–1371
Welker NE, Campbell LL (1967) Unrelatedness of Bacillus amyloliquefaciens and Bacillus subtilis. J Bacteriol 94:1124–1130
Kim KM, Kim MJ, Kim DH, Park YS, Kang JS (2009) Characterization of Bacillus polyfermenticus KJS-2 as a Probiotic. J Microbiol Biotechnol 19:1013–1018
Richard A, Margaritis A (2003) Optimization of cell growth and poly(glutamic acid) production in batch fermentation by Bacillus subtilis. Biotechnol Lett 25:465–468
Wu Q, Xu H, Ying HJ, Ouyang PK (2010) Kinetic analysis and pH-shift control strategy for poly(gamma-glutamic acid) production with Bacillus subtilis CGMCC 0833. Biochem Eng J 50:24–28
Chen X, Chen S, Sun M, Yu Z (2005) High yield of poly-[gamma]-glutamic acid from Bacillus subtilis by solid-state fermentation using swine manure as the basis of a solid substrate. Bioresour Technol 96:1872–1879
Chen X, Chen S, Sun M, Yu Z (2005) Medium optimization by response surface methodology for poly-γ-glutamic acid production using dairy manure as the basis of a solid substrate. Appl Microbiol Biotechnol 69:390–396
Bajaj IB, Singhal RS (2010) Effect of aeration and agitation on synthesis of poly (gamma-glutamic acid) in batch cultures of Bacillus licheniformis NCIM 2324. Biotechnol Bioprocess Eng 15:635–640
García-Ochoa F, Gomez E, Santos VE, Merchuk JC (2010) Oxygen uptake rate in microbial processes: an overview. Biochem Eng J 49:289–307
García-Ochoa F, Castro EG, Santos VE (2000) Oxygen transfer and uptake rates during xanthan gum production. Enzyme Microb Technol 27:680–690
Bandaiphet C, Prasertsan P (2006) Effect of aeration and agitation rates and scale-up on oxygen transfer coefficient, kLa in exopolysaccharide production from Enterobacter cloacae WD7. Carbohydr Polym 66:216–228
Chun BH, Lee YK, Chung N (2012) Poly-γ-glutamic acid enhances the growth and viability of Chinese hamster ovary cells in serum-free medium. Biotechnol Lett pp 1–4
García-Ochoa F, Santos VE, Casas JA, Gomez E (2000) Xanthan gum: production, recovery, and properties. Biotechnol Adv 18:549–579
Casas JA, Santos VE, Garcia-Ochoa F (2000) Xanthan gum production under several operational conditions: molecular structure and rheological properties. Enzyme Microb Technol 26:282–291
Giavasis I, Harvey LM, McNeil B (2006) The effect of agitation and aeration on the synthesis and molecular weight of gellan in batch cultures of Sphingomonas paucimobilis. Enzyme Microb Technol 38:101–108
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
da Silva, S.B., Cantarelli, V.V. & Ayub, M.A.Z. Production and optimization of poly-γ-glutamic acid by Bacillus subtilis BL53 isolated from the Amazonian environment. Bioprocess Biosyst Eng 37, 469–479 (2014). https://doi.org/10.1007/s00449-013-1016-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-013-1016-1