Abstract
Although fibrous dysplasia (FD) is a benign fibro-osseous lesion, locally aggressive behaviour has rarely been described but is poorly characterised. In this study, we document clinical, radiological and pathological (including molecular genetics) findings in three cases of locally aggressive FD, two of which involved the ribs. Lesions in these cases, one of which was a recurrent lesion, were followed up for 2–7 years. All of the lesions showed typical histological features of FD but were characterised by extension through the bone cortex into the extra-osseous soft tissue. The lesions did not exhibit overexpression/amplification of CDK4 and MDM2; in two of the cases, a GNAS mutation was identified. Our findings confirm that FD can rarely exhibit locally aggressive behaviour with extension beyond the bone compartment into the surrounding soft tissue; these lesions can be distinguished from low-grade intramedullary osteosarcoma by lack of amplification/overexpression of CDK4 and MDM2 and the presence of a GNAS mutation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fibrous dysplasia (FD) is a relatively common benign fibro-osseous lesion in which irregularly distributed spicules of woven bone lie in a cellular fibrous stroma [1, 2]. Most cases involve only a single bone (monostotic FD), but in a significant percentage of cases, more than one bone is involved (polyostotic FD). FD usually presents as pain, swelling, deformity or a pathological fracture. It may present over a wide age range but is most often diagnosed in the first three decades of life. Patients with rib lesions alone tend to be older and are often asymptomatic. Males and females are affected equally. Activating missense mutations in the GNAS gene (20q13) that encodes the alpha subunit of the stimulatory G-protein, GS alpha (GSα), have been identified in monostotic and polyostotic FD [3–5].
FD, in most cases, is a relatively well-circumscribed intramedullary lesion that expands the bone and typically is surrounded by a relatively wide rim of sclerotic bone. FD enters into the differential diagnosis of many benign and malignant bone tumours. Growth of FD often stabilises when skeletal maturity is reached, but in some cases, there may be continued growth [2, 6]. Distinction of FD from other bone-forming tumours, such as low-grade intramedullary osteosarcoma, can in some cases be problematic. FD is typically confined to the affected bone, but lesions that extend beyond the normal confines of the bone compartment have rarely been documented [7–15]. Most of these cases have been reported in the craniofacial skeleton, particularly the maxilla and mandible, common sites of FD development [7–12]. This extra-osseous extension of FD needs to be distinguished not only from other aggressive bone-forming tumours but also rare sarcomatous transformation of FD.
In this report, we document the clinical, radiological and pathological findings in three cases of FD which exhibited extension of the lesion into the extraosseous tissues. We have examined whether these lesions showed evidence of the GNAS mutation characteristic of FD. We also determined whether these lesions expressed CDK4 and showed MDM2 amplification, markers which have recently been documented in low-grade forms of osteosarcoma [16–18].
Materials and methods
Clinical details of the three cases presented in this report are shown in Table 1. Imaging was not available for review in case 1, but the rib lesions of cases 2 and 3 were investigated radiologically using plain X-ray, CT and MRI.
For pathological investigation, 5-μm haematoxylin- and eosin-stained sections of the formalin-fixed, decalcified lesion were examined histologically. Immunohistochemistry was carried out using an indirect immunoperoxidase technique with the antibodies MIB-1 (Dako, UK) and anti-CDK4 (clone DCS-31), (Invitrogen, CA, USA) directed against Ki-67 and CDK4, respectively.
Molecular genetic investigations included polymerase chain reaction (PCR) and mutation-specific restriction enzyme digestion (MSRED) for GNAS1 mutation. DNA was extracted from 5-μm sections cut from the tissue blocks and MSRED of exon 8 or exon 9 PCR products used to screen for the ten previously reported GNAS1 codon 201 and 227 mutations as previously described [19]. In addition, MDM2 FISH using the ZytoLight SPEC MDM2/CEN12 Dual Color Probe kit (Zyto Vision GmbH, Bremerhaven, Germany) was performed and evaluated on a full tissue section as previously described [20]. All FISH findings were reviewed by at least two individuals experienced in interpreting FISH.
Results
Clinical findings
As shown in Table 1, all cases in our series were adults (age range 32–72 years). One patient had a lesion of the left maxilla; the other two patients had lesions in the right 11th rib and right 7th rib. Cases 1 and 2 presented without a prior history of FD; case 3 had multiple rib lesions and was considered a case of polyostotic FD.
Radiological findings
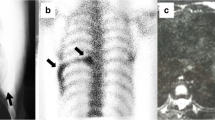
In case 2, MRI showed a single expansile lesion arising from the posterior portion of the 11th rib with uniform intermediate signal on T1-weighted sequence and mixed high and low signal on the STIR sequence. CT showed an expansile lytic lesion with multiple breaches of the thinned cortex (Fig. 1).
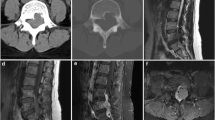
In case 3, CT showed two lytic lesions arising from the 7th rib at the resection margins and an additional lesion involving the posterior end of the rib. The small posterior lesion was lytic and non-expansile and had a sclerotic border with a breach in the cortex anteriorly. The other two lesions were expansile and were surrounded by a thin cortex with intermittent breaches. The more anterior of the two lesions was spherical and measured 5 cm in diameter. The smaller more posterior lesion showed modest expansion of the rib along a 5-cm length (Fig. 2).
Histological and molecular genetic findings
The diagnosis of FD was confirmed in all cases histologically. The fibro-osseous lesions contained abundant cellular fibrous tissue in which there were scattered irregular woven bone trabeculae (Fig. 3). Bone trabeculae were lined by flattened cells, and there was no marked cellular or nuclear pleomorphism. Mitotic activity was not pronounced and atypical mitotic figures were not identified. Focal myxoid change was seen in the fibrous stroma in which there were many small collections of osteoclastic giant cells, some of which were associated with small areas of haemorrhage; neighbouring woven bone trabeculae in the lesion often showed evidence of osteoclastic bone remodelling (Fig. 3). Other morphological changes which have been noted in FD such as cementum-like bodies, island of cartilage or extensive cystic change within the fibrous stroma were not seen in these lesions.
In all three cases, there was extension of FD through the bone cortex into the covering soft tissue. In cases 1 and 2, the cortex had been breached but the lesion was still covered by periosteum (Fig. 4). In case 3, the lesion extended into extraosseous fat, fibrous tissue and muscle (Fig. 5).
Immunohistochemistry showed a low proliferating fraction (<5 %) on Ki-67 staining and no evidence of CDK4 expression in all cases.
Molecular genetic investigation showed evidence of a R201H GNAS mutation typical of FD in cases 1 and 2 (Fig. 6). There was no evidence of MDM2 amplification by FISH. Molecular genetic investigation was not informative due to inadequate DNA quality in case 3.
Discussion
In this report, we document three cases of FD, two arising in the rib and one in the maxilla, where unusually there was histological evidence of extension of the lesion through the bone cortex into the covering soft tissues. The lesions showed typical histological features of FD with disorganised, irregular woven bone trabeculae lying in a cellular fibrous stroma. There was no marked nuclear pleomorphism or mitotic activity. Focal myxoid change and small collections of osteoclast-like giant cells were noted in the fibrous stroma, and there was prominent osteoclast remodelling of FD woven bone trabeculae. All cases were negative for CDK4 and did not have a high proliferating fraction. Two of these cases were negative for MDM2 amplification and showed a R201H GNAS mutation.
Most of the previously published reports of aggressive behaviour or soft tissue extension of typical FD have been noted in the craniofacial skeleton, particularly in the maxilla and mandible [7–12]. These cases arose mainly in young patients in whom FD had recurred locally following an initial surgery. We also report a case (case 1) of locally aggressive FD that arose in the maxilla, but in our case, the patient had reached skeletal maturity and the lesion was not a recurrence. The other two cases in this series arose in the ribs of patients that had reached skeletal maturity. One of these (case 3) was a recurrent lesion developing in the seventh rib of a 72-year-old female; the other (case 2) was a primary lesion arising in the 11th rib of a 60-year-old male. Spread beyond the bone cortex into the surrounding connective tissue was relatively limited in cases 1 and 2 where the lesion was largely covered by periosteal fibrous tissue. Infiltration beyond the periosteum was seen in case 3 where the extraosseous component was covered by muscle and fat.
FD usually presents in the first three decades of life; it may often remain asymptomatic but can present later in life as an incidental radiological finding or as pathological fracture. Most monostotic lesions of FD cease to enlarge after skeletal growth has ended. Lesions are usually confined to bone and have a sclerotic border. It should be noted that FD can rarely be attached to bone by a bony stalk, producing an exophytic lesion on the bone surface (fibrous dysplasia protuberans); this occurs mainly in short tubular bones of the hands and feet but has also been reported in the ribs [21]. In this series of lesions which showed a locally aggressive behaviour, typical histological features of FD were noted. No evidence of cystic change was seen, but focal myxoid change and small collections of osteoclastic giant cells were noted in the fibrous stroma; the latter were often associated with small areas of stromal haemorrhage and in most cases with osteoclastic activity on the surface of the neighbouring woven bone trabeculae. It is possible that stromal haemorrhage may have resulted from lesional trauma and that this increased osteoclast activity is significant with regard to osteolysis of the cortex and expansion of the lesion into extraosseous soft tissue.
As all these FD cases had extended through the bone cortex, the principal differential diagnosis was an aggressive bone-forming tumour, such as aggressive osteoblastoma or low-grade (FD-like) intramedullary osteosarcoma. However, unlike osteoblastoma, these lesions did not show evidence of prominent osteoblastic rimming of woven bone and had an inter-trabecular stroma composed mainly of cellular fibrous tissue. The possibility of low-grade osteosarcoma was a concern in these cases as a FD-like variant of this tumour is known to exist [22, 23]. However, the absence of nuclear pleomorphism or increased mitotic activity made a diagnosis of osteosarcoma difficult to substantiate morphologically. Immunohistochemistry showed a low proliferating fraction on Ki-67 staining, and CDK4 expression and MDM2 amplification, which have recently been described as markers of low-grade osteosarcoma [16, 17], were not identified in this case or any of the other lesions examined. Evidence of a R201H GNAS mutation typical of FD was seen in two of our cases where there was DNA of adequate quality. Pollard et al. also concluded that mutational analysis for GNAS mutation was useful in the differential diagnosis of FD and low-grade central osteosarcoma [24].
One of the features that distinguish FD from other bone-forming tumours is that even when it is large and has replaced cancellous bone, there is a rim of cortical bone which limits the lesion to the bone compartment. This series of cases provides evidence that FD is not always confined to bone and can rarely extend through the bone cortex into extra-osseous soft tissues. Full-thickness cortical bone destruction is a feature that characterises aggressive or malignant lesions of bone. With regard to FD, it is important to bear in mind that such cortical penetration could represent rare (approximately 0.5 % of monostotic cases) sarcomatous transformation [25]. Distinction of locally aggressive FD from a low-grade (FD-like) osteosarcoma can be difficult but, as this study shows, absence of cellular and nuclear pleomorphism and mitotic activity, a low-proliferating fraction on Ki-67 staining, absence of CDK4 overexpression and MDM2 amplification, and the presence of GNAS mutation should be useful in distinguishing rare examples of locally aggressive FD.
References
Siegal GP, Bianco P, Dal Cin P (2013) Fibrous dysplasia. In: Fletcher CD, Bridge JA, Hogendoorn PCW, Mertens F (eds) Pathology and genetics of tumours of soft tissue and bones. IARC, Lyon, pp 352–353
DiCaprio MR, Enneking WF (2005) Fibrous dysplasia. Pathophysiology, evaluation, and treatment. J Bone Joint Surg (Am) 87:1848–1864
Weinstein LS, Shenker A, Gejman PV, Merino MJ, Friedman E, Spiegel AM (1991) Activating mutations of the stimulatory G protein in the McCune–Albright syndrome. N Engl J Med 325:1688–1695
Schwindinger WF, Francomano CA, Levine MA (1992) Identification of a mutation in the gene encoding a subunit of the stimulatory G protein of adenylyl cyclase in McCune–Albright syndrome. Proc Natl Acad Sci USA 89:5152–5156
Shenker A, Weinstein LS, Sweet DE, Spiegel AM (1994) An activating Gs alpha mutation is present in fibrous dysplasia of bone in the McCune–Albright syndrome. J Clin Endocrinol Metab 79:750–755
Henry A (1969) Monostotic fibrous dysplasia. J Bone Joint Surg (Br) 51:300–306
Schofield DF (1974) An aggressive fibrous dysplasia. Oral Surg, Oral Med, Oral Pathol 38:29–35
Gille P, Beugnet D, Carbillet JP, Giordan H (1987) Aggressive fibrous dysplasia of the mandible. Resection of the lower portion of the mandible. Chir Paediatr 28:262–265
Gambhir G, Batra R, Sethi N, Bansal A (2011) A rare case of fibrous dysplasia in an elderly patient, with brief review of literature of monostotic fibrous dysplasia of maxilla. Indian J Med Sciences 3:68–70
Gupta MK, Mhaske S (2011) Aggressive fibrous dysplasia of mandible—a case report. JIDA 5:503–505
Olasoji HO, Ugboko VI, Nggada HA (2006) Aggressive form of fibrous dysplasia of the mandible in childhood: case report. Otorhinolaryngology 1:89–92
Shapeero LG, Vanel D, Ackerman LV, Terrier-Lacombe MJ, Housin D, Schwaab G, Sigal R, Masselot J (1993) Aggressive fibrous dysplasia of the maxillary sinus. Skeletal Radiol 22:563–568
Vanel D, Couanet D, Micheau C, Piekarski JD, Schwaab G, Masselot J (1980) Pseudotumoural fibrous dysplasia of the maxilla: radiological studies and computed tomography contribution. Skeletal Radiol 5:99–103
Latham PD, Athanasou NA, Woods CG (1992) Fibrous dysplasia with locally aggressive malignant change. Arch Orthop Trauma Surg 111:183–186
Yao L, Eckardt JJ, Seeger LL (1994) Fibrous dysplasia associated with cortical bony destruction: CT and MR findings. J Comput Assist Tomography 8:91–94
Yoshida A, Ushiku T, Motoi T, Shibata T, Beppu Y, Fukayama M, Tsuda H (2012) MDM2 and CDK4 immunohistochemical coexpression in high-grade osteosarcoma: correlation with a dedifferentiated subtype. Am J Surg Pathol 36:423–431
Wunder JS, Eppert K, Burrow SR, Gokgoz N, Bell RS, Andrulis IL (1999) Co-amplification and overexpression of CDK4, SAS and MDM2 occurs frequently in human parosteal osteosarcomas. Oncogene 21(18):783–788
Tarkkanen M, Bohling T, Gamberi G, Ragazzini P, Benassi MS, Kivioja A, Kailio P, Elomaa I, Picci P, Knuutila S (1998) Comparative genomic hybridization of low-grade central osteosarcoma. Mod Pathol 11:421–426
Idowu BD, Al-Adnani M, O’Donnell P, Yu L, Odell E, Diss T, Gale RE, Flanagan AM (2007) A sensitive mutation-specific screening technique for GNAS1 mutations in cases of fibrous dysplasia: the first report of a codon 227 mutation in bone. Histopathology 50:691–704
Kashima T, Halai D, Ye H, Hing SN, Delaney D, Pollock R, O’Donnell P, Tirabosco R, Flanagan AM (2012) Sensitivity of MDM2 amplification and unexpected multiple faint alphoid 12 (alpha 12 satellite sequences) signals in atypical lipomatous tumour. Mod Pathol 25:1384–1396
Dorfman HD, Ishida T, Tsuneyoshi M (1994) Exophytic variant of fibrous dysplasia (fibrous dysplasia protuberans). Hum Pathol 25:1234–1237
Franceschina MJ, Hankin RC, Irwin RB (1997) Low-grade central osteosarcoma resembling fibrous dysplasia. A report of two cases. Am J Orthop 26:432–440
Wenger DE, Sundaram M, Unni KK, Janney CG, Merkel K (2002) Microscopic correlation of radiographically disparate appearing well differentiated osteosarcoma. Skeletal Radiol 31:488–492
Pollard K, Engels C, Kaiser E, Werner M, Delling G (2001) Gsalpha gene mutations in monostotic fibrous dysplasia of bone and fibrous dysplasia-like low-grade central osteosarcoma. Virchows Arch 439:170–175
Ruggieri P, Sim FH, Bond JR, Unni KK (1994) Malignancies in fibrous dysplasia. Cancer 73:1411–1424
Acknowledgments
We would like to thank Chris Lowe for typing the manuscript.
Conflict of interest
The authors declare there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kashima, T.G., Gamage, N.M., Ye, H. et al. Locally aggressive fibrous dysplasia. Virchows Arch 463, 79–84 (2013). https://doi.org/10.1007/s00428-013-1437-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-013-1437-x