Abstract
We investigated the significance of PI3K/AKT/mTOR pathway and its interactions with MAPK, JAK/STAT and Notch pathways in meningioma progression. Paraffin-embedded tissue from 108 meningioma patients was analysed for the presence of mutations in PIK3CA and AKT1. These were correlated with the expression status of components of the PI3K/AKT/mTOR pathway, including p85α and p110γ subunits of PI3K, phosphorylated (p)-AKT, p-mTOR, p-p70S6K and p-4E-BP1, as well as of p-ERK1/2, p-STAT3 and Notch-1, clinicopathological data and patient survival. A mutation in PIK3CA or AKT1 was found in around 9 % of the cases. Higher grade meningiomas displayed higher nuclear expression of p-p70S6K; higher nuclear and cytoplasmic expression of p-4E-BP1 and of Notch-1; lower cytoplasmic expression of p85αPI3K, p-p70S6K and p-ERK1/2; and lower PTEN Histo-scores (H-scores). PTEN H-score was inversely correlated with recurrence probability. In univariate survival analysis, nuclear expression of p-4E-BP1 and absence of p-ERK1/2 expression portended adverse prognosis, whereas in multivariate survival analysis, p-ERK1/2 expression emerged as an independent favourable prognostic factor. Treatment of the human meningioma cell line HBL-52 with the PI3K inhibitor LY294002 resulted in reduction of p-AKT, p-p70S6K and p-ERK1/2 protein levels. The complex interactions established between components of the PI3K/AKT/mTOR pathway, or with components of the MAPK, JAK/STAT and Notch-1 pathways, appear to be essential for facilitating and fuelling meningioma progression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meningiomas are among the most common types of primary intracranial tumour in adults. Although most are benign and slowly growing, some are highly recurrent and associated with poor prognosis [1]. In particular, anaplastic (WHO grade 3) meningiomas share clinical features, such as infiltrating capacity and metastasis, similar to other malignant brain neoplasms [2]. Despite the remarkable progress made in clarifying the molecular mechanisms that underlie meningioma pathogenesis and progression [3, 4], deregulated signalling pathways in meningioma of therapeutic and prognostic significance remain to be elucidated.
Disruption of phosphoinositide 3-kinase (PI3K)/v-akt murine thymoma viral oncogene homolog 1 (AKT, also known as protein kinase B, PKB)/mammalian target of rapamycin (mTOR) signalling pathway (Fig. 1) is a major regulator of crucial cell functions such as cell growth, survival and proliferation, and reportedly contributes to the pathogenesis of several types of tumour [for review, 5]. This pathway is strongly interconnected at multiple steps with the mitogen-activated protein kinase (MAPK) pathway [6, 7], the Janus-activated kinase (JAK)/signal transducer and activator of transcription (STAT) pathway [7, 8] and the Notch-1 pathway [9, 10]. Complex interactions and cooperation between these aberrantly activated pathways fuel growth and survival of neoplastic cells.
The PI3K/AKT/mTOR pathway and its interactions with MAPK, JAK/STAT and Notch pathways. The initial step of PI3K/AKT/mTOR pathway activation takes place at the cell membrane and is propagated through PI3K class IA. The PI3K product (phosphatidylinositol-3,4,5-triphosphate (PIP3)) binds to 3′-phosphoinositide-dependent kinase 1 (PDK-1) and AKT through the pleckstrin homology domains (PH), allowing translocation of both proteins to the cell membrane, followed by their activation. As consequence of this colocalization, AKT is fully activated. PI3K is antagonized by PTEN (phosphatase and tensin homolog deleted on chromosome 10) through dephosphorylation of PIP3, thereby preventing AKT activation. Upon activation, AKT act by activating or inhibiting many downstream targets implicated in various cellular functions including protein synthesis and cell cycle progression. One of the most studied AKT’s targets is the serine/threonine kinase mTOR. mTOR participates as member of two complexes: mTORC1 (mTOR binding to Raptor) and mTORC2 (mTOR binding to Rictor). Two downstream pathways of mTORC1 involved in the translation machinery are responsible for ribosome recruitment to mRNA: phosphorylation and inactivation of 4E-BP1 (eukaryotic translation initiation factor 4E-binding protein 1), the repressor of mRNA translation, and phosphorylation and activation of S6K1 (ribosomal S6 kinase 1), the promoter of mRNA translation [5]. PI3K/AKT/mTOR can interact directly or indirectly with the MAPK, JAK/STAT and Notch pathways (for details see the text). The analysed molecules in our study are shown in grey shape fill. Dashed arrows indicate indirect or probable interactions between molecules. Black lightenings show molecules that were analysed also for mutations
Among the three classes of PI3Ks, only the altered expression of class I is implicated in tumourigenesis. The gene (PIK3CA) encoding for p110α, the catalytic subunit of class I PI3Ks, is frequently found either amplified or mutated in several types of tumours [6]. The majority of PIK3CA gain-of-function mutations cluster in exons 9 and 20, which encode for the helical and kinase domain of p110α subunit, respectively. In exon 9, the most frequent mutations are E542K and E545K, whereas in exon 20, the most frequent mutation is H1047R. Several other rare gain-of-function mutations have been recorded in tumours [11]. As yet, the role of p85α regulatory subunit in oncogenesis has not been studied extensively. The AKT serine/threonine kinase, which is encoded by three distinct genes (AKT1, AKT2 and AKT3), can be aberrantly activated either by gene amplification or mainly as a critical target of PI3K in human tumours [12]. However, a new gain-of-function somatic mutation affecting amino acid 17 (E17K) in the lipid-binding pocket of AKT1 has been recently reported [13]. Negative regulation of AKT is indirectly mediated by PTEN (phosphatase and tensin homolog deleted on chromosome 10), the loss of which appears to be an alternative mechanism of activation of the PI3K pathway in human tumours [14].
Studies looking into the interactions of several signalling pathways in meningiomas are rare. In order to treat these tumours more effectively, the complex signalling networks should be deciphered and clarified. For this purpose, we first analysed the mutation status of PIK3CA and AKT1 genes in relation to the expression status of several components of the PI3K/AKT/mTOR pathway, namely p85α and p110γ subunits of PI3K, phosphorylated (p)-AKT, p-mTOR, p-p70S6K, p-4E-BP1, as well as p-ERK1/2, p-STAT3 and Notch-1 expression. Immunohistochemistry was validated by Western blot analysis in five meningioma specimens. In addition, we tested the effect of an inhibitor of PI3K/AKT pathway on the expression levels of p-AKT, p-ERK and p-p70S6K, the three most commonly expressed proteins. To this end, we chose LY294002, an inhibitor of PI3K lying upstream of AKT, and the human meningioma cell line HBL-52. We also examined correlations between the aforementioned components of signalling pathways and clinicopathological data. Finally, we determined the potential prognostic utility of these molecules in meningiomas.
Materials and methods
Patients
This is a retrospective study of 108 consecutive cases (69 female, 39 male) with meningioma (intracranial and SS) diagnosed at the Department of Pathology of the University of Athens as well as the Metropolitan Hospital between 2002 and 2009. Only primary tumours at diagnosis were included in this investigation. Informed consent was obtained from all patients before their enrolment in the study. All available histological slides, routinely stained with haematoxylin and eosin, were reviewed and evaluated for tumour histological grade. Sections were immunohistochemically stained for GFAP, S100 protein and epithelial membrane antigen (EMA). Using the criteria established in the WHO classification of 2007, the diagnosis was confirmed by two experienced neuropathologists (PK, EP), who also graded the tumours as follows: grade 1, 73 cases; grade 2, 29 cases; and grade 3, 6 cases. Evidence of brain invasion was observed in 9/29 (31 %) of grade 2 meningiomas. Information on total resection was based on the surgeon’s assessment at the time of surgery and post-operative computerized tomography with contrast, whenever considered necessary. Simpson grade was 1 to 4. None of the cases had received radiotherapy before surgery.
The presence of recurrence or death from disease was based on data obtained by clinical and histological records, office notes on follow-up visits and telephone contact whenever possible. Follow-up period ranged from 0.7 to 146.3 months (median 74.7 months) and was available in 71 patients, a cohort of which the main characteristics did not differ substantially from those of the whole series (Supplementary Table 1). Radiotherapy was administered in 35/108 (32 %) cases. During this period of time, 7 patients died of disease, i.e. as a result of meningioma progression, after a medial follow-up period of 23 months (range 0.7–81 months), whereas the remaining 65 cases were followed up for a median period of 76.5 months (range 2.7–146.3 months). The patients’ clinicopathological data are shown in Supplementary Table 1.
PIK3CA and AKT1 mutational analysis
DNA extraction from paraffin-embedded tissues
Sections 10 μm thick were cut from paraffin-embedded tissue blocks after tumour enrichment by light microscopy. DNA was extracted from the selected tissue areas following a standard DNA extraction kit protocol (NucleoSpin tissue, Macherey–Nagel, Duren, Germany). The extracted DNA was quantitated on a Picodrop Microliter spectrophotometer.
High-resolution melting analysis
PIK3CA exons 9 and 20 and AKT1 exon 4 were screened for mutations in 100 tumour specimens (grade 1, 67 cases; grade 2, 27 cases; grade 3, 6 cases) using high-resolution melting analysis (HRMA) on a LightCycler 480 (Roche Diagnostics, GmbH, Germany). Each reaction consisted of 20 ng DNA, 200 nmol/L of each primer, 10 μL LightCycler 480 HRM Master Mix (Roche), and 3.5 mM MgCl2, in a total volume of 20 μL. The profile used in the LightCycler was as follows: 95 °C for 10 min, followed by 50 cycles of 95 °C for 10 s, 58–60 °C for 15 s, and 72 °C for 7 s. DNA samples from colorectal cancers or cell lines were used as positive control for HRMA. In detail, for PIK3CA exon 20 mutational analysis, we used DNA extracted from colon cancer cell line HCT-116 (PIK3CA mutation, p.H1047R). For AKT1, previously identified mutant samples from colorectal cancers were used. Primers for PIK3CA gene exon 9 were designed in order to exclude the amplification of a pseudogene. The sequences of the primers for PIK3CA and AKT1 genes have been published previously [15].
Sequencing
PIK3CA (exons 9 and 20) PCR products positive by HRMA were sequenced using the BigDye terminator cycle sequencing kit (Applied Biosystems, CA, USA) in order to confirm the presence of mutations. The sequencing products were analysed on an ABI Prism 310 Genetic Analyzer (Applied Biosystems). PCR primers were also used for sequencing analysis. Results were verified by sequencing analysis of at least two independent PCR products. None of the sequences of exon 9 showed amplification of the pseudogene. AKT1 mutations were identified using pyrosequencing with the Q24 pyrosequencer according to the manufacturer’s protocol (Qiagen Gmblt, Hilden, Germany).
Immunohistochemical analysis
Immunostaining was performed on paraffin-embedded 4-μm sections of formalin-fixed tumour tissue using the two-step peroxidase-conjugated polymer technique (DAKO Envision kit, DAKO, Carpinteria, CA). The primary antibodies used are listed in Supplementary Table 2.
Evaluation of immunohistochemical staining was performed by two pathologists (PK, ET), without knowledge of the clinical information. They viewed the first 20 cases for each antibody together to obtain consensus regarding the evaluation, without knowledge of the clinical information. The remaining cases were then examined by one pathologist (ET) and of those another 20 cases were checked for reproducibility by the second pathologist (PK). Nuclear and cytoplasmic immunoreactivity was recorded separately. A Histo-score (H-score) based on the percentage of stained neoplastic cells (labelling index, LI) multiplied by staining intensity was calculated, as previously described [16].
Cell culture
The meningioma cell line HBL-52 (Cell Line Services, Heidelberg, Germany), originally established from a transitional meningioma grade 1 localized at the optic canal, was grown in McCoy’s 5a Medium (CLS), supplemented with 2 mM L-Glutamine (Gibco, Life Technologies), 10 % foetal bovine serum (FBS; Gibco, Life Technologies) and 1 % penicillin–streptomycin mixture (10,000 U/mL of penicillin and 10,000 μg/mL of streptomycin; Gibco, Life Technologies). Cell cultures were incubated in a humidified incubator at 37 °C and 5 % CO2.
When cell cultures reached 70 % confluency, LY294002 PI3K inhibitor (Calbiochem, Germany) was added for 3 h in a final concentration of 100 μM. All cell culture experiments were performed at least in triplicate.
Western blot analysis
Western blot analysis was performed on extracts from HBL-52 cells as well as on extracts from a subset of frozen meningioma samples (five samples) which were also analysed immunohistochemically. More specifically, 50 mg of tumour tissue, fresh frozen and stored at −80 °C, was homogenized in ice-cold lysis buffer (RIPA Lysis Buffer, Thermo Scientific) containing protease inhibitors (1 % aprotinin, 2 mmol/L of phenylmethylsulfonyl fluoride, 1 mmol/L DTT and 10 μg/mL of leupeptin) and phosphatase inhibitors (2 mmol/L of sodium orthovanadate, 10 mmol/L of sodium fluoride, 2 mmol/L beta-glycerophosphate and 2 mmol/L sodium pyrophosphate, decahydrate). Following the homogenization, analysis of protein levels was performed. More specifically, samples containing 100 μg of protein were separated on an 8 % (for p-AKT) and 10 % (for p-p70S6K, p-ERK1/2 and actin) SDS polyacrylamide gel, blotted onto nitrocellulose membranes (Porablot NCP) and blocked for 1 h in room temperature in phosphate-buffered saline–Tween-20 (PBST) with 5 % non-fat milk. Subsequently, membranes were incubated overnight at 4 °C with the primary antibodies listed in Supplementary Table 2. Primary antibodies were diluted in PBST containing 1 % non-fat milk. The membranes were then incubated with the HRP-conjugated secondary antibodies (1:1,000–1:2,000) for 1 h in room temperature. Immunoreactive bands were detected with the LumiSensor Chemiluminescent HRP Substrate kit (GenScript).
Statistical analysis
Statistical analysis was performed by an MSc Biostatistician (GL). In the basic statistical analysis, PTEN, p85aPI3K, p110γPI3K, p-mTOR, p-p70S6K, p-4E-BP1, p-AKT, p-ERK1/2, p-STAT3 and Notch-1 H-scores were treated as continuous variables. Correlations between immunohistochemical expression levels of the investigated proteins and clinic pathological parameters were tested using non-parametric tests (Kruskal–Wallis ANOVA, Mann–Whitney U test, Spearman’s rank correlation coefficient, Fisher’s exact test and chi-square, as appropriate).
Survival analysis was performed using death from disease as endpoint for overall survival. Overall survival was measured from the date of surgical resection to the last follow-up visit or death. The effect of various parameters (age, sex, grade, Simpson grade, radiotherapy, along with PTEN, p85aPI3K, p110γPI3K, p-mTOR, p-p70S6K, p-4E-BP1, p-AKT, p-ERK1/2, p-STAT3 and Notch-1 H-scores and the presence of PIK3CA and AKT mutations) on clinical outcome was assessed by comparing groups using the log-rank test. Receiver operating characteristics (ROC) analysis was performed for the selection of the cut-off values, which are indicated in Table 1. For multivariate analysis, Cox’s proportional hazards estimation model with forward selection of variables was used and only those variables significant in univariate analysis were included. For statistical calculations, the statistical package STATA 11.0 for Windows was used. All results with a two-sided p level ≤0.05 were considered statistically significant.
Results
PIK3CA and AKT1 mutations in meningioma
Of the 100 cases studied, molecular analysis was successful with regard to the presence of activating mutations at exons 9 and 20 of PIK3CA gene in 91 and 88 cases, respectively, and for mutations at exon 4 of AKT1 gene in 99 cases. PIK3CA mutations were observed in 12 cases, three of which in exon 9 (E547K, S541F and L540L), the latter synonymous (Fig. 2). We found nine mutations in exon 20, two concerning the most common gain-of-function mutation H1047R, four concerning rare missense mutations (A1046T, E1034K, M1043I, R1023Q) and three concerning a known polymorphism (T1025T). The E17K mutation of AKT1 was observed in nine cases, one of which occurred simultaneously with PIK3CA mutation L540L. AKT1 mutation correlated with higher PTEN and lower pERK1/2 cytoplasmic H-score (Mann–Whitney U test, p = 0.0239 and p = 0.0359, respectively). Trends also emerged regarding correlations between AKT1 mutations and cytoplasmic expression of p-AKT, p-mTOR and p-4E-BP1, but these did not reach significance (p = 0.0995, p = 0.0534 and p = 0.0932, respectively). Moreover, there was a trend for PIK3CA or AKT1 mutation to occur more often in men than in women (9/29 vs 9/58, chi-square test p = 0.0921).
AKT/mTOR pathway components, PTEN, p-ERK1/2, p-STAT3 and Notch-1 expression in meningioma
Coexpression rates among analysed molecules are shown in Supplementary Table 3. p-mTOR immunoreactivity was cytoplasmic and/or membranous in 52/107 cases (49 %) (Fig. 3). Nuclear p-p70S6K immunoreactivity was seen in all cases, 44 (41 %) of which also showed cytoplasmic immunoreactivity (Fig. 3). Nuclear p-4E-BP1 immunoreactivity was seen in 41 % of cases, of which 68 (64 %) also showed cytoplasmic immunoreactivity (Fig. 3). Endothelial cells were immunoreactive for both proteins and served as internal positive control. In 52 cases (52/106, 49 %), nuclear coexpression of p-p70S6K and p-mTOR was found, most p-p70S6K (nuclear)-positive cases but also p-mTOR-negative cases (36/57, 67 %). Coexpression of p-mTOR and p-4E-BP1, either nuclear or cytoplasmic, was rather infrequent (22 and 33 %, respectively), but most (70 %) p-4E-BP1-positive/p-mTOR-negative cases expressed p-ERK1/2. Although simultaneous expression of all three components of mTOR cascade was rather uncommon (23/105, 22 %), no case was triple negative for p-mTOR, nuclear p-p70S6K and nuclear p-4E-BP1 and only 13 % were simultaneously negative for p-mTOR, cytoplasmic p-p70S6K and cytoplasmic p-4E-BP1.
Immunohistochemical expression of p85aPI3K, p110γPI3K, p-AKT, p-mTOR, p-p70S6K and p-4E-BP1 in benign and non-benign meningiomas. Non-benign meningiomas displayed higher nuclear p-p70S6K, nuclear and cytoplasmic p-4E-BP1 and lower cytoplasmic p85αPI3K and cytoplasmic p-p70S6K H-scores when compared to benign meningiomas
p-AKT immunoreactivity was mainly cytoplasmic (77/108, 71 %), with nuclear expression in only 31 % of cases (Fig. 3). Cytoplasmic p-AKT was coexpressed with p-mTOR in 38 % of cases, but along with nuclear expression of p-p70S6K in 18 % and cytoplasmic expression of p-p70S6K and p-4E-BP1 in 17 %, with even lower percentages for nuclear p-AKT (9 and 5 %, respectively). PTEN nuclear expression was observed in all cases (Fig. 4).
Cytoplasmic expression of p85αPI3K was observed in 38/108 cases (35 %) and nuclear expression in 33/108 cases (31 %) (Fig. 3). Cytoplasmic coexpression of p-AKT and p85apI3K was found in 28 % of cases. Coexpression of nuclear or cytoplasmic p85αPI3K with cytoplasmic p-ERK1/2 was observed in 43/108 (40 %) of cases and of nuclear or cytoplasmic p85aPI3K and Notch-1 in 24/108 (22 %) of cases.
Cytoplasmic expression for p110γPI3K was observed in 45/106 (42 %) of cases, but in only 19/106 (18 %) nuclear expression (Fig. 3). Of the p110γPI3K-positive cases, 80 % showed cytoplasmic p-AKT expression. Coexpression of nuclear or cytoplasmic p110γPI3K with cytoplasmic p-ERK1/2 was observed in 37 % of cases. Moreover, in 30 % of the examined cases, either nuclear or cytoplasmic coexpression of p110γPI3K and Notch-1 was found.
Expression of p-ERK1/2 was mostly cytoplasmic in 88/108 cases (81 %) (Fig. 4). Coexpression of cytoplasmic p-ERK1/2 with all three mTOR pathway components was observed only in 18 % of cases.
Cytoplasmic Notch-1 expression was found in 54/107 cases (50 %) (Fig. 4). Coexpression of Notch-1 with mTOR pathway components was uncommon (19 %), but 44 % of Notch-1-positive cases also expressed p-STAT3. Nuclear expression of p-STAT3 was recorded in 90/108 (83 %) of cases, most of which (60 %) with cytoplasmic coexpression of p-AKT.
Correlation coefficients among analysed molecules are shown in Supplementary Table 4. Expression of p-mTOR was positively correlated with its downstream effectors and with pERK1/2, Notch-1 and p-STAT3, some which of marginal statistical significance. Significant positive correlations also emerged between nuclear expression of p-p70S6K and Notch-1, as well as between cytoplasmic expression of p-AKT and p-4E-BP1. Cytoplasmic expression of p-p70S6K positively correlated with that of p-ERK1/2, p85aPI3K, p110γPI3K as well as p-STAT3. Moreover, cytoplasmic expression of p-4E-BP1 was positively correlated with that of Notch-1, p-AKT, p-STAT3, as well as p110γPI3K and p-ERK1/2. Expression of Notch-1 and p-STAT3 correlated with nuclear expression of p-4EBP1, which also showed a marginally significant inverse correlation with cytoplasmic expression of p85aPI3K and p-ERK1/2. Expression of both subunits of PI3K and p-ERK1/2 was significantly correlated, whereas expression of p85aPI3K was negatively correlated with that of Notch-1. Interestingly, PTEN expression was inversely correlated with that of p-STAT3, nuclear and cytoplasmic p-4E-BP1 as well as with cytoplasmic p110γPI3K. Expression of p-STAT3 was also significantly correlated with that of Notch-1 and marginally significantly with cytoplasmic p-ERK1/2 and p-AKT.
The expression levels of p-AKT, p-p70S6K and p-ERK1/2, by Western blot of fresh frozen tissue from five randomly selected meningioma samples, were found to correlate with immunohistochemical expression (Supplementary Fig. 1), which validates the specificity of the immunohistochemical results. Treatment of HBL-52 cells with the PI3K inhibitor LY294002 (100 μΜ) reduced protein levels of p-AKT, p-p70S6K and p-ERK1/2 in comparison to untreated cells (Supplementary Fig. 2).
Nuclear expression of p-p70S6K, nuclear and cytoplasmic expression of p-4E-BP1, Notch-1 H-scores and histological grade appeared to be positively correlated (Kruskal–Wallis ANOVA, 1 vs 2 vs 3, p = 0.0001, p = 0.0001, p = 0.0055 and p = 0.0089, respectively). A negative correlation was found between cytoplasmic expression of p-p70S6K and p-ERK1/2, PTEN H-score and histological grade (Kruskal–Wallis ANOVA, 1 vs 2 vs 3, p = 0.0223, p = 0.0067 and p = 0.0046, respectively) (Figs. 3, 4 and 5 and Table 1).
Nuclear expression of p-p70S6K and p-ERK1/2 H-scores were higher in women than in men (Mann–Whitney U test, p = 0.0569 and p = 0.0357). PTEN H-score was lower in recurring cases (p = 0.0587). No other significant correlations between protein expression levels and clinicopathological features were found.
Survival analysis
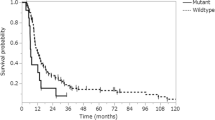
In univariate survival analysis (Table 2), histological grade (1 vs 2 vs 3, p < 0.0001), nuclear p-4E-BP1 expression (p = 0.0486, Fig. 6) and absence of cytoplasmic p-ERK1/2 expression (p = 0.0046, Fig. 6) correlated with adverse prognosis. Upon stratification according to grade (grade 1 meningiomas vs meningiomas grade 2/3), p-ERK1/2 remained significant (p = 0.0047). For all other parameters, significant correlations were not found.
In multivariate survival analysis including only those parameters that were proven to be significant in univariate analysis (Table 3), cytoplasmic p-ERK1/2 expression (HR = 0.078, p = 0.029) along with histological grade (p = 0.030) emerged as independent predictors of prognosis.
Discussion
The standard treatment for meningioma patients consists of surgical resection, followed by radiotherapy for all grade 3 tumours and for those grade 2 tumours for which complete resection (Simpson grade 1 or 2) has not been achieved [17]. However, the role of radiotherapy remains controversial and no effective therapies exist for meningiomas recurring after initial treatment [18]. Despite the identification of key genetic alterations associated with meningioma tumourigenesis and progression, such as the biallelic inactivation of the NF2 gene, the existence of prognostic and predictive biomarkers that might identify therapeutic targets remains an open issue. On a series of well-characterized meningiomas of different grade, our study analysed simultaneously expression of all key members of the PI3K/AKT/mTOR pathway, associations with clinical characteristics and prognosis, and interactions with the MAPK, Notch and JAK/STAT pathways.
A novel finding emerging from our study is that 9 % of meningiomas contain rare oncogenic gain-of-function mutations in exons 9 and 20 of PIK3CA gene, which contrasts with a single previous paper reporting a much lower frequency (<1 %) [19]. Although early reports failed to identify AKT1 mutations in meningioma [20], we found 9 % of our cases mutated, confirming the results of recent studies [21, 22]. AKT1 mut cases displayed a lower cytoplasmic p-ERK1/2 H-score, a finding which has also been observed in urothelial bladder cancer [23]. Interestingly, simultaneous PIK3CA and AKT1 mutations were recorded in one case, indicating that they are not always mutually exclusive [24]. Moreover, mutations were marginally more frequent in men than in women, which accounts for the higher p-ERK1/2 levels in female cases and suggests that the underlying molecular events may vary according to gender [25]. Other mechanisms could be involved in the activation of the PI3K pathway in meningioma, taking into account that mutational activation of PIK3CA was found in a small subset and p110γPΙ3Κ overexpression in approximately 40 % of our cases. These include copy number gains of PIK3CA gene, which have been recorded in around 90 % of grade 2/3 meningiomas [26] or alternatively, upstream events involving RTKs such as PDGFR or FGFR3 or even a direct interaction with the RAS/RAF/MEK/ERK pathway [27–29]. The latter hypothesis is supported by a positive correlation between p110γPI3K and p-ERK1/2 in our cohort.
The number of cases expressing AKT (either cytoplasmic or nuclear) far exceeded the mutation rate of AKT1 gene or the number expressing p110γPI3K, which acts upstream of AKT, although a significant association was documented between the two proteins. In this context, AKT activation might be attributed to other pathways, e.g. STAT3 [30], which we found in the vast majority of cases to be expressed simultaneously with p-AKT at correlating levels, confirming previous studies [31]. It is of interest that, although cytoplasmic expression of p-AKT correlated positively with nuclear expression of p-p70S6K, nuclear levels of these proteins were inversely correlated. In this context, p-AKT seems to play different roles according to its subcellular localization, which has also been observed in other tumours [32, 33].
Contrary to what we expected, we did not find a significant correlation between p-mTOR and p-AKT. The positive correlation of the expression of p-mTOR with that of Notch-1, p110γPI3K or p-STAT3, and marginally with p-ERK1/2, illustrates the complexity of the molecular cues regulating mTOR activation, as has been reported also in other human malignancies [12, 34–38]. All our cases displayed nuclear expression of p-p70S6K and 41 % also cytoplasmic expression, as expected since one of the genetic abnormalities characteristic of meningioma progression is the amplification of the 17q23 locus, in which the RPS6KB1 gene encoding for p70S6K protein is located [28, 39]. In our series, expression of p-4E-BP1 was more often cytoplasmic than nuclear. Furthermore, nuclear expression of p-p70S6K and cytoplasmic expression of p-4E-BP1 was significantly higher than that of p-mTOR. Moreover, no significant correlation was observed between the former and the latter proteins, which implies that mTOR may not be the primary or the sole contributor to p70S6K and 4E-BP1 activation. In fact, p-AKT directly phosphorylates p70S6K via TSC1/2 [40], which might correspond to our findings, since we observed that the majority of nuclear p-p70S6K-positive/p-mTOR-negative cases coexpressed cytoplasmic p-AKT, and a marginally significant positive correlation existed between their expression levels. Inhibition of PI3K with LY294002 in HBL-52 cells reduced both p-AKT and p-p70S6K levels, which further supports the liaison between p-AKT and p-p70S6K. Moreover, nuclear and cytoplasmic expression of p-p70S6K and p-4E-BP1 was positively correlated with that of p-STAT3, whereas p-STAT3 expression was strongly correlated with that of Notch-1, which is in accordance with the notion that Notch-1 is an upstream activator of STAT3 [41]. Collectively, our findings raise the hypothesis that in a subset of meningiomas, Notch-1 phosphorylates and activates STAT3 which in turn promotes AKT activation, resulting in activation of both p70S6K and 4E-BP1.
Interestingly, cytoplasmic expression of p-p70S6K and p-4E-BP1 positively correlated with p-mTOR and p110γPI3K expression, whereas expression of p-p70S6K correlated with that of p85αPI3K and p-4E-BP1 with p-AKT. These observations suggest that PI3K/AKT/mTOR signalling activates both cytoplasmic p70S6K and 4E-BP1 and also that nuclear and cytoplasmic expression of p-p70S6K is differentially regulated, since only cytoplasmic expression of p-p70S6K correlated with p-ERK1/2 expression, in agreement with the reported activation of p70S6K by MAPK through ribosomal p90rsk [42]. The borderline negative correlation of nuclear p-4E-BP1 expression with that of p-ERK1/2, and its borderline positive correlation with cytoplasmic expression of p-4E-BP1, suggests that the MAPK pathway may either activate [43] or inhibit 4E-BP1 [44], depending on its subcellular localization.
We found nuclear and cytoplasmic expression of p-p70S6K to be inversely correlated. Although nuclear p-p70S6K expression by immunohistochemistry is rather unusual, it has been reported in various tumours including astrocytomas [45]. Moreover, for p-p70S6K, distinct correlations with components of the examined pathways have been identified, depending on its subcellular localization, which highlights separate roles of nuclear and cytoplasmic p-p70S6K as has been reported previously for gastric carcinoma [46].
We found, as previously reported [47], that the MAPK pathway plays an essential role in meningioma initiation but not progression. In contrast, high p-p70S6K and p-4E-BP1 expression in grade 2/3 meningiomas fits with amplification of the 17q23 locus during meningioma progression [28, 39] also reported in gliomas [45]. Furthermore, high Notch-1 expression in grade 2/3 meningiomas fits with its involvement into meningioma progression similar to what has been reported for gliomas [37, 48]. Our finding of nuclear PTEN staining in all meningioma cases is intriguing. Several reports point to the distinct functions of PTEN according to its subcellular localization [49–51]. While cytoplasmic PTEN acts primarily as phosphatase to inhibit PI3K signalling, nuclear PTEN is implicated in many functions which are unrelated to this activity [51]. Our finding of a negative correlation between the expression of PTEN and p-STAT3 is consistent with PTEN operating as a negative regulator of STAT3 activation [38]. Importantly, PTEN expression was found to inversely correlate with meningioma grade and the probability of recurrence, in agreement with observations on other tumours [52–54].
In terms of prognostic significance, we found in univariate survival analysis grade 2/3 histology and nuclear p-4E-BP1 expression associated with poor survival, as has been reported previously for other malignancies [16, 45, 55]. In contrast, high cytoplasmic p-ERK1/2 expression associated with improved overall survival in the entire cohort as well as in grade 1 meningioma, which is in harmony with the reported association between high expression of p-ERK1/2 and the likelihood of meningioma recurrence [47]. The prognostic impact of activated ERK1/2 in tumourigenesis is controversial with published studies highlighting elevated p-ERK1/2 as a favourable as well as an unfavourable prognosticator according to tumour type or subcellular localization [56–62]. The latter has been proposed to define its activities, i.e. targeting of proteins localized in either the cytoplasm or the nucleus [reviewed in 63]. Interestingly, in multivariate survival analysis, tumour grade as well as p-ERK1/2 expression retained their statistical significance as independent prognosticators, indicating that the relationship of p-ERK1/2 with prognosis cannot be attributed to its correlation with histological grade.
In summary, we describe for the first time an almost 9 % frequency of gain-of-function mutations within PIK3CA gene and confirm recently published findings regarding AKT1 mutations in meningioma. Moreover, we show that components of the PI3K/AKT/mTOR pathway establish complex interactions either with each other or with components of MAPK-, JAK/STAT- and Notch-1-mediated pathways, which appear to facilitate and fuel tumour progression. The results obtained with LY294002 in the meningioma cell line encourage further investigations into the potential use of PI3K pathway inhibitors for the treatment of meningioma. Finally, our survival analysis identifies expression of p-ERK1/2 as a favourable prognostic factor for meningioma patient survival.
References
Pham MH et al (2011) Molecular genetics of meningiomas: a systematic review of the current literature and potential basis for future treatment paradigms. Neurosurg Focus 30:E7
Riemenschneider MJ, Perry A, Reifenberger G (2006) Histological classification and molecular genetics of meningiomas. Lancet Neurol 5:1045–1054
Jaskolski DJ et al (2013) Molecular alterations in meningiomas: association with clinical data. Clin Neuropathol 32:114–121
Barbera S et al (2013) Genetic changes with prognostic value in histologically benign meningiomas. Clin Neuropathol 32:311–317
Yuan TL, Cantley LC (2008) PI3K pathway alterations in cancer: variations on a theme. Oncogene 27:5497–5510
De Luca A, Maiello MR, D’Alessio A, Pergameno M, Normanno N (2012) The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin Ther Targets 16:S17–S27
Logue JS, Morrison DK (2012) Complexity in the signaling network: insights from the use of targeted inhibitors in cancer therapy. Genes Dev 26:641–650
Vogt PK, Hart JR (2011) PI3K and STAT3: a new alliance. Cancer Discov 1:481–486
Gutierrez A, Look AT (2007) NOTCH and PI3K-AKT pathways intertwined. Cancer Cell 12:411–413
Wong GW, Knowles GC, Mak TW, Ferrando AA, Zúñiga-Pflücker JC (2012) HES1 opposes a PTEN-dependent check on survival, differentiation, and proliferation of TCRβ-selected mouse thymocytes. Blood 120:1439–1448
Gymnopoulos M, Elsliger MA, Vogt PK (2007) Rare cancer-specific mutations in PIK3CA show gain of function. Proc Natl Acad Sci U S A 104:5569–5574
Steelman LS et al (2011) Roles of the Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR pathways in controlling growth and sensitivity to therapy—implications for cancer and aging. Aging (Albany NY) 3:192–222
Carpten JD et al (2007) A transforming mutation in the pleckstrin homology domain of AKT1 in cancer. Nature 448:439–444
Chetram MA, Hinton CV (2012) PTEN regulation of ERK1/2 signaling in cancer. J Recept Signal Transduct Res 32:190–195
Korkolopoulou P et al (2012) A comprehensive immunohistochemical and molecular approach to the PI3K/AKT/mTOR (phosphoinositide 3-kinase/v-akt murine thymoma viral oncogene/mammalian target of rapamycin) pathway in bladder urothelial carcinoma. BJU Int 110:E1237–E1248
Rojo F et al (2007) 4E-binding protein 1, a cell signaling hallmark in breast cancer that correlates with pathologic grade and prognosis. Clin Cancer Res 13:81–89
Mair R, Morris K, Scott I, Carroll TA (2011) Radiotherapy for atypical meningiomas. J Neurosurg 115:811–819
Vranic A, Peyre M, Kalamarides M (2012) New insights into meningioma: from genetics to trials. Curr Opin Oncol 24:660–665
Pang JC et al (2006) Rare mutation of PIK3CA in meningiomas. Acta Neuropathol 111:284–285
Eom HS, Kim MS, Hur SY, Yoo NJ, Lee SH (2009) Absence of oncogenic AKT1 E17K mutation in prostate, esophageal, laryngeal and urothelial carcinomas, hepatoblastomas, gastrointestinal stromal tumors and malignant meningiomas. Acta Oncol 48:1084–1085
Brastianos PK et al (2013) Genomic sequencing of meningiomas identifies oncogenic SMO and AKT1 mutations. Nat Genet 45:285–289
Clark VE et al (2013) Genomic analysis of non-NF2 meningiomas reveals mutations in TRAF7, KLF4, AKT1, and SMO. Science 339:1077–1080
Juanpere N et al (2012) Mutations in FGFR3 and PIK3CA, singly or combined with RAS and AKT1, are associated with AKT but not with MAPK pathway activation in urothelial bladder cancer. Hum Pathol 43:1573–1582
McCubrey JA et al (2012) Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR cascade inhibitors: how mutations can result in therapy resistance and how to overcome resistance. Oncotarget 3:1068–1111
Konstantinidou AE et al (2003) Hormone receptors in non-malignant meningiomas correlate with apoptosis, cell proliferation and recurrence-free survival. Histopathology 43:280–290
Beckner ME et al (2013) Low-level amplification of oncogenes correlates inversely with age for patients with nontypical meningiomas. World Neurosurg 79:313–319
Johnson MD, O’Connell MJ, Pilcher W, Reeder JE (2010) Fibroblast growth factor receptor-3 expression in meningiomas with stimulation of proliferation by the phosphoinositide 3 kinase-Akt pathway. J Neurosurg 112:934–939
Johnson MD, Okedli E, Woodard A, Toms SA, Allen GS (2002) Evidence for phosphatidylinositol 3-kinase-Akt-p7S6K pathway activation and transduction of mitogenic signals by platelet-derived growth factor in meningioma cells. J Neurosurg 97:668–675
Roberts PJ, Der CJ (2007) Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 26:3291–3310
Xu Q et al (2005) Targeting Stat3 blocks both HIF-1 and VEGF expression induced by multiple oncogenic growth signaling pathways. Oncogene 24:5552–5560
Johnson MD, O’Connell M, Vito F, Bakos RS (2009) Increased STAT3 and synchronous activation of Raf-1-MEK-1-MAPK, and phosphatidylinositol 3-Kinase-Akt-mTOR pathways in atypical and anaplastic meningiomas. J Neurooncol 92:129–136
Le Page C, Koumakpayi IH, Alam-Fahmy M, Mes-Masson AM, Saad F (2006) Expression and localisation of Akt-1, Akt-2 and Akt-3 correlate with clinical outcome of prostate cancer patients. Br J Cancer 94:1906–1912
Badve S et al (2010) Subcellular localization of activated AKT in estrogen receptor- and progesterone receptor-expressing breast cancers: potential clinical implications. Am J Pathol 176:2139–2149
Dobashi Y et al (2009) Critical and diverse involvement of Akt/mammalian target of rapamycin signaling in human lung carcinomas. Cancer 115:107–118
Johnson SM et al (2010) Novel expression patterns of PI3K/Akt/mTOR signaling pathway components in colorectal cancer. J Am Coll Surg 210:767–778
Levidou G et al (2013) A comprehensive immunohistochemical approach of AKT/mTOR pathway and p-STAT3 in mycosis fungoides. J Am Acad Dermatol 69:375–384
Zhao N, Guo Y, Zhang M, Lin L (2010) AKT-mTOR signalling is involved in Notch-1-mediated gliomas cell survival and proliferation. Oncol Rep 23:1443–1447
Zhou J et al (2007) Activation of the PTEN/mTOR/STAT3 pathway in breast cancer stem-like cells is required for viability and maintenance. Proc Natl Acad Sci U S A 104:16158–16163
Pachow D et al (2013) mTORC1 inhibitors suppress meningioma growth in mouse models. Clin Cancer Res 19:1180–1189
Riemenschneider MJ, Betensky RA, Pasedag SM, Louis DN (2006) AKT activation in human glioblastomas enhances proliferation via TSC2 and S6 kinase signaling. Cancer Res 66:5618–5623
Ge W, Ren J (2012) mTOR-STAT3-notch signalling contributes to ALDH2-induced protection against cardiac contractile dysfunction and autophagy under alcoholism. J Cell Mol Med 16:616–626
Pende M et al (2004) S6K1(−/−)/S6K2(−/−) mice exhibit perinatal lethality and rapamycin-sensitive 5′-terminal oligopyrimidine mRNA translation and reveal a mitogen-activated protein kinase-dependent S6 kinase pathway. Mol Cell Biol 24:3112–3124
Cohen JD et al (2011) ERK crosstalks with 4EBP1 to activate cyclin D1 translation during quinol-thioether-induced tuberous sclerosis renal cell carcinoma. Toxicol Sci 124:75–87
Rolli-Derkinderen M et al (2003) ERK and p38 inhibit the expression of 4E-BP1 repressor of translation through induction of Egr-1. J Biol Chem 278:18859–18867
Korkolopoulou P et al (2012) Phosphorylated 4E-binding protein 1 (p-4E-BP1): a novel prognostic marker in human astrocytomas. Histopathology 61:293–305
Xiao L, Wang YC, Li WS, Du Y (2009) The role of mTOR and phospho-p70S6K in pathogenesis and progression of gastric carcinomas: an immunohistochemical study on tissue microarray. J Exp Clin Cancer Res 28:152
Mawrin C et al (2005) Different activation of mitogen-activated protein kinase and Akt signaling is associated with aggressive phenotype of human meningiomas. Clin Cancer Res 11:4074–4082
Jiang L et al (2011) Notch 1 expression is upregulated in glioma and is associated with tumor progression. J Clin Neurosci 18:387–390
Chung JH, Eng C (2005) Nuclear-cytoplasmic partitioning of phosphatase and tensin homologue deleted on chromosome 10 (PTEN) differentially regulates the cell cycle and apoptosis. Cancer Res 65:8096–8100
Gimm O et al (2000) Differential nuclear and cytoplasmic expression of PTEN in normal thyroid tissue, and benign and malignant epithelial thyroid tumors. Am J Pathol 156:1693–1700
Song MS, Salmena L, Pandolfi PP (2012) The functions and regulation of the PTEN tumor suppressor. Nat Rev Mol Cell Biol 13:283–296
Choucair K et al (2012) PTEN genomic deletion predicts prostate cancer recurrence and is associated with low AR expression and transcriptional activity. BMC Cancer 12:543
McMenamin ME et al (1999) Loss of PTEN expression in paraffin-embedded primary prostate cancer correlates with high Gleason score and advanced stage. Cancer Res 59:4291–4296
Mueller S et al (2012) PTEN promoter methylation and activation of the PI3K/Akt/mTOR pathway in pediatric gliomas and influence on clinical outcome. Neuro Oncol 14:1146–1152
Castellvi J et al (2006) Phosphorylated 4E binding protein 1: a hallmark of cell signaling that correlates with survival in ovarian cancer. Cancer 107:1801–1811
Adeyinka A et al (2002) Activated mitogen-activated protein kinase expression during human breast tumorigenesis and breast cancer progression. Clin Cancer Res 8:1747–1753
Chadha KS et al (2006) Activated Akt and Erk expression and survival after surgery in pancreatic carcinoma. Ann Surg Oncol 13:933–939
Givant-Horwitz V et al (2003) Mitogen-activated protein kinases (MAPK) as predictors of clinical outcome in serous ovarian carcinoma in effusions. Gynecol Oncol 91:160–172
Milde-Langosch K et al (2005) Expression and prognostic relevance of activated extracellular-regulated kinases (ERK1/2) in breast cancer. Br J Cancer 92:2206–2215
Mizumoto Y et al (2007) Activation of ERK1/2 occurs independently of KRAS or BRAF status in endometrial cancer and is associated with favorable prognosis. Cancer Sci 98:652–658
Schmitz KJ et al (2007) Activation of extracellular regulated kinases (ERK1/2) but not AKT predicts poor prognosis in colorectal carcinoma and is associated with k-ras mutations. Virchows Arch 450:151–159
Vicent S et al (2004) ERK1/2 is activated in non-small-cell lung cancer and associated with advanced tumors. Br J Cancer 90:1047–1052
Saetta AA et al (2011) Expression of pERK and pAKT in human astrocytomas: correlation with IDH1-R132H presence, vascular endothelial growth factor, microvascular characteristics and clinical outcome. Virchows Arch 458:749–759
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Elias A. El-Habr, Georgia Levidou, Eleni-Andriana Trigka and Joanna Sakalidou have equally contributed to this manuscript.
Angelica A. Saetta and Penelope Korkolopoulou are senior authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOC 60 kb)
Supplementary Table 2
(DOC 67 kb)
Supplementary Table 3
(DOC 106 kb)
Supplementary Table 4
(DOC 189 kb)
Fig. S1
p-AKT, p-p70S6K, and p-ERK1/2 protein levels in fresh frozen tissue specimens by Western blot and immunohistochemical expression of p-AKT, p-p70S6K and p-ERK1/2 in formalin-fixed tissue from the same five specimens. (A) Detection of p-AKT in the five cases. Tumours #2, 5 showed the highest p-AKT levels in frozen and paraffin-embedded samples. (B) Detection of p-p70S6K in the five cases. Tumours #1, 3, 5 showed the highest p-p70S6K levels in frozen and paraffin-embedded samples. (C) Detection of p-ERK1/2 in the five cases. Tumours #1, 2, 5 showed the highest p-ERK1/2 levels in frozen and paraffin-embedded samples (GIF 257 kb)
Fig. S2
Detection of p-AKT, p-p70S6K and p-ERK1/2 protein levels in the meningioma cell line HBL-52 before and after 3 h treatment with PI3K inhibitor LY294002 (100 μM). Actin was used as an internal control for Western blot analysis in all experiment (GIF 24 kb)
Rights and permissions
About this article
Cite this article
El-Habr, E.A., Levidou, G., Trigka, EA. et al. Complex interactions between the components of the PI3K/AKT/mTOR pathway, and with components of MAPK, JAK/STAT and Notch-1 pathways, indicate their involvement in meningioma development. Virchows Arch 465, 473–485 (2014). https://doi.org/10.1007/s00428-014-1641-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-014-1641-3