Abstract
Varroa destructor is the most serious pest of honeybee (Apis mellifera), causing high economic losses in the beekeeping industry worldwide. The intensive utilization of many chemical substances against the mites resulted in resistance development. One of the applicable and alternative treatments being used for their control is plant-derived products (PDSS). The aim of this study was to evaluate the acaricidal activity of Lepidium latifolium and Zataria multiflora leaf extracts on V. destructor in field conditions. Four different concentrations (100, 200, 400, and 500 ppm) of the methanolic extracts were sprayed to treat each colony. The efficacy and side effects on adult bees were compared to Apistan chemical strips (ACSS). The acaricidal activity was the highest (100 %) for L. latifolium extract at 500 ppm after 12 days and 86.26 % for Z. multiflora. The infestation rate was decreased to 0.0 % with L. latifolium and to 13.74 % with Z. multiflora. The highest reduction was observed with L. latifolium followed by Z. multiflora extract at 500 ppm concentration. Both of the extracts showed negligible effect on bees, and it can be concluded that these PDSS as biodegradable agents could be used for V. destructor control in honeybee colonies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Varroa destructor (Anderson and Trueman 2000) (Acari: Varroidae) is the most serious pest of honeybee (Apis mellifera), causing considerable economic losses such as malformation, weight loss, and a shortened life span in the bee throughout the world. It also serves as a vector of diseases that may lead to 100 % bee mortality (Kanga and James 2002). There are several synthetic acaricides against V. destructor. The most effective agents are the organophosphate coumaphos (Checkmite®, Asuntol®, Perizin®), the pyrethroids tau-fluvalinate (Apistan®, Klartan®, Mavrik®) and flumethrin (Bayvarol®), and the formamidine amitraz (Milani and Barbattini 1988; Milani and Della Vedova 1996; Ritter 1988). Although these compounds are very effective in most situations, significant drawbacks including contamination of honey, wax, and pollen have been reported (Chauzat et al. 2009; Johnson et al. 2009; Lodesani et al. 2008; Martel et al. 2007; Wallner 1999). Furthermore, much evidence of drug resistance was recorded in many cases (Milani and Della Vedova 1996; Milani and Vedova 2002; Sammataro et al. 2005). Therefore, investigations on appropriate alternative compounds to develop new treatment strategies are necessary (Lodesani 2004). Recently, natural compounds such as plant-derived products (PDPs) were introduced as good candidates to safer control agents that may provide acaricidal activity and few drawbacks (Aydin et al. 2007; Batish et al. 2008; Cakmak et al. 2006; De Ruijter 1982; Eischen and Vergara 2004; Gregorc and Poklukar 2003; Satta et al. 2008; Shaddel-Telli et al. 2008). The acaricidal activities of some PDPs or essential oil-based products against honeybee mite were investigated (Calderone et al. 1997; Semmler et al. 2009). Lepidium latifolium is known as perennial pepper weed and belongs to family Brassicaceae. This plant is native to southern Europe and Asia (Francis and Warwick 2007; Hultén and Fries 1986). The members of genus Zataria are widely distributed in Iran, Afghanistan, and Pakistan (Mahmoudabadi et al. 2007; Shaiq Ali et al. 2000), and Z. multiflora is one the most common herbal medicines in Iranian traditional medicine (Khalili and Vahidi 2006). Several studies have been performed on different medicinal properties of Z. multiflora, and its antimicrobial, antitoxigenic, insecticidal, and scolicidal activities has been confirmed (Lindberg et al. 2000; Moazeni and Roozitalab 2012; Najari et al. 2014). Several studies have been done on acaricidal potential of PDPs (Calderone et al. 1997; Flamini 2003 ; Floris et al. 2004; Mattila et al. 2000), such as compounds from L. Latifolium and Z. multiflora (Mohagheghzadeh et al. 2000; Navaei and Mirza 2007). The objective of the present study was the field assessment of L. latifolium and Z. multiflora methanolic extracts against V. destructor in A. mellifera colonies.
Materials and methods
The study area
The present cross-sectional study was carried out on A. mellifera colonies in Shahrekord (32° 19′ 32″ N, 50° 51′ 52″ E), capital city of Chaharmahal and Bakhtiari Province, Iran, in summer 2014. Its weather is dry, cold in winter, and mild in summer. The annual average temperature in Shahrekord is about 5.11 °C, but the minimum and maximum absolute temperatures during the last 30 years have been −32 °C and 42 °C, respectively (http://www.weatherzone.com.au/world/middle-east/iran/shahrekord).
Plant materials and extraction
Methanolic extracts of L. latifolium and Z. multiflora (Fig. 1) were prepared as follows:
L. latifolium (a) Z. multiflora (b). L. latifolium (perennial pepper weed) is an herbaceous perennial that can grow up to 1.5 m (5 ft.) tall. Plants emerge from thick, minimally branched roots or semi-woody crowns. In the late fall to early spring, a rosette of leaves develops with 4–12 in. (10–30 cm) long and 1–2 in. (2.5–5 cm) wide, toothed leaves. Lepidium latifolium is native to southeast Europe, North Africa, and southwest Asia. Z. multiflora is a genus of flowering plant in the Lamiaceae family. It is a shrub to a height of 40–80 cm, with twisted branches, native to southwestern Asia (Iran, Afghanistan, Pakistan, and Kashmir). Small leaves are almost rounded and in the surface are covered with a large number of follicles. Plant powder is silvery green with aromatic smell and astringent and spicy taste. Its harvest time is in June when it is blossoming
After removing and collecting the leaves of both plants, they were dried in the shade. After that, the dried leaves were ground into powder by electric blender, and then 100 g of each was dissolved in 400 ml of pure methanol separately. The solution was kept for 24 h at room temperature after mixing with a magnetic stirrer. The solvent was removed by evaporator system after filtering. Finally, the dry powder was obtained using lyophilization of the semisolid material. The obtained residue was placed in a sterile glass container and stored at 4 °C till use (Moazeni and Nazer 2010).
Insects
Thirty hybrid honeybee colonies, A. mellifera infested with V. destructor, were used in the present study. Each colony consisted of ten full-depth combs. The hives were placed in a farm apiary near Shahrekord, capital city of Chaharmahal and Bakhtiari Province, Iran
Mean infestation rate
The percentage of Varroa infestation in the tested colonies before and after treatments was determined in approximately 100 living worker bees taking directly from the combs using icing sugar (sugar shake method) (Mark and Cliff 2001). We calculated the infestation rate by Alloui et al. (2002) formula (Alloui et al. 2002).
Field application and experimental design
In this study, the acaricidal effect of L. latifolium and Z. multiflora methanolic extracts were tested and compared with Apistan chemical strip (ACS) against V. destructor. Four concentrations (100, 200, 400, and 500 ppm) of the methanolic extracts were used, and each concentration was evaluated in three colonies (El Zalabani et al. 2012). Treatments were performed by means of 500-ml plastic sprayer. To treat each colony, different dilutions of plant extracts (100 ml each) were sprinkled over the bees inside the colonies (three colonies for each concentration). Every comb in the colony were exposed to test sample by rising up separately and sprinkled over the bees which covered the comb. ACS by slow release polymer strip formulation was applied in form of polymer beehive strips. Each 8 g (±0.8 g) strip containing 824 mg tau-fluvalinate, which was hanged between the middle combs as positive control. Three colonies were used as a control. Technical floors were used in the hives to record natural mite mortality. The mites which fallen from the adult bees and showed no movements were considered dead. The mean of mortality of mites and bees following treatment by different concentrations were recorded 4, 8, and 12 days after treatment. Three colonies were treated by distilled water using the same method and were regarded as negative control (El Zalabani et al. 2012).
Statistical analysis
Statistical analysis was performed using SPSS software version 21. One-way ANOVA and Duncan tests were used for comparison of measured parameters among control and treatment groups. Kruskal-Wallis, a nonparametric test, was used to compare the mortality rates of bees and mites among different groups. All values in tables were presented as mean and standard error of mean (SEM), and P < 0.05 was considered as statistically significant.
Results
To evaluate the acaricidal effect of L. latifolium and Z. multiflora methanolic extracts, the different dilutions of two plant extracts were applied to all 30 colonies and the effect of each dilution was evaluated in three colonies. Treatments were carried out at 12-day intervals and repeated three times for each extract. The mortality rate of Varroa and honeybees was calculated 4, 8, and 12 days after treatment. The colonies treated by ACS and distilled water were regarded as positive and negative controls, respectively.
The preliminary phytochemical screening of both plants showed a similarity in response. Our results showed that both extracts were effective against Varroa in a dose-dependent manner and had a negligible effect on bees. The extract of L. latifolium was the most effective, since its 500 ppm concentration caused 100 % mortality in mites after 12 days compared to Z. multiflora and ACS-treated colonies during the same period. The Varroa mortality rates after treatment with different extracts are presented in Table 1. The data in Table 1 showed that the high concentration of L. latifolium and Z. multiflora extracts had significant acaricidal effects on Varroa with the lowest mortality rate on bee colonies. The results of field application of two extracts are shown in Table 2 and their respective effects on honeybees in Table 3.
The data in Table 2 showed that the mean infestation rate of colonies before treatment with 500 ppm of L. latifolium (18.66 ± 0.72) was decreased to zero after 12 days with minimal effect on bees compared to the other groups (see Table 3). This finding means that compared to ACS and Z. multiflora, L. latifolium extract had more and safer therapeutic effects.
The number of mite drop is depicted in Fig. 2. The highest mite drops occurred 12 days after treatment by 500 ppm of L. latifolium which was significantly higher than the other groups.
The number of mite drop after treatment with different concentrations of L. latifolium and Z. multiflora extracts compared to controls. L. latifolium: 100, 200, 400, and 500 ppm were demonstrated by A1, A2, A3, and A4, respectively. Z. multiflora: 100, 200, 400, and 500 ppm were demonstrated by B1, B2, B3, and B4, respectively
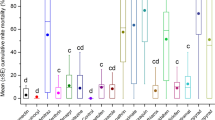
The effects of two plant extracts and ACS on honeybees are shown in Fig. 3. L. latifolium had the least mortality effect on bees compared with Z. multiflora and ACS. Compared to control, the mortality effect of L. latifolium was significantly decreased by increasing concentration and time.
The number of dead bees after treatment with different concentrations of L. latifolium and Z. multiflora extracts compared to controls. L. latifolium: 100, 200, 400, and 500 ppm were demonstrated by A1, A2, A3, and A4, respectively. Z. multiflora: 100, 200, 400, and 500 ppm were demonstrated by B1, B2, B3, and B4, respectively
Discussion
The application of PDPs is a new alternative method for the control of arthropod infestation, especially against Varroa mite in bee colonies (Calderone et al. 1997; El Zalabani et al. 2012). Natural products such as essential oils and methanolic extracts offer a highly desirable alternative to synthetic products. These substances are commonly used because they are generally inexpensive and safe to both man and honeybees (Eischen and Vergara 2004; El Zalabani et al. 2012; Gregorc and Poklukar 2003; Shaddel-Telli et al. 2008). Recently, some extracts have been tested for the control of different honeybee pathogens (Damiani et al. 2009; El Zalabani et al. 2012). Thyme, mint, lemon juice, camphor, eucalyptus essential oils, and tobacco smoke were demonstrated to be effective against Varroa with no or low harmful effects on bees (Abdel-Rahman 2008; Calderone 1999; Calderone et al. 1997; Damiani et al. 2009; De Ruijter 1982).
The control of parasites and pathogens in A. mellifera colonies can be performed more effectively by integrated pest management (IPM). According to the IPM strategies, pest control drugs should be environmentally friendly. In all cases, controls of Varroa mite by natural PDPs are recommended more than chemical acaricides (Dimetry et al. 2005; Flamini 2003; Ruffinengo et al. 2015), not only to keep the social life of honeybee away from any harmful effect but also for food safety and economic issues.
Nowadays, many researchers tend to return to investigations involving plant extracts for safe control of parasites and pest (Damiani et al. 2009; Semmler et al. 2009). Numerous investigations about the biological activity of PDPs against V. destructor and other pathogens in bee colonies have previously been performed (Damiani et al. 2011; George et al. 2008; Porrini et al. 2011; Umpiérrez et al. 2013).
In the present study, the acaricidal activities of botanical extracts obtained from L. latifolium and Z. multiflora were evaluated on V. destructor. Furthermore, the lethal effects of such extracts on bees were also examined.
Based on our results, compared to ACS which is used frequently as commercially available products, L. latifolium and also Z. multiflora with fewer side effects were more effective to treat infested colonies and in effective levels had no significant mortality on treated bees. Compared to ACS, L. latifolium extract was more effective, since its 500 ppm concentration caused 100 % acaricidal effect after 12 days. In the present study, Z. multiflora showed less acaricidal activity than L. latifolium, but more than ACS.
Several studies have been performed on different medicinal properties of Z. multiflora and its antimicrobial, antitoxigenic, insecticidal, and scolicidal activities have been confirmed (Lindberg et al. 2000; Moazeni and Roozitalab 2012; Najari et al. 2014). Our findings showed that the infestation rate was decreased by increasing concentration of both extracts, and this was in agreement with other studies (Abdel-Rahman 2008; El Zalabani et al. 2012). In addition, the results (Table 3) showed that both extracts appeared to be nontoxic for honeybees.
Conclusion
Our study showed that both L. latifolium and Z. multiflora at high concentrations are nontoxic for bee colonies and can decrease the overall population of Varroa mites, significantly. However, L. latifolium was more effective and its application can be considered as a simple, effective, safe, and cheap treatment for controlling Varroa in bee colonies.
References
Abdel-Rahman M (2008) Evaluation of lemon juice for controlling Varroa destructor in honeybee colonies. Ass J Agri Sci 39(2):195–206
Alloui N, Boucherit MR, Nouicer F (2002) Effect of flumethrine on Varroa destructor in honeybee colonies. Bull Vet Inst Pulawy 46(2):233–238
Anderson D, Trueman J (2000) Varroa jacobsoni (Acari: Varroidae) is more than one species. Exp Appl Acarol 24(3):165–189
Aydin L, Gulegen E, Cakmak I, Girisgin AO (2007) The occurrence of Varroa destructor on Honey Bees (Apis mellifera) in Turkey. Turk J Vet Anim Sci 31:189–191
Batish DR, Singh HP, Kohli RK, Kaur S (2008) Eucalyptus essential oil as a natural pesticide. Forest Ecol Manag 256(12):2166–2174
Cakmak I, Aydin L, Wells H (2006) Walnut leaf smoke versus mint leaves in conjunction with pollen traps for control of Varroa destructor. Bull Vet Inst Pulawy 50:477–479
Calderone NW (1999) Use of essential oils for the control of Varroa jacobsoni Oud. in honey bee colonies. Apidologie 30:209–228
Calderone NW, Wilson WT, Spivak M (1997) Plant extracts used for control of the parasitic mites Varroa jacobsoni (Acari: Varroidae) and Acarapis woodi (Acari: Tarsonemidae) in colonies of Apis mellifera (Hymenoptera: Apidae). J Econ Entomol 90(5):1080–1086
Chauzat MP et al (2009) Influence of pesticide residues on honey bee (Hymenoptera: Apidae) colony health in France. Environ Entomol 38(3):514–523
Damiani N, Gende LB, Bailac P, Marcangeli JA, Eguaras MJ (2009) Acaricidal and insecticidal activity of essential oils on Varroa destructor (Acari: Varroidae) and Apis mellifera (Hymenoptera: Apidae). Parasitol Res 106(1):145–152
Damiani N, Gende LB, Maggi MD, Palacios S, Marcangeli JA, Eguaras MJ (2011) Repellent and acaricidal effects of botanical extracts on Varroa destructor. Parasitol Res 108(1):79–86
De Ruijter A (1982) Tobacco smoke can kill Varroa mites. Bee World 63:138
Dimetry NZ, El-Wahab TA, Zakaria ME (2005) Effective control of varroa mite Varroa destructor infesting honey bee colonies Apis mellifera by some natural products. Bull Facul Agriculture, Cairo University 56:295–308
Eischen FA, Vergara CH (2004) Natural products smoke and its effect on Acarapis woodi and honey bees. Apidologie 35:341–350
El Zalabani SM, El-Askary HI, Mousa OM, Issa MY, Zaitoun AA, Abdel-Sattar E (2012) Acaricidal activity of Swietenia mahogani and Swietenia macrophylla ethanolic extracts against Varroa destructor in honeybee colonies. Exp Parasitol 130(2):166–170
Flamini G (2003) Acaricides of natural origin, personal experiences and review of literature (1990–2001). Stud Natur Prod Chem 28:381–451
Floris I, Satta A, Cabras P, Garau VL, Angioni A (2004) Comparison between two thymol formulations in the control of Varroa destructor: effectiveness, persistence, and residues. J Econ Entomol 97(2):187–191
Francis A, Warwick SI (2007) The biology of invasive alien plants in Canada. 8. Lepidium latifolium L. Can J Plant Sci 87(3):639–658
George DR et al (2008) Use of Plant derived Products to Control Arthropods of Veterinary Importance: A Review. Ann NY Acad Sci 1149(1):23–26
Gregorc A, Poklukar J (2003) Rotenone and oxalic acid as alternative acaricidal treatments for Varroa destructor in honeybee colonies. Vet Parasitol 111(4):351–360
Hultén E, Fries M (1986) Atlas of North European vascular plants, part I-III, maps and commentaries. Koeltz Scientific Books, Königstein, Germany, p 1172
Johnson RM, Pollock HS, Berenbaum MR (2009) Synergistic interactions between in-hive miticides in Apis mellifera. J Econ Entomol 102(2):474–479
Kanga LH, James RR (2002) Varroa control with fungal pathogens may be and option soon. Am Bee J 142(7):519–519
Khalili M, Vahidi A (2006) The anti-microbial effect of Zataia multiflora drops on three bacterial species cause gastrointestinal disease. World J Med Sci 1(2):162–163
Lindberg CM, Melathopoulos AP, Winston ML (2000) Laboratory evaluation of miticides to control Varroa jacobsoni (Acari: Varroidae), a honey bee (Hymenoptera: Apidae) parasite. J Econ Entomol 93(2):189–198
Lodesani M (2004) Control strategies against Varroa mites. Parassitologia 6(1–2):277–279
Lodesani M, Costa C, Serra G, Colombo R, Sabatini AG (2008) Acaricide residues in beeswax after conversion to organic beekeeping methods. Apidologie 39(3):324–333
Mahmoudabadi AZ, Dabbagh MA, Fouladi Z (2007) In vitro anti-Candida activity of Zataria multiflora Boiss. Evidence-Based Complementary Alternative Med 4(3):351–353
Mark G, Cliff V (2001) Control of varroa (A guide for New Zealand beekeepers). N Z Ministry Agriculture and Forestry: 29–38
Martel A-C, Zeggane S, Aurières C, Drajnudel P, Faucon J-P, Aubert M (2007) Acaricide residues in honey and wax after treatment of honey bee colonies with Apivar® or Asuntol® 50. Apidologie 38(6):534–544
Mattila HR, Otis GW, Daley J, Schulz T (2000) Trials of apiguard, a thymolbased miticide part 2. Non-target effects on honey bees. Am Bee J 140(1):68–70
Milani N, Barbattini R (1988) Effectiveness of Apistan (fluvalinate) in the control of Varroa jacobsoni Oudemans and its tolerance by Apis mellifera Linnaeus. Apicoltura 4:39–58
Milani N, Della Vedova G (1996) Determination of the LC50 in the mite Varroa jacobsoni of the active substances in Perizin® and Cekafix®. Apidologie 27:175–184
Milani N, Vedova GD (2002) Decline in the proportion of mites resistant to fluvalinate in a population of Varroa destructor not treated with pyrethroids. Apidologie 33(4):417–422
Moazeni M, Nazer A (2010) In vitro effectiveness of garlic (Allium sativum) extract on scolices of hydatid cyst. World J Surg 34(11):2677–2681
Moazeni M, Roozitalab A (2012) High scolicidal effect of Zataria multiflora on protoccoleces of hydatid cyst: an in vitro study. Comp Clin Pathol 21(1):99–104
Mohagheghzadeh A, Shams-Ardakani MR, Ghannadi A (2000) Volatile constituents of callus and flower‐bearing tops of Zataria multiflora Boiss. (Lamiaceae). Flavour Frag J 15(6):373–376
Najari R, Alizadeh A, Samani RB (2014) Antimicrobial activity and essential oil composition of Zataria multiflora Boiss in two regions of Iran. Int J of Biosci (IJB) 4(6):147–152
Navaei MN, Mirza M (2007) Chemical composition of the essential oils from the aerial parts, roots and seed of Lepidium latifolium L. from Iran. A review of Forest, Wood Products and Wood Biotechnology of Iran and Germany., p 127
Porrini MP, Fernández NJ, Garrido PM, Gende LB, Medici SK, Eguaras MJ (2011) In vivo evaluation of antiparasitic activity of plant extracts on Nosema ceranae (Microsporidia). Apidologie 42(6):700–707
Ritter W (1988) Medications registered in Western Europe for Varroatosis control. Apidologie 19(2):113–116
Ruffinengo S, Maggi MD, Marcangeli JA, Eguaras MJ, Principal J, Barrios C, Piano FD, Giullia M (2015) Integrated Pest Management to control Varroa destructor and its implications to Apis mellifera colonies. Zootec Trop 32(2)
Sammataro D, Untalan P, Guerrero F, Finley J (2005) The resistance of varroa mites (Acari: Varroidae) to acaricides and the presence of esterase. Int J Acarol 31(1):67–74
Satta A, Floris I, Caboni P, Cabras P, Eguaras M, Velis G (2008) New experimental data on use of rotenone as an acaricide for control of Varroa destructor in honey bee colonies. J Econ Entomol 101(4):1075–1080
Semmler M, Abdel-Ghaffar F, Al-Rasheid K, Mehlhorn H (2009) Nature helps: from research to products against blood-sucking arthropods. Parasitol Res 105(6):1483–1487
Shaddel-Telli A-A, Maheri-Sis N, Aghajanzadeh-Golshani A, Asadi-Dizaji A, Cheragi H, Mousavi M (2008) Using medicinal plants for controlling Varroa mite in honey bee colonies. J Anim Vet Adv 7(3):328–330
Shaiq Ali M, Saleem M, Ali Z, Ahmad VU (2000) Chemistry of Zataria multiflora (Lamiaceae). Phytochemistry 55(8):933–936
Umpiérrez ML, Santos E, Mendoza Y, Altesor P, Rossini C (2013) Essential oil from Eupatorium buniifolium leaves as potential varroacide. Parasitol Res 112(10):3389–3400
Wallner K (1999) Varroacides and their residues in bee products. Apidologie 30:235–248
Acknowledgments
The authors acknowledge Dr. M. Riyahi for his kind assistance and Mr. A. Motabi Alavi for technical helping during this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Razavi, S.M., Asadpour, M., Jafari, A. et al. The field efficacy of Lepidium latifolium and Zataria multiflora methanolic extracts against Varroa destructor . Parasitol Res 114, 4233–4238 (2015). https://doi.org/10.1007/s00436-015-4661-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4661-2