Abstract.
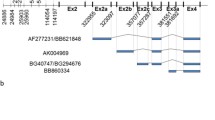
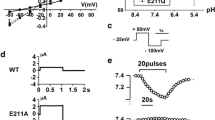
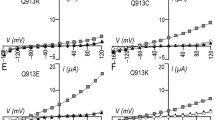
The term Bartter syndrome encompasses a heterogeneous group of autosomal recessive salt-losing nephropathies that are caused by disturbed transepithelial sodium chloride reabsorption in the distal nephron. Mutations have been identified in the NKCC2 (Na+-K+-2Cl–) cotransporter and ROMK potassium channel, which cooperate in the process of apical sodium chloride uptake, and ClC-Kb chloride channels, which mediate basolateral chloride release. Recently, mutations in barttin, a protein not related to any known ion transporter or channel, were described in BSND, a variant of Bartter syndrome associated with sensorineural deafness. Here we show that barttin functions as an activator of ClC-K chloride channels. Expression of barttin together with ClC-K in Xenopus oocytes increased ClC-K current amplitude, changed ClC-K biophysical properties, and enhanced ClC-K abundance in the cell membrane. Co-immunoprecipitation revealed a direct interaction of barttin with ClC-K. We performed in situ hybridization on rat kidney slices and RT-PCR analysis on microdissected nephron segments to prove co-expression of barttin, ClC-K1 and ClC-K2 along the distal nephron. Functional analysis of BSND-associated point mutations revealed impaired ClC-K activation by barttin. The results demonstrate regulation of a CLC chloride channel by an accessory protein and indicate that ClC-K activation by barttin is required for adequate tubular salt reabsorption.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Waldegger, S., Jeck, N., Barth, P. et al. Barttin increases surface expression and changes current properties of ClC-K channels. Pflügers Arch - Eur J Physiol 444, 411–418 (2002). https://doi.org/10.1007/s00424-002-0819-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-002-0819-8