Abstract
The dynamics of the passage of conidia, chlamydospores, and mycelia of the fungus Duddingtonia flagrans through the digestive tracts of goats was evaluated. Four groups with five goats each were formed. In the group conidia, each animal received 1 × 106 D. flagrans conidia per kilogram of live weight. In the group chlamydospore, each animal received 1 × 106 chlamydospores per kilogram of live weight. In the group mycelia, each animal received 1 g of mycelium mass per kilogram of live weight. In the control group, the animals received no fungal structure. Feces were obtained 3 h before and 12, 24, 30, 36, 42, 48, 60, 72, 84, and 96 h after the inoculation. The feces were placed in Petri dishes containing water-agar. The Petri dishes were examined to detect the fungus and trapped nematodes. A second trial evaluated the effect of the fungal structures on the number of gastrointestinal larvae of Haemonchus contortus and Strongyloides papillosus harvested from the fecal cultures of the goats. The feces were obtained from the goats in the 12–24, 24–30, 30–36, 42–48, 60–72, 72–84, and 84–96 intervals after the inoculation. D. flagrans survived the digestive process of the goats and maintained its predatory activity, being observed from 12 to 96 h before inoculation in the animals that received chlamydospores and conidia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastrointestinal parasitism with nematodes is one of the main limiting factors in raising small ruminants. Haemonchus contortus is considered the most important gastrointestinal nematode parasite in small ruminants, causing large economic losses worldwide. The damages caused by this parasite are associated to their hematophagous habit, which leads to severe anemia a few days after infection, loss of weight and appetite, weakness, and death in some cases (Urquhart et al. 1996).
Strongyloides papillosus is an important gastrointestinal parasite in ruminants, mainly in tropical and subtropical regions of the world. This nematode can develop both parasitic and free-living reproductive life cycles. Females are responsible for the embryonic egg production that occurs by parthenogenesis during the parasitic phase (Chandrawathani et al. 1998; Urquhart et al. 1996). In some cases, the infection by S. papillosus may lead to diarrhea, loss of appetite, growth retardation, and sudden death (Taira et al. 1992; Pienaar et al. 1999).
Predatory nematophagous fungi have been shown as a viable alternative to the control of the pre-parasitic stage of several species of gastrointestinal nematodes in ruminants (Larsen 1999).
Several studies have demonstrated the viability of Duddingtonia flagrans for this use. This species can produce not only mycelia and conidia but also thick-walled chlamydospores that have been shown to survive passage through the gastrointestinal tract of ruminant and subsequently germinate in the voided feces to attack the developing larvae (Larsen 2000).
Although there is an agreement that the production of chlamydospores is the ideal form of administration of the fungi as a biological control, there are drawbacks to the development of efficient methodologies for mass production. It is also necessary to determine if the chlamydospore is the only stage responsible for the high fungal viability (Araújo et al. 2004a).
The objective of this work was to evaluate the predatory effect of D. flagrans (CG-722) isolates on H. contortus and S. papillosus L3, after the passage of mycelia, conidia, and chlamydospores of this species through the gastrointestinal tract of goats in confinement.
Materials and methods
The isolate of D. flagrans (CG-722) was supplied by CENARGEM, kept at the mycological collection of the Universidade Federal de Viçosa (Parasitology Laboratory, Veterinary Department) at 4°C in culture tubes containing corn meal agar (CMA, Difco) in the dark.
Chlamydospore production
The D. flagrans fungal isolates were kept in culture tubes containing 2% CMA culture medium, and culture disks of isolate, 4 mm in diameter, containing 2% CMA, were transferred with a sterile platinum loop to 50 × 10 mm Petri dishes containing 2% water-agar (2% WA, Sigma) culture medium. After mycelial growth of this new culture, a 5-mm-diameter culture disk was transferred to a 90 × 15-mm Petri dish containing a 2% WA.
These plates were incubated in a biochemical oxygen demand (BOD) chamber at 25°C in the dark for 5 days. Agar disks of 4 mm in diameter were scraped with a sterile platinum loop from the edges of the fungal colonies, free from contamination, and inoculated onto 30 Petri dishes containing 20 ml of yeast-phosphate-sulfate-sucrose-agar (YPSSA) culture medium (yeast extract, 4 g; K2HPO4, 1 g; MgSO4, 0,5 g; soluble starch, 20 g; agar, 20 g; water for 1 L solution). Next, the plates were incubated in BOD chamber at 25°C for 28 days in the dark. After this period, the plate surfaces were washed with 10 ml of sterile distilled water using a paintbrush. The suspension in the plates was sieved through a drum sieve coupled to a plastic container to eliminate mycelium fragments. The recovered spores were counted ten times in a Neubauer chamber and classified (conidia or chlamydospores) in three 10-μl aliquots. After counting and characterizing the spores, the dilution was prepared to the desired concentration and administered to each animal.
Conidial production
CMA disks (4 mm in diameter) containing D. flagrans were inoculated on 30 Petri dishes (9-cm diameter) with YPSSA medium. The plates were placed in a BOD chamber at 25°C for 7 days. Following incubation, the plate surfaces were washed with 10 ml of water and a paintbrush. Spore counting and characterization were carried out as described above, followed by dilution to the desired concentration and administration to each animal.
Mycelial mass production
Five 4-mm-diameter WA disks containing D. flagrans mycelia and spores were inoculated in 250 ml Erlenmeyer flasks containing 150 ml of glucose-peptone-yeast medium (15 g glucose, 2 g peptone, and 5 g yeast extract). The flasks were incubated for 10 days in a BOD chamber at 25°C in the dark on a rotary shaker (120 rpm). Following incubation, the mycelial mass was harvested by filtration on filter paper, separated from the culture medium, and washed three times with sterile distilled water. The excess moisture was removed by hand pressing between filter paper three times, and the mycelial mass was weighed in a precision scale. After weighing, five samples were collected from the total mycelial mass, mounted on a glass slide overlayed with a cover glass, and examined at ×100 and ×400 magnifications to eliminate the possibility of formation of conidia and chlamydospores in the liquid medium.
Nematodes
Infective larvae (L3) of H. contortus and S. papillosus were recovered by coproculture of 20 g feces from goats presenting monospecific infection and hospitalized in the clinic of the Veterinary Department of the Universidade Federal de Viçosa.
The larvae were recovered from the coprocultures by the Baermann method, washed five times by centrifugation (1,500 rpm) for 5 min, and the supernatant discarded. The L3 were filtered in Kimwipes (Kimberly-Clark) antistatic film according to the methodology described by Barçante et al. (2003) to eliminate residues and bacteria and kept in 0.05% (w/v) streptomycin sulfate and 0.05% (p/v) chloramphenicol solution for 1 h. Then, the previous washing procedure was repeated.
Animals
Twenty-eight-month-old females of Saanem goats infected with H. contortus and S. papillosus were used in the experiment. The animals were purchased from the goat-breeding sector of the Animal Science Department of the Federal University of Viçosa. The animals were kept in concrete floor stalls at the Veterinary Department of the Federal University of Viçosa. During the experimental period, the goats were fed on chopped elephant grass (Pennisetum purpureum), Tifton hay (Cynodon dactylon), and water ad libitum.
Experimental procedure
In vitro predatory activity of D. flagrans on infective larvae of H. contortus and S. papillosus
Predation tests were carried out in Petri dishes (6 cm in diameter) containing 2% WA. Four groups were formed with five repetitions each:
-
Group 1: fungal colony grown for 7 days at 25°C and approximately 103 L3 of H. contortus.
-
Group 2: 103 L3 of H. contortus.
-
Group 3: fungal colony grown for 7 days at 25°C and approximately 103 L3 of S. papillosus.
-
Group 4: 103 L3 of S. papillosus.
The plates were incubated in a BOD chamber for 7 days at 25°C in the dark. Over the incubation time, the plates were removed from the chamber daily so that trap formation and capture of L3 by fungi could be observed. After 7 days, the agar was removed from the plates with a metal spatula, and predation-free L3 were recovered in hemolysis tubes by the Baermann method for 12 h. The content in the hemolysis tubes was transferred to centrifuge tubes and were centrifuged for 10 min at 1,500 rpm. After centrifugation, the volume was adjusted to 1 ml with a Pasteur pipette attached to a vacuum pump. The L3 number estimate was calculated as the average of five 50-μl aliquots and extrapolated to 1 ml in the tubes.
The fungus predation rate for each nematode species was calculated using the following formula: [(mean number of larvae recovered in the control group − mean number of larvae recovered in the treated group)/mean number of larvae recovered in the control group].
Predatory ability on larvae of H. contortus and S. papillosus in goat feces following the oral administration of conidia, chlamydospores, and mycelia
The animals were randomly assigned to four groups on the basis of eggs per gram feces (EPG), with five animals each:
-
Conidia: Each animal received 120 ml of oral suspension containing approximately 1 × 106 spores (95.28% conidia and 4.72% chlamydospores) of D. flagrans.
-
Chlamydospores: Each animal received 120 ml of oral suspension containing approximately 1 × 106 spores (93.37% chlamydospores and 6.63% conidia) of D. flagrans per kilogram of live weight.
-
Mycelia: Each animal received 120 ml of oral suspension containing 1 g of fresh D. flagrans mycelial mass per kilogram of live weight.
-
Control: Each animal received 120 ml of sterile distilled water.
Fecal collections were carried out in cotton bags, coated internally with plastic tape to facilitate cleaning between collections, in intervals of 12–24, 24–30, 30–36, 42–48, 60–72, 72–84, and 84–96 h after administration of the fungal material. The collected feces were homogenized, and 20 g samples from each animal of the four experimental groups were removed and mixed to 10 g of vermiculite and 25 ml of water, for coproculture, in 200 ml polyvinyl chloride (PVC) glasses incubated in a BOD chamber at 26°C. Coprocultures were visually examined over the incubation time every 3 days. After 18 days of incubation and starting from the time of sample collection, the L3 were recovered in hemolysis tubes by the Baermann method with water at 42°C for 12 h. The predation-free larvae were counted using an optical microscope (×100) in five 100-μl aliquots taken from the initial sample and then extrapolated to the total volume in the hemolysis tubes.
For EPG determination, the feces were collected from the animals’ rectum three times a day at 08:00, 14:00, and 18:00 hours during the experimental period. The EPG counting was performed following each fecal collection (Gordon and Whitlock 1939).
The percentage of larval development was determined for each group and each collection interval using the formula (mean lpg/mean epg) × 100 described by Paraud et al. (2004).
The percentage of reduction in the larval development in the groups treated with conidia, chlamydospores, and mycelia was calculated with the formula: [(mean lpg of the control group/mean epg of the control group) − (mean lpg of the fungus group/mean epg of the fungus group)]/(mean lpg of the control group/mean epg of the control group) × 100.
Dynamics of conidium, chlamydospore, and mycelium elimination in the goat feces after passing through the gastrointestinal tract of goats
To eliminate the possibility of fungal presence in the animal’s feces before the experiment and to evaluate the fungal presence after treatment, feces samples were collected in plastic bags directly from the animal’s rectum 3 h before and 12, 24, 30, 36, 42, 48, 60, 72, 84, and 96 h after the administration of fungal structures. Two grams of these samples were crushed with a glass rod and inoculated on 9-cm Petri dishes with 2% WA. Three thousand H. contortus larvae were dropped onto the agar to stimulate fungal growth and trap development. The plates were sealed with Parafilm (Sigma®) and incubated at 25°C in the dark. The plates were examined daily for 30 days using a stereoscopic microscope to visualize traps, conidia, and chlamydospores typical of the isolate and nematodes trapped on plate surface.
Statistical analysis
The experiment was arranged in a split plot design, with the treatments (chlamydospore, conidia, mycelia, and control) as the main plots and the collection intervals (12–24, 24–30, 30–36, 36–48,48–60, 60–72, 72–84, and 84–96 h) as the sub-plots in a complete randomized design with 20 repetitions (five animals per treatment). The data of the percentage of larval development were transformed (Arcsin), examined by analysis of variance, and the means were compared using the Tukey test at 5% probability level using the SAEG statistical package.
Results and discussion
Conidia, chlamydospores, and mycelia production
Chlamydospores (93.37%) and conidia (6.63%) were recovered from the plates used for the production of chlamydospores. In the plates used for the production of conidia, 95.28% of the collected spores were conidia, and 4.72% were chlamydospores. Mycelium fragments were observed in suspensions of conidia and chlamydospores, even after successive passages through the sieve.
In vitro predatory activity of D. flagrans on H. contortus and S. papillosus infective larvae
The structures used for predation were three-dimensional adhesive nets, which started to be produced 10 h after the addition of nematodes to the plates. The first trapped nematodes were observed 12 h after plate incubation.
Table 1 shows the mean number of H. contortus and S. papillosus infective larvae recovered after 7 days in plates with D. flagrans (CG-722) and in control plates.
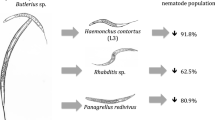
D. flagrans showed higher predatory activity on S. papillosus. Experiments with other D. flagrans isolates had already demonstrated the high predatory effectiveness of this species on H. contortus L3 (Mendoza-de-Gives et al. 1998; Chandrawathani et al. 2002).
No reports have been found in the literature concerning the use of D. flagrans against S. papillosus. The predation rate observed in the present work is higher than the rate reported by Gonzalez-Cruz et al. (1998), who found that the percentage of predation of S. papillosus L3 by Monacrosporium gehyropagum was 93.1% and by Arthrobotrys robusta was 32.3%.
Studies have demonstrated that variations in fungal predatory activity on nematodes are common (Mendoza-de-Gives et al. 1994; Larsen 2000), independently of the fungus or nematode species used in the experiments. These variations can be related to different factors such as the experimental design, losses of viability during storage in laboratory, and genetic factors inherent to the assessed fungal strains as reported by (Gronvold et al. 1996).
In vitro experiments do not expose the fungi to the adverse conditions imposed by the gastrointestinal tract of the animal and environmental biotic and abiotic factors such as temperature variation, humidity, pH, and competition with other organisms of the coprophagic fauna and flora. Therefore, the results obtained in this kind of test cannot be extrapolated for in vivo experiments. On the other hand, we can confirm that the fungi-nematode interaction is occurring, which is important for the initial stages of the microorganism selection for biological control programs.
Considering that the production of chlamydospores is desirable in fungi used in biological control programs, it is important to select isolates with higher chlamydospore production associated with high predatory capacity.
Dynamics of conidium, chlamydospore, and mycelium elimination after passing through the gastrointestinal tract of goats
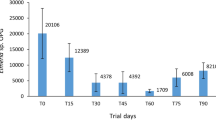
D. flagrans was not found in any of the fecal samples collected before the administration of the fungal structures or in the plates from the control group during the entire experimental period (Table 1). Chlamydospores, conidia, and trapped larvae were seen on the surface of the plates inoculated with feces collected from the animals of the groups treated with chlamydospores, conidia, and mycelia. However, there were variations among treatments and among times (Table 1). The fungus was detected in 84.44% of the plates inoculated with feces from the animals treated with chlamydospores, in 68.88% of the plates from the group treated with conidia, and in 40.0% of the plates from the group treated with mycelia.
The results obtained for the animals treated with chlamydospores confirm the passage of chlamydospores of the CG-722 isolate and confirm the findings of Larsen et al. (1998), who isolated fungi from sheep feces 12 h after oral administration of D. flagrans chlamydospores.
This is the first report of the dynamics of D. flagrans conidia and mycelia through the gastrointestinal tract of goats.
Comparing the results of this work with other studies, we found that the time for fungus elimination after the oral administration of conidia and chlamydospores can vary significantly. Previous studies reported activity of D. flagrans from 14–116 h in sheep and goat feces (Llerandi-Juárez and Mendoza-de-Gives 1998; Ojeda-Robertos et al. 2005).
Other factors related to the diet used during the experiments may have influenced the retention time of the digest in the gastrointestinal tract of the animals. This may also be influenced by the content and quality of fiber, food particle size, amount of ingested water, and occasional variations in the digestive physiology.
Studying the elimination of fungal material in animals treated with mycelia, conidia, and chlamydospores, we found that in up to 36 h post-administration of fungal structures, the results were similar. Starting from this time, there was a clear reduction in fungus recovery in the animals treated with mycelia (Table 2).
During the transit through the gastrointestinal tract of ruminants, the ingesta are subjected to mechanical, microbial, and chemical actions (Ribeiro 1997) that are adverse to fungus survival. Thus, the longer the retention time in the digestive tract, the greater is the possibility of changes in the fungal structures. The structural composition of conidia and chlamydospores possibly allowed them to remain intact, thus facilitating their isolation until the last collection. On the other hand, mycelium fragments may have been damaged and turned unviable.
Predatory ability on larvae of H. contortus and S. papillosus in goat feces following the administration of conidia, chlamydospores, and mycelia
The percentage means of larval development of H. contortus and S. papillosus in coproculture are shown in Tables 3 and 4.
On the average of the collection intervals, the percentage of larval development of H. contortus for the groups control, conidia, chlamydospores, and mycelia were 3.486%, 2.653%, 1.327%, and 2.306%, respectively; and the percentage of reduction in larval development for the conidia, chlamydospores, and mycelia groups were 23.89%, 61.23%, and 33.84%, respectively.
The percentage of larval development obtained for H. contortus is similar to the percentage found by Ojeda-Robertos et al. (2005; 0.9–11.1%) and lower than the results of Terril et al. (2004; 3.9–100%) in goat coprocultures.
The mean percentage of larval development of S. papillosus at the end of the experiment for the control, conidia, chlamydospores, and mycelia groups were 15.95%, 13.417%, 4.97%, and 12.82%, respectively; and the percentage of reduction in larval development for the groups treated with conidia, chlamydospores, and mycelia were 15.88%, 68.84%, and 19.62%, respectively.
The results of the percentage of larval development for H. contortus and S. papillosus show that the treatment with chlamydospores was the most effective in controlling the two species. Except for the first interval of collection of S. papillosus and the sixth interval for H. contortus, the reductions followed the dynamics of the chlamydospore elimination described in Table 2.
In the group of animals treated with conidia, the percentage of larval development for H. contortus was significantly lower (p < 0.05) than the percentage for the control group and similar to the groups treated with chlamydospores and mycelia (up to 36 h post-administration). However, the reductions were not correlated with the dynamics of conidium elimination, showing that damage may have occurred during the passage through the animal gastrointestinal tract, which did not harm the fungus germination capacity, but did harm its predatory activity. The results obtained in this group for S. papillosus corroborate this hypothesis because no significant differences (p < 0.05) were found among the percentage means of larval development when compared with the control group.
The mycelium treatment significantly reduced (p < 0.05) the percentage of larval development of H. contortus only in the first two collection intervals, but it was not possible to demonstrate its effect on S. papillosus larvae.
These results demonstrated that the administration of chlamydospores from the Brazilian isolate of D. flagrans CG-722 is effective in controlling H. contortus and S. papillosus larvae in goats.
Further studies are recommended to evaluate the effectiveness of this isolate in the field, in different environmental conditions, and after the passage through the gastrointestinal tract of another animal species.
The administration of conidia and mycelia to the animals resulted in a short period of efficacy. The use of formulations that can protect these structures from the adverse conditions in the gastrointestinal tract of animals is a possibility. Alves et al. (2003), Araújo et al. (2004b), and Assis et al. (2005) obtained significant reductions of infective larvae of gastrointestinal parasites in bovines after the administration of fungal mycelia of Monacrosporium isolates encapsulated in sodium alginate to bovines and goats.
Although chlamydospores are very resistant to the passage through the gastrointestinal tract of animals and can maintain their predatory activity, their production requires a longer period of time when compared with the production of mycelia and conidia.
The use of nematophagous fungi in the biological control of nematode parasites of ruminants will only be possible after the involvement of industries in the development of fungal formulations. It will be then necessary to study the cost/benefit relationship for the industrial-scale production of fungal structures and the development of methodologies to improve production.
References
Alves PH, Araújo JV, Guimarães MP, Assis RCL, Sarti P, Campos AK (2003) Aplicação de formulação do fungo predador de nematóides Monacrosporium thaumasium (Drechsler, 1937) no controle de nematóides de bovinos. Arq Bras Med Vet Zootec 55:568–573
Araújo JV, Guimarães MP, Campos AK, Sá NC, Sarti P, Assis RCL (2004a) Control of bovine gastrointestinal nematodes parasites using pellets of the nematode-trapping fungus Monacrosporium thaumasium. Cienc Rural 34:457–463
Araújo JV, Mota MA, Campos AK (2004b) Controle biológico de helmintos parasitos de animais por fungos nematófagos. Rev Bras Parasitol Vet 13(1):165–171
Assis RCL, Araújo JV, Gandra JR, Campos AK (2005) Avaliação de fungos predadores do gênero Monacrosporium sobre larvas infectantes de Haemonchus contortus de caprinos. Rev Bras Cienc Vet 12:42–45
Barçante JMP, Barçante TA, Dias SRC, Lima WS, Negrão-Corrêa DA (2003) A method to obtain axenic Angiostrongylus vasorum first-stage larvae from dog feces. Parasitol Res 89:89–93
Chandrawathani P, Jamnah O, Waller PJ (1998) The control of the free-living stages of Strongyloides papillosus by the nematophagous fungus, Arthrobotrys oligospora. Vet Parasitol 76:321–325
Chandrawathani P, Jamnah O, Waller PJ, Höglund J, Larsen M, Zahari WM (2002) Nematophagous fungi as a biological control agent for nematode parasites of small ruminants in Malaysia; a special emphasis on Duddingtonia flagrans. Vet Res 33:685–696
Gonzalez-Cruz ME, Mendoza-de-Gives P, Quiroz-Romero H (1998) Comparison of the ability of Arthrobotrys robusta and Monacrosporium gehyropagum on infective larvae of Strongyloides papillosus. J Helminthol 72:209–213
Gordon HM, Whitlock HV (1939) A new technique for counting nematode eggs in sheep faeces. J Counc Sci Ind Res 12:50–52
Gronvold J, Henriksen SA, Larsen M, Nansen P, Wolstrup J (1996) Aspects of biological control—with special reference to arthropods, protozoans and helminths of domesticated animals. Vet Parasitol 64:47–64
Larsen M (1999) Biological control of helminths. Int J Parasitol 29:139–146
Larsen M (2000) Prospects for controlling animal parasitic nematodes by predacious microfungi. Parasitology 120:121–131
Larsen M, Faedo M, Waller PJ (1998) The potential of nematophagous fungi to control the free living stages of nematode parasites of sheep: studies with Duddingtonia flagrans. Vet Parasitol 76:121–128
Llerandi-Juárez RD, Mendoza-de-Gives P (1998) Resistance of chlamydospores of nematophagous fungi to the digestive processes of sheep in Mexico. J Helminthol 72:155–158
Mendoza-de-Gives P, Zavaleta-Mejia E, Herrera-Rodrigues D, Quiroz-Romero H (1994) In vitro trapping capability of Arthrobotrys spp on infective larvae of Haemonchus contortus and Nacobus aberrans. J Helminthol 68:223–229
Mendoza-de-Gives P, Flores-Crespo J, Herrera-Rodrigues D, Vasquez-Prats V, Liebano Hernandez E, Ontiveros-Fernadez GE (1998) Biological control of Haemonchus contortus infective larvae in ovine faeces by administering an oral suspension of Duddingtonia flagrans chlamydospores to sheep. J Helminthol 72:343–347
Ojeda-Robertos NF, Mendoza-de-Gives P, Torres-Acosta JFJ, Rodríguez-Vivas RI, Aguillar-Caballero AJ (2005) Evaluating the effectiveness of a Mexican strain of Duddingtonia flagrans as a biological control agent against gastrointestinal nematodes in goat faeces. J Helminthol 79:151–157
Paraud C, Pors I, Chartier C (2004) Activity of Duddingtonia flagrans on Trichostrongylus colubriformis larvae in goat feces and interaction with a benzimidazole treatment. Small Rumin Res 55:199–209
Pienaar JG, Basson PA, Plessis JLD, Collins HM, Naude TW, Boyazoglu PA, Boomker J, Reyers F, Pienaar WL (1999) Experimental studies with Strongyloides papillosus in goats. Onderstepoort J Vet Res 66:191–235
Ribeiro SDA (1997) Caprinocultura: Criação racional de caprinos. Nobel, São Paulo
Taira N, Nakamura Y, Tsuji N, Kubo M, Ura S (1992) Sudden death of calves by experimental infection with Strongyloides papillosus. I. Parasitological observations. Vet Parasitol 42:247–256
Terril TH, Larsen M, Samples O, Husted S, Miller JE, Kaplan RM, Gelaye S (2004) Capability of the nematode-trapping fungus Duddingtonia flagrans to reduce infective larvae of gastrointestinal nematodes in goat faeces in the southeastern United States: dose titration and dose time interval studies. Vet Parasitol 120:285–296
Urquhart GM, Armour J, Duncan JL (1996) Veterinary parasitology. Blackwell Science, London, UK, p 307
Acknowledgments
The authors are grateful to CNPq and Fapemig for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Campos, A.K., Araújo, J.V., Guimarães, M.P. et al. Resistance of different fungal structures of Duddingtonia flagrans to the digestive process and predatory ability on larvae of Haemonchus contortus and Strongyloides papillosus in goat feces. Parasitol Res 105, 913–919 (2009). https://doi.org/10.1007/s00436-009-1476-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-009-1476-z