Abstract
Background
Prospective multicenter trials have shown the feasibility of sentinel node (SN) navigation surgery using a dual tracer of dye and radioisotope for early gastric cancer. However, comparable trials using the indocyanine green (ICG) and the infrared ray laparoscopic system (IRLS) have not been reported. On the basis of our cohort studies, we assumed that the ICG imaging with the IRLS is as effective as the dual tracer in detecting SNs. Therefore, we conducted a prospective multicenter trial to assess the effectiveness and generalizability of the infrared ICG technique in patients with early gastric cancer.
Patients and methods
Patients with previously untreated cT1 gastric adenocarcinomas less than 4 cm in gross diameter were eligible for inclusion in this study. SN mapping was performed by using ICG combined with IRLS technique. Following biopsy of the identified SNs, D2 or modified D2 laparoscopic gastrectomy was performed according to the current Japanese Gastric Cancer Association guidelines.
Results
Among the 47 patients who gave written informed consent, 44 were eligible from the surgical findings, for whom SN biopsy was performed. The detection rate of the lymphatic basin by the ICG with IRLS was 100% (44/44). The accuracy was also 100% (7/7) for detecting metastatic lymph node, which was verified on the permanent pathologic examination. Following the median follow-up of 114 months (range, 60 to 120 months), no recurrence (0/40) was observed. Although the number of patients was unequally distributed among the hospitals participating in the trial, the detection rates of SNs in low-volume hospitals were comparable to those in high-volume hospitals. Consequently, there was no learning curve in this trial.
Conclusion
The infrared ICG imaging accurately detected the lymphatic basin and SNs with occult metastasis in SN-navigated gastrectomy for gastric cancer. This method was feasible even for low-volume surgeons and hospitals.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sentinel node (SN) navigation surgery has been well investigated and adopted in melanoma and breast cancer, allowing a reduction in the extent of surgical intervention with limited lymph node dissection [1, 2]. However, the clinical application of SN biopsy in early gastric cancer has long been debated due to the complicated lymphatic flow around the stomach and the inconsistent results. Some studies suggested the limitation of SN navigation surgery for early gastric cancer, because it could overlook a number of positive SNs [3, 4]. For the past decade, clinical trials using a dual tracer with dye and radioisotope [5] revealed that the dual tracer is better than the single tracer [6, 7] in detecting SNs in early gastric cancer. For instance, in a study using a dual tracer by Kitagawa et al. [8], the detection rate was 96%, the mean number of retrieved SNs was 4.1, sensitivity was 93%, and accuracy was 99%. In a retrospective study not using the radioisotope, we demonstrated that high detection and accuracy rates could be achieved with the indocyanine green (ICG) and infrared ray laparoscopic system (IRLS) in patients with early gastric cancer [9, 10]. In general, the use of radioactive substances was associated with high costs and potential biohazard, restricting its clinical application. ICG plus infrared ray method seems safer, more convenient, and less expensive compared with the method using radioisotope as a tracer. As far as we know, prospective multicenter studies using the infrared ICG imaging have not been published. Therefore, we conducted a trial to evaluate the clinical significance of SN mapping with the ICG and IRLS. The primary aim of this study was to assess the effectiveness of the infrared ICG technique across the hospitals with different experiences in SN navigation surgery for early gastric cancer. The secondary aim was to clarify the long-term outcome of the participants.
Patients and methods
Indication for sentinel node biopsy
Patients with previously untreated cT1N0M0 gastric adenocarcinomas less than 4 cm in gross diameter were eligible for inclusion in this study. Clinical staging was determined by routine preoperative endoscopy and computed tomography, while endoscopic ultrasound was not routinely performed. Patients with apparent T3/T4 tumors, nodal or distant metastasis diagnosed intraoperatively, and extensive abdominal adhesion were excluded from the study. Four institutions participated in this multicenter prospective study. Prior to entry in the trial, all surgeons were trained to be a surgeon for SN navigation surgery for early gastric cancer, but there were considerable differences in surgical volume among them. From July 2005 to June 2007, 47 patients were enrolled onto this study, all of whom were preoperatively registered in a central data center after obtaining written informed consent. This study was approved by all local institutional review boards and conducted in accordance with the Good Clinical Practice guidelines and the Declaration of Helsinki.
Sentinel node mapping, pathological evaluation, and learning curve
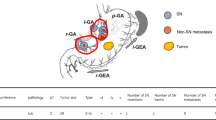
SN navigation surgery was performed as previously described [9]. Briefly, prior to ICG injection, the stomach was mobilized by dissecting through the gastrocolic ligament without disrupting the gastroepiploic vessels. ICG (5 mg/ml; Diagnogreen™, Daiichi Pharmaceutical, Tokyo, Japan) of 0.5 ml was injected endoscopically in four quadrants of the submucosa surrounding the tumor with an endoscopic puncture needle. At 20 min after the injection, SNs stained with ICG were observed with the naked eye (ordinary light observation) followed by the IRLS (Olympus Optical, Tokyo, Japan) (infrared light observation). Positive or negative staining was judged in concordance with at least two surgeons and one endoscopist. The lymphatic vessels and lymph nodes were dyed green by the ordinary light observation and they were seen in black by IRLS (Fig. 1). The detected lymphatic basin including SNs were resected laparoscopically and recorded according to the lymph node station numbers as described in The Japanese Classification of Gastric Carcinoma. Suspected SNs were then histologically examined intraoperatively in one plane (with the largest dimension) by frozen section with H&E staining. Laparoscopic gastrectomy with lymphadenectomy was performed regardless of the metastatic status of SNs on the frozen section examination. Postoperatively, permanent pathological examination was performed by the experienced pathologist, who was blinded to the SN status and detection methods. Both SNs and non-SNs were diagnosed by one plane (with the largest dimension).
To evaluate the presence of a learning curve, we analyzed the primary outcome for each surgeon. We attempted to follow up all patients on the outpatient basis for a long-term survival analysis.
Results
As shown in Fig. 2, one patient was preoperatively excluded and two others were excluded because of intraoperative findings ineligible for the trial. Finally, 44 patients underwent SN biopsies (Table 1). Adverse events associated with the SN biopsy did not occur during the study period. The detection rate of the lymphatic basin by the infrared light observation was much higher than that by the ordinary light observation (100 vs. 57%). At the back-table, the detection rate of the SN by the infrared light was 100%, while the SN detection rate determined by the ordinary light observation alone was 80% (35/44). Lymph node metastasis was pathologically confirmed in 16% of patients (7/44). The proportion of false negatives by the ordinary light observation was 57% (4/7); however, that by the infrared light observation was 0% (0/7) (Table 2). In five of these seven patients (71%), metastatic spread was limited to the SNs, and in the remaining two patients, metastases were limited to the area within the SN basins.
The number of patients enrolled in the trial were unevenly distributed among the surgeons and institutions (Table 3). There was no learning curve, although the sample size was too small to obtain a sufficient statistical power (Table 3). Four patients were lost to follow-up. Following the median follow-up of 114 months (range, 60 to 120 months), no recurrence was observed.
Discussion
In the present study, SNs were detected by the infrared light observation in all the patients during laparoscopic gastrectomy for early gastric cancer, while the SN detection rate was 80% in the ordinary light alone. In addition, the sensitivity of the infrared ICG for detecting SNs was 100%, which was much higher than the 43% achieved by naked eye observation. There was no learning curve in the trial.
In the clinical trials using a dual tracer, efficacy of SN biopsy for early gastric cancer has been reported [11, 12]. Kitagawa et al. [11] suggested that SN mapping for gastric cancer with the dual tracer method was a feasible and safe procedure, because the detection rate of SNs was 97.5% and the false negative rate was 7% in their trial. In a more recent study, sentinel basins were detected in 100 of the 108 patients (92.6%), and all patients with nodal metastasis (10/10), which was confirmed on permanent section, were identified during surgery [12]. Oppositely, in a multicenter trial using the non-infrared ICG as a single tracer by Miyashiro et al. [13], a high false negative rate (46%) on interim analysis stopped patient accrual. The present study also revealed the suboptimal ability of the non-infrared ICG to detect SN and predict nodal metastasis, whereas the infrared ICG did not miss the SNs and nodal metastasis (Table 2).
Tonouchi et al. [14] conducted a literature review to determine the cause of false negative SN biopsies in gastric cancer and concluded that the common cause was lymphatic occlusion due to massive lymph node metastasis. Careful attention should be paid to the preoperative diagnosis of the T factor. Notably, the diagnostic accuracy of SN mapping for cT2 tumors is currently suboptimal; therefore, we believe that the clinical application of SN mapping should be limited to cT1 tumors. In a single-center study including 16 patients with early gastric cancer, the feasibility and usefulness of SN mapping with the infrared ICG was shown [15]. In our retrospective cohort study of 84 patients, SNs detected by ICG injection alone did not include metastases in 4 of 11 patients, whereas the infrared ICG imaging detected SNs containing lymph node metastases in all 11 patients [9]. Therefore, we conducted this multicenter trial, assuming that outcomes of SN mapping by ICG combined with IRLS would be comparable to those of the dual tracer method.
Lee et al. [16] speculated that the learning period for identification of sentinel nodes in gastric cancer would be 26 cases, and the learning curve should be considered to minimize bias due to surgical factors in clinical trials for gastric cancer with SN mapping. In the present trial, the lymphatic basin was detected in all 44 cases, which was achieved by a combined team of well- and less-experienced surgeons. Although firm conclusion cannot be drawn from our study with a small sample size, it could be assumed that infrared ICG is feasible for less-experienced surgeons to perform SN navigation surgery under supervision after a short period of training. We acknowledge that the current study does not completely erase the concern about a considerable learning time required for achieving better and sustainable outcomes of SN-navigated gastrectomy with the infrared ICG imaging. There are a number of factors which may shorten the learning curve, including patient selection, the site of injection of ICG (submucosal or subserosal), and the observation timing after injection. Long-term outcomes of SN navigation surgery for early gastric cancer have been poorly studied. In 40 patients who could be followed-up for the median period of 114 months in the trial, no recurrence was observed. The use of infrared ICG imaging is being extended particularly to SN navigation surgery for early breast cancer [17] and early endometrial cancer [18] with high detection and accuracy rates. Since the first description of ICG fluorescence imaging with a charge-coupled device detecting fluorescence signals from SNs in gastric cancer and colorectal cancer by Kusano et al. in 2008. [19], the scope of fluorescence-based SN mapping has expanded from the stomach, breast, skin, and lower digestive tract to the lung, head and neck, aerodigestive tract, and genitourinary system [20]. The present findings suggest that infrared ICG imaging is feasible for SN mapping in minimally invasive surgery for early gastric cancer, as is ICG fluorescence imaging.
In conclusion, the infrared ICG observation was much superior to ICG observation with naked eyes in detection of SNs, irrespective of the surgeon’s experience. These findings suggest that SN biopsy using the infrared ICG is an effective and useful procedure to identify patients with cT1 N0 gastric cancer who have occult nodal metastasis.
Abbreviations
- ICG:
-
Indocyanine green
- SN:
-
Sentinel node
- SNNS:
-
Sentinel node navigation surgery
- IRLS:
-
Infrared ray laparoscopic system
References
Veronesi U, Paganelli G, Viale G, Luini A, Zurrida S, Galimberti V, Intra M, Veronesi P, Robertson C, Maisonneuve P, Renne G, De Cicco C, De Lucia F, Gennari R (2003) A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. N Engl J Med 349:546–553
Morton DL, Thompson JF, Essner R, Elashoff R, Stern SL, Nieweg OE, Roses DF, Karakousis CP, Mozzillo N, Reintgen D, Wang HJ, Glass EC, Cochran AJ (1999) Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for early-stage melanoma: a multicenter trial. Ann Surg 230:453–463
Ryu KW, Eom BW, Nam BH, Lee JH, Kook MC, Choi IJ, Kim YW (2011) Is the sentinel node biopsy clinically applicable for limited lymphadenectomy and modified gastric resection in gastric cancer? A meta-analysis of feasibility studies. J Surg Oncol 104:578–584
Kitagawa Y, Fujii H, Kumai K, Kubota T, Otani Y, Saikawa Y, Yoshida M, Kubo A, Kitajima M (2005) Recent advances in sentinel node navigation for gastric cancer: a paradigm shift of surgical management. J Surg Oncol 90:147–151
Kinami S, Fujimura T, Ojima E, Fushida S, Ojima T, Funaki H, Fujita H, Takamura H, Ninomiya I, Nishimura G, Kayahara M, Ohta T, Yoh Z (2008) PTD classification: proposal for a new classification of gastric cancer location based on physiological lymphatic flow. Int J Clin Oncol 13:320–329
Miwa K, Kinami S, Taniguchi K, Fushida S, Fujimura T, Nonomura A (2003) Mapping sentinel nodes in patients with early-stage gastric carcinoma. Br J Surg 90:178–182
Hiratsuka M, Miyashiro I, Ishikawa O, Furukawa H, Motomura K, Ohigashi H, Kameyama M, Sasaki Y, Kabuto T, Ishiguro S, Imaoka S, Koyama H (2001) Application of sentinel node biopsy to gastric cancer surgery. Surgery 129:335–340
Kitagawa Y, Saikawa Y, Takeuchi H, Mukai M, Nakahara T, Kubo A, Kitajima M (2006) Sentinel node navigation in early stage gastric cancer—updated data and current status. Scand J Surg 95:256–259
Nimura H, Narimiya N, Mitsumori N, Yamazaki Y, Yanaga K, Urashima M (2004) Infrared ray electronic endoscopy combined with indocyanine green injection for detection of sentinel nodes of patients with gastric cancer. Br J Surg 91:575–579
Kelder W, Nimura H, Takahashi N, Mitsumori N, van Dam GM, Yanaga K (2010) Sentinel node mapping with indocyanine green (ICG) and infrared ray detection in early gastric cancer: an accurate method that enables a limited lymphadenectomy. Eur J Surg Oncol 36:552–558
Kitagawa Y, Takeuchi H, Takagi Y, Natsugoe S, Terashima M, Murakami N, Fujimura T, Tsujimoto H, Hayashi H, Yoshimizu N, Takagane A, Mohri Y, Nabeshima K, Uenosono Y, Kinami S, Sakamoto J, Morita S, Aikou T, Miwa K, Kitajima M (2013) Sentinel node mapping for gastric cancer: a prospective multicenter trial in Japan. J Clin Oncol 31:3704–3710
Lee YJ, Jeong SH, Hur H, Han SU, Min JS, An JY, Hyung WJ, Cho GS, Jeong GA, Jeong O, Park YK, Jung MR, Kim YW, Yoon HM, Eom BW, Park JY, Ryu KW; Sentinel Node Oriented Tailored Approach (SENORITA) Study Group (2015) Prospective multicenter feasibility study of laparoscopic sentinel basin dissection for organ preserving surgery in gastric cancer: quality control study for surgical standardization prior to phase III trial. Medicine (Baltimore).;bb94:e1894. doi: 10.1097/MD.0000000000001894
Miyashiro I, Hiratsuka M, Sasako M, Sano T, Mizusawa J, Nakamura K, Nashimoto A, Tsuburaya A, Fukushima N, Gastric Cancer Surgical Study Group (GCSSG) in the Japan Clinical Oncology Group (JCOG) (2014) High false-negative proportion of intraoperative histological examination as a serious problem for clinical application of sentinel node biopsy for early gastric cancer: final results of the Japan clinical oncology group multicenter trial JCOG0302. Gastric Cancer 17:316–323
Tonouchi H, Mohri Y, Tanaka K, Kobayashi M, Ohmori Y, Kusunoki M (2005) Laparoscopic lymphatic mapping and sentinel node biopsies for early-stage gastric cancer: the cause of false negativity. World J Surg 29:418–421
Ishikawa K, Yasuda K, Shiromizu A, Etoh T, Shiraishi N, Kitano S (2007) Laparoscopic sentinel node navigation achieved by infrared ray electronic endoscopy system in patients with gastric cancer. Surg Endosc 21:1131–1134
Lee JH, Ryu KW, Lee SE, Cho SJ, Lee JY, Kim CG, Choi IJ, Kook MC, Kim MJ, Park SR, Lee JS, Nam BH, Kim YW (2009) Learning curve for identification of sentinel lymph node based on a cumulative sum analysis in gastric cancer. Dig Surg 26:465–470
Verbeek FP, Troyan SL, Mieog JS, Liefers GJ, Moffitt LA, Rosenberg M, Hirshfield-Bartek J, Gioux S, van de Velde CJ, Vahrmeijer AL, Frangioni JV (2014) Near-infrared fluorescence sentinel lymph node mapping in breast cancer: a multicenter experience. Breast Cancer Res Treat 143:333–342
Plante M, Touhami O, Trinh XB, Renaud MC, Sebastianelli A, Grondin K, Gregoire J (2015) Sentinel node mapping with indocyanine green and endoscopic near-infrared fluorescence imaging in endometrial cancer. A pilot study and review of the literature. Gynecol Oncol 137:443–447
Kusano M, Tajima Y, Yamazaki K, Kato M, Watanabe M, Miwa M (2008) Sentinel node mapping guided by indocyanine green fluorescence imaging: a new method for sentinel node navigation surgery in gastrointestinal cancer. Dig Surg 25:103–108
Zelken JA, Tufaro AP. 2015. Current trends and emerging future of indocyanine green usage in surgery and oncology: an update Ann Surg Oncol; Suppl 3:S1271-S1283.
Acknowledgements
The authors thank the following members for their help in collecting the data in the trial: Kazuhiro Yasuda, Haruhiro Inoue, Junichi Tanaka, Shinei Kudo, and Tomoki Koyama.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics and consent
The study protocol and publication of the study results including the personal details were approved by the Institutional Review Board of the Jikei University Scholl of Medicine (Reference No.15-53) and all local institutional review boards. Written informed consent was obtained from all patients.
Conflict of interest
All authors declare that they have no conflict of interest.
Author’s contributions
Naoto Takahashi, Hiroshi Nimura, Seigo Kitano, and Katsuhiko Yanaga conceived the study and analyzed the data. Norio Mitsumori, Norio Shiraishi, and Hitoshi Satodate helped collect the clinical data. Naoto Takahashi, Hiroshi Nimura, and Tetsuji Fujita drafted and revised the manuscript on behalf of all authors. All authors read and approved the final manuscript.
Availability of data and materials
We clarified that the datasets supporting the conclusions of this article are included within the article.
Funding
We received no support from any funding.
Rights and permissions
About this article
Cite this article
Takahashi, N., Nimura, H., Fujita, T. et al. Laparoscopic sentinel node navigation surgery for early gastric cancer: a prospective multicenter trial. Langenbecks Arch Surg 402, 27–32 (2017). https://doi.org/10.1007/s00423-016-1540-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-016-1540-y