Abstract
Background
The therapeutic regimen for patients suffering of HCC in liver cirrhosis must pay attention to the underlying liver disease. Surgical resection is often limited by liver function and transplantation, as an optimal therapy for many early diagnosed HCC, by the availability of organs. Due to three prospective, randomized trials radiofrequency ablation (RFA) is the standard method of local ablation. RFA compared with resection for HCC in liver cirrhosis yields similar results concerning overall survival but a lower rate of complications. The laparoscopic approach may be advantageous concerning the major drawback of RFA which is still the rate of local failure as shown by a meta-analysis of local recurrences.
Method
Indication for RFA was HCC in liver cirrhosis either as a definite therapy or as a bridging procedure for transplantation if the expected waiting time exceeded 6 months. Laparoscopic ultrasound, standardized algorithm of laparoscopic RFA procedure, track ablation and a Trucut biopsy were performed. The postoperative follow-up was done according to institutional standards. Patient data and parameters of laparoscopic RFA were prospectively documented, analyzed and compared with the results of previously published series found in a Medline search.
Results
34 patients were treated by laparoscopic RFA. The average time of follow-up was 36.9 ± 28.3 months. There was no procedure-related mortality or surgical complications. An upstaging of the tumor stage by laparoscopic ultrasound was achieved in 32 % of the patients. The overall survival of these patients was 44.7 ± 6.9 months. The intrahepatic recurrence rate was 61.8 % based on the number of patients treated. The results have been analyzed and compared with six independent papers identified in a Medline search that report on the treatment of patients with HCC in a liver cirrhosis by laparoscopic RFA with a mean follow-up of 12 or more months.
Conclusions
Laparoscopic RFA is a feasible and reliable therapy for unresectable HCCs in patients with cirrhosis. The laparoscopic RFA combines the advantage of a minimally invasive procedure concerning liver dysfunction with the ability of an accurate intraoperative staging by laparoscopic ultrasound.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) accounts for more than 1 million deaths annually [1]. In Western countries with an established program of hepatitis B vaccination chronic hepatitis C is the predominate risk factor for HCC [2]. Any therapeutic regimen for patients suffering of HCC must pay attention to the underlying liver disease. Liver transplantation is an optimal therapy for many early diagnosed HCC, but limited by the availability of organs. Resection is often contraindicated due to the limited liver function. Radiofrequency ablation (RFA) has replaced percutaneous ethanol injection as the standard method of local ablation [3] due to three prospective, randomized trials that showed superiority of RFA concerning local recurrence and overall survival [4–6]. Recently, two trials compared RFA with resection for HCC in liver cirrhosis yielding similar results concerning local tumor control but a lower rate of complications [7, 8]. The major drawback of RFA still is the rate of local failure as shown by a meta-analysis of local recurrences after RFA [9, 10]. In this meta-analysis the laparoscopic approach yielded a significant lower rate of local recurrence compared to the percutaneous approach. However, this analysis is biased, since the published clinical experience using the laparoscopic approach for RFA of HCC nodules is very limited [11–15]. The purpose of this paper is to describe and discuss indication and technique of laparoscopic RFA for HCC and report the clinical results achieved with this technique. The results are compared to previously published series with a median follow-up of at least 12 months which resulted from a Medline search entering hepatocellular cancer, radiofrequency ablation and laparoscopic (and synonyms).
Material and methods
Patient selection and description
Only patients were included that had a diagnosis of liver cirrhosis and/or chronic viral hepatitis. Diagnosis of HCC was made by biopsy or by imaged based criteria (preferentially) as published by the EASL consensus [16]. Briefly, a HCC was diagnosed when a nodule (diameter > 2 cm) showed early arterial enhancement in two different image modalities, or if α-fetoprotein was above 400 ng/ml in the presence of an early enhancing nodule in one modality or if biopsy showed a HCC. If diagnosis of HCC was not based on preoperative biopsy, a Trucut biopsy was taken immediately after RFA to confirm the diagnosis. A biopsy taken after the RFA decreases the risk of tumor seeding due to the biopsy [17]. Thus, all patients included had a biopsy proven HCC.
Indication for RFA as opposed to surgery was considered, when one of the following criteria was met:
-
CHILD B patient
-
CHILD A patients with a liver function that made a postoperative hepatic decompensation likely (thrombocyte count below 100,000/μl, signs of portal hypertension on imaging and/or MELD score > 10)
-
The extension of the necessary resection made a postoperative liver failure likely,
-
More than one resection necessary to clear all HCC nodules,
The size and number of the HCC nodules had to be suitable for RFA:
-
The largest HCC nodule on preoperative imaging had to be smaller than 3.5 cm in diameter,
-
The number of HCC nodules less or equal to three.
Indication for laparoscopic approach in contrast to percutaneous approach was based on the following criteria:
-
Localization of the HCC within 1 cm of the liver capsule,
-
Localization in the dome of the liver (difficult needle placement in axial plane),
-
Size of the nodule to treat was larger than 2.5 cm.
-
Bridge to transplantation as therapeutic aim of RFA
This stratification was chosen according to published data about the size of the zone of ablation that can be achieved by the specific RFA applicator and generator used for percutaneous and laparoscopic RFA [18, 19].
Between February 2002 and February 2008, 172 patients were treated for HCC by either transplantation (without bridging), resection, RFA or transarterial chemoembolisation. According to the above mentioned criteria 34 consecutive patients were stratified to be treated by laparoscopic radiofrequency ablation.
Description of patients’ risk factors was done by classifying the stage of the HCC according to the Cancer of the Liver Italian Program (CLIP) staging system [20]. The liver function was evaluated by the Child–Turcotte–Pugh Score [21] and calculation of the MELD score [22].
The characteristics of the 34 treated patients are summarized in Table 1.
Laparoscopic RFA procedure
The first trocar (camera) was introduced by subumbilical mini laparotomy to avoid portovenous collaterals to the umbilicus. A second trocar (ultrasound) was placed in the right upper quadrant at least 8 to 10 cm below the rip bow. The position of the second trocar facilitated an ultrasound examination of both liver lobes. A 7.5-MHz linear probe (Siemens Sonoline Sienna, Siemens Medical Systems, Inc., Issaquah, WA, USA) was utilized. The 4 cm long scanner is mounted on a double joint allowing for 4 degrees of freedom (up/down, right/left, rotation, forward/back). The highly flexible scanner facilitated a perfect acoustic coupling to any part of the liver. The placement of the RFA applicator was performed as a freehand procedure by one surgeon (D.L.S.). To enable a secure placement of the applicator the HCC nodule has place in the far end of the scanned liver section. Using a 20-gauge needle a puncture position on the abdominal wall was located by visual control of the laparoscopic camera image from where the applicator would be inline with the scanner. Depending on the distance between liver surface and the HCC the angle within the ultrasound image from the right upper corner to the targeted position of the applicator tip was estimated. With approximately the same angle, the applicator was introduced into the liver surface right beside the joint of the ultrasound probe. Rotating the ultrasound probe facilitates visualization of the applicator. Corrections concerning the angle between the liver surface and the applicator were made either by pushing the liver with the ultrasound or by directing the extra corporal part of the applicator in the opposite direction of the direction needed in the liver. To ensure reliable placement of the applicator a long enough path (>2 cm) through liver tissue was important. If the applicator position needed correction a track ablation was performed even if the applicator was not in contact with the HCC.
RFA was performed using a Rita generator model 1500XTM (RITA Medical Systems, Mountain View, CA, USA), and either a Rita XL or a Rita XLi applicator. The applicator specific standard algorithm of energy deposition as supplied by the company was applied, as described before [18]. When pulling the applicator back out track ablation was performed until the applicator had left the body. Thus the abdominal wall was always subject to track ablation.
Postoperative imaging and follow-up
The postoperative follow-up was done according to institutional standards. Ultrasound (contrast enhanced, if needed) examination and α-fetoprotein level were checked every three months. Magnetic resonance imaging (MRI), or in selected cases computer tomography scanning, was performed once a year. In addition to this standard procedure, the patients included in this prospective study were evaluated for complete ablation or local recurrence of HCC by MRI on day 7 and every 3 months during the first year. The MRI on day 7 was used for comparison with the follow-up scans. Any new enhancement in the vicinity of the zone of ablation was considered a local recurrence. Further treatment was initiated according to interdisciplinary consultation including all surgical, radiological, and medical options.
Documentation and analysis of data
Patient data and parameters of laparoscopic RFA were prospectively documented in a dedicated database. All calculations were performed using SPSS for Windows version 19.0.0 (SPSS Inc., an IBM Company). Values are given as mean ± standard deviation, unless mentioned otherwise. Survival analysis was done according to the Kaplan–Meier method.
Medline search
A Medline search was done using the keywords “hepatocellular or HCC” and “radiofrequency or radio frequency or radio-frequency” and “laparoscopic or laparoscopy”. The 137 articles identified by this search were further evaluated whether they met the following criteria: (1) data on group or a subgroup of patients consisting only of patients with HCC, (2) treated by laparoscopic RFA and (3) a mean follow-up of at least 12 months is reported.
Results
Results of the Medline search
The primary search identified 137 articles published before January 1, 2012. Fifty-two papers report on RFA by open or percutaneous approach with only a minor number of laparoscopic procedures that are not described in detail, or the method of therapy employed is different than RFA. The topic of further 27 papers is the laparoscopic treatment of different liver tumors others than HCC or only a minor number of HCC. Three papers could not be evaluated due to language. Forty articles reported technical details of the procedure and perioperative results without any or a too short follow-up or without any analysis of survival. Fifteen of these papers report on laparoscopic RFA of HCC with a sufficient follow-up (≥12 months) but some are from one institution or relate to identical cohorts of patients. From these 15, a total of 6 independent papers could be identified that report on the treatment of patients with HCC in a liver cirrhosis by laparoscopic RFA with a mean follow-up of 12 or more months [15, 23–27]. Table 2 summarizes the data reported in these articles.
Procedure-related mortality and morbidity
There was no procedure-related mortality. The 30 days and hospital mortality were zero. There were no surgical complications, no blood transfusions and no secondary complications of RFA such as biliary tract problems or portal vein thrombosis. However, one patient suffered a transitory decrease in liver function, the main symptom being increased ascites. Due to decreased liver function, the hospital stay of three patients was prolonged (average 8.3 ± 4.9 days).
Upstaging due to intraoperative imaging
Laparoscopic ultrasound did change the staging concerning the number of HCC nodules (14 additional were found) in 7 patients or the size of the largest nodules in 15 patients. In three patients multiple additional nodes were identified. In three patients the size of the nodules and multiple additional nodules rendered a complete ablation impossible. None of these three patients are among those that received a liver transplantation.
Overall survival and recurrences of HCC
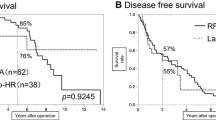
Figure 1 shows the overall survival of the patient cohort. The average time of follow-up was 36.9 ± 28.3 months. Out of the 27 patients that died during follow-up, 3 died of reasons independent of primary liver disease or HCC (1× myocardial infarction, 1× lung TBC, 1× pneumonia). The mean survival of these patients was 44.7 ± 6.9 months (95%CI, 31.2–58.1 months), the mean age at time of death 65.7 ± 9.8 years. The influence of liver transplantation is examined below. Figure 2 displays the overall survival stratified for this factor.
The local recurrence rate was 15 % based on the number of ablations performed. Local recurrence, as defined by radiological criteria, occurred in 2 out of 34 patients. Local recurrence according to histological evaluation of the liver specimen after transplantation was present in 8 out of 14 patients and 9 out of 16 ablations. Thus, in a total of 10 out of 34 patients (29 %), local recurrence was proven. In 18 patients, new HCC nodules in a location different from the treated HCC were diagnosed. Out of these patients, six could be treated by repeated RFA.
Liver transplantation during follow-up
Fifteen patients were on the waiting list for a liver transplantation at the time of laparoscopic RFA. Fourteen patients received a liver graft during follow-up.
Patients that were listed on the waiting list were younger (57 ± 9 years) than those not (67 ± 8 years). The difference was even larger in those that received a transplantation (54 ± 9 years versus 66 ± 8 years) (p < 0.001). Concerning the MELD score, there was no difference between patients receiving a liver graft versus not (mean 10.2 ± 3.2). The average CLIP score was lower in the patients receiving a liver graft (0.8) versus not (1.3), without reaching statistical significance (p = 0.07).
At the time of the first RFA, there was no significant difference in either number of HCC nodules (1.8 versus 1.8) or maximum diameter of the nodule (30.2 versus 33.2 mm) between patients that received a liver graft and those that did not.
The average waiting time was 8.5 ± 7.1 months. No patient intended for transplantation suffered a complication or a seeding of HCC. Histopathologic examination of the liver specimen revealed a complete ablation of the HCC nodule treated in 6 of 14 patients and 7 out of 16 nodules treated. In 7 cases, 13 additional HCC nodules were found on thorough histological examination of the specimen. Due to these additional HCC’s, only one of the 14 patients was without evidence of HCC at the time of transplantation. The average diameter of the HCC’s with complete ablation according to histology was 29 mm at time of RFA and 28 mm at time of histology. The average diameter of those HCC’s that were not completely necrotic was 31 mm at time of RFA and 58 mm at time of histology. Liver transplantation exerted an influence on longtime overall survival with reaching statistical significance. The corresponding values for the mean survival and 95 % confidence interval are with transplantation 70.5 ± 10.9 months (95%CI, 49.2–91.7 months), without transplantation 22.8 ± 4.3 (95%CI, 14.5–31.2 months). The patients receiving a liver transplantation were significantly younger than those not being listed for transplantation.
Discussion
The published clinical experiences using the laparoscopic approach for RFA of HCC nodules are very limited [11–14]. A total of six independent papers could be identified in a Medline search that report on the treatment of patients with HCC in a liver cirrhosis by laparoscopic RFA with a mean follow-up of 12 or more months [15, 23–27]. RFA performed in a laparoscopic way is a safe treatment of liver tumors with deep locations, superficial nodules adjacent to diaphragm and organs or multiple lesions [14, 28]. There is no limitation to treat tumors located centrally in the liver parenchyma or beneath the diaphragm as it is in the percutaneous access. The intraoperative ultrasound in the laparoscopic approach allows a much more accurate staging than the preoperative imaging and an adaptation of the treatment. Laparoscopy enables the visualization and protection of surrounding structures (bowel, stomach) through some laparoscopic maneuvers. According to our results and the cited series, there have not been any procedure-related complications (visceral damage, intraperitoneal bleeding, seeding) as observed in percutaneous RFA [29]. A Pringle maneuver to increase the zone of ablation is contraindicated in liver cirrhosis due to the risk of portal thrombosis. However, an enlargement of the zone of ablation can be reached by reduction of liver blood flow by up to 30–40 % caused by pneumoperitoneum [30]. The Trucut biopsy after RFA allows a safe histological diagnosis without the risk of intraperitoneal seeding of the tumor. The postoperative hospital stay after a laparoscopic RFA is remarkable short (8.3 ± 4.9 days) compared with other studies [26]. Unfortunately, the RFA has a higher incidence of local recurrence compared to liver resection but this is not the only factor that affects the overall survival [23]. A single nodule, a nodule size of 3 cm and more are certainly factors favoring resection [31, 32]. Further data is needed to allow a better stratification for surgery versus RFA [33]. We observed a local recurrence of 15 % of the ablations and an intrahepatic recurrence of new HCCs of 61.8 % during the follow-up due to multifocal character of the disease. Compared with the published survival rates of patients with HCC [29, 34], a better median survival could be achieved.
Conclusion
Laparoscopic RFA is a feasible and reliable therapy for unresectable HCCs especially in patients with cirrhosis when a surgical resection is not possible. It allows an effective local control of the disease and can be performed safely with a low rate of complications. In comparison to percutaneous access, difficulties due to localization of the nodule within the liver are not observed and the size of the zone of ablation can be increased. The laparoscopic RFA combines the advantage of a minimally invasive procedure with the ability of an accurate intraoperative staging by laparoscopic ultrasound. The laparoscopic RFA can provide significant survival for patients with hepatocellular cancer while also forming a bridge to liver transplantation.
References
Teo EK, Fock KM (2001) Hepatocellular carcinoma: an Asian perspective. Dig Dis 19:263–268
Monto A, Wright TL (2001) The epidemiology and prevention of hepatocellular carcinoma. Semin Oncol 28:441–449
Llovet JM (2005) Updated treatment approach to hepatocellular carcinoma. J Gastroenterol 40:225–235
Shiina S, Teratani T, Obi S, Sato S, Tateishi R, Fujishima T, Ishikawa T, Koike Y, Yoshida H, Kawabe T, Omata M (2005) A randomized controlled trial of radiofrequency ablation with ethanol injection for small hepatocellular carcinoma. Gastroenterology 129:122–130
Lin SM, Lin CJ, Lin CC, Hsu CW, Chen YC (2005) Randomised controlled trial comparing percutaneous radiofrequency thermal ablation, percutaneous ethanol injection, and percutaneous acetic acid injection to treat hepatocellular carcinoma of 3 cm or less. Gut 54:1151–1156
Lencioni RA, Allgaier HP, Cioni D, Olschewski M, Deibert P, Crocetti L, Frings H, Laubenberger J, Zuber I, Blum HE, Bartolozzi C (2003) Small hepatocellular carcinoma in cirrhosis: randomized comparison of radio-frequency thermal ablation versus percutaneous ethanol injection. Radiology 228:235–240
Chen MS, Li JQ, Zheng Y, Guo RP, Liang HH, Zhang YQ, Lin XJ, Lau WY (2006) A prospective randomized trial comparing percutaneous local ablative therapy and partial hepatectomy for small hepatocellular carcinoma. Ann Surg 243:321–328
Lu MD, Kuang M, Liang LJ, Xie XY, Peng BG, Liu GJ, Li DM, Lai JM, Li SQ (2006) Surgical resection versus percutaneous thermal ablation for early-stage hepatocellular carcinoma: a randomized clinical trial. Zhonghua Yi Xue Za Zhi 86:801–805
Mulier S, Ni Y, Jamart J, Ruers T, Marchal G, Michel L (2005) Local recurrence after hepatic radiofrequency coagulation: multivariate meta-analysis and review of contributing factors. Ann Surg 242:158–171
Mulier S, Ruers T, Jamart J, Michel L, Marchal G, Ni Y (2008) Radiofrequency ablation versus resection for resectable colorectal liver metastases: time for a randomized trial? An update. Dig Surg 25:445–460
Berber E, Rogers S, Siperstein A (2005) Predictors of survival after laparoscopic radiofrequency thermal ablation of hepatocellular cancer: a prospective study. Surg Endosc 19:710–714
Kim RD, Nazarey P, Katz E, Chari RS (2004) Laparoscopic staging and tumor ablation for hepatocellular carcinoma in Child C cirrhotics evaluated for orthotopic liver transplantation. Surg Endosc 18:39–44
Santambrogio R, Costa M, Barabino M, Opocher E (2008) Laparoscopic radiofrequency of hepatocellular carcinoma using ultrasound-guided selective intrahepatic vascular occlusion. Surg Endosc 22:2051–2055
Chung MH, Wood TF, Tsioulias GJ, Rose DM, Bilchik AJ (2001) Laparoscopic radiofrequency ablation of unresectable hepatic malignancies. A phase 2 trial. Surg Endosc 15:1020–1026
Karabulut K, Aucejo F, Akyildiz HY, Siperstein A, Berber E (2012) Resection and radiofrequency ablation in the treatment of hepatocellular carcinoma: a single-center experience. Surg Endosc 26:990–997
Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, Christensen E, Pagliaro L, Colombo M, Rodes J (2001) Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol 35:421–430
Stigliano R, Marelli L, Yu D, Davies N, Patch D, Burroughs AK (2007) Seeding following percutaneous diagnostic and therapeutic approaches for hepatocellular carcinoma. What is the risk and the outcome? Seeding risk for percutaneous approach of HCC. Cancer Treat Rev 33:437–447
Stippel DL, Bangard C, Prenzel K, Yavuzyasar S, Fischer JH, Holscher AH (2009) Which parameters are needed for targeting a multitined radiofrequency device—an approach to a simple algorithm. Langenbecks Arch Surg 394:671–679
Bangard C, Rosgen S, Wahba R, Wiemker R, Hellmich M, Reiter H, Fischer JH, Stippel DL, Lackner KJ (2009) Large-volume multi-tined expandable RF ablation in pig livers: comparison of 2D and volumetric measurements of the ablation zone. Eur Radiol 20:1073–1078
The Cancer of the Liver Italian Program (CLIP) Investigators (2000) Prospective validation of the CLIP score: a new prognostic system for patients with cirrhosis and hepatocellular carcinoma. Hepatology 31:840–845
Infante-Rivard C, Esnaola S, Villeneuve JP (1987) Clinical and statistical validity of conventional prognostic factors in predicting short-term survival among cirrhotics. Hepatology 7:660–664
Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D’Amico G, Dickson ER, Kim WR (2001) A model to predict survival in patients with end-stage liver disease. Hepatology 33:464–470
Montorsi M, Santambrogio R, Bianchi P, Donadon M, Moroni E, Spinelli A, Costa M (2005) Survival and recurrences after hepatic resection or radiofrequency for hepatocellular carcinoma in cirrhotic patients: a multivariate analysis. J Gastrointest Surg 9:62–67, discussion 67–68
Hsieh CB, Chang HM, Chen TW, Chen CJ, Chan DC, Yu JC, Liu YC, Chang TM, Shen KL (2004) Comparison of transcatheter arterial chemoembolization, laparoscopic radiofrequency ablation, and conservative treatment for decompensated cirrhotic patients with hepatocellular carcinoma. World J Gastroenterol 10:505–508
Santambrogio R, Opocher E, Costa M, Cappellani A, Montorsi M (2005) Survival and intra-hepatic recurrences after laparoscopic radiofrequency of hepatocellular carcinoma in patients with liver cirrhosis. J Surg Oncol 89:218–225, discussion 225–216
Tanaka S, Shimada M, Shirabe K, Taketomi A, Maehara S, Tsujita E, Ito S, Kitagawa D, Maehara Y (2009) Surgical radiofrequency ablation for treatment of hepatocellular carcinoma: an endoscopic or open approach. Hepatogastroenterology 56:1169–1173
Sakaguchi H, Seki S, Tsuji K, Teramoto K, Suzuki M, Kioka K, Isoda N, Ido K (2009) Endoscopic thermal ablation therapies for hepatocellular carcinoma: a multi-center study. Hepatol Res 39:47–52
Santambrogio R, Podda M, Zuin M, Bertolini E, Bruno S, Cornalba GP, Costa M, Montorsi M (2003) Safety and efficacy of laparoscopic radiofrequency ablation of hepatocellular carcinoma in patients with liver cirrhosis. Surg Endosc 17:1826–1832
Curley SA, Izzo F, Ellis LM, Nicolas Vauthey J, Vallone P (2000) Radiofrequency ablation of hepatocellular cancer in 110 patients with cirrhosis. Ann Surg 232:381–391
Jakimowicz J, Stultiens G, Smulders F (1998) Laparoscopic insufflation of the abdomen reduces portal venous flow. Surg Endosc 12:129–132
Ueno S, Sakoda M, Kubo F, Hiwatashi K, Tateno T, Baba Y, Hasegawa S, Tsubouchi H (2009) Surgical resection versus radiofrequency ablation for small hepatocellular carcinomas within the Milan criteria. J Hepatobiliary Pancreat Surg 16:359–366
Crocetti L, de Baere T, Lencioni R (2010) Quality improvement guidelines for radiofrequency ablation of liver tumours. Cardiovasc Intervent Radiol 33:11–17
Cho YK, Kim JK, Kim WT, Chung JW (2010) Hepatic resection versus radiofrequency ablation for very early stage hepatocellular carcinoma: a Markov model analysis. Hepatology 51:1284–1290
Nagasue N, Kohno H, Tachibana M, Yamanoi A, Ohmori H, El-Assal ON (1999) Prognostic factors after hepatic resection for hepatocellular carcinoma associated with Child-Turcotte class B and C cirrhosis. Ann Surg 229:84–90
Conflicts of interest
All authors (Till Herbold, Roger Wahba, Christopher Bangard, Münevver Demir, Uta Drebber, Dirk L. Stippel) have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Herbold, T., Wahba, R., Bangard, C. et al. The laparoscopic approach for radiofrequency ablation of hepatocellular carcinoma—indication, technique and results. Langenbecks Arch Surg 398, 47–53 (2013). https://doi.org/10.1007/s00423-012-1018-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-012-1018-5