Abstract
Both atmospheric [CO2] and average surface temperatures are predicted to increase with potentially different, additive or opposing, effects on leaf quality and insect herbivore activity. Few studies have directly measured the interactive effects of concurrent changes in [CO2] and temperature on insect herbivores. None have done so over the entire developmental period of a tree-feeding insect, and none have compared responses to low pre-industrial [CO2] and present day [CO2] to estimate responses to future increases. Eucalypt herbivores may be particularly sensitive to climate-driven shifts in plant chemistry, as eucalypt foliage is naturally low in [N]. In this study, we assessed the development of the eucalypt herbivore Doratifera quadriguttata exposed concurrently to variable [CO2] (290, 400, 650 μmol mol−1) and temperature (ambient, ambient +4 °C) on glasshouse-grown Eucalyptus tereticornis. Overall, insects performed best on foliage grown at pre-industrial [CO2], indicating that modern insect herbivores have already experienced nutritional shifts since industrialisation. Rising [CO2] increased specific leaf mass and leaf carbohydrate concentration, subsequently reducing leaf [N]. Lower leaf [N] induced compensatory feeding and impeded insect performance, particularly by prolonging larval development. Importantly, elevated temperature dampened the negative effects of rising [CO2] on larval performance. Therefore, rising [CO2] over the past 200 years may have reduced forage quality for eucalypt insects, but concurrent temperature increases may have partially compensated for this, and may continue to do so in the future. These results highlight the importance of assessing plant–insect interactions within the context of multiple climate-change factors because of the interactive and potentially opposing effects of different factors within and between trophic levels.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
As a result of radiative forcing associated with anthropogenic greenhouse gas emissions, future rates of global climate change will be significantly greater than at any time in the recent geological past (Overpeck et al. 1991). By 2100, global atmospheric carbon dioxide concentrations [CO2] will be double pre-industrial levels, and mean surface temperatures will rise by up to 4.5 °C (Solomon et al. 2007). The ecological consequences of such multifactorial changes will depend on complex multitrophic interactions between affected species, yet impacts on organisms other than plants have so far rarely been studied. In particular, insect herbivores, which play key roles in nutrient cycling and energy transfer from plants to higher organisms, will experience direct physiological effects of climate change (Fajer et al. 1989; Massad and Dyer 2010) but also indirect ecological effects. Specifically, the additive or opposing effects of concurrent increases in [CO2] and temperature on the plants that herbivores consume will fundamentally alter their distribution, quality and quantity as a food resource. Plant growth in elevated [CO2] (reviews by Ainsworth and Long 2005; Ainsworth and Rogers 2007) and elevated temperature (review by Way and Oren 2010) has been studied extensively, although rarely in combination. Elevated [CO2] increases foliar biomass and specific leaf mass (SLM) due to the accumulation of leaf carbohydrates, which dilute [N] and increase the C:N ratio (Coley et al. 2002; Ainsworth and Rogers 2007). Warming generally accelerates plant development, increasing plant and leaf biomass, and may affect SLM and leaf [N] (Way and Oren 2010), but to a lesser degree than elevated [CO2] (Ghannoum et al. 2010b).
Although studies have reported interactive effects of concurrent increases in [CO2] and temperature on foliar traits of importance to herbivores (see Massad and Dyer 2010 and Robinson et al. 2012), a recent meta-analysis (Zvereva and Kozlov 2006) cites just seven that directly measured insect responses to these concurrent changes (Williams et al. 2000, 2003; Johns and Hughes 2002; Veteli et al. 2002; Johns et al. 2003; Chong et al. 2004; Flynn et al. 2006). Only three involved insect species feeding on woody plants (all deciduous, Northern-Hemisphere temperate forest trees) (Williams et al. 2000, 2003; Veteli et al. 2002), and only Williams et al. (2003) measured responses over multiple larval instars (excluding the first instar), despite the potential for cumulative effects over an insect’s development. The limited empirical data for herbivores exposed to concurrent increase in temperature and [CO2], especially over their entire development, greatly restricts any reliable prediction of ecologically meaningful responses to future climate change.
Comparing species responses to past vs. present-day climate conditions can inform predictions of responses to future environmental change. Vascular plants, and their associated herbivores, evolved during a time of significant variation in atmospheric [CO2] (170–3,600 μmol mol−1) and temperature (Gerhart and Ward 2010). Radiocarbon-dated paleoclimate and pollen records have been used to reconstruct past vegetation patterns and build models of future change (e.g. Prentice et al. 2011), but similar geological abundance and distribution records for insects are relatively scarce (but see Axford et al. 2011; Wappler and Denk 2011). Data on insect responses to recent warming have shown latitudinal and altitudinal range shifts (Hill et al. 1999; Parmesan et al. 1999; Beaumont and Hughes 2002), and increased asynchrony between insect and host-plant phenology (Dewar and Watt 1992; Hill and Hodkinson 1995; Visser and Holleman 2001). The few studies of modern plants grown in pre-industrial [CO2] have shown reduced foliar biomass and photosynthetic rates, lower SLM, leaf carbohydrates and secondary metabolites, and higher leaf [N] than ambient-grown plants (Sage and Coleman 2001; Gerhart and Ward 2010; Ghannoum et al. 2010a; Tissue and Lewis 2010). This implies a reduction in the nutritional quality of plants for insects since the Industrial Revolution, but this has not been tested. Studies have, however, shown that insect herbivores generally respond negatively to reduced foliar quality generated by increasing [CO2] from present to future predicted levels (e.g. Lincoln et al. 1986; Fajer et al. 1989; Lindroth et al. 1993; Lawler et al. 1997; Stiling and Cornelissen 2007). Assessing insect responses to the full range of [CO2] experienced in the past 200 years and projected to occur within this century will improve our understanding of the trajectory of responses to rising [CO2] (Tissue and Lewis 2012). Interpreting these in light of the potential effects of concurrent warming will provide a more realistic insight into how insect herbivores are likely to cope with future environments.
Eucalyptus L’Herit (Myrtaceae) species dominate Australia’s 149 million ha of forests and woodlands spanning 35° of latitude (MPIGA 2008), and their ecological and global economic importance make them particularly relevant host plants with which to study multitrophic impacts of climate change. In general, eucalypt leaves are sclerotic and low in nitrogen relative to most angiosperms (Springett 1978; White 1993), and contain high concentrations of secondary metabolites that also directly reduce their nutritive value or act as toxins (Ohmart and Edwards 1991; Henery et al. 2008). Growth of eucalypts in both elevated [CO2] and temperature generally reduces leaf [N] (Ayub et al. 2011), thereby further reducing nutritive quality for feeding insects. Thus, projected increases in [CO2] and temperature could significantly reduce the nutritional resources available to eucalypt herbivores and may stimulate particularly high levels of compensatory feeding. In the absence of increased plant biomass this could reduce tree productivity. Alternatively, these insects may be so well adapted to assimilating limited nutrients from their “suboptimal” food source (Fox and Macauley 1977) that compensatory feeding may be minimal, with negligible impact on tree productivity.
In this study we assess developmental responses of the four-spotted cup moth, Doratifera quadriguttata (Walker) (Lepidoptera: Limacodidae), to combined changes in [CO2] and temperature. Doratifera spp. are widespread in Australia and exhibit occasional localised outbreaks (Froggatt 1923; Common 1990; Young 2011). This is the first study to evaluate insect responses to the interactive effects of rising [CO2] and temperature that includes a pre-industrial [CO2] treatment and second study that investigates interactive responses of a tree-feeding insect herbivore from egg hatch to pupation (also see Williams et al. 2003). One previous study (Lawler et al. 1997) measured the responses of a eucalypt-feeding insect to rising [CO2], but did not include a temperature treatment and, being restricted to the last larval instar, was limited in its capacity to predict ecological impacts. Our objectives were to assess the interactive effects of rising [CO2] and temperature on key leaf- and insect-performance traits throughout larval development, in order to reveal potential impacts of climate change on future plant–insect interactions.
Methods
Insect and plant growth conditions
Adult D. quadriguttata were collected in January 2010 from a Eucalyptus saligna Sm. plantation (Richmond, NSW, 33°36′4″0S, 150°44′26.5″E) within the Hawkesbury Forest Experiment (HFE) (see Barton et al. 2010). This site is adjacent to a natural woodland remnant where D. quadriguttata is frequently observed feeding on the dominant E. tereticornis Sm., and is the likely origin of the plantation moth population. Moths were held in 30 cm3 ventilated plastic oviposition cages (25 °C, ambient R. H., natural light) with E. saligna foliage. Larvae were reared in 70 L ventilated plastic bins (22 ± 2 °C, 55 % R. H., 12L/12D) and fed excised E. saligna foliage until pupation. Eggs from the second generation were used in experiments.
Plant material and insects were maintained in three atmospheric [CO2] (290, 400, 650 μmol mol−1, hereafter C low, C ambient, and C elevated, respectively) and two temperature (ambient and ambient +4 °C, T ambient and T elevated, respectively) treatments in a full-factorial design in six sun-lit glasshouse chambers (see Ghannoum et al. 2010a for full description). T ambient (mean 26/18 °C day/night) was based on the 30-year growing season (November to May) daily average temperature in Richmond (Australian Bureau of Meteorology, station 067105) and T elevated was maintained at 4 °C above T ambient (mean 30/22 °C day/night). Eucalyptus tereticornis, grown from seed (open pollination, Cecil Hills, Western Sydney Parklands), were transplanted into 10 L pots of native, loamy-sand soil from the A-horizon of the HFE site (see Barton et al. 2010 for description of soil characteristics). Four seedlings were randomly allocated to each experimental treatment in order to reduce potential effects on insect larvae (see below) due to genetic diversity between seedlings. Trees were watered daily and supplemented monthly with liquid fertiliser (Aquasol®, N:P:K 23:4:18 %).
Insect responses to [CO2] and temperature
At hatching, D. quadriguttata larvae from individual egg batches were divided between all six treatments to control for maternal effects. Larvae were initially placed in groups of 20 on excised leaves (petioles secured with Blu-Tack® into 1.5 ml water-filled microcentrifuge tubes) from each treatment, as early instars suffered significant mortality if fed in isolation. Each group was maintained in a 14-cm-diameter Petri dish in the same chamber as their leaves were grown. Water and leaves were replenished every 1–3 days. Upon moulting to the third instar, individuals were transferred onto newly expanded leaves on trees within the same chamber in which the larvae had initially been reared, such that there were 30 larval replicates per treatment. Each larva was confined to a separate leaf in a gauze bag plugged with cotton. Care was taken to maintain similar herbivore loads on all four trees per treatment. Throughout their development, individual larvae were regularly moved between the four trees within their assigned treatment to ensure constant food supply and to reduce any effects of differences in the inherent foliar chemistry (i.e. non-treatment effects) of individual trees. Development time (egg hatch to pupation), pupal fresh weight, and frass dry weight (oven-dried, 48 h, 70 °C) were recorded for each larva. Frass dry weight was previously identified as an accurate estimate of relative consumption for this species under glasshouse conditions (Murray, unpublished data). Sex was determined from adult antennal morphology at eclosion.
Foliar traits
Five fully expanded leaves per treatment were randomly collected (including one from each of the four trees) on day 17 of the experiment when most insects had moulted to the third instar. Fresh weight, surface area, and dry weight were recorded to determine specific leaf mass (SLM), fresh mass/area (FMA) and dry matter content (DMC). Leaves were pooled by treatment, ground in a ball mill, and analysed to determine [N] and C:N ratio using a CHN analyser (LECO TruSpec Micro, LECO Corporation, St. Joseph, MI, USA). Foliar total non-structural carbohydrates (TNC) were determined by quantifying soluble sugars using a modified anthrone method (Ebell 1969) and starch was enzymatically assayed on the residual pellet using a total starch kit (Megazyme International Ireland Ltd.). As leaves were inadvertently pooled during grinding, [N] and TNC were analysed based on samples that were not properly replicated. To partially compensate for this, a second set of leaves that was collected at the end of the experiment following the same regime was also analysed, and values presented for [N] and TNC are the average of these two collections.
Statistical analysis
SLM, FMA and DMC treatment means were compared using ANOVA and Tukeys post-hoc test (R, 2.13.2, R Development Core Team 2009). The number of larvae of each sex per treatment could not be controlled because sex could only be determined at the adult stage. To ensure that any between-treatment differences in development time and frass production were independent of sex, a linear regression was conducted between each response variable and pupal weight, which was significantly correlated to sex (see “Results”). ANCOVA with pupal weight as the co-variate was used to examine [CO2] and temperature effects on the slope and intercept of each regression.
Results
Insect responses to [CO2] and temperature
The sex of D. quadriguttata was correlated with pupal weight (females heavier, F = 94.067, df = 1, P < 0.001, r 2 = 0.503), frass production (F = 35.654, df = 1, P < 0.001, r 2 = 0.592) and development time (F = 4.987, df = 1, P = 0.028, r 2 = 0.734). Frass production was also correlated with pupal weight (F = 95.689, df = 1, P < 0.001, r 2 = 0.739) and development time (F = 152.273, df = 1, P < 0.001, r 2 = 0.647). Neither [CO2] (F = 1.448, df = 2, P = 0.24) nor temperature (F = 0.601, df = 1, P = 0.44) significantly affected pupal weight (Table 1).
Development time and frass production increased with rising [CO2] (Fig. 1). When regressed against pupal weight, [CO2] had a significant effect on the intercept for both development time and frass production, but slope only varied for frass production (Fig. 2; Table 2). At T elevated, development time and frass production were 12–33 and 10–24 % lower than at T ambient, respectively (Fig. 1); both reductions were significant when regressed against pupal weight (Fig. 2; Table 2). There was a significant [CO2] × temperature interaction for both development and frass production (Table 2). At T elevated, larvae developed more slowly (19 %) and produced 29 % more frass when foliage was grown at C ambient compared to C low, but a further rise to C elevated had no additional effect (Fig. 1a, b). At T ambient, there was minimal increase in development time (6 %) and frass production (9 %) from C low to C ambient, but substantial increases (26 and 31 %, respectively) from C ambient to C elevated (Fig. 1a, b).
Mean (±SE) a development time and b frass production of D. quadriguttata larvae when fed directly on E. tereticornis grown at pre-industrial, present-day ambient and elevated [CO2] (290, 400, 650 μmol mol−1, respectively) and ambient (dashed line with open circles) or elevated (ambient +4 °C) (solid line with filled circles) temperature. n = 13–25 larvae per treatment
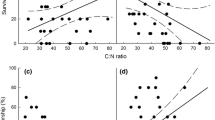
Linear regression showing the effects of [CO2] and temperature treatments on correlations between pupal weight and a, b development time and c, d frass production of D. quadriguttata larvae fed directly on E. tereticornis growing at pre-industrial (squares), present-day ambient (triangles) and elevated (circles) [CO2] (290, 400 and 650 μmol mol−1, respectively) and ambient (open symbols) or elevated (ambient +4 °C) (filled symbols) temperature. Significant differences between treatment slopes and intercepts are indicated in Table 2 under “ANCOVA”
Foliar traits
[CO2] had a significant effect on specific leaf mass (SLM), fresh mass/area (FMA) and dry matter content (DMC; Table 2). At T ambient, raising [CO2] from C low to C elevated resulted in an overall accumulation of foliar total non-structural carbohydrates (TNC) in leaves of 21 %, increases in SLM (59 %), FMA (28 %) and DMC (26 %), and a subsequent reduction in leaf [N] of 32 % (Figs. 3, 4). Similarly, at T elevated, TNC increased by 34 %, SLM by 56 %, FMA by 35 % and DMC by 17 % and leaf [N] was subsequently reduced by 42 %. The stepwise increases between [CO2] treatments were significant for SLM from C low to C ambient and for SLM, DMC and FMA from C ambient to C elevated (Fig. 3). Overall, T elevated significantly reduced SLM (up to 15 %), but FMA and DMC were unaffected (Fig. 3; Table 2). TNC did not differ between T ambient and T elevated (Fig. 4a). There was an interaction between temperature and [CO2], such that leaf [N] at T ambient was similar at C low and C ambient but decreased substantially from C ambient to C elevated (36 %); whereas, at T elevated, leaf [N] decreased to a greater degree from C low to C ambient (35 %) than from C ambient to C elevated (10.5 %, Fig. 4b).
Mean (±SE) a specific leaf mass (SLM), b fresh mass/area (FMA) and c dry matter content (DMC) of E. tereticornis leaves grown at pre-industrial, present-day ambient and elevated [CO2] (290, 400, 650 μmol mol−1, respectively) and ambient (dashed line with open circles) or elevated (ambient +4 °C) (solid line with filled circles) temperature. n = 5. Different letters indicate significant differences (P < 0.05) between [CO2] treatments
Total non-structural carbohydrates (TNC) and total [N] for E. tereticornis leaves grown at pre-industrial, present-day ambient, and elevated [CO2] (290, 400, 650 μmol mol−1, respectively) and ambient (dashed line with open circles) or elevated (ambient +4 °C) (solid line with filled circles) temperature. n = 2 pooled subsamples of five leaves per treatment
Insect performance as a function of leaf traits
Overall, as [CO2] was increased from C low to C elevated, reductions in leaf [N] (T ambient = 32 %, T elevated = 38 %) combined with increased SLM (T ambient = 42 %, T elevated = 56 %) slowed insect development (Fig. 5a, c, T ambient = 33 %, T elevated = 16 %) and increased frass production (Fig. 5b, d. T ambient = 42 %, T elevated = 52 %).
Relationships of a, b [N] and c, d specific leaf mass (SLM) to development time and frass production of D. quadriguttata larvae fed on E. tereticornis grown at pre-industrial, present-day and elevated [CO2] (290, 400, 650 μmol mol−1, respectively) and ambient (dashed line with open circles) or elevated (ambient +4 °C) (solid line with filled circles) temperature
Discussion
This study clearly shows that future climate change will influence eucalypt-feeding insects directly, through temperature-driven increases in their metabolism and development, and indirectly through [CO2]-driven reductions in the nutritional value of their food resource. We have demonstrated that the direct effects of elevated temperature interact with, and dampen, the negative nutritional effects of rising [CO2]. The detection of this interaction was largely enabled by assessing insect responses across their entire larval development, including the first instar. While some of these outcomes agree with the few factorial studies undertaken with insect herbivores of herbaceous plants (e.g. Johns and Hughes 2002; Johns et al. 2003), this study is first to demonstrate interactive effects on insect herbivores that feed on tree foliage. These findings are thus an important step forward in understanding the mechanisms and potential ecological impacts of climate change on insect herbivores of trees, and consequently on forest productivity and carbon balance (Pinkard et al. 2011).
In this study, foliar traits responded more strongly to rising [CO2] than to elevated temperature. As [CO2] was increased from 290–650 μmol mol−1, all foliar traits became less favourable for herbivores. Reduced leaf [N] and increased SLM subsequently increased herbivore consumption and developmental time, which has important ecological implications such as extended exposure to antagonists (Stiling et al. 1999), but the magnitude of this effect was highly dependent on temperature. Although total [N] does not equate precisely to [N] available for assimilation by herbivores (Wallis et al. 2010), the observed decline in leaf [N] at ambient temperature (32 %) strongly reflected the magnitude by which insect development was subsequently delayed (33 %). In contrast, at elevated temperature, there was a similar reduction in foliar quality from low to elevated [CO2], but the delay in larval development was only half as long. Thus, the negative effect of reduced foliar [N] on larval performance in rising [CO2] was outweighed by a positive effect of higher temperature on larval physiology. Future increases in temperature may therefore improve insect tolerance of reduced foliar quality in rising [CO2], while enhanced plant growth may allow plants to sustain and recover from increased defoliation resulting from compensatory feeding or insect outbreaks. In particularly warm regions such as Australia, however, rising temperatures might exceed the thermal optima of plants or insects, thereby limiting the positive effects of elevated [CO2] on plant growth and water use efficiency, or reducing geographical ranges suitable for the survival of particular insect species (e.g. Beaumont and Hughes 2002).
In the only previous study to assess responses of an eucalypt-feeding insect to [CO2] (but not temperature), Lawler et al. (1997) also reported significant reductions in leaf [N] and increases in SLM and TNC for E. tereticornis in elevated [CO2], but no subsequent significant decline in the performance of the leaf beetle Chrysophtharta flaveola (Chapuis). This was attributed to confounding effects of other experimental parameters (soil nutrient and light treatments) obscuring the effects of elevated [CO2] on foliar quality. It was also noted that [CO2] effects on insect performance may have been underestimated because only the last larval instar was included in the experiment. Late-instar larvae can compensate for reduced foliar quality to a greater degree than early instars by increasing foliar consumption (Fajer et al. 1989). Compensatory feeding is a common response to foliage grown at elevated [CO2] (e.g. Lindroth et al. 1993) and was clearly evident in the present study, with frass production increasing by 42 and 52 % at ambient and elevated temperature, respectively. By limiting [CO2] exposure to larvae of a single late developmental stage, effects of rising [CO2] on foliar parameters fundamental to the establishment of young larvae may also be missed. For example, increased SLM is associated with increased leaf toughness (Steinbauer 2001), which can inhibit the establishment and performance of early-instar eucalypt-feeding insects (Nahrung et al. 2001). Assessing responses over the entire larval development period and subsequently determining the sex of each individual also allowed us to confidently exclude the possibility that between-treatment differences in foliar consumption and insect development time resulted from unequal representation of each sex. Furthermore, because female pupal weight was greater than that of males, and not influenced by [CO2] or temperature, it was clear that treatments altered the time and resources required by larvae to complete development, rather than directly affecting pupal weight, which is a commonly used proxy for reproductive fitness (but see Leather 1988). Overall, our study demonstrated the importance of assessing insect response over their entire development, including the first instar. The magnitude and ecological consequences of these responses to the interactive effects of CO2 and temperature may not have been detected if only a fraction of the larval period had been assessed.
Assessing the effects of both low and elevated [CO2] compared to ambient [CO2] allowed us to simulate insect responses to past changes of a similar magnitude to those predicted for the future. This may help predict future plant–insect interactions by more accurately estimating the overall trajectory of responses to climate change. Previous studies utilising this approach have focused on fast-growing, short-lived plants (e.g. Dippery et al. 1995), whereas tree studies are limited (e.g. Ghannoum et al. 2010a; Lewis et al. 2010; Tissue and Lewis 2010; Ayub et al. 2011), and no insect studies have been conducted. In single factor low [CO2] studies, a proportionally larger plant response is often observed in the transition from low to modern [CO2] compared to modern to future [CO2] (Gerhart and Ward 2010). Our results for both foliar nutritional quality and insect performance were consistent with this at T elevated, but not T ambient. Ghannoum et al. (2010a, b) previously observed that at T elevated leaf [N] of E. saligna and E. sideroxylon decreased as [CO2] was increased from 290 to 650 μmol mol−1. However, at T ambient, leaf [N] decreased between 400 and 650 μmol mol−1 in both species, but did not decrease between 290 and 400 μmol mol−1 in E. sideroxylon. These results (see also Lewis et al. 2010; Ayub et al. 2011) highlight the nonlinear and species-specific nature of plant responses to rising [CO2], as the primary limitation on plant performance shifts from carbon to other resources (Sage and Coleman 2001; Gerhart and Ward 2010; Tissue and Lewis 2012). Interactions between [CO2] and other environmental variables, especially temperature and precipitation, may be extremely important in N-limited environments if, for example, they result in foliar [N] declining below a threshold critical for insect survival at lower [CO2] than otherwise predicted. For example, Lawler et al. (1997) suggested that plant-mediated effects of elevated [CO2] might be particularly important for eucalypt herbivores because the high light and low soil nutrient conditions common to Australia result in foliage that is already nutritionally marginal.
C3 plants have persisted through substantial variations in [CO2] over geological time, and are therefore likely to have sufficient plasticity to acclimate or adapt to future change (Ward and Kelly 2004). However, the current rate and magnitude of climate change far exceeds that of the recent past. Long-lived woody plants, in particular, are not expected to migrate rapidly enough to keep up with shifting climate zones (Hughes et al. 1996). Insects may respond more quickly as behavioural changes, dispersal and rapid generation times facilitate adaptation when genetic diversity is sufficient for selection to occur (but see Hoffmann et al. 2003). Poleward range shifts (Parmesan et al. 1999) and compensatory feeding (Stiling and Cornelissen 2007) have already been observed, but evidence for adaptation and acclimation to modern climate change is limited due to a lack of long-term ecological data (but see Docherty et al. 1997; Brooks and Whittaker 1998; van Asch et al. 2007). As specialists on long-lived, nutrient-limited trees, eucalypt herbivores may be particularly sensitive to changes in the chemistry, distribution and abundance of their host plants. In this study, larvae performed best on foliage grown at elevated temperature and low [CO2] (with highest [N]), rather than on foliage grown under present-day conditions. It is likely that eucalypt herbivores are better adapted to subsisting on a suboptimal diet and taking advantage of temporary increases in leaf [N] than they are to optimal performance under current conditions. As such, sufficient plasticity may exist to facilitate acclimation to predicted future reductions in food quantity and quality.
By assessing insect responses to concurrent increases in [CO2] and temperature, this study has shown that measuring changes in foliar chemistry alone is not sufficient to accurately predict the impacts of climate change on insect herbivores. Rising [CO2] delayed insect development and stimulated compensatory feeding by reducing foliage quality. However, a concurrent increase in temperature reduced these negative effects by accelerating insect development. Exposure to low [CO2] reflecting pre-industrial conditions revealed consistent but non-linear plant and insect responses to rising [CO2], suggesting that with rising temperature, insects may be less challenged by future [CO2]-induced reductions in foliar quality than during the rise of [CO2] since the industrial revolution. To further improve our understanding of the character and magnitude of herbivore responses to climate change, and provide insight into the future capacity of plants and insects to adapt or acclimate, a greater emphasis on multigenerational, multifactorial studies is required. Of particular importance for warm, nutrient-limited, dry-land ecosystems such as Australia’s extensive eucalypt woodlands is whether interactions with temperature and water availability will cause available leaf [N] to drop below critical thresholds for insect development. Such studies will substantially improve our understanding of the potential multitrophic consequences of future climate change.
References
Ainsworth EA, Long SP (2005) What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol 165(2):351–371. doi:10.1111/j.1469-8137.2004.01224.x
Ainsworth EA, Rogers A (2007) The response of photosynthesis and stomatal conductance to rising CO2: mechanisms and environmental interactions. Plant Cell Environ 30(3):258–270. doi:10.1111/j.1365-3040.2007.01641.x
Axford Y, Briner JP, Francis DR, Miller GH, Walker IR, Wolfe AP (2011) Chironomids record terrestrial temperature changes throughout Arctic interglacials of the past 200,000 years. Geol Soc Am Bull 123(7–8):1275–1287. doi:10.1130/b30329.1
Ayub G, Smith RA, Tissue DT, Atkin OK (2011) Impacts of drought on leaf respiration in darkness and light in Eucalyptus saligna exposed to industrial-age atmospheric CO2 and growth temperature. New Phytol 190(4):1003–1018. doi:10.1111/j.1469-8137.2011.03673.x
Barton CVM, Ellsworth DS, Medlyn BE, Duursma RA, Tissue DT, Adams MA, Eamus D, Conroy JP, McMurtie RE, Parsaby J, Linder S (2010) Whole-tree chambers for elevated atmospheric CO2 experimentation and tree scale flux measurements in south-eastern Australia: the Hawkesbury Forest Experiment. Agr Forest Meteorol 150:941–951
Beaumont LJ, Hughes L (2002) Potential changes in the distributions of latitudinally restricted Australian butterfly species in response to climate change. Glob Change Biol 8(10):954–971
Brooks GL, Whittaker JB (1998) Responses of multiple generations of Gastrophysa viridula, feeding on Rumex obtusifolius, to elevated CO2. Glob Change Biol 4(1):63–75. doi:10.1046/j.1365-2486.1998.00111.x
Chong JH, van Iersel MW, Oetting RD (2004) Effects of elevated carbon dioxide levels and temperature on the life history of the Madeira mealybug (Hemiptera: Pseudococcidae). J Entomol Sci 39(3):387–397
Coley PD, Massa M, Lovelock CE, Winter K (2002) Effects of elevated CO2 on foliar chemistry of saplings of nine species of tropical tree. Oecologia 133(1):62–69. doi:10.1007/s00442-002-1005-6
Common I (1990) Moths of Australia. Melbourne University Press, Carlton
Dewar RC, Watt AD (1992) Predicted changes in the synchrony of larval emergence and budburst under climatic warming. Oecologia 89(4):557–559
Dippery JK, Tissue DT, Thomas RB, Strain BR (1995) Effects of low and elevated CO2 on C-3 and C-4 annuals. 1. Growth and biomass allocation. Oecologia 101(1):13–20. doi:10.1007/bf00328894
Docherty M, Wade FA, Hurst DK, Whittaker JB, Lea PJ (1997) Responses of tree sap-feeding herbivores to elevated CO2. Glob Change Biol 3(1):51–59
Ebell LF (1969) Variation in total soluble sugars of conifer tissues with method of analysis. Phytochemistry 8:227–233
Fajer ED, Bowers MD, Bazzaz FA (1989) The effects of enriched carbon dioxide atmospheres on plant–insect herbivore interactions. Science 243:1198–1200
Flynn DFB, Sudderth EA, Bazzaz FA (2006) Effects of aphid herbivory on biomass and leaf-level physiology of Solanum dulcamara under elevated temperature and CO2. Environ Exp Bot 56(1):10–18. doi:10.1016/j.envexpbot.2004.12.001
Fox LR, Macauley BJ (1977) Insect grazing on Eucalyptus in response to variation in leaf tannins and nitrogen. Oecologia 29(2):145–162
Froggatt WW (1923) Forest insects of Australia. Government Printer, Sydney
Gerhart LM, Ward JK (2010) Plant responses to low [CO2] of the past. New Phytol 188(3):674–695. doi:10.1111/j.1469-8137.2010.03441.x
Ghannoum O, Phillips NG, Conroy JP, Smith RA, Attard RD, Woodfield R, Logan BA, Lewis JD, Tissue DT (2010a) Exposure to preindustrial, current and future atmospheric CO2 and temperature differentially affects growth and photosynthesis in Eucalyptus. Glob Change Biol 16(1):303–319. doi:10.1111/j.1365-2486.2009.02003.x
Ghannoum O, Phillips NG, Sears MA, Logan BA, Lewis JD, Conroy JP, Tissue DT (2010b) Photosynthetic responses of two eucalypts to industrial-age changes in atmospheric [CO2] and temperature. Plant Cell Environ 33:1671–1681
Henery ML, Wallis IR, Stone C, Foley WJ (2008) Methyl jasmonate does not induce changes in Eucalyptus grandis leaves that alter the effect of constitutive defences on larvae of a specialist herbivore. Oecologia 156(4):847–859
Hill JK, Hodkinson ID (1995) Effects of temperature on phenological synchrony and altitudinal distribution of jumping plant lice (Hemiptera: Psylloidea) on dwarf willow (Salix lapponum) in Norway. Ecol Entomol 20(3):237–244
Hill JK, Thomas CD, Huntley B (1999) Climate and habitat availability determine 20th century changes in a butterfly’s range margin. P Roy Soc Lond B Bio 266(1425):1197–1206
Hoffmann AA, Hallas RJ, Dean JA, Schiffer M (2003) Low potential for climatic stress adaptation in a rainforest Drosophila species. Science 301(5629):100–102. doi:10.1126/science.1084296
Hughes L, Cawsey EM, Westoby M (1996) Climatic range sizes of Eucalyptus species in relation to future climate change. Global Ecol Biogeogr 5(1):23–29. doi:10.2307/2997467
Johns CV, Hughes A (2002) Interactive effects of elevated CO2 and temperature on the leaf-miner Dialectica scalariella Zeller (Lepidoptera: Gracillariidae) in Paterson’s Curse, Echium plantagineum (Boraginaceae). Glob Change Biol 8(2):142–152
Johns CV, Beaumont LJ, Hughes L (2003) Effects of elevated CO2 and temperature on development and consumption rates of Octotoma championi and O. scabripennis feeding on Lantana camara. Entomol Exp Appl 108:169–178
Lawler IR, Foley WJ, Woodrow IE, Cork SJ (1997) The effects of elevated CO2 atmospheres on the nutritional quality of Eucalyptus foliage and its interaction with soil nutrient and light availability. Oecologia 109(1):59–68
Leather SR (1988) Size, reproductive potential and fecundity in insects: things aren’t as simple as they seem. Oikos 51(3):386–389
Lewis JD, Ward JK, Tissue DT (2010) Phosphorus supply drives nonlinear responses of cottonwood (Populus deltoides) to increases in CO2 concentration from glacial to future concentrations. New Phytol 187(2):438–448. doi:10.1111/j.1469-8137.2010.03307.x
Lincoln DE, Couvet D, Sionit N (1986) Responses of an insect herbivore to host plants grown in carbon-dioxide enriched atmospheres. Oecologia 69(4):556–560. doi:10.1007/bf00410362
Lindroth RL, Kinney KK, Platz CL (1993) Responses of deciduous trees to elevated atmospheric CO2: productivity, phytochemistry, and insect performance. Ecology 74(3):763–777. doi:10.2307/1940804
Massad TJ, Dyer LA (2010) A meta-analysis of the effects of global environmental change on plant–herbivore interactions. Arthropod-Plant Inte 4(3):181–188. doi:10.1007/s11829-010-9102-7
MPIGA (2008) Montreal process implementation group for Australia, Australia’s state of the forests report 2008. Bureau of Rural Science, Canberra
Nahrung HF, Dunstan PK, Allen GR (2001) Larval gregariousness and neonate establishment of the eucalypt-feeding beetle Chrysophtharta agricola (Coleoptera: Chrysomelidae: Paropsini). Oikos 94(2):358–364
Ohmart CP, Edwards PB (1991) Insect herbivory on Eucalyptus. Annu Rev Entomol 36:637–657
Overpeck JT, Bartlein PJ, Webb T (1991) Potential magnitude of future vegetation change in eastern North America: comparisons with the past. Science 254(5032):692–695
Parmesan C, Ryrholm N, Stefanescu C, Hill JK, Thomas CD, Descimon H, Huntley B, Kaila L, Kullberg J, Tammaru T, Tennent WJ, Thomas JA, Warren M (1999) Poleward shifts in geographical ranges of butterfly species associated with regional warming. Nature 399(6736):579–583
Pinkard EA, Battaglia M, Roxburgh S, O’Grady AP (2011) Estimating forest net primary production under changing climate: adding pests into the equation. Tree Physiol 31(7):686–699
Prentice IC, Harrison SP, Bartlein PJ (2011) Global vegetation and terrestrial carbon cycle changes after the last ice age. New Phytol 189(4):988–998. doi:10.1111/j.1469-8137.2010.03620.x
R Development Core Team (2009) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. ISBN 3-90051-07-0. http://www.R-project.org
Robinson EA, Ryan GD, Newman JA (2012) A meta-analytical review of the effects of elevated CO2 on plant–arthropod interactions highlights the importance of interacting environmental and biological variables. New Phytol 194(2):321–336. doi:10.1111/j.1469-8137.2012.04074.x
Sage RF, Coleman JR (2001) Effects of low atmospheric CO2 on plants: more than a thing of the past. Trends Plant Sci 6(1):18–24. doi:10.1016/s1360-1385(00)01813-6
Solomon SD, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Toignor M, Miller HL (eds) (2007) Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, 2007. Cambridge University Press, Cambridge
Springett BP (1978) On the ecological role of insects in Australian eucalypt forests. Aust J Ecol 3:129–139
Steinbauer MJ (2001) Specific leaf weight as an indicator of juvenile leaf toughness in Tasmanian bluegum (Eucalyptus globulus ssp. globulus): implications for insect defoliation. Austral For 64:32–37
Stiling P, Cornelissen T (2007) How does elevated carbon dioxide (CO2) affect plant–herbivore interactions? A field experiment and meta-analysis of CO2-mediated changes on plant chemistry and herbivore performance. Glob Change Biol 13(9):1823–1842. doi:10.1111/j.1365-2486.2007.01392.x
Stiling P, Rossi AM, Hungate B, Dijkstra P, Hinkle CR, Knott WM III, Drake B (1999) Decreased leaf-miner abundance in elevated CO2: reduced leaf quality and increased parasitoid attack. Ecol Appl 9:240–244
Tissue DT, Lewis JD (2010) Photosynthetic responses of cottonwood seedlings grown in glacial through future atmospheric CO2 vary with phosphorus supply. Tree Physiol 30(11):1361–1372. doi:10.1093/treephys/tpq077
Tissue DT, Lewis JD (2012) Learning from the past: how low [CO2] studies inform plant and ecosystem response to future climate change. New Phytol 194(1):4–6
van Asch M, Tienderen PH, Holleman LJM, Visser ME (2007) Predicting adaptation of phenology in response to climate change, an insect herbivore example. Glob Change Biol 13(8):1596–1604. doi:10.1111/j.1365-2486.2007.01400.x
Veteli TO, Kuokkanen K, Julkunen-Tiitto R, Roininen H, Tahvanainen J (2002) Effects of elevated CO2 and temperature on plant growth and herbivore defensive chemistry. Glob Change Biol 8(12):1240–1252
Visser ME, Holleman LJM (2001) Warmer springs disrupt the synchrony of oak and winter moth phenology. P Roy Soc Lond B Bio 268(1464):289–294
Wallis IR, Nicolle D, Foley WJ (2010) Available and not total nitrogen in leaves explains key chemical differences between the eucalypt subgenera. Forest Ecol Manag 260:814–821
Wappler T, Denk T (2011) Herbivory in early tertiary Arctic forests. Palaeogeogr Palaeoecol 310(3–4):283–295. doi:10.1016/j.palaeo.2011.07.020
Ward JK, Kelly JK (2004) Scaling up evolutionary responses to elevated CO2: lessons from Arabidopsis. Ecol Lett 7(5):427–440. doi:10.1111/j.1461-0248.2004.00589.x
Way DA, Oren R (2010) Differential responses to changes in growth temperature between trees from different functional groups and biomes: a review and synthesis of data. Tree Physiol 30(6):669–688. doi:10.1093/treephys/tpq015
White TCR (1993) The inadequate environment: nitrogen and the abundance of animals. Springer, Berlin
Williams RS, Norby RJ, Lincoln DE (2000) Effects of elevated CO2 and temperature-grown red and sugar maple on gypsy moth performance. Glob Change Biol 6(6):685–695. doi:10.1046/j.1365-2486.2000.00343.x
Williams RS, Lincoln DE, Norby RJ (2003) Development of gypsy moth larvae feeding on red maple saplings at elevated CO2 and temperature. Oecologia 137(1):114–122. doi:10.1007/s00442-003-1327-z
Young CJ (2011) News bulletin of the Australian Entomological Society. Myrmecia 47(2):15
Zvereva EL, Kozlov MV (2006) Consequences of simultaneous elevation of carbon dioxide and temperature for plant–herbivore interactions: a meta-analysis. Glob Change Biol 12(1):27–41. doi:10.1111/j.1365-2486.2005.01086.x
Acknowledgments
This research was funded by the Australian Research Council (DP1095972 to MR and DSE; DP0879531 to DTT). We thank Sarah Bingham, Rosemary Freeman, Goran Lopaticki, Jenny Shanks and Renee Smith for technical assistance, Remko Duursma for statistical advice and Taronga Zoo for access to the E. tereticornis plantation on the UWS Hawkesbury campus. We gratefully acknowledge the helpful comments and suggestions of the editorial team and two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Phyllis Coley.
Rights and permissions
About this article
Cite this article
Murray, T.J., Tissue, D.T., Ellsworth, D.S. et al. Interactive effects of pre-industrial, current and future [CO2] and temperature on an insect herbivore of Eucalyptus . Oecologia 171, 1025–1035 (2013). https://doi.org/10.1007/s00442-012-2467-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-012-2467-9