Abstract
Purpose
Circulating insulin-like growth factor-1 (IGF-1) is positively associated with the risk of BC recurrence, and is more frequently dysregulated in older people, especially in those with metabolic syndrome (MetS) and obesity. This study aimed to analyze the association between IGF-1 levels and indices of MetS and insulin resistance in BC survivors.
Methods
Baseline data of 563 BC survivors enrolled in the DIet and ANdrogen-5 (DIANA-5; NCT05019989) study were analyzed.
Results
Lower circulating IGF-1 levels in subjects with MetS than in those without MetS were found. After stratification of the patients according to the diagnosis of MetS, we highlighted that the insulin was the main predictor of elevated IGF-1 levels only in subjects without MetS. Moreover, we found an interaction between high-density lipoprotein cholesterol (HDL-C), glycemia, and IGF-1 levels, showing a positive correlation between HDL-C and IGF-1, especially in subjects with higher values of glycemia and without a diagnosis of MetS.
Conclusions
While IGF-1 levels appear to be much more impaired in subjects diagnosed with MetS, in non-MetS subjects, IGF-1 levels may respond better to metabolic parameters and lifestyle changes. Further studies are needed to analyze the role of physical activity and/or dietary intervention in modulating IGF-1 concentrations in BC survivors.
Implications for cancer survivors
These results could have important clinical implications for planning customized strategies aimed at modulating IGF-1 levels in BC survivors. In fact, while the IGF-1 system seems to be much more compromised in subjects with a diagnosis of MetS, in noMetS subjects, IGF-1 levels could better respond to lifestyle changes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epidemiological evidence shows that metabolic syndrome (MetS), fasting hyperinsulinemia, and high levels of circulating insulin-like growth factor-1 (IGF-1) are risk factors for the development and recurrence of breast cancer (BC) (Andò et al. 2019; Calori et al. 2011; Duggan et al. 2011; Endogenous Hormones and Breast Cancer Collaborative Group et al. 2010; Goodwin et al. 2002; Pasanisi et al. 2008).
Insulin and IGF-1 signaling systems are involved in energy metabolism and growth. There is substantial experimental and clinical evidence that cancer cells express insulin and IGF-1 receptors, which are important activators of the Akt and mitogen-activated protein kinase signaling networks in neoplastic tissues (Pollak 2008; De Santi et al. 2016).
MetS is characterized by the presence of at least three out of five dysmetabolic traits according to the consensus definition (incorporating the IDF and AHA/NHLBI definitions) (i.e., abdominal obesity, high blood pressure, low plasma high-density lipoprotein cholesterol (HDL-C), high plasma fasting glucose, and high triglycerides) (Alberti et al. 2009). In epidemiological studies, MetS and its individual components have been associated with BC risk, recurrence, and distant metastasis, after adjusting for stage and hormonal receptor expression (Agnoli et al. 2010; Berrino et al. 2014; Biganzoli et al. 2017; Esposito et al. 2012; Hwang et al. 2020).
Although the exact role of MetS components is largely uncertain, insulin resistance is considered a common mechanism underlying metabolic derangements associated with the syndrome (Cornier et al. 2008; Nelson and Bremer 2010), and it is the most important link between MetS and cancer according to low-grade inflammatory processes (Capasso et al. 2013; Devericks et al. 2022; Duggan et al. 2011; Goodwin et al. 2002). Chronically elevated insulin levels are associated with cell proliferation and survival and contribute to the migration, invasiveness, and metastasis of cancerous cells (Cevenini et al. 2018; Pollak 2008).
The IGF-1 system may also be involved in the effects of hyperinsulinemia on carcinogenic pathways. Indeed, owing to its structural homology to IGF-1, insulin may bind and activate IGF-1 receptor signaling (Doyle et al. 2012; Hankinson et al. 1998; Kaaks 2001). Furthermore, insulin may control IGF-1 production by altering the levels of IGF-1 binding proteins (Lukanova et al. 2001) and/or by hyperinsulinemia-induced promotion of hepatic growth hormone receptor expression and IGF-1 synthesis (Brauna et al. 2011). Accordingly, activated insulin and IGF-1 receptors were detected in all BC subtypes and linked to poor survival in patients with BC (Law et al. 2008).
In the last decade, several studies have revealed an inverse association between circulating IGF-1 levels and MetS in elderly subjects (Brugts et al. 2010) and in large population studies (Oh et al. 2012; Parekh et al. 2010). Moreover, it has been observed that the concentration of IGF-1 decreases with increasing MetS factors (Oh et al. 2012). However, the association between IGF-1 and MetS criteria in BC survivors is not well-known and raises the apparently contradictory finding of a higher risk of BC recurrence in subjects with MetS. Investigation of metabolic and clinical variables in relation to IGF-1 may improve our understanding of this complex onco-metabolic scenario.
Given the role of IGF-1 as a risk factor for BC recurrences, we aimed this study to investigate the association between insulin resistance and MetS risk factors with IGF-1 circulating levels in women with and without MetS (noMetS) from a cohort of BC survivors enrolled in the DIet and ANdrogen-5 (DIANA-5) study (Villarini et al. 2012).
Materials and methods
Subjects and methods
The present report concerns the baseline data of 563 women, aged 35–70 years, with early stage invasive BC within the previous 5 years (1.76 years on average), progression-free survivors, recurrence-free, enrolled in the DIANA-5 study, an ongoing multi-center randomized controlled trial aimed at testing the hypothesis that a lifestyle change based on the Mediterranean diet and macrobiotic principles, together with daily moderate intensity physical activity, can reduce the incidence of BC recurrences (ClinicalTrials.gov Identifier: NCT05019989). In the DIANA-5 study were enrolled women at high metabolic and hormonal risk of BC recurrences: ER-negative tumors or serum testosterone level ≥ 0.4 ng/mL (1.338 nmol/mL), and/or serum insulin ≥ 7 μU/mL (50 pmol/L) and/or MetS diagnosis (3/5 risk factors), without a diabetes diagnosis. At sampling time, all enrolled women were in natural or induced menopausal status. Before enrollment, all participants signed an informed consent form, including authorization, to obtain blood samples to study serum parameters. The trial design has been described in detail previously (Villarini et al. 2012).
This study was approved by the Institutional Review Board and Ethical Committee of the National Cancer Institute of Milan (No. 37/07).
Laboratory analysis
Plasma glucose, triglyceride, and HDL-C levels were measured by routine laboratory techniques. Insulin levels were measured using an immunoradiometric kit (Immunotech, Prague, Czech Republic), which showed intra- and interassay coefficients of variation of 2.2% and 5.1%, respectively, with a mean insulin value of 10 μU/mL.
Serum IGF-1 was measured using commercial radioimmunoassay kits (Biosource, Nivelles, Belgium). Samples were analyzed blinded; the technicians who analyzed the serum samples were unaware of the patients’ disease status. The analyses were organized into 16 analytical batches each consisting of 38 serum samples and 3 quality controls provided by the kit. The interbatch coefficients of variation were 9.29%, 9.61% and 5.19% for IGF I values of 75.16 ng/mL, 144.41 ng/mL and 422.84 ng/mL, respectively.
Definition of MetS
MetS was defined on the basis of the Consensus definition (incorporating IDF and AHA/NHLBI definitions) with the presence of at least three out of five components, according to the thresholds proposed by the International Diabetic Federation: systolic blood pressure > 130/85 mmHg, fasting plasma glucose ≥ 100 mg/dL (5.6 mmol/L), fasting plasma triglycerides ≥ 150 mg/dL (1.7 mmol/L), HDL-C < 50 mg/dL (1.03 mmol/L), waist circumference ≥ 80 cm (Alberti et al. 2009). As reported in the DIANA-5 study (Villarini et al. 2012), we considered MetS components as well as the presence of treatments for dyslipidemia, hypertension, and hyperglycemia.
Statistical analysis
Age, anthropometric values, clinical parameters, and therapies were evaluated in the entire group and separately in women with and without MetS. For each numerical variable, the distribution was checked for unimodal and symmetric shapes using graphical procedures (histograms and normal quantile–quantile plots). Variables with approximately unimodal and symmetrical distributions were summarized using means and standard deviations, otherwise through medians and quartiles (Q1, Q3). The relationship between IGF-1 levels, MetS diagnosis, and the number of MetS components was evaluated using graphical methods (boxplots).
The analyses described below were performed separately for the 206 and 357 women with and without MetS, respectively. The main aim was to evaluate the association of IGF-1 levels with MetS risk factors: waist circumference (cm), IGF-1 concentration (ng/mL), plasma insulin concentration (μU/mL), HOMA1-IR index (fasting plasma glucose (mg/dL) × fasting insulin concentration)/405) (Matthews et al. 1985), systolic and diastolic blood pressure (mm/hg), HDL-C (mg/dL), triglycerides (mg/dL), and glycemia (mg/dL) fasting concentrations. To such end, linear regression models were used, with IGF-1 concentration as the response variable and the remaining variables as independent variables. The association was assessed by evaluating the estimates of the slopes of the regression lines with 95% confidence intervals and association tests (Wald test).
To investigate the joint effects of the above variables on IGF-1, a multivariable model-building procedure based on Harrell’s guidelines (Harrell 2015) was adopted to pursue model robustness and higher generalizability concerning the set of variables and respective effects included in the model. The procedure is outlined as follows:
-
1.
To prevent overfitting, the maximum number of regression coefficients to be estimated by the model was fixed a priori using the 10 subjects-per-coefficient rule.
-
2.
Putative non-linear effects of the independent variables were assessed in univariate and multivariable analysis;
-
3.
A “maximal” model, including the interaction effects of the independent variables, was fitted. Nonlinear effects were no longer considered, because no evidence was found at point 2.
-
4.
The interaction effects were evaluated in the first step using a global ANOVA-like test against the null hypothesis that they were absent in the maximal model. If the null hypothesis could not be rejected, all interactions were removed from the model. Otherwise, if the null hypothesis was rejected, tests for assessing single interactions were performed to determine their presence in the model. Subsequently, a backward variable selection procedure was applied to further reduce the complexity of the model.
During Steps (3) and (4), residual analysis techniques were adopted to check the homoscedasticity, leverage points, and robustness of the model estimates with respect to outliers. The results were reported in terms of estimated regression coefficients and Wald tests, as previously described for univariate models. The interaction effects were represented graphically (effect plots). For each test, statistical significance was set at P < 0.05. The analyses were performed using R software version 4.2.1 (R Core Team (2020)—European Environment Agency n.d.) with the rms package (CRAN 2022—Package rms n.d.) and the Knime Analytic Platform version 3.6.0 (Berthold et al. 2021).
Results
IGF-1 levels were analyzed in 563 women enrolled in the DIANA-5 study. Age, anthropometric values, clinical parameters, and therapies for the MetS and non-MetS groups are shown in Table 1.
The average age at study entry was 52.2 years (± 8.3) years, with significantly younger women in the noMetS group (p < 0.0001). Of the 563 patients enrolled in the study, 206 (36.6%) had a MetS diagnosis, 241 (43.8%) had only one or two MetS traits, and 116 (20.6%) had none. As expected, the risk factors for MetS and fasting insulin levels were significantly lower in the noMetS group, while the levels of IGF-1 were higher.
Although the relationship between tamoxifen and IGF-1 is complex and not yet completely described, evidence suggests that tamoxifen suppresses IGF-1 plasma levels in early and advanced BC patients (Mandalà et al. 2001). Notably, the percentage of women taking drugs, such as tamoxifen, which can affect IGF-1 levels, was not significantly different between the two groups (46.6 and 46.2 in MetS and NoMetS, respectively, p = 0.995).
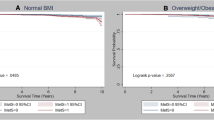
As shown in Fig. 1A, IGF-1 levels decreased with an increase in MetS factor numbers, and were globally lower in women with MetS than in those without MetS (p < 0.0001) (Fig. 1B).
The results of univariate analysis are shown in Table 2. In women with and without MetS, there was a significant inverse association between IGF-1 concentration and waist circumference (p < 0.0001 and p = 0.0098, respectively). The model estimated an average IGF-1 decrease of 1.31 ng/mL (95% CI; − 1.96 to − 0.65 ng/mL) in MetS group and 0.84 ng/mL (95% CI; − 1.46 to − 0.20 ng/mL) in noMetS group for each unit of waist circumference. No significant relationships emerged with the other metabolic characteristics in the MetS group. In the noMetS subjects, a negative association was found for SBP, while a positive association emerged for HDL-C and insulin concentrations. The estimated average IGF-1 decrease was 0.42 ng/mL for a unit increase of SBP (95% CI; − 0.82 to − 0.02 ng/mL; p = 0.0377) and an average IGF-1 increase of 0.88 ng/mL for a unit increase of HDL-C (95% CI 0.40–1.36 ng/mL; p = 0.0004) and 1.75 ng/mL for a unit increase of insulin (95% CI; 0.32–3.17 ng/mL; p = 0.0160). We also observed a positive correlation between HOMA1-IR and IGF-1 levels in noMetS women (Table 2).
In the multivariable regression analysis, HOMA1-IR was excluded from the model building procedure because of its extremely high correlation with insulin (r = 0.98 for noMetS women; r = 0.93 for the MetS women), thus causing multicollinearity-related issues. During the model building phase, the tests for overall interaction effects showed no statistical evidence (p = 0.4252 and p = 0.0729 for women with and without MetS, respectively). However, in both cases, evidence of an interaction effect between HDL-C concentration and glycemia was found (tests of association under the maximal model: p = 0.0012 and p = 0.0478 for women with and without MetS, respectively). Therefore, we decided to keep these effects were maintained within the models.
Table 2 presents the results of the final model. The multivariable model revealed that IGF-1 levels were negatively correlated with waist circumference (p = 0.0002) and SBP (p = 0.0133) in the MetS and noMetS groups, respectively. IGF-1 showed a positive relationship with insulin level (p = 0.0111) only in noMetS women. Finally, an interaction between HDL-C and glycemia was found in both groups (MetS, p = 0.0132; noMetS, p = 0.0114).
To analyze the interaction effect between HDL-C level and glycemia, the effect plots are shown in Fig. 2. Linear interaction effects, shown in Fig. 2A, B revealed that in subjects with MetS and low HDL-C (e.g., 42 mg/dL), as glycemia increased, a reduction in average IGF-1 levels was observed; however, for higher values of HDL-C (e.g., HDL-C = 56 mg/dL), IGF-1 was positively related to glycemia (Fig. 2A). Similarly, IGF-1 was positively or negatively correlated with HDL-C in subjects with higher (e.g., 107 mg/dL) or lower values of glycemia (e.g., 91 mg/dL) (Fig. 2A). In noMets women, the relationship between average IGF-1 and glycemia was negative for lower HDL-C concentrations (e.g., 55 mg/dL) and positive for higher values (e.g., 75 mg/dL) (Fig. 2B). In Fig. 2B, it may be seen that the relationship between average IGF-1 and HDL-C is positive both for lower (e.g., 85 mg/dL) and higher values (e.g., 96 mg/dL) of glycemia. However, it may be seen that higher slopes of the straight lines within the figure correspond to higher values of glycemia: this suggests that the modification effect of glycemia on the relationship between IGF-1 and HDL-C is analogous to that previously shown for noMets women, although with a lower effect strength.
Effect plots showing the joint effect between HDL-C and glycemia on IGF-1 levels according to a linear (A, B) and semi-parametric smmothing model (C–F). A, B Linear interaction effect plots; C, D, surface plots; E, F contour plots showing the estimates of the average IGF-1 as a function of HDL-C and glycemia. Threshold for glycemia and HDL are shown (dashed lines)
For a detailed insight into how HDL-C and glycemia are related to IGF-1 levels, a semi-parametric smoothing method (Wood 2017) was used to check for evidence of the linear interaction effect in the parametric model. The surface and contour plots in Fig. 2 show IGF-1 estimates as a function of HDL-C and glycemia. For women with MetS (Fig. 2C, E), the estimated values of IGF-1 were relatively low (approximately within 100–120 ng/mL) when glycemia was within the range of 150–180 mg/dL and HDL-C was lower than 60 mg/dL. When glycemia lies in the range of 100–130 mg/dL, the estimated IGF-1 lies within 140–180 ng/mL and tends to increase with higher values of HDL-C. For lower values of glycemia (lower than 100 mg/dL), the average IGF-1 level again has relatively lower values, approximately 140 ng/mL or lower. For noMetS women, the estimated average IGF-1 showed a different behavior (Fig. 2D, E). In particular, when glycemia is within 80 and 120 mg/dL, average IGF-1 levels increase with HDL-C and can reach levels far higher than those in women with MetS, up to approximately 250 mg/mL (Fig. 2D).
Discussion
In this study, we explored the relationship between insulin resistance, metabolic indices that characterize MetS, and IGF-1 levels in BC survivors. To this end, we evaluated the relationship between IGF-1 levels and relevant metabolic parameters in women with BC with and without MetS.
Our main findings showed that IGF-1 levels correlated differently with metabolic and insulin resistance-related variables in subjects with or without MetS. Indeed, we found that the insulin level was predictive of elevated circulating IGF-1 levels in women with BC without MetS. We found different levels of IGF-1 associated with glucose and HDL-C levels; in particular, there was a positive association between HDL-C and IGF-1, especially in subjects with higher blood glucose values and without a diagnosis of MetS. In agreement with previous studies, we confirmed lower levels of circulating IGF-1 in participants with MetS and a decrease in IGF-1 levels associated with an increase in MetS criteria (Brugts et al. 2010; Oh et al. 2012; Parekh et al. 2010).
These aspects are particularly interesting in BC survivors, since it is well-known that marked impairments in metabolic function are associated with more aggressive postmenopausal breast tumor biology (Dong et al. 2021; Healy et al. 2010). Jones et al. 2012 (Jones et al. 2012) found that on average, cardiometabolic conditions in BC women are less than those in age-matched sedentary but otherwise healthy women without a history of BC. Remarkably, patients with BC reach a predicted cardiorespiratory condition in a particular age group (e.g., 40 years) approximately 20–30 years earlier than healthy women without a history of BC. It has also been reported that elderly cancer survivors (of mixed diagnoses) have the most functional limitations (are less likely to be able to perform heavy household tasks, walk half a mile, or walk up and down stairs) compared to women without a history of cancer (Elad et al. 2022; Sweeney et al. 2006). In our study, we found a negative correlation between IGF-1 levels and age, BMI, and waist circumferences. The negative correlation between IGF-1 levels and anthropometric measurements is consistent with the results of several epidemiological studies (Gram et al. 2006; Parekh et al. 2010; Roberts et al. 2010; Sandhu et al. 2004; Sesti et al. 2005; Sherlala et al. 2021; Slattery et al. 2005; Succurro et al. 2009). Although the mechanisms involved are not fully understood, it has been proposed that they interact with other metabolic hormones, such as insulin, GH, and leptin (Parekh et al. 2010).
Epidemiological evidence has shown that high circulating IGF-1 levels are positively associated with BC risk (Endogenous Hormones and Breast Cancer Collaborative Group et al. 2010) and prognosis (Murphy et al. 2020; Pasanisi et al. 2008). Moreover, it has been shown that high IGF-1 levels were protective in non-overweight patients but a risk factor for overweight patients (Tong et al. 2020). Notably, as metabolic disorders are frequently associated with low IGF-1 levels in subjects with MetS, it could be hypothesized that IGF-1 is probably not involved as a proliferation inducer but might have other autocrine and/or paracrine actions. In future, we intend to deepen this aspect of the entire cohort of women recruited in the DIANA-5 study.
Interestingly, we observed that IGF-1 levels correlated differently with metabolic and insulin resistance-related variables in subjects with or without MetS. Insulin resistance is often described as a condition of subclinical MetS. Patients can be classified as insulin-resistant, with a HOMA1-IR index ≥ 2.50. An interesting study (Capasso et al. 2013) showed that most BC survivors have normal glucose and insulin levels. However, the measurement of glycemia or insulin alone could underestimate the prevalence of insulin resistance in this population of BC survivors. On the other hand, finding physiological predictors of metabolic changes (e.g., IGF-1), considering glycemia and insulin levels alone, could be more informative than the HOMA1-IR index. This aspect might be relevant for better defining the complicated relationship between MetS, insulin resistance, and IGF-1.
In this study, we observed a positive relationship between HOMA1-IR index and IGF-1 levels in subjects without MetS. In fact, HOMA1-IR was identified as a strong predictor of IGF-1 levels only in the noMetS group. These results are consistent with studies that showed a positive correlation between insulin resistance measures (i.e., HOMA1-IR indices) and levels of circulating IGF-1 (Matsumoto et al. 2018). Furthermore, other studies have highlighted a non-linear (i.e., U-shaped) relationship between insulin resistance and IGF-1 levels in patients with MetS (Friedrich et al. 2012; Oh et al. 2012). It should be noted that there is evidence showing a U-shaped relationship between IGF-1 and general mortality and several chronic degenerative diseases (Zhang et al. 2021). Although this aspect is age-dependent and slightly related to cancer risk, it deserves further investigation.
Interestingly, considering glycemia and insulin levels separately, instead of HOMA1-IR in the regression models, we observed that only insulin levels predicted IGF-1 levels in subjects without MetS, but not in subjects with MetS. In vivo studies suggest that despite the increased uptake of glucose by the tumor, hyperglycemia alone may not increase tumor growth without hyperinsulinemia (Giovannucci et al. 2010; Muti et al. 2002; Vigneri et al. 2009). This suggests that, although many cancer cells rely on glucose metabolism, glucose is not the only key driver of cancer growth and progression in metabolic disorders (Kang et al. 2018), in which other factors such as IGF-1 may be involved.
In this study, we also demonstrated a possible interaction between glycemia and HDL-C levels in relation to circulating IGF-1 levels. Although some studies have failed to find associations between lipoproteins and BC, some large clinical studies have demonstrated a direct association between LDL cholesterol levels and BC risk, and an inverse association between HDL-C and BC risk (Cedó et al. 2019; Strohmaier et al. 2013) and that low HDL-C levels are a single MetS component independently associated with BC recurrence in the DIANA-5 cohort (Berrino et al. 2014). We observed different interaction effects between HDL-C concentration and glycemia in relation to IGF-1 in participants with or without MetS. In women with MetS, we observed that IGF-1 was positively related to HDL-C in those with higher glycemic levels and negatively related to HDL-C in those with lower glycemic levels. In contrast, in subjects without MetS, we observed a strong positive correlation between HDL-C and circulating IGF-1 levels, especially in those with relatively higher values of glycemia. These results highlight the importance of glucose metabolism in the modulation of circulating IGF-1, especially in an oncological context. Intriguingly, an additional complex level of IGF-1 modulation is the presence of a glycosylation site in the Ea peptide of the IGF-1 pro-hormone (Annibalini et al. 2016; Philippou et al. 2014; De Santi et al. 2016). In this context, we recently showed that N-linked glycosylation regulates the stability and secretion of the IGF-1Ea pro-hormone, acting as a glucose-sensitive flexible tail that controls mature IGF-1 production (Annibalini et al. 2018). Moreover, there is mounting evidence for the role of HDL-C in the control of glucose metabolism (Siebel et al. 2015). Although the mechanisms by which insulin resistance contributes to low HDL-C are known, there are also indications that low HDL-C may actually promote the development of diabetes (Drew et al. 2012) and that HDL-C elevation can increase insulin sensitivity in peripheral tissues (Carey et al. 2013). However, there is little evidence of a correlation between IGF-1 levels and circulating lipoproteins. It has been hypothesized a possible role of IGF-1 in regulating HDL-C levels as they show a positive correlation between serum IGF-1 and HDL-C (Song et al. 2016).
Although our results showed an interaction between HDL-C and glycemia, the interpretation is largely incomplete and requires further analysis in a larger population to confirm or exclude the possible role of HDL-C in modulating IGF-1 and its role in BC recurrence. HDL-C is considered as “good cholesterol,” but recent research suggests that this might not always be the case. HDL-C particle composition and biological functions may change under particular conditions, such as metabolic disorders and inflammatory diseases, and a fundamental reassessment of the clinical significance of HDL-C is warranted (März et al. 2017).
An intrinsic limitation of this study is that it does not allow us to clarify cause and effect. However, from a perspective point of view, we hypothesized that lifestyle strategies should focus on the regulation of IGF-1 levels in the normal range rather than on reducing them. The DIANA-5 study will allow us to more thoroughly analyze which factors act as circulating IGF-1 modulators. In the future, we will examine in all women recruited in the Diana-5 study how IGF-1 levels, insulin resistance, and fasting insulin levels in women without MetS may influence the incidence of relapses, secondary BC, and metastases compared to women diagnosed with MetS.
In conclusion, the main findings of this study showed that overall, in women with MetS, mean IGF-1 values are lower and less modulated by metabolic factors than in women without MetS. In particular, findings showing a positive correlation between IGF-1 levels and insulin resistance only in subjects without MetS, a predictive value of circulating IGF-1 for insulin levels, and a possible role of HDL-C in IGF-1 modulation could have important clinical implications for planning customized strategies aimed at modulating IGF-1 levels in BC survivors. In fact, while the IGF-1 system seems to be much more compromised in subjects with a diagnosis of MetS, in noMetS subjects, IGF-1 levels could better respond to lifestyle changes. Further analyses of all DIANA-5 populations are needed to evaluate the role of physical activity and/or dietary intervention in the modulation of IGF-1 concentrations and its implications in BC survivors.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors without any undue reservation.
References
Agnoli C et al (2010) Metabolic syndrome and postmenopausal breast cancer in the ORDET cohort: a nested case-control study. Nutr Metab Cardiovasc Dis 20(1):41–48
Alberti KGMM et al (2009) Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120(16):1640–1645
Andò S et al (2019) Obesity, leptin and breast cancer: epidemiological evidence and proposed mechanisms. Cancers 11(1):1–27
Annibalini G et al (2016) MIR retroposon exonization promotes evolutionary variability and generates species-specific expression of IGF-1 splice variants. Biochimica Et Biophysica Acta Gene Regul Mech 1859(5):757–768
Annibalini G et al (2018) The intrinsically disordered E-domains regulate the IGF-1 prohormones stability, subcellular localisation and secretion. Sci Rep 8(1):1–13
Berrino F et al (2014) Metabolic syndrome and breast cancer prognosis. Breast Cancer Res Treat 147(1):159–165
Berthold MR et al (2021) KNIME-The Konstanz information miner version 2.0 and beyond. ACM SIGKDD Explor Newsl 11(1):26–31
Biganzoli E et al (2017) Recurrence dynamics of breast cancer according to baseline body mass index. Eur J Cancer 87:10–20
Brauna S, Bitton-Worms K, le Roith D (2011) The link between the metabolic syndrome and cancer. Int J Biol Sci 7(7):1003–1015
Brugts MP et al (2010) IGF-I bioactivity in an elderly population: relation to insulin sensitivity, insulin levels, and the metabolic syndrome. Diabetes 59(2):505–508
Calori G et al (2011) Prevalence, metabolic features, and prognosis of metabolically healthy obese Italian individuals: the cremona study. Diabetes Care 34(1):210–215
Capasso I et al (2013) Homeostasis model assessment to detect insulin resistance and identify patients at high risk of breast cancer development: National Cancer Institute of Naples Experience. J Exp Clin Cancer Res 32(1):1
Carey AL et al (2013) Skeletal muscle insulin resistance associated with cholesterol-induced activation of macrophages is prevented by high density lipoprotein. PLoS One 8(2):e56601
Cedó L et al (2019) HDL and LDL: potential new players in breast cancer development. J Clin Med 8(6):853
Cevenini A et al (2018) Molecular signatures of the insulin-like growth factor 1-mediated epithelial-mesenchymal transition in breast, lung and gastric cancers. Int J Mol Sci 19(8):1–24
Cornier MA et al (2008) The metabolic syndrome. Endocr Rev 29(7):777–822
“CRAN (2022) Package Rms. https://cran.r-project.org/web/packages/rms/index.html. Accessed 23 Nov 2022
De Santi M et al (2016) Human IGF1 pro-forms induce breast cancer cell proliferation via the IGF1 receptor. Cell Oncol 39(2):149–159
Devericks EN et al (2022) The obesity-breast cancer link: a multidisciplinary perspective. Cancer Metastasis Rev 41(3):607–625
Dong S, Wang Z, Shen K, Chen X (2021) Metabolic syndrome and breast cancer: prevalence, treatment response, and prognosis. Front Oncol 11:629666
Doyle SL, Donohoe CL, Lysaght J, Reynolds JV (2012) Visceral obesity, metabolic syndrome, insulin resistance and cancer. Proc Nutr Soc 71:181–189
Drew BG et al (2012) The emerging role of HDL in glucose metabolism. Nat Rev Endocrinol 8(4):237–245
Duggan C et al (2011) Associations of insulin resistance and adiponectin with mortality in women with breast cancer. J Clin Oncol 29(1):32–39
Elad B, Habib M, Caspi O (2022) Cardio-oncology rehabilitation-present and future perspectives. Life (Basel) 12(7):1006
Endogenous Hormones and Breast Cancer Collaborative Group et al (2010) Insulin-like growth factor 1 (IGF1), IGF binding protein 3 (IGFBP3), and breast cancer risk: pooled individual data analysis of 17 prospective studies. Lancet Oncol 11(6): 530–542
Esposito K et al (2012) Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care 35(11):2402–2411
Friedrich N et al (2012) The association between IGF-I and insulin resistance: a general population study in danish adults. Diabetes Care 35(4):768–773
Giovannucci E et al (2010) Diabetes and cancer: a consensus report. CA Cancer J Clin 60(4):207–221
Goodwin PJ et al (2002) Fasting insulin and outcome in early-stage breast cancer: results of a prospective cohort study. J Clin Oncol 20(1):42–51
Gram IT et al (2006) Body mass index, waist circumference and waist-hip ratio and serum levels of IGF-I and IGFBP-3 in European women. Int J Obes 30(11):1623–1631
Hankinson SE et al (1998) Circulating concentrations of insulin-like growth factor-I and risk of breast cancer. Lancet 351(9113):1393–1396
Harrell FE (2015) Regression modeling strategies. Springer International Publishing, Cham. https://doi.org/10.1007/978-3-319-19425-7
Healy LA et al (2010) Metabolic syndrome, central obesity and insulin resistance are associated with adverse pathological features in postmenopausal breast cancer. Clin Oncol (r Coll Radiol (great Britain)) 22(4):281–288
Hwang K-T et al (2020) Influence of metabolic syndrome on risk of breast cancer: a study analyzing nationwide data from Korean National Health Insurance Service. Cancer Epidemiol Biomark Prev 29(10):2038–2047
Jones LW et al (2012) Cardiopulmonary function and age-related decline across the breast cancer survivorship continuum. J Clin Oncol 30(20):2530–2537
Kaaks R (2001) Plasma insulin, IGF-I and breast cancer. Gynecologie Obstetrique Et Fertilite 29(3):185–191
Kang C, LeRoith D, Gallagher EJ (2018) Diabetes, obesity, and breast cancer. Endocrinology 159(11):3801–3812
Law JH et al (2008) Phosphorylated insulin-like growth factor-I/insulin receptor is present in all breast cancer subtypes and is related to poor survival. Cancer Res 68(24):10238–10246
Lukanova A et al (2001) A cross-sectional study of IGF-I determinants in women. Eur J Cancer Prev 10(5):443–452
Mandalà M et al (2001) Effect of tamoxifen on GH and IGF-1 serum level in stage I–II breast cancer patients. Anticancer Res 21:585–588
März W et al (2017) HDL cholesterol: reappraisal of its clinical relevance. Clin Res Cardiol 106(9):663–675
Matsumoto R et al (2018) Factors correlated with serum insulin-like growth factor-I levels in health check-up subjects. Growth Hormone IGF Res 40:55–60
Matthews DR et al (1985) Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7):412–419
Murphy N et al (2020) Insulin-like growth factor-1, insulin-like growth factor-binding protein-3, and breast cancer risk: observational and mendelian randomization analyses with ∼430 000 women. Ann Oncol 31(5):641–649
Muti P et al (2002) Fasting glucose is a risk factor for breast cancer. Cancer Epidemiol Prev Biomark 11(11):1361–8
Nelson RA, Bremer AA (2010) Insulin resistance and metabolic syndrome in the pediatric population. Metab Syndrome Relat Disord 8(1):1–14
Oh J et al (2012) The relationship between insulin-like growth factor-1 and metabolic syndrome, independent of adiponectin. Clin Chim Acta 413(3–4):506–510
Parekh N et al (2010) Lifestyle, anthropometric, and obesity-related physiologic determinants of insulin-like growth factor-1 in the third national health and nutrition examination survey (1988–1994). Ann Epidemiol 20(3):182–193
Pasanisi P et al (2008) Serum insulin-like growth factor-I and platelet-derived growth factor as biomarkers of breast cancer prognosis. Cancer Epidemiol Biomark Prev 17(7):1719–1722
Philippou A, Maridaki M, Pneumaticos S, Koutsilieris M (2014) The complexity of the IGF1 gene splicing, posttranslational modification and bioactivity. Mol Med 20(1):202–214
Pollak M (2008) Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer 8(12):915–928
R Core Team (2020) European Environment Agency. https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006. Accessed 23 Nov 2022
Roberts DL, Dive C, Renehan AG (2010) Biological mechanisms linking obesity and cancer risk: new perspectives. Annu Rev Med 61:301–316
Sandhu MS et al (2004) Association between insulin-like growth factor-I: insulin-like growth factor-binding protein-1 ratio and metabolic and anthropometric factors in men and women. Cancer Epidemiol Biomark Prev 13(1):166–170
Sesti G et al (2005) Plasma concentration of IGF-I is independently associated with insulin sensitivity in subjects with different degrees of glucose tolerance. Diabetes Care 28(1):120–125
Sherlala RA et al (2021) Relationship between serum IGF-1 and BMI differs by age. J Gerontol Ser A Biol Sci Med Sci 76(7):1303–1308
Siebel AL, Heywood SE, Kingwell BA (2015) HDL and glucose metabolism: current evidence and therapeutic potential. Front Pharmacol 31;6:258
Slattery ML et al (2005) Genetic, anthropometric, and lifestyle factors associated with IGF-1 and IGFBP-3 levels in hispanic and non-hispanic white women. Cancer Causes Control 16(10):1147–1157
Song X et al (2016) Positive correlation between serum IGF-1 and HDL-C in type 2 diabetes mellitus. Diabetes Res Clin Pract 118:44–49
Strohmaier S et al (2013) Total serum cholesterol and cancer incidence in the metabolic syndrome and cancer project (me-can). PLoS ONE 8(1):e542428
Succurro E et al (2009) Low plasma insulin-like growth factor-1 levels are associated with reduced insulin sensitivity and increased insulin secretion in nondiabetic subjects. Nutr Metab Cardiovasc Dis 19(10):713–719
Sweeney C et al (2006) Functional limitations in elderly female cancer survivors. J Natl Cancer Inst 98(8):521–529
Tong Y et al (2020) IGF-1 Interacted With obesity in prognosis prediction in HER2-positive breast cancer patients. Front Oncol 10:550
Vigneri P et al (2009) Diabetes and cancer. Endocr Relat Cancer 16(4):1103–1123
Villarini A et al (2012) Lifestyle and breast cancer recurrences: the DIANA-5 trial. Tumori 98(1):1–18
Wood SN (2017) Generalized additive models: an introduction with R, 2nd edn. Chapman and Hall, Boca Raton
Zhang WB, Ye K, Barzilai N, Milman S (2021) The antagonistic pleiotropy of insulin-like growth factor 1. Aging Cell 20(9):e13443
Acknowledgements
This study was partially supported by the project “IGF-1 isoforms and breast cancer,” RF-2009-1532789. We thank the NuMeLab laboratory (Ssd. Nutritional Research and Metabolomic, IRCCS Foundation of the National Cancer Institute of Milan) for carrying out the serum IGFI measurement.
Funding
Open access funding provided by Università degli Studi di Urbino Carlo Bo within the CRUI-CARE Agreement. This study was partially supported by the project “IGF-1 isoforms and breast cancer,” Ministry of Health, RF-2009-1532789.
Author information
Authors and Affiliations
Contributions
AV, EBa, and GBa contributed to the study conception and design. AV and EV planning of data collection, and laboratory analysis. EBi, GM, and GBi performed the statistical analysis. MDS and GA written the first draft of the manuscript. MP and FL made important intellectual contribution to the manuscript revision. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
De Santi, M., Annibalini, G., Marano, G. et al. Association between metabolic syndrome, insulin resistance, and IGF-1 in breast cancer survivors of DIANA-5 study. J Cancer Res Clin Oncol 149, 8639–8648 (2023). https://doi.org/10.1007/s00432-023-04755-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04755-6