Abstract
Taste receptors and their downstream signaling molecules are activated by sugars and sweeteners in the gut and participate in the regulation of glucose transport into enterocytes. The glucose transporter families GLUT and SGLT are responsible for the absorption of glucose, GLUT4 and SGLT1 being expressed preferentially in T1R3-positive taste cells. However, the expression patterns of the other glucose transporters in mouse gustatory tissues have not yet been elucidated. Therefore, we have examined the expression patterns of the glucose transporters (GLUT1-4 and SGLT1-3) in mouse gustatory tissues. Reverse transcription/polymerase chain reaction assays have revealed that GLUT1, 3, and 4 and SGLT1 mRNAs are expressed in the circumvallate papillae. Immunohistochemical analysis has shown that SGLT1 is expressed in a subset of the epithelial cells: from the basal cell layer to the prickle cell layer and in intragemmal and extragemmal epithelium cells in the circumvallate, foliate, and fungiform papillae. GLUT1, GLUT3, and GLUT4 are expressed in the prickle cell layers and/or basal cell layers in these papillae. Moreover, GLUT1, but not GLUT3 or GLUT4, is expressed in a subset of intragemmal and extragemmal epithelium cells in these papillae. Double-labeling experiments have demonstrated that GLUT1-positive taste bud cells coexpress gustducin and inositol 1,4,5-triphosphate receptor type III. These results suggest that SGLT1 and GLUT1 play a role in glucose-sensing and/or transport in mouse taste buds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mammalian cells take up glucose from the extracellular fluid into the cell through two families of structurally related glucose transporters (Thorens and Mueckler 2010; Wright et al. 2007; Zhao and Keating 2007): the facilitated glucose transporters, the GLUT family, and the sodium-coupled glucose cotransporters, the SGLT family. The GLUT family mediates a bidirectional and energy-independent process of glucose transport in most tissues and cells, whereas the SGLT family mediates an active sodium-linked transport process against an electrochemical gradient. The GLUT family consists of 14 members (GLUT1-14). In this family, GLUTs 1–4 are well-established glucose transporter isoforms and have distinct regulatory and/or kinetic properties that reflect their specific roles in cellular and whole-body glucose homeostasis. The SGLT family consists of six members (SGLT1-6). In this family, two members function as sugar transporters (SGLT1 and SGLT2), and SGLT3 is a sugar sensor (Wright et al. 2007).
The sweet receptor, which detects sugars including sucrose and glucose in taste buds, is composed of T1R2 and T1R3 (Chandrashekar et al. 2006; Chaudhari and Roper 2010; Roper 2007). Several types of enteroendocrine cells throughout the gut express T1R2 and T1R3, together with the signal-transduction molecules gustducin, Gβ3, Gγ13, phospholipase Cβ2 (PLCβ2), and the TRPM5 Ca2+-activated Na+/K+ channel (Cummings and Overduin 2007). T1R3 senses intestinal sugar levels and regulates the glucose transporters, SGLT1 and GLUT2, in enterocytes (Mace et al. 2007; Margolskee et al. 2007). Recently, Yee et al. (2011) have reported that GLUT2, GLUT4, GLUT8, GLUT9, SGLT1, and two components of the ATP-gated K+ (KATP) metabolic sensor [sulfonylurea receptor (SUR) 1 and potassium inwardly rectifying channel (Kir) 6.1] are expressed selectively in taste cells. Of these molecules, GLUT4, SGLT1, and SUR1 are expressed exclusively in T1R3-positive taste cells. However, the expression patterns of the other glucose transporters in mouse gustatory tissues have not yet been elucidated. Therefore, we have examined the expression patterns of the glucose transporters (GLUT1-4 and SGLT1-3) in mouse gustatory tissues.
In the present study, we have used reverse transcription/polymerase chain reaction (RT-PCR), immunoblot analysis, and immunohistochemistry to examine the expression patterns of the GLUT and SGLT glucose transporter families (GLUT1-4 and SGLT1-3) in mouse gustatory tissues. We have also compared the expression pattern of GLUT1 with those of gustducin and inositol 1,4,5-triphosphate receptor type III (IP3R3), which is the downstream molecule of PLCβ2 (Chaudhari and Roper 2010).
Materials and methods
All the methods used in the present study were approved by the Institutional Animal Care and Use Committee at Kyushu Dental College.
RT-PCR analysis
Adult male C57BL6/J mice were killed by sodium pentobarbital injection. Their small intestines and kidneys were quickly removed. Their tongues were dissected proximal to the circumvallate papillae and immediately placed in an isolation buffer (26 mM NaHCO3, 2.5 mM NaH2PO4, 20 mM glucose, 65 mM NaCl, 20 mM KCl, and 1 mM EDTA) for 10 min on ice. The preparations were then removed from ice, and 1 ml isolation buffer containing 1.5 mg/ml pronase E (Sigma-Aldrich, St. Louis, Mo., USA) and 1 mg/ml elastase (Worthington Biochemical, Lakewood, N.J., USA) was injected with a 25-gauge syringe under the lingual epithelium of the circumvallate papillae of the dissected tongues. After 20 min of incubation in isolation buffer at room temperature, the epithelium was gently peeled from the underlying muscle layer under a dissecting microscope. Tongue epithelium containing circumvallate papillae and tongue epithelium with no taste papillae were removed from the peeled epithelium. Their total RNAs were isolated by using the GenElute Mammalian Total RNA Miniprep Kit (Sigma-Aldrich) according to the manufacturer's instructions. Following incubation with TURBO DNase (Applied Biosystems, Foster City, Calif., USA) to remove any contaminating genomic DNA, first-strand cDNA synthesis was performed by RT of 100 ng each total RNA sample by using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems). PCR amplifications were performed under the following conditions with the primers listed in Table 1: 94°C for 20 s, 53°C for 30 s, 72°C for 40 s for a total of 40 cycles for β-actin; 94°C for 20 s, 56°C for 30 s, 72°C for 60 s for a total of 35 cycles for SGLT1, SGLT2, and GLUT2; 94°C for 20 s, 56°C for 30 s, 72°C for 60 s for a total of 40 cycles for SGLT3; 94°C for 20 s, 58°C for 30 s, 72°C for 30 s for a total of 35 cycles for GLUT1; 94°C for 20 s, 58°C for 30 s, 72°C for 60 s for a total of 40 cycles for GLUT3 and GLUT4. The reverse transcriptase step was omitted in controls to confirm removal of all genomic DNA. All PCR products were subcloned and sequenced.
Immunoblot analysis
Adult male C57BL6/J mice were killed by sodium pentobarbital injection. The tissues containing circumvallate papillae and foliate papillae and kidney were removed and homogenized in a Polytoron homogenizer (Kinematica, Switzerland) with 5 volumes of ice-cold buffer comprising 0.32 M sucrose, 50 mM TRIS-HCl (pH 7.4), 1 mM EDTA, 1 mM EGTA, and 0.25 mM dithiothreitol in the presence of protease and phosphatase inhibitor cocktails (Sigma-Aldrich) and centrifuged at 2000g for 5 min at 4°C. The supernatant was subsequently centrifuged at 13,000g for 30 min. The membrane fractions were resuspended in the homogenization buffer, and the samples were suspended in SDS sample buffer containing 100 mM dithiothreitol. Equal amounts (20 μg) of the samples were subjected to 10% (w/v) SDS-polyacrylamide gel electrophoresis, and the separated proteins were transferred electrophoretically to a polyvinylidine difluoride (PVDF) filter (Millipore, Mass., USA). Non-specific binding sites on the PVDF filter were blocked by incubation with 1% bovine serum albumin for 1 h. The filter was then incubated with the anti-GLUT1 rabbit polyclonal antibody (#RB-9052-P0; Thermo Fisher Scientific, Waltham, Mass., USA), the anti-GLUT3 rabbit polyclonal antibody (53520; AnaSpec, Fremont, Calif., USA), and the anti-GLUT4 rabbit polyclonal antibody (BS3680; Bioworld Technology, Mineapolis, Minn., USA) at dilutions of 1:500 for 5 h at room temperature. After being washed with 0.01 M phosphate-buffered saline (PBS) containing 0.03% (v/v) Triton X-100 (PBST), the filter was incubated with peroxidase-conjugated goat anti-rabbit IgG (GE healthcare Japan, Tokyo, Japan) for 1 h. After three rinses, the immunoreactive bands were made visible with a Immobilon Western horseradish peroxide substrate (Millipore) according to the manufacturer’s standard method.
Immunohistochemistry
Adult male C57BL6/J mice were anesthetized with chloral hydrate (350 mg/kg) and transcardially perfused with a fixative containing 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.3) for 15 min. The areas of fungiform, foliate, and circumvallate papillae were removed and rinsed overnight with phosphate buffer containing 30% sucrose. The tissues were then embedded in Tissue-Tek OCT compound (Sakura Finetechnical, Tokyo, Japan) and snap-frozen in a dry ice/isopentane mixture. Sections (8 μm thick) were cut with a cryostat, thaw-mounted onto MAS-coated glass slides (MATSUNAMI, Tokyo, Japan), air-dried for 60 min, and washed twice in PBS for 5 min each. The frozen sections were treated with 10 mM citrate buffer (pH 6.0) in an autoclave at 121°C for 10 min and then processed for indirect single- or double-immunofluorescence staining for GLUT1 and GLUT4. The frozen sections were treated with 10 mM TE buffer pH 9.0 (10 mM TRIS-HCl pH 9.0, 1 mM EDTA) at 95°C for 40 min and then processed for indirect single- or double-immunofluorescence staining for GLUT3 and SGLT1.
Double-immunofluorescence staining involved incubations with the anti-GLUT1 rabbit polyclonal antibody (#RB-9052-P0; 1:100 dilution; Thermo Fisher Scientific), the anti-GLUT3 rabbit polyclonal antibody (53520; 1:100 dilution; AnaSpec), the anti-GLUT4 rabbit polyclonal antibody (BS3680; 1:100 dilution; Bioworld Technology), the anti-SGLT1 rabbit polyclonal antibody (ab14686; 1:100 dilution; Abcam, Cambridge, UK), anti-IP3R3 (610312; 1:800 dilution; BD Transduction laboratories), and the anti-cytokeratin 18 mouse monoclonal antibody (Ks18.04; 1:100 dilution; Progen) as the primary antibodies and subsequent incubations with Alexa-Fluor-488-conjugated goat anti-rabbit IgG (1:500 dilution; Invitrogen, Carlsbad, Calif., USA) and Alexa-Fluor-546-conjugated goat anti-mouse IgG (1:500 dilution; Invitrogen) as the secondary antibodies. For the double-immunofluorescence staining of GLUT1 and gustducin, immunostaining for GLUT1 was initially carried out with an Alexa-Fluor-488-conjugated goat anti-rabbit IgG (1:500 dilution; Invitrogen) as the secondary antibody, followed by immunostaining with an Alexa-Fluor-546-labeled anti-gustducin rabbit polyclonal antibody (1:500 dilution; Santa Cruz Biotechnology, Santa Cruz, Calif., USA). This labeled antibody was prepared by using a Zenon rabbit IgG labeling kit (Invitrogen) according to the manufacturer’s instructions.
Images of the immunostained sections were obtained by changing the filter cube without altering the portion of the section or focus, by using a cooled charge-coupled device camera (DP72; OLYMPUS, Tokyo, Japan). Images of the double-immunostained sections were overlaid with Adobe Photoshop CS3 (Adobe Systems, San Jose, Calif., USA).
Renal tubules in the kidney were used as a positive control to determine whether the immunoreactivities of GLUT1, GLUT3, GLUT4, and SGLT1 were specific. In the gustatory papillae, PBS was substituted for the primary antibodies to check for antibody specificity.
Results
RT-PCR analysis for SGLT and GLUT families
We used RT-PCR to examine the presence of mRNAs of SGLT and GLUT families in mouse circumvallate papillae. As shown in Fig. 1, SGLT1, GLUT1, GLUT3, and GLUT4 mRNAs were expressed in the circumvallate papillae and non-taste epithelium, whereas SGLT2, SGLT3, and GLUT2 mRNAs were not expressed in either the circumvallate papillae or non-taste epithelium. The amplification products were the expected sizes, and their identities were confirmed by DNA sequencing. These results indicated that SGLT1, GLUT1, GLUT3, and GLUT4 mRNAs were expressed in the lingual epithelium including the circumvallate papillae.
SGLT1, GLUT1, GLUT3, and GLUT4 mRNAs are expressed in mouse circumvallate papillae. Reverse transcription/polymerase chain reaction was performed with total RNAs prepared from the circumvallate papillae (CV), taste epithelium lacking taste buds (NT non-taste epithelium), kidney, and small intestine. Amplified products of SGLT1 (501 bp), SGLT2 (707 bp), SGLT3 (506 bp), GLUT1 (292 bp), GLUT2 (273 bp), GLUT3 (370 bp), GLUT4 (410 bp), and β-actin (573 bp) were obtained with specific primer sets, separated by gel electrophoresis, and visualized by UV illumination after ethidium bromide staining. The expression level of the mRNA for β-actin (bottom) was used as a control. The reverse transcriptase step was omitted in controls to confirm removal of genomic DNA. mRNAs for SGLT1, GLUT1, GLUT3, GLUT4, and β-actin were detected in the CV, NT, kidney, and/or small intestine
Immunoblot analyses for GLUT1, GLUT3, and GLUT4
In order to confirm the specificities of GLUT1, GLUT3, and GLUT4 antibodies, immunoblotting of mouse taste tissue extracts was performed, together with kidney as a positive control. With regard to the immunoblot analysis for GLUT1, GLUT1 has been demonstrated to be a broad band of a 45–60 kDa form (Asano et al. 1991; Heilig et al. 1995); this broad band is produced by glycosylation. The broad bands between 45–60 kDa indicating GLUT1 appeared in both the kidney and taste tissues containing foliate and circumvallate papillae (Fig. 2). In the immunoblot analysis for GLUT3, an immunoreactive band of molecular mass around 46 kDa representing GLUT3 (Heilig et al. 1995) was recognized in both the kidney and taste tissues (Fig. 2). In the immunoblot analysis for GLUT4, an immunoreactive band of molecular mass around 45 kDa representing GLUT4 (Clancy and Czech 1990) was recognized in both the kidney and taste tissues (Fig. 2).
GLUT1, GLUT3, and GLUT4 proteins are expressed in mouse taste tissues containing foliate and circumvallate papillae. An immunoreactive broad band of 45–60 kDa indicating GLUT1 is observed in taste tissue. GLUT3 is identified as a 46-kDa protein in taste tissues. GLUT4 is identified as a 45-kDa protein in taste tissues. Kidney provides a positive control. The positions of molecular mass markers of standard proteins are indicated left
Immunohistochemistry for SGLT1, GLUT1, GLUT3, and GLUT4
In the mouse circumvallate papillae, PBS was substituted for the anti-SGLT1, GLUT1, GLUT3, and GLUT4 antibodies in order to check for antibody specificities. Immunoreactivities were not observed when PBS was substituted for these antibodies (Figs. 3m, 4m).
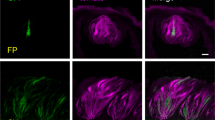
Expression pattern of SGLT1 in the mouse gustatory papillae. Double-immunofluorescence staining for SGLT1 (green) and cytokeratin 18 (red) in the mouse circumvallate (a–f), foliate (g–i), and fungiform (j–l) papillae. a, d, g, j Anti-SGLT1 antibody labels a subset of epithelial cells from the basal cell layer to the prickle cell layer and the muscle fibers in these papillae (arrows). The SGLT1 antibody also labels a subset of intragemmal (arrowheads) and extragemmal epithelium cells (arrows). b, e, h, k Anti-cytokeratin 18 antibody labels of a subset of taste bud cells. c, f, i, l Merged fluorescent images of SGLT1 immunoreactivity and cytokeratin 18 immunoreactivity illustrating the coexpression of SGLT1 and cytokeratin 18 (arrowheads). m Anti-SGLT1 antibody was replaced with PBS to check for antibody specificity in the mouse circumvallate papillae. Bars 100 μm (a–c), 50 μm (d–m)
Expression pattern of GLUT3 in the mouse gustatory papillae. Double-immunofluorescence staining for GLUT3 (green) and cytokeratin 18 (red) in the mouse circumvallate (a–f), foliate (g–i), and fungiform (j–l) papillae. a, d, g, j Anti-GLUT3 antibody labels the basal cell layers and prickle cell layer (arrows) in the mouse circumvallate, foliate, and fungiform papillae. b, e, h, k Anti-cytokeratin 18 antibody labels a subset of taste bud cells. c, f, i, l Merged fluorescent images of GLUT3 immunoreactivity and cytokeratin 18 immunoreactivity illustrating the expression patterns of GLUT3 and cytokeratin 18. m Anti-GLUT3 antibody was replaced with PBS to check for antibody specificity in the mouse circumvallate papillae. Bars 100 μm (a–c), 50 μm (d–m)
The anti-SGLT1 antibody strongly labeled the muscle fibers beneath the fungiform, foliate, and circumvallate papillae (Fig. 3a, b, d, j) and weakly labeled a subset of epithelial cells from the basal cell layer to the prickle cell layer and in the intragemmal and extragemmal epithelium cells in the circumvallate, foliate, and fungiform papillae (Fig. 3c, f, i, l). GLUT3-positive signals were detected in the prickle cell layers and basal cell layers in the mouse circumvallate, foliate, and fungiform papillae (Fig. 4a, d, g, j) but were not detected in the intragemmal and extragemmal epithelium cells in these papillae (Fig. 4a, d, g, j). GLUT4-positive signals were detected in the basal cell layers and muscle fibers in the circumvallate, foliate, and fungiform papillae (Fig. 5a, d, g, j) but were not detected in the intragemmal and extragemmal epithelium cells in these papillae (Fig. 5a, d, g, j). GLUT1-positive signals were detected in the blood vascular layer, muscle fibers, prickle cell layers, and basal cell layers in the circumvallate, foliate, and fungiform papillae (Fig. 6a, d, g, j) and were also detected in a subset of intragemmal and extragemmal epithelium cells in these papillae (Fig. 6a, d, g, j). In addition, a few strong GLUT1-positive signals were observed in the intragemmal cells in these papillae (Fig. 6c, f, i, l).
Expression pattern of GLUT4 in the mouse gustatory papillae. Double-immunofluorescence staining for GLUT4 (green) and cytokeratin 18 (red) in the mouse circumvallate (a–f), foliate (g–i), and fungiform (j–l) papillae. a, d, g, j Anti-GLUT4 antibody labels the prickle cell layers, basal cell layers, and muscle fibers (arrows) in the mouse circumvallate, foliate, and fungiform papillae. b, e, h, k Anti-cytokeratin 18 antibody labels a subset of taste bud cells. c, f, i, l Merged fluorescent images of GLUT4 immunoreactivity and cytokeratin 18 immunoreactivity showing the expression patterns of GLUT4 and cytokeratin 18; GLUT4 was not detected in the intragemmal and extragemmal epithelium cells in these papillae (oval of dotted lines in j–l region of taste buds in fungiform papillae). Bars 100 μm (a–c), 50 μm (d–l)
Expression pattern of GLUT1 in the mouse gustatory papillae. Double-immunofluorescence staining for GLUT1 (green) and cytokeratin 18 (red) in the mouse circumvallate (a–f), foliate (g–i), and fungiform (j–l) papillae. a, d, g, j Anti-GLUT1 antibody labels the blood vascular layers, prickle cell layers, basal cell layers, and muscle fibers (arrows) in the mouse circumvallate, foliate, and fungiform papillae. GLUT1-positive signals also were detected in a subset of intragemmal (arrowheads) and extragemmal epithelium cells (arrows). b, e, h, k Anti-cytokeratin 18 antibody labels a subset of taste bud cells. c, f, i, l Merged fluorescent images of GLUT1 immunoreactivity and cytokeratin 18 immunoreactivity depicting the coexpression of GLUT1 and cytokeratin 18 (arrowheads). Bars 100 μm (a–c), 50 μm (d–l)
IP3R3, a marker of type II taste cells (Chaudhari and Roper 2010), was expressed in a large subset of mouse circumvallate taste buds (Fig. 7b). In the double-immunofluorescence staining for GLUT1 and IP3R3 in the circumvallate papillae, all GLUT1-expressing cells also showed IP3R3 immunoreactivity (Fig. 7a–c). Gustducin, a marker of a subset of type II taste cells (Yang et al. 2000), was expressed in a subset of mouse circumvallate taste buds (Fig. 7e). In double-immunofluorescence staining for GLUT1 and gustducin in the circumvallate papillae, all GLUT1-expressing cells also showed gustducin immunoreactivity (Fig. 7d–f).
Expression pattern of GLUT1, IP3R3, and gustducin in the mouse gustatory papillae. Double-immunofluorescence staining for GLUT1 (green) and IP3R3 (red), and for GLUT1 (green) and gustducin (red) in mouse circumvallate papillae. a, d Anti-GLUT1 antibody labels a subset of taste bud cells (arrowheads). b Anti-IP3R3 antibody labels a subset of taste bud cells. c Merged fluorescent image of GLUT1 immunoreactivity and of IP3R3 immunoreactivity illustrating the coexpression of GLUT1 and IP3R3 (arrowheads). e Anti-gustducin antibody labels a subset of taste bud cells. f Merged fluorescent image of GLUT1 immunoreactivity and of gustducin immunoreactivity depicting the coexpression of GLUT1 and gustducin (arrowheads). Bars 25 μm
Discussion
In the present study, SGLT1, GLUT1, GLUT3, and GLUT4 antibodies have been used for the immunohistochemical investigation of taste tissue. Of these antibodies, the specificity of the SGLT1 antibody (ab14686; Abcam) in taste tissue has been previously confirmed (Yee et al. 2011), but not of the GLUT1, GLUT3, and GLUT4 antibodies. Therefore, we have attempted to verify the specificities of these antibodies with immunoblot analyses in taste tissue containing foliate and circumvallate papillae. Immunoblot analyses have shown that GLUT1, GLUT3, and GLUT4 proteins are detectable in taste tissues at the expected sizes. Our data therefore confirm the specificities of these antibodies in taste tissue.
In our hands, immunohistochemistry has shown that GLUT4 is expressed only in basal layers and muscle and not in taste buds, whereas Yee et al. (2011) have reported that GLUT4 is expressed in taste receptor cells. The specificity of the results obtained by immunohistochemistry depends on the specificity of the antibody (Burry 2000). We have used one human GLUT4 antibody (BS3680, Bioworld Technology) in our immunohistochemical studies, whereas Yee et al. (2011) have used an alternative human GLUT4 antibody (ab33780, Abcam). Therefore, the discrepancy in the expression pattern of GLUT4 might be attributable to the different specificities of these antibodies for GLUT4.
Our RT-PCR analysis has demonstrated that SGLT1, GLUT1, GLUT3, and GLUT4 mRNAs, but not SGLT2, SGLT3, and GLUT2 mRNAs, are expressed in the circumvallate papillae. Analyses of T1R3-null mice have revealed that sucrose can be detected by taste receptors other than T1R2 and T1R3 in taste buds (Damak et al. 2003; Delay et al. 2006). Another type of sugar sensor might be expressed in taste bud cells. In the SGLT family, SGLT2 is expressed in various tissues throughout the body, including the renal proximal tubule in which it appears to play a major role in the reabsorption of glucose from the glomerular filtrate (Wright et al. 2007). SGLT3 might be a sugar sensor rather than a transporter in cholinergic neurons and skeletal muscle (Diez-Sampedro et al. 2003). In the GLUT family, GLUT2 plays an essential role in glucose-sensing in pancreatic β-cells and in the hypothalamus (Zhao and Keating 2007). However, SGLT2, SGLT3, and GLUT2 presumably do not play a role as another type of sugar sensor in taste tissue, because SGLT2, SGLT3, and GLUT2 mRNAs are not expressed in circumvallate papillae. In addition to the results of RT-PCR analyses, immunohistochemical analyses have shown that SGLT1 and GLUT1 are expressed in a subset of intragemmal and extragemmal cells in the gustatory papillae. Taken together, these data indicate that SGLT1 and GLUT1 might play a role in glucose-sensing in the mouse circumvallate taste buds.
Our RT-PCR analysis suggests that GLUT2 mRNA is not expressed in the circumvallate papillae. In contrast, Yee et al. (2011) have shown that GLUT2 mRNA is expressed in the circumvallate papillae, by using RT-PCR. These contradictory results might have arisen because of different PCR amplification efficiencies. PCR amplification efficiency is affected by diverse PCR conditions, including primer positions and PCR enzymes, with those of this study being different from those of Yee et al. (2011). Therefore, the RT-PCR analyses of this study might not have detected the expression of GLUT2 in taste tissue, because the PCR amplification efficiency in this study might have been lower than that of Yee et al. (2011).
In the SGLT family, only SGLT1 has been found to be expressed in a subset of epithelial cells from the basal cell layer to prickle cell layer in gustatory papillae. SGLT1 is also expressed in the subset of intragemmal and extragemmal cells in the gustatory papillae. In the small intestine, SGLT1 is expressed in the apical domain and intracellular organelles in the enteroendocrine cells and the enterocytes (Balen et al. 2008). Enteroendocrine cells express taste-signaling molecules, including sweet (T1R) and bitter (T2R) taste receptors and gustducin (Sternini et al. 2008). These cells produce and release brain–gut peptides, including cholecystokinin, gastric inhibitory polypeptide, gastric peptide YY, and glucagon-like peptide 1, all of which are involved in the regulation of gastrointestinal secretion and motility, food intake and satiety, and glucose homeostasis (Sternini et al. 2008). The similarity of SGLT1 expression between the enteroendocrine cells and taste bud cells suggests that SGLT1 plays a role in the taste-signaling cascade in these cells.
GLUT1 is expressed in the prickle cell layers and basal cell layers in the mouse circumvallate, foliate, and fungiform papillae. GLUT1 is also expressed in a subset of intragemmal or extragemmal epithelium cells in these papillae. In the buccal mucosal epithelium, marked expression of GLUT1 has been observed in the prickle cell layer immediately above the basal layer, and the immunoreactivity decreases along the differentiation gradient (Kuroki et al. 2009). GLUT1 expression probably depends on epithelial cell differentiation in the oral mucosa (Gherzi et al. 1991; Kuroki et al. 2009). Therefore, we hypothesize that GLUT1 expression might depend on epithelial cell differentiation in the gustatory papillae with a similar expression pattern of epithelial cells in the oral mucosa.
GLUT1, GLUT3, and GLUT4 are expressed in the prickle cell layers and/or basal cell layers in the gustatory papillae. As mentioned above, GLUT1 is also expressed in a subset of intragemmal and extragemmal epithelium cells in these papillae. GLUT1, GLUT3, and GLUT4 are expressed in glomeruli and renal tubules (Heilig et al. 1995). The unique GLUT expression patterns along the renal tubules suggest unique functional roles for these isoforms. Like the GLUT expression patterns in glomeruli and renal tubules, the unique GLUT expression patterns in the gustatory papillae suggest common or unique functional roles for GLUT1, GLUT3, and GLUT4 in gustatory papillae.
The double-immunofluorescence staining of GLUT1 and gustducin has demonstrated the co-expression of GLUT1 and gustducin in a subset of taste bud cells. In previous studies involving immunohistochemistry and in situ hybridization analyses, the localization of T1R and T2R taste receptor proteins and of downstream signaling effectors for bitter, sweet, and umami taste-stimuli-positive cells was approximately equivalent to that of type II cells (Clapp et al. 2001, 2004; Miyoshi et al. 2001). Gustducin is also expressed in type II cells (Yang et al. 2000). Therefore, GLUT1 is expressed in a subset of gustducin- or IP3R3-expressing type II cells in the circumvallate papillae.
GLUT2, GLUT4, GLUT8, GLUT9, SGLT1, and two components of the KATP metabolic sensor (SUR1 and Kir 6.1) are expressed selectively in taste cells (Yee et al. 2011). Although GLUT1 and GLUT3 in taste tissue have not been investigated by Yee et al.(2011), our RT-PCR and immunohistochemical analyses have shown that GLUT1 and GLUT3 mRNAs are expressed in the taste tissue, and that GLUT1 is expressed in a subset of gustducin- or IP3R3-expressing type II cells in circumvallate papillae. SGLT1 is also expressed in a subset of intragemmal and extragemmal cells in the gustatory papillae. Yee et al. (2011) have reported that SGLT1 is expressed exclusively in a T1R3-positive subset of type II taste cells. Collectively, these data suggest that GLUT1 and SGLT1 might be expressed in type II taste cells. However, the detailed expression patterns of GLUT1 and SGLT1 in type II taste cells have not been elucidated. We speculate that GLUT1 and SGLT1 might act as glucose sensors and/or transporters in type II taste cells. Further studies regarding this aspect are necessary.
References
Asano T, Katagiri H, Takata K, Lin JL, Ishihara H, Inukai K, Tsukuda K, Kikuchi M, Hirano H, Yazaki Y, Oka Y (1991) The role of N-glycosylation of GLUT1 for glucose transport activity. J Biol Chem 266:24632–24636
Balen D, Ljubojević M, Breljak D, Brzica H, Žlender V, Koepsell H, Sabolić I (2008) Revised immunolocalization of the Na+-D-glucose cotransporter SGLT1 in rat organs with an improved antibody. Am J Physiol Cell Physiol 295:C475–C489
Burry RW (2000) Specificity controls for immunocytochemical methods. J Histochem Cytochem 48:163–166
Chandrashekar J, Hoon MA, Ryba NJ, Zuker CS (2006) The receptors and cells for mammalian taste. Nature 444:288–294
Chaudhari N, Roper SD (2010) The cell biology of taste. J Cell Biol 190:285–296
Clancy BM, Czech MP (1990) Hexose transport stimulation and membrane redistribution of glucose transporter isoforms in response to cholera toxin, dibutyryl cyclic AMP, and insulin in 3T3-L1 adipocytes. J Biol Chem 265:12434–12443
Clapp TR, Stone LM, Margolskee RF, Kinnamon SC (2001) Immunocytochemical evidence for co-expression of type III IP3 receptor with signaling components of bitter taste transduction. BMC Neurosci 2:6
Clapp TR, Yang R, Stoick CL, Kinnamon SC, Kinnamon JC (2004) Morphologic characterization of rat taste receptor cells that express components of the phospholipase C signaling pathway. J Comp Neurol 468:311–321
Cummings DE, Overduin J (2007) Gastrointestinal regulation of food intake. J Clin Invest 117:13–23
Damak S, Rong M, Yasumatsu K, Kokrashvili Z, Varadarajan V, Zou S, Jiang P, Ninomiya Y, Margolskee RF (2003) Detection of sweet and umami taste in the absence of taste receptor T1r3. Science 301:850–853
Delay ER, Hernandez NP, Bromley K, Margolskee RF (2006) Sucrose and monosodium glutamate taste thresholds and discrimination ability of T1R3 knockout mice. Chem senses 31:351–357
Diez-Sampedro A, Hirayama BA, Osswald C, Gorboulev V, Baumgarten K, Volk C, Wright EM, Koepsell H (2003) A glucose sensor hiding in a family of transporters. Proc Natl Acad Sci USA 100:11753–11758
Gherzi R, Melioli G, De Luca M, D'Agostino A, Guastella M, Traverso CE, D'Anna F, Franzi AT, Cancedda R (1991) High expression levels of the "erythroid/brain" type glucose transporter (GLUT1) in the basal cells of human eye conjunctiva and oral mucosa reconstituted in culture. Exp Cell Res 195:230–236
Heilig C, Zaloga C, Lee M, Zhao X, Riser B, Brosius F, Cortes P (1995) Immunogold localization of high-affinity glucose transporter isoforms in normal rat kidney. Lab Invest 73:674–684
Kuroki S, Yokoo S, Terashi H, Hasegawa M, Komori T (2009) Epithelialization in oral mucous wound healing in terms of energy metabolism. Kobe J Med Sci 55:E5–E15
Mace OJ, Affleck JA, Patel N, Kellett GL (2007) Sweet taste receptors in rat small intestine stimulate glucose absorption through apical GLUT2. J Physiol (Lond) 582:379–392
Margolskee RF, Dyer J, Kokrashvili Z, Salmon KS, Ilegems E, Daly K, Maillet EL, Ninomiya Y, Mosinger B, Shirazi-Beechey SP (2007) T1R3 and gustducin in gut sense sugars to regulate expression of Na+-glucose cotransporter 1. Proc Natl Acad Sci USA 104:15075–15080
Miyoshi MA, Abe K, Emori Y (2001) IP3 receptor type 3 and PLCβ2 are co-expressed with taste receptors T1R and T2R in rat taste bud cells. Chem Senses 26:259–265
Roper SD (2007) Signal transduction and information processing in mammalian taste buds. Pflügers Arch 454:759–776
Sternini C, Anselmi L, Rozengurt E (2008) Enteroendocrine cells: a site of “taste” in gastrointestinal chemosensing. Curr Opin Endocrinol Diabetes Obes 15:73–78
Thorens B, Mueckler M (2010) Glucose transporters in the 21st Century. Am J Physiol Endocrinol Metab 298:E141–E145
Wright EM, Hirayama BA, Loo DF (2007) Active sugar transport in health and disease. J Intern Med 261:32–43
Yang R, Tabata S, Crowley HH, Margolskee RF, Kinnamon JC (2000) Ultrastructural localization of gustducin immunoreactivity in microvilli of type II taste cells in the rat. J Comp Neurol 425:139–151
Yee KK, Sukumaran SK, Kotha R, Gilbertson TA, Margolskee RF (2011) Glucose transporters and ATP-gated K+ (KATP) metabolic sensors are present in type 1 taste receptor 3 (T1r3)-expressing taste cells. Proc Natl Acad Sci USA 108:5431–5436
Zhao FQ, Keating AF (2007) Functional properties and genomics of glucose transporters. Curr Genomics 8:113–128
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported in part by a research grant from the Society for Research on Umami Taste.
Rights and permissions
About this article
Cite this article
Toyono, T., Seta, Y., Kataoka, S. et al. Differential expression of the glucose transporters in mouse gustatory papillae. Cell Tissue Res 345, 243–252 (2011). https://doi.org/10.1007/s00441-011-1210-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-011-1210-x