Abstract
Steatosis in donor livers is an accepted adverse prognostic factor after liver transplantation. While its semiquantitative assessment shows varying reproducibility, it is questioned as a standard method. Additionally, the influence of hepatic steatosis on ischemia/reperfusion injury (I/R injury) has not been evaluated in biopsies after reperfusion. We compared different staining and analyzing methods for the assessment of donor liver steatosis in order to predict I/R injury and clinical outcome after transplantation. To do this, 56 paired pre- and post-reperfusion liver biopsies were analyzed for macro- (MaS)/micro- (MiS) and total steatosis in cryo and permanent sections by special fat (Oil Red O or ORO) and standard stains. Computerized morphometrical analyses were compared to the semiquantitative assessment by a pathologist. I/R injury was determined histopathologically and by M30 immunohistochemistry. We found ORO to be more sensitive in detecting hepatic steatosis with higher reproducibility for MaS. Semiquantitative analyses were highly reproducible and not inferior to computerized morphometry. Categorized MaS as determined by ORO correlated with the extent of I/R injury, initial poor function, liver enzymes, and survival. Therefore fat stains like ORO are a reliable and easy method comprising significant advantages in the evaluation of hepatic steatosis and are thereby of prognostic value. Computerized analysis is a precise tool though not superior to semiquantitative analyses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Quality determination of the donor liver is important to extend the pool for liver transplantation [1–4]. The acceptability of donor livers for transplantation is generally assessed by the combination of donor blood tests and clinical data/patient history, ultrasound, gross anatomical appearance judged by the surgeons, and histo-pathological microscopic evaluation. Significant liver steatosis is a risk factor for operative complications after major hepatectomy and liver transplantation [5]. Mild macrosteatosis (MaS) will not affect the clinical outcome [1], while more severe steatosis (MaS > 30 %) is often associated with graft dysfunction within 7 days after transplantation [6]. Because of the limited period of time between liver explantation and transplantation, the determination of the degree of steatosis mostly depends on frozen sections, if a pathological service is available [3]. However, the major limitations of frozen sections are lower histopathological definition compared to permanent paraffin sections (FFPE) and the risk of underestimating macrosteatosis and overestimating microsteatosis (MiS) [7]. Up to now, the influence of MiS versus MaS on post-transplant outcome remains debatable [8]. While some authors showed that a moderate degree of MiS increases the rate of organ failure after transplantation [9], other groups found no negative impact on graft outcome regardless of the degree of MiS [10, 11].
One limitation of many studies is the single use of hematoxylin–eosin (HE)-stained tissue slides to evaluate the lipid content of the liver [8]. Only few authors applied specific fat stainings such as Oil red O (ORO), which are more sensitive than conventional HE stainings to discriminate and evaluate the extent of MaS and MiS [11, 12]. Another point of debate is the interobserver variability seen in quantitative and qualitative assessments of steatosis by pathologists. While some authors demonstrated a high degree of congruency between different pathologists [6], others even questioned the use of histopathological examinations due to poor agreement among pathologists and called for a computerized system to assess lipid content [13].
Due to a permanent full-time and year-long pathology service, our institution is able to provide frozen sections for the evaluation of liver grafts any time. This results in high rates of availability of both pre- and 1-h post-reperfusion biopsies of the same patient. Our approach enables us to evaluate the role of steatosis on ischemia–reperfusion (I/R) injury and, due to the availability of fresh frozen tissue, to compare conventional HE with special fat stains (ORO). Additionally, we compared the usual semiquantitative analysis as performed by a pathologist to a computerized morphometrical quantification of liver steatosis.
We demonstrate that MaS in ORO-stained pre-reperfusion biopsies correlates significantly with I/R injury and clinical outcome with high interobserver concordance. Furthermore, ORO-based assessment of steatosis proves to be superior to HE-stained frozen sections. Computerized morphometrical assessment failed to prove superiority over semiquantitative analyses.
Methods
Study design
From April 2010 to December 2011, 56 paired cases of pre- and post-reperfusion liver biopsies from patients who received orthotopic liver transplantations (OLT) at the University Hospital of Essen due to different causative factors were available for further analyses. Corresponding recipient and donor data were gathered. The institutional review board approved the study (12-5195-BO).
Recipient and donor characteristics
Recipient data included age, gender, body mass index (BMI), indication for OLT, pre-operative figures as Model of End Stage Liver Disease (MELD) score, intensive care unit (ICU), ward stay, and post-operative figures such as concentration of alanine transaminase (ALT (IU/L)) and aspartate transaminase (AST (IU/L)) within 72 h after OLT, initial poor function (IPF; ALT and/or AST ≥ 1,500 IU/L within 72 h after OLT) [14], primary non-function (PNF; re-transplantation or death within 7 days of initial OLT) [15], and death of the recipient in the observed time period. Donor data involved age, gender, BMI, and warm and cold ischemic time (WIT/CIT).
Tissue samples
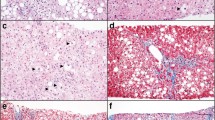
The pre-reperfusion biopsy (Fig. 1a–f) was provided by the explanting surgeon via the German Organ Transplantation Foundation (DSO), and the post-reperfusion biopsy (Fig. 1g–h) was taken during the transplantation process 1 h after re-establishing blood flow. The pre-reperfusion biopsy was split into two parts if possible, one of which was handled to frozen section and the other formalin fixed. Due to possible high fat content, freezing process of the tissue was conducted in a cryostat at −40 °C in the fast-freezing position on a metal disc. Air temperature within the cryostat itself was −20 °C, and the frozen sections were cut to 3-μm thickness. The frozen sections were stained with HE (Fig. 1a) and ORO (Online Resource 1; Fig. 1b). The staining process needed approximately 12–15 min for ORO and 7–8 min for HE. After frozen-section processing, the second part was transferred into formalin as well. Afterwards, both parts were separately embedded in paraffin and HE stained (3-μm slide thickness) according to institutional standards (Fig. 1e).
Hepatic steatosis and PMNs as indicators of I/R injury in pre- (a–f) and post-reperfusion biopsies (g–h). Displayed is the case with highest MaS (40 % in ORO). All × 400 (d magnified detail). a Frozen section (HE): As in other organs, frozen section displays low histopathological definition with higher rate of technique-related artifacts compared to definitive permanent section (e). b Corresponding frozen section (ORO): Lipid vacuoles appear as red droplets distinctly contrasting to the blue, hematoxylin-stained background which allows identification of cellular structures. c Computerized morphometrical image of b: ORO positive, red-stained lipid droplets are recognized by the software, and green pixels are superimposed, thus allowing computerized calculation of the area covered by lipid vacuoles in relation to the lobular, cell covered, non-portal tissue area. By adjusting the cut-off value of lipid-covered area size, the software is able to discriminate between MiS and MaS. Displayed d is a magnified detail in which macro-steatotic droplets are automatically encircled yellow, and accordingly, micro-steatotic droplets are encircled green. Computerized processing including encirculation and calculation of one ORO-stained image takes approximately 5 s. e Permanent section of a: Morphology is improved compared to frozen section. f Analysis of PMNs as indicators of I/R injury in pre-reperfusion biopsies. Although already markedly steatotic, a low rate of lobular PMNs is found compared to the 1-h post-reperfusion biopsy (h). g One-hour post-reperfusion biopsy in FFPE permanent section. h Analysis of PMNs in the corresponding 1-h post-reperfusion biopsy with marked increase in lobular PMNs compared to the pre-reperfusion biopsy (f). f, h: Naphthol-AS-D-chloracetate-esterase staining (ASDCL)
Accordingly, the post-reperfusion biopsy was instantly fixed in formalin, embedded in paraffin (FFPE), and HE stained (Fig. 1g). The average length was 12.7 mm (median 12 mm) for the pre- and 12.5 mm (median 12 mm) for the post reperfusion biopsies.
Histopathological semiquantitative evaluation of steatosis
Steatosis as a percentage of total hepatocytes was determined by two different experienced pathologists (HB and PTP) in terms of MaS and MiS. Both observers were blinded from each other and evaluated the biopsies separately. MaS was classified as “mild” if less than 30 % of hepatocytes were affected and “moderate” in cases of 30–60 %. The classification of “severe” (>60 %) was not applied due to the absence of such a degree of MaS in the present transplanted organs. Total steatosis (ToS) was defined as the sum of MaS and MiS.
Rules for classification of MaS and MiS were applied both in ORO and HE stains. MaS was defined as mostly a single droplet in a hepatocyte, displacing the nucleus to the periphery and at least the size of the nucleus. MiS was defined as at least one, usually numerous intracytoplasmatic droplets in hepatocytes not displacing and of smaller size than the nucleus [8].
Computerized morphometrical evaluation of steatosis
Computerized morphometrical quantification of liver steatosis of pre-reperfusion ORO-stained frozen sections (Fig. 1c–d) was performed using a camera-equipped microscope (Zeiss Axioplan, Carl Zeiss Microscopy GmbH, Jena, Germany) “AxioVision” (V4.6.3.0, Carl Zeiss Microscopy GmbH). Eight representative images per biopsy made with × 40 objective were evaluated and averaged. The images were chosen not to contain portal areas. The software was able to detect characteristic intense red (ORO) signals and recognized them as fat droplets. In the first step, the fat droplets were automatically overlaid green by the software (Fig. 1c), thus allowing the calculation of the area covered by fat and the calculation of the total tissue area from the negative image. Non-cell covered, empty tissue/slide areas were automatically subtracted from the total tissue area by the software. Due to variation in nuclear size, two different analyses were conducted for each case with cut-off values of 20 and 40 μm2, defining cut-off values for MiS and MaS, respectively. Micro-steatotic droplets were encircled green, and macro-steatotic droplets yellow (Fig. 1d), thus allowing the software to discriminate MiS and MaS. The results of MiS and MaS were expressed as percentages of tissue area.
For semiquantitative and morphometrical evaluation, the same slides were used throughout. Altogether, the whole process of digital evaluation of hepatic steatosis took approximately 20 min per case including recording of digital images.
Histopathological evaluation of ischemia–reperfusion injury
I/R injury represented by the presence of polymorphonuclear leukocytes (PMNs), apoptosis, and hepatocellular ballooning was determined in both pre- and post-reperfusion biopsies [16]. Other indicators of I/R injury as cholestasis or necrosis [17] were not taken into consideration as quantification of these parameters is non-objective.
PMNs were counted in Naphthol-AS-D-chloracetate-esterase staining (ASDCL) in three representative fields (periportal, lobular, and pericentral) in × 400 magnification (Fig. 1f,h). Apoptotic cells were detected by M30 immunohistochemistry. It identifies a neo-epitope of cytokeratin 18 indicating caspase activity typically present in apoptotic cells [18]. Paraffin sections were dewaxed, and antigen retrieval was carried out with Target Retrieval (Dako, S2367, Glostrup, Denmark) at pH 9.0 for 20 min in a hot water bath (96 °C). The primary antibody (M30, #10700, TECO medical, Switzerland) was incubated for 30 min at room temperature (dilution 1:8,000). Antibody binding was detected with the Zytochem Plus AP Polymer Kit (Zytomed Systems, Berlin, Germany). Negative controls were included in every run.
The apoptotic cells themselves exhibited a distinct and intense red cytoplasmatic staining. Only apoptotic hepatocytes were taken into consideration. M30-positive hepatocytes were manually counted in four representative areas (×200 magnification), and a mean value was calculated. Hepatocellular ballooning was determined in a four-tiered scheme (none, mild, moderate, and strong) by two blinded observers (PTP and HR).
Statistical analyses
For statistical analyses, SPSS (V19, IBM, Armonk, NY, USA) was used. Graphics were created with GraphPad Prism version 6.00 (GraphPad Software, La Jolla California, USA). The Wilcoxon signed-rank test (Wilcoxon) was used to assess the significance of the differences between the parameters obtained by pre- and post-reperfusion biopsies. Due to not normally distributed values, the Spearman's rank correlation coefficient (Spearman) was chosen to reveal the statistical dependence between steatosis and histological damage as well as between steatosis and clinical outcome. It was also used to calculate the interobserver reliability. When appropriate, the values were dichotomized at median level. In those cases, the Mann–Whitney U test (Mann–Whitney) was chosen to evaluate statistical significance. For determination of survival, Kaplan–Meier analyses were performed. P values <0.05 were regarded statistically significant.
Results
Recipients' characteristics and clinical outcome
From the nine patients deceased in the analyzed period after OLT, three died within the first week. The person who required re-transplantation deceased in the course of observation. Median follow-up time was 233 days (SD 199). One-year survival rate was 84 %. Occurrence of IPF and PNF was tightly correlated to shortened survival (p < 0.0001). Moreover, the necessity of prolonged intensive care therapy was an adverse prognostic factor for survival (p = 0.051). Four recipients exhibited MELD scores ≥30 at the time of transplantation, of which three (75 %) deceased in the course of observation. For detailed information on patients' characteristics, see Table 1.
Donor characteristics
Donor livers were procured from 56 deceased patients (29 females, 27 males) with a median age of 61 years (SD 18.7) and a median BMI of 26.5 kg/m2 (SD 5.4). In four cases, the donors were 18 years or younger. The median CIT was 410 min (SD 75.5), and the median WIT was 33 min (SD 7.2).
Semiquantitative evaluation of steatosis in pre-reperfusion biopsies
In semiquantitative analyses of cryo tissue, all kinds of steatosis (ToS, MaS, and MiS) were evaluated higher in ORO compared to those in HE-stained pre-reperfusion biopsies (Table 2, Fig. 2). MiS was strongly underestimated in cryo HE stains compared to ORO stains (Fig. 2). Also, low-level concordance in MiS evaluation was found between HE stains in cryo-tissue and FFPE (Table 2, Fig. 2), displaying an overestimation of MiS in cryo HE compared to FFPE. MaS was underestimated in cryo HE stains compared to both ORO and FFPE (Table 2, Fig. 2). ToS figures form an intermediate group mostly parallel to MiS due to much higher impact of MiS than MaS on ToS (Table 2, Fig. 2).
Semiquantitative evaluation of steatosis using standard (HE) and special fat stains (ORO) in cryo and FFPE-tissue. Higher sensitivity in detection of steatosis of ORO compared to HE staining is observable. MiS is overestimated, and MaS underestimated in HE-stained frozen sections compared to permanent, FFPE sections
Semiquantitative evaluation of steatosis in post-reperfusion biopsies
Median values in HE-stained, FFPE-based permanent sections in post-reperfusion biopsies were 15 % for MiS (SD 35.5, range 0–90 %), 2 % for MaS (SD 7, range 0–32 %), and 23 % for ToS (SD 38.6, range 0–100 %) (Fig. 2). There was a significant increase in MiS from pre- to post-reperfusion biopsies (HE, FFPE; factor 2.1 for mean value; p < 0.0001) and a non-significant borderline decrease in MaS (HE, FFPE; factor 1.2 for mean value). Figures for ToS run parallel to those for MiS (HE, FFPE; p = 0.003) due to high impact of MiS on ToS in relation to MaS.
Morphometrical evaluation of steatosis in pre-reperfusion biopsies
In computerized morphometrical evaluation of ORO-stained pre-reperfusion biopsies, good to very good correlations to semiquantitative analyses of the same slides were noticed (Table 3).
Reproducibility
Moderate to good correlation was seen for MiS (cryo ORO r = 0.576, cryo HE r = 0.660, FFPE HE r = 0.714) with very good values for MaS (cryo ORO r = 0.881, cryo HE r = 0.835, FFPE HE r = 0.923) regarding semiquantitative analyses of the two blinded observers (HB and PTP).
Donor data and steatosis
When dichotomized at 25.0 kg/m2, higher donor BMI was correlated with higher ToS in post-reperfusion biopsies (FFPE, HE; p = 0.017). Older age was partly correlated with lower degrees of MiS and ToS in pre-reperfusion biopsies (FFPE, HE; p values 0.003 and 0.006, respectively) and with MiS in morphometrical analyses of the same biopsies (ORO, cryo; p values 0.049 (20 μm2) and 0.035 (40 μm2)). Male gender was associated with higher degree of ToS (ORO, cryo; p = 0.041) and MiS (HE, cryo; p = 0.015) in pre-reperfusion biopsies. CIT exhibited correlations with MiS and ToS in post-reperfusion biopsies (FFPE, HE; p values 0.011 and 0.025, respectively). No association was found with WIT.
I/R injury
The median number of PMNs rose significantly from pre- to post-reperfusion biopsies (median values 45 ± 26.8 SD vs. 77.5 ± 72.7 SD; Wilcoxon p < 0.0001; Fig. 1f,h, Fig. 3a). The same was true for the rate of apoptotic cells identified by M30 immunohistochenistry which increased significantly (mean values 1.25 ± 0.955 SE vs. 10.38 ± 4.05; Wilcoxon p < 0.0001; Fig. 3b). Also, the extent of hepatocellular ballooning significantly rose from pre- to post-reperfusion biopsies (32 positive, 5 negative deviations; Wilcoxon p < 0.0001). No significant impact of parameters of I/R injury on survival was noted.
Steatosis and I/R injury
In categorized analyses of MaS (mild vs. moderate; cut-off 30 %; ORO frozen) and parameters of I/R injury, a significant association with dichotomized values of PMNs was detected (p = 0.03, Fig. 3c). Parallel analyses in HE-stained frozen sections were not feasible, due to only one single case of moderate MaS in HE staining evaluation. No associations of steatosis and M30-based apoptotic rate or hepatocellular ballooning were found.
Donor/graft data and I/R injury
Donor age and BMI were correlated in a positive fashion to the number of PMNs in post-reperfusion biopsies (p = 0.009 and p = 0.015, respectively). Additionally, older donor age was associated with stronger hepatocellular ballooning in post-reperfusion biopsies (p = 0.013). No other associations were noted.
Steatosis and clinical outcome
No correlations were found between degree of steatosis and length of ward stay, and PNF and requirement for Re-OLT. However, correlations between morphometrical values of MiS and ToS (ORO, cryo) in pre-reperfusion biopsies with length of ICU stay were detected (p = 0.013 (20 μm2)/p = 0.017 (40 μm2) and p = 0.028, respectively). These results were confirmed in dichotomized analyses (p = 0.02/p = 0.042 and p = 0.042, respectively).
Additionally, categorized (mild vs. moderate) data of MaS (ORO, cryo) exhibited a significant correlation with AST on first and borderline association on second post-OLT day (p = 0.037, p = 0.068). The same was true for ALT (day 1 p = 0.028, day 2 p = 0.066, day 3 p = 0.062). In dichotomized analyses, MaS evaluated by morphometry was associated with dichotomized AST on first post-OLT day (ORO, cryo; p = 0.041). Furthermore, categorized MaS (ORO cryo) was associated with occurrence of IPF (p = 0.006) and survival in Kaplan–Meier analyses (p = 0.045). Dichotomized MaS at median level in pre- and post-reperfusion biopsies had borderline influence on survival (both HE, FFPE; p = 0.06, p = 0.056).
Discussion
Due to a shortage of livers for transplantation, extended donor criteria livers are frequently accepted [1–4]. In these organs, the histopathological evaluation of steatosis is a major factor in quality assessment. While diverse techniques have been suggested in the evaluation of hepatic steatosis, a combination of the surgeon's macroscopic and the pathologist's microscopic assessment by frozen section is still considered to be a gold standard [19, 20].
Due to the availability of a pathologist's service day and night all year long, our institution is able to provide frozen section evaluation of grafts for transplantation any time. The availability of this service and of pre- and 1-h post-reperfusion biopsies put us in a position to (1) compare two staining methods (HE and ORO) in the setting of frozen section of pre-reperfusion biopsies, (2) compare two methods/inter-observer variability of steatosis evaluation (pathologist's semiquantitative and computerized morphometrical), and (3) relate the results to I/R injury in post-reperfusion biopsies and clinical outcome.
Regarding established criteria [19], macro-steatosis (MaS) was predominantly mild (<30 %) in pre-reperfusion, HE-stained frozen section evaluation. Only in one case, MaS was classified as moderate (35 %). In semiquantitative ORO analyses of the same biopsies, MaS was lower than 30 % in 47 cases (85 %), while 8 cases (15 %) displayed moderate steatosis with a maximum of 40 % (Table 2).
In general, all varieties of steatosis (MaS, MiS, and ToS) were estimated lower in HE compared to ORO staining (Table 2, Fig. 1a,b and 2), i.e., HE-stained frozen section analyses display a lower sensitivity for the detection of steatosis than special fat stains like ORO [11, 12, 19, 21]. Possible reasons in case of MiS might be the inability to discriminate fat vacuoles from glycogen accumulation in HE-stained frozen sections [7]. In case of MaS entrapment of water droplets during the freezing process, reactive ballooning of hepatocytes and the lower histopathological definition of frozen sections in general might add to misinterpreting HE-stained slides. Special fat stains like ORO are able to resolve these problems due to the high contrast of deep red-stained fat vacuoles to faint background tissue staining (Fig. 1b).
Additionally, we found an overestimation of MiS (factor 2.0) and underestimation of MaS (factor 1.6) in HE-stained frozen section compared to FFPE-based, HE-stained permanent sections (Table 2, Fig. 1a,e, Fig. 2) which were in line with the results of Lo et al. [7]. This discrepancy adds to the advantage of special fat stains like ORO in the evaluation of liver steatosis in frozen sections.
Since all varieties of steatosis are detected slightly (MaS) or markedly (MiS) higher in ORO compared to HE in frozen sections, in our opinion, the applied method of steatosis evaluation has to be discussed with the transplanting surgeons. The aim is to timely detect steatosis with the highest possible accuracy and sensitivity without increasing the rate of discarded organs due to higher rates of steatosis. The parallel usage of HE and ORO stainings in the evaluation of liver steatosis by frozen section and an appropriate communication of its results to the transplanting surgeon, as is done at our center, constitute a practicable procedure.
However, insufficiencies in special fat stainings like ORO have been reported [3, 21, 22]. Displacements of fat droplets and non-hepatocellular/sinusoidal staining were noted. In our experience, those problems are reproducible in part. We do not find sinusoidal or non-hepatocellular-related staining. However, displacement of fat droplets can occur, especially with larger, MaS-related droplets (Fig. 1b). According to the experience with other fatty types of tissue in frozen section, this might be related to different freezing temperatures of the incorporated fat droplets in relation to the surrounding, water-rich tissue. Depending on the adjusted temperature of the cryostat and handling/expertise of the operator in the rinsing and staining process, too, such artifacts can occur, as is the case with standard HE staining in frozen section. However, for the evaluation of steatosis, we find ORO staining to be consistent in quality and simple in preparation and use also during service hours.
Since special fat stains like ORO directly stain fat which is washed out in the further processing of permanent, paraffin-based sections (FFPE), their use is limited to the acute diagnostic situation of frozen sections. For the detection of fat vacuoles in further processed FFPE tissue, indirect methods like adipophilin/perilipin-2 immunohistochemistry have proven both effective and sensitive [23]. However, the aim of the present study was not to compare different methods of indirect measurement of steatosis in permanent sections but to compare useful and applicable methods in the acute diagnostic setting of frozen sections.
In the second part of the present study, we found the reproducibility of the semiquantitative analyses of frozen sections to be moderate/good to very good between the two different experienced and blinded observers. ORO staining proved to be slightly superior to HE in the evaluation of MaS (ORO r = 0.881, HE r = 0.835), while correlation regarding MiS was higher in HE staining (ORO r = 0.576, HE r = 0.660). However, the advantage of ORO staining in the evaluation of MaS seems to be of more significance since the extent of MaS is a commonly accepted risk factor for clinical outcome, while the impact of MiS is under debate [8, 19]. Additionally, ORO facilitates the recognition of fat vacuoles in hepatocytes due to its distinct staining and high contrast (Fig. 1b), a fact which is helpful especially during service hours and in institutions with low rates of liver tissue samples in their diagnostic spectrum.
Morphometrical analyses (Fig. 1c,d) generally exhibited much lower rates of MiS, MaS, and ToS due to different figures of steatosis. In pathologists' semiquantitative analyses, the percentage of steatotic hepatocytes was evaluated, whereas morphometrical analyses showed percentages of the steatotic area in relation to the total lobular, non-portal tissue area. However, good to very good correlations between semiquantitative and morphometrical analyses were detected (Table 3), thus confirming the accuracy of semiquantitative evaluation of steatosis. Considering this and the fact of good/very good correlation between two different experienced observers, semiquantitative analysis seems to be a reliable and robust method of steatosis quantification. Therefore, we consider morphometry to be reliable, but not mandatory in the evaluation of liver steatosis in frozen sections as suggested by El-Badry et al. [13].
Furthermore, the study design enabled us to compare the pre- and post-reperfusion biopsies of the same patients (Fig. 1a–f/g,h). In histopathological analyses, I/R injury is among the accompanying factors defined as an increased occurrence of granulocyte/PMN infiltration and aggregation as well as increased apoptotic rate and hepatocellular ballooning [16, 17]. We found these indicators to be significantly elevated in post-reperfusion biopsies, thus indicating I/R injury on the histopathological level (Fig. 3a,b). Donor age and BMI were risk factors for increased I/R injury. These factors plus CIT were also found to be associated with the extent of steatosis, which are influencing factors regularly detected [19, 20, 24–26].
Steatosis itself proved to be associated with the extent of I/R injury. More severe steatosis (MaS) semiquantitatively detected in ORO-stained frozen sections (categorized mild vs. moderate) was associated with higher infiltration of PMNs (p = 0.03) (Fig. 3c). However, no further association between steatosis and factors of I/R injury were detected. These results on the one hand add to the relevance of special fat stains. On the other, we expected to detect more frequent associations between steatosis and I/R injury. The predominance of mild steatosis (MaS <30 %) in our cohort with few cases of moderate steatosis and a maximum of 40 % may be viewed as a causative factor. Several previous studies have highlighted the prognostic benignity of minor steatosis (MaS <30 %) [19, 27]. It seems that this minor steatosis (MaS) exhibits a non-fatal influence on hepatocellular homeostasis which is detected in more severe steatosis [27, 28]. Supporting this theory, we detected a moderate (30–60 %) extent of MaS (ORO, cryo) to be associated with an elevated post-operative course of AST and ALT, occurrence of IPF (p = 0.006), and shortened survival times (p = 0.045) in categorized analyses. No comparable results were found for mild (MaS <30 %) steatosis. Additionally, higher rates of MiS and ToS (ORO, cryo) were detected to be associated with longer ICU stays, thus giving support to the observation of an adverse prognostic effect of MiS [29].
However, a limiting factor of the present study is its low rate of higher steatotic cases (>30 % MaS). A study with more cases of moderate and/or severe steatosis would be desirable to verify our results. Nevertheless, we found that the application of special fat stains like ORO is a reliable and an easy method comprising significant advantages in the evaluation of hepatic steatosis. In addition, computerized analyses proved to be a precise tool, which is, however, not superior to semiquantitative analyses. These conclusions are emphasized by clinical, prognostic, and histopathological correlations particularly in terms of I/R injury.
References
Soejima Y, Shimada M, Suehiro T, Kishikawa K, Yoshizumi T, Hashimoto K, Minagawa R, Hiroshige S, Terashi T, Ninomiya M, Shiotani S, Harada N, Sugimachi K (2003) Use of steatotic graft in living-donor liver transplantation. Transplantation 76(2):344–348. doi:10.1097/01.TP.0000071205.52835.A4
Sotiropoulos GC, Paul A, Molmenti E, Lang H, Frilling A, Napieralski BP, Nadalin S, Treckmann J, Brokalaki EI, Gerling T, Broelsch CE, Malago M (2005) Liver transplantation for hepatocellular carcinoma in cirrhosis within the Eurotransplant area: an additional option with "livers that nobody wants". Transplantation 80(7):897–902
Rey JW, Wirges U, Dienes HP, Fries JW (2009) Hepatic steatosis in organ donors: disparity between surgery and histology? Transplant Proc 41(6):2557–2560. doi:10.1016/j.transproceed.2009.06.121
Sotiropoulos GC, Paul A, Gerling T, Molmenti EP, Nadalin S, Napieralski BP, Treckmann J, Lang H, Saner F, Frilling A, Broelsch CE, Malago M (2006) Liver transplantation with "rescue organ offers" within the Eurotransplant area: a 2-year report from the University Hospital Essen. Transplantation 82(3):304–309. doi:10.1097/01.tp.0000229447.37333.ed
Clavien PA, Petrowsky H, DeOliveira ML, Graf R (2007) Strategies for safer liver surgery and partial liver transplantation. N Engl J Med 356(15):1545–1559. doi:10.1056/NEJMra065156
Fiorentino M, Vasuri F, Ravaioli M, Ridolfi L, Grigioni WF, Pinna AD, D'Errico-Grigioni A (2009) Predictive value of frozen-section analysis in the histological assessment of steatosis before liver transplantation. Liver Transpl 15(12):1821–1825. doi:10.1002/lt.21948
Lo IJ, Lefkowitch JH, Feirt N, Alkofer B, Kin C, Samstein B, Guarrera JV, Renz JF (2008) Utility of liver allograft biopsy obtained at procurement. Liver Transpl 14(5):639–646. doi:10.1002/lt.21419
Selzner M, Clavien PA (2001) Fatty liver in liver transplantation and surgery. Semin Liver Dis 21(1):105–113. doi:10.1055/s-2001-12933
Yoong KF, Gunson BK, Neil DA, Mirza DF, Mayer AD, Buckels JA, McMaster P (1999) Impact of donor liver microvesicular steatosis on the outcome of liver retransplantation. Transplant Proc 31(1–2):550–551
Fishbein TM, Fiel MI, Emre S, Cubukcu O, Guy SR, Schwartz ME, Miller CM, Sheiner PA (1997) Use of livers with microvesicular fat safely expands the donor pool. Transplantation 64(2):248–251
Urena MA, Ruiz-Delgado FC, Gonzalez EM, Segurola CL, Romero CJ, Garcia IG, Gonzalez-Pinto I, Gomez Sanz R (1998) Assessing risk of the use of livers with macro and microsteatosis in a liver transplant program. Transplant Proc 30(7):3288–3291
Frongillo F, Avolio AW, Nure E, Mule A, Pepe G, Magalini SC, Agnes S (2009) Quantification of degree of steatosis in extended criteria donor grafts with standardized histologic techniques: implications for graft survival. Transplant Proc 41(4):1268–1272. doi:10.1016/j.transproceed.2009.03.096
El-Badry AM, Breitenstein S, Jochum W, Washington K, Paradis V, Rubbia-Brandt L, Puhan MA, Slankamenac K, Graf R, Clavien PA (2009) Assessment of hepatic steatosis by expert pathologists: the end of a gold standard. Ann Surg 250(5):691–697. doi:10.1097/SLA.0b013e3181bcd6dd
Nanashima A, Pillay P, Verran DJ, Painter D, Nakasuji M, Crawford M, Shi L, Ross AG (2002) Analysis of initial poor graft function after orthotopic liver transplantation: experience of an Australian single liver transplantation center. Transplant Proc 34(4):1231–1235
Ploeg RJ, D'Alessandro AM, Knechtle SJ, Stegall MD, Pirsch JD, Hoffmann RM, Sasaki T, Sollinger HW, Belzer FO, Kalayoglu M (1993) Risk factors for primary dysfunction after liver transplantation—a multivariate analysis. Transplantation 55(4):807–813
Weigand K, Brost S, Steinebrunner N, Buchler M, Schemmer P, Muller M (2012) Ischemia/reperfusion injury in liver surgery and transplantation: pathophysiology. HPB Surg 2012:176723. doi:10.1155/2012/176723
Longerich T, Schirmacher P (2006) General aspects and pitfalls in liver transplant pathology. Clin Transplant 20(Suppl 17):60–68. doi:10.1111/j.1399-0012.2006.00602.x
Leers MP, Kolgen W, Bjorklund V, Bergman T, Tribbick G, Persson B, Bjorklund P, Ramaekers FC, Bjorklund B, Nap M, Jornvall H, Schutte B (1999) Immunocytochemical detection and mapping of a cytokeratin 18 neo-epitope exposed during early apoptosis. J Pathol 187(5):567–572. doi:10.1002/(SICI)1096-9896(199904)187:5 < 567::AID-PATH288 > 3.0.CO;2-J
McCormack L, Dutkowski P, El-Badry AM, Clavien PA (2011) Liver transplantation using fatty livers: always feasible? J Hepatol 54(5):1055–1062. doi:10.1016/j.jhep.2010.11.004
Silva MA (2009) Putting objectivity into assessment of steatosis. Transplantation 88(5):620–621. doi:10.1097/TP.0b013e3181b39085
Markin RS, Wisecarver JL, Radio SJ, Stratta RJ, Langnas AN, Hirst K, Shaw BW Jr (1993) Frozen section evaluation of donor livers before transplantation. Transplantation 56(6):1403–1409
McCormack L, Petrowsky H, Jochum W, Mullhaupt B, Weber M, Clavien PA (2007) Use of severely steatotic grafts in liver transplantation: a matched case–control study. Ann Surg 246(6):940–946. doi:10.1097/SLA.0b013e31815c2a3f, discussion 946–948
Straub BK, Gyoengyoesi B, Koenig M, Hashani M, Pawella LM, Herpel E, Mueller W, Macher-Goeppinger S, Heid H, Schirmacher P (2013) Adipophilin/perilipin-2 as a lipid droplet-specific marker for metabolically active cells and diseases associated with metabolic dysregulation. Histopathology 62(4):617–631. doi:10.1111/his.12038
Imber CJ, St Peter SD, Lopez I, Guiver L, Friend PJ (2002) Current practice regarding the use of fatty livers: a trans-Atlantic survey. Liver Transpl 8(6):545–549. doi:10.1053/jlts.2002.31747
Iwasaki M, Takada Y, Hayashi M, Minamiguchi S, Haga H, Maetani Y, Fujii K, Kiuchi T, Tanaka K (2004) Noninvasive evaluation of graft steatosis in living donor liver transplantation. Transplantation 78(10):1501–1505
Heuer M, Kaiser GM, Kahraman A, Banysch M, Saner FH, Mathe Z, Gerken G, Paul A, Canbay A, Treckmann JW (2012) Liver transplantation in nonalcoholic steatohepatitis is associated with high mortality and post-transplant complications: a single-center experience. Digestion 86(2):107–113. doi:10.1159/000339344
Patkowski W, Zieniewicz K, Skalski M, Krawczyk M (2009) Correlation between selected prognostic factors and postoperative course in liver transplant recipients. Transplant Proc 41(8):3091–3102. doi:10.1016/j.transproceed.2009.09.038
Vetelainen R, van Vliet A, Gouma DJ, van Gulik TM (2007) Steatosis as a risk factor in liver surgery. Ann Surg 245(1):20–30. doi:10.1097/01.sla.0000225113.88433.cf
Cieslak B, Lewandowski Z, Urban M, Ziarkiewicz-Wroblewska B, Krawczyk M (2009) Microvesicular liver graft steatosis as a risk factor of initial poor function in relation to suboptimal donor parameters. Transplant Proc 41(8):2985–2988. doi:10.1016/j.transproceed.2009.08.019
Acknowledgments
We thank Dorothe Möllmann, Laura Malkus, and Nicole Macha for their skillful technical assistance, and Achim Konietzko for his assistance.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 127 kb)
Rights and permissions
About this article
Cite this article
Reis, H., Peterek, P.T., Wohlschlaeger, J. et al. Oil Red O-assessed macrosteatosis in liver transplant donor biopsies predicts ischemia–reperfusion injury and clinical outcome. Virchows Arch 464, 165–174 (2014). https://doi.org/10.1007/s00428-013-1512-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-013-1512-3