Abstract
Purpose
Betalains are indole-derived pigments found in beet root, and recent studies suggest that they may exert ergogenic effects. Herein, we examined if supplementation for 7 days with betalain-rich beetroot concentrate (BLN) improved cycling performance or altered hemodynamic and serum analytes prior to, during and following a cycling time trial (TT).
Methods
Twenty-eight trained male cyclists (29 ± 10 years, 77.3 ± 13.3 kg, and 3.03 ± 0.62 W/kg) performed a counterbalanced crossover study whereby BLN (100 mg/day) or placebo (PLA) supplementation occurred over 7 days with a 1-week washout between conditions. On the morning of day seven of each supplementation condition, participants consumed one final serving of BLN or PLA and performed a 30-min cycling TT with concurrent assessment of several physiological variables and blood markers.
Results
BLN supplementation improved average absolute power compared to PLA (231.6 ± 36.2 vs. 225.3 ± 35.8 W, p = 0.050, d = 0.02). Average relative power, distance traveled, blood parameters (e.g., pH, lactate, glucose, NOx) and inflammatory markers (e.g., IL-6, IL-8, IL-10, TNFα) were not significantly different between conditions. BLN supplementation significantly improved exercise efficiency (W/ml/kg/min) in the last 5 min of the TT compared to PLA (p = 0.029, d = 0.45). Brachial artery blood flow in the BLN condition, immediately post-exercise, tended to be greater compared to PLA (p = 0.065, d = 0.32).
Conclusions
We report that 7 days of BLN supplementation modestly improves 30-min TT power output, exercise efficiency as well as post-exercise blood flow without increasing plasma NOx levels or altering blood markers of inflammation, oxidative stress, and/or hematopoiesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Beetroot contains multiple phytochemical compounds along with bioactive pigments known as betalains (Lee et al. 2005; Clifford et al. 2015). Betalains are classified as nitrogen-containing pigments with two distinct chemical structural groups, betacyanins and betaxanthins, which are responsible for their respective red/violet and yellow color (Delgado-Vargas et al. 2000; Strack et al. 2003; Kujala et al. 2001). Betalains have been shown to be absorbed through paracellular junctions of human intestinal epithelial cells in vitro (Tesoriere et al. 2013), although in vivo analysis in humans has provided mixed results regarding the bioavailability of betalains (Tesoriere et al. 2004; Clifford et al. 2017). In vivo and in vitro studies have revealed that betalains possess general health-related benefits including antioxidant (Tesoriere et al. 2003, 2004; Butera et al. 2002; Escribano et al. 1998; Kanner et al. 2001; Zielinska-Przyjemska et al. 2009; Albano et al. 2015), anti-nitrosative (Sakihama et al. 2012) and anti-inflammatory (Vidal et al. 2014; Zielinska-Przyjemska et al. 2009; Pietrzkowski et al. 2010) properties. In this regard, betalains have been well documented to be robust free radical and nitrogen scavengers (Gandia-Herrero et al. 2009, 2010; Sakihama et al. 2012). Additionally, betalains have been reported to protect red blood cells against oxidative damage and hemolysis (Tesoriere et al. 2005), and some supporting literature suggests that betalains can promote hematopoiesis (Cho et al. 2017). Betalains have also been shown to reduce the production of superoxide anions and cytokines such as TNF-α and interleukins (Martinez et al. 2015).
Exercise has been shown to increase the production of free radicals and reactive oxygen species (Reid et al. 1992; O’Neill et al. 1996; Powers et al. 2004; Yavari et al. 2015) and cytokine production (Pedersen 2000; Pedersen et al. 2001). Specifically, exercise-induced increases in reactive oxygen species and cytokines have been linked to skeletal muscle fatigue and decrements in performance (Barclay and Hansel 1991; Vincent et al. 2000; Allen et al. 2008; Burt and Twist 2011), although the repeated oxidative stress response to exercise promotes increases in free radical defense systems chronically (Criswell et al. 1993; Powers et al. 1994; Vincent et al. 1999). Exercise has also been shown to increase nitric oxide bioavailability, and impairments in this process may limit exercise capacity (Vincent et al. 1999). There has been recent interest in the potential ergogenic effects of betalain supplementation given that these compounds elicit a wide variety of purported biological and physiological effects, which may offset physiological perturbations up-regulated during exercise (e.g., increased cytokine production, increased oxidative stress, etc.). In this regard, two recent investigations reported that short-term (7-day) betalain-rich beet root concentrate (BLN) supplementation improved 5- and 10-km time trial (TT) performance in competitive male runners (Van Hoorebeke et al. 2016) and male and female competitive triathletes (Montenegro et al. 2017), respectively. The improvement in TT performance in both aforementioned studies was attributed to a number of potential possibilities such as decreased muscle damage, inflammation and oxidative stress. However, with the limited number of investigations with regard to the effects of BLNs on exercise performance, mechanisms underlying these effects are still unclear and under-investigated.
Beet root juice supplementation has been shown to improve exercise efficiency (Bailey et al. 2009; Lansley et al. 2011) as well as TT performance in both runners and cyclists (Bailey et al. 2009; Cermak et al. 2012; Lansley et al. 2011); effects thought to be largely due to improved blood flow and oxygen delivery via the high nitrate content of beet root juice (Bailey et al. 2009; Cermak et al. 2012; Lansley et al. 2011). Given that the BLN supplementation tested in the studies of Van Hoorebeke et al. (2016) and Montenegro et al. (2017) were free of nitrates, these authors suggested that the observed performance improvements were not necessarily due to increased nitric oxide (NO) availability. However, these authors could not rule out potential improvements in blood flow due to a lack of measuring hemodynamic characteristics. Therefore, given the limited number of investigations regarding BLN supplementation and exercise performance, the purpose of the current study was twofold: (1) to investigate the physiological and TT performance effects of a similar short-term (7 days) BLN supplementation protocol using well-trained cyclists, and (2) investigate if BLN supplementation alters hemodynamic variables including blood flow, serum NO availability and serum markers of inflammation and oxidative stress in attempt to identify and/or rule out underlying mechanisms of how BLNs are improving exercise performance. Based upon previous performance studies, we hypothesized that BLN supplementation would increase TT cycling performance and modify post-exercise markers of oxidative stress and inflammation.
Methods
Subjects
Twenty-eight male cyclists (age 28.8 ± 9.8 years; height 180.63 ± 6.53 cm; body mass 77.3 ± 13.3 kg) were recruited from the Auburn University cycling community, local cycling clubs and bike shops. Participants were included if they were: (a) not currently consuming beet roots; (b) cycling 3–7 days per week and totaling a weekly mileage of greater than 100 miles; (c) 19–50 years of age; (d) free of any known cardiovascular, respiratory, metabolic, or musculoskeletal ailments, and (e) were not taking any other supplements or ergogenic aids. Prior to initiating the study, participants were informed of the experimental procedures, read and signed Informed Consent forms, and filled out a medical history questionnaire. Notably, all consent and experimental procedures were approved through the Auburn University Institutional Review Board (protocol #: 15-533 MR 1601), and conformed to the standards set by the latest revision of the Declaration of Helsinki.
Experimental protocol
This study implemented a randomized, double-blind, placebo controlled crossover design whereby participants performed two separate experimental 30-min TTs following 1 week of either BLN or PLA supplementation (Fig. 1).
For the initial baseline visit, participants reported to the laboratory following a minimum of a 4-h fast. Participants’ height and weight were recorded, and participants were then administered a standardized meal consisting of a protein pudding (MHP, West Caldwell, NJ, USA), granola bar (Quaker, Chicago, IL, USA) and 20 oz water (calorie content: 380 kcal, 37 g of carbohydrates, 11.5 g of fats and 33 g of protein), while resting quietly for 105 min. Thereafter, blood samples were collected via a finger stick for capillary blood/gas analysis (described in greater detail below) and venipuncture from the antecubital vein using a 10-mL serum separator tube and a 10 mL EDTA tube (BD Vacutainer, Franklin Lakes, NJ, USA). Participants were then instructed to rest quietly in a supine position for 10 min followed by blood pressure, heart rate (HR) and brachial artery flow-mediated dilation (FMD) measurements. A subset of participants (n = 18) performed a familiarization (FAM) 30-min TT (described in greater detail below). The purpose of the FAM TT was to examine if statistical differences in performance existed between the PLA and FAM TTs to ensure a learning effect had not occurred Thereafter, all participants were randomized using a simple randomization procedure whereby participants received 6 days of either BLN (betalain-rich beetroot concentrate, 100 mg/day, FutureCeuticals, Momence, IL, USA) or PLA (silica dioxide, 100 mg/day) supplementation in pill form, and instructed to take one pill (50 mg/pill) in the morning and one in the evening. The supplementation protocol was replicated from Montenegro et al. (2017). Participants were then scheduled to return to the laboratory 7 days later at the same time of day for the first experimental 30-min TT.
Upon arrival for the first experimental TT, participants provided a urine sample for hydration status, body weight was recorded, a single 50-mg dose of BLN was administered (seventh day of supplementation) and then sat quietly for 15 min. Thereafter, the exact same procedures described during the baseline visit were followed regarding the administration of a standardized meal, sitting quietly for 105 min, collection of blood samples, resting quietly for 10 min, and blood pressure, HR and FMD measurements. Participants were then fit on either their personal road bike or a provided road bike. For all TTs, the rear wheel was equipped with a CycleOps power meter (Madison, WI, USA) and synced to a GPS device (Garmin Edge 500, Olathe, KS, USA). The rear wheel was attached to a Computrainer (Racermate, Inc. Seattle, WA, USA) for adjustment of resistance with a magnetic braking system as previously described by our laboratory (Kephart et al. 2016). Participants were then outfitted with a finometer and mouth piece attached to a metabolic cart (both described in greater detail below) and instructed to sit quietly on the bike for 5 min to obtain pre-exercise resting values. Thereafter, a 5-min warm-up of self-selected intensity was performed prior to initiating the 30-min TT. Immediately following the warm-up, the 30-min TT commenced whereby power (W), distance (km) and metabolic cart measures were recorded every 5 min and capillary blood samples were collected every 10 min. Notably, the TT was performed in an environmental chamber (Panelized Walk-In Chamber, ESPEC, US) set to a temperature of 20 °C and a relative humidity of 35%. Upon completion of the TT, an immediate antecubital vein venipuncture was performed along with a finger stick and metabolic cart measures were collected at 5 and 15 min following the TT to obtain post-exercise values. Thereafter, FMD was performed and a second (1-h) post-exercise venipuncture was obtained.
Participants were then scheduled for a second baseline visit for the following week whereby no supplementation occurred (i.e., a 7-day washout period). The second baseline visit was procedurally identical to the initial baseline visit. Upon completion of the second baseline visit, participants were given the supplement that was not previously consumed to consume over the following 6 days and then scheduled for the second 30-min TT the following week. Thereafter, the second 30-min TT procedures were performed identical to the first experimental TT. More in-depth procedures are described below.
Anthropometric and urine assessments
Standing height was measured to the nearest 0.5 cm using a calibrated stadiometer and body mass was measured to the nearest 0.01 kg using a calibrated clinical scale with participants wearing minimal clothing. Systolic and diastolic brachial blood pressure and HR were measured via an automatic blood pressure cuff (model BP785N; Omron Healthcare, INC., Lake Forest, IL, USA) in a supine position after resting quietly for 10 mins. Hydration status was assessed by measuring urine specific gravity (USG) via a handheld refractometer (model CLX-1, precision = 0.001 T 0.001; VEE GEE Scientific, Inc., Kirkland, WA, USA). A reading between 1.000 and 1.029 for USG was considered adequately hydrated (Casa et al. 2000; Mumford et al. 2016). If a USG reading was at or above 1.030, participants were instructed to drink additional water while resting comfortably until a second urine sample could be provided. Hydration was assessed to ensure participants were adequately hydrated and to minimize the effects of dehydration on performance.
Exercise performance measures
As mentioned prior, a CycleOps power meter (Madison, WI, USA) was synced to a GPS device (Garmin Edge 500, Olathe, KS, USA) and was used for analysis of watts and distance cycled during the 30-min TT. Watts and distance were measured per second and were averaged over the 30-min TT. Finger cuff photoplethysmography was used during the TT to measure HR, beat-by-beat arterial blood pressure, stroke volume and cardiac output (CO) via a Finometer PRO (Finapres Medical Systems, Amsterdam, The Netherlands) as previously described (Haun et al. 2016). Oxygen uptake (VO2) and respiratory gas exchange ratio (RER) were analyzed using a Parvo Medics TrueOne 2400 metabolic cart (Parvo Medics, Sandy, UT, USA) at minutes 3–5, 8–10, 13–15, 18–20, 23–25 and 28–30 during the TT, and exercise efficiency was calculated as W/VO2 for each 5-min stage during each TT. Metabolic cart measures were additionally sampled at minutes 3–5 and 13–15 following the TT.
Blood gas analytes
Approximately 65 µL of capillary blood was obtained by a small puncture to the tip of the finger using a lancet, collected via a lithium heparin microcapillary tube (Instrumentation Laboratory, Bedford, MA, USA) for analysis of pH, lactate and glucose using a GEM Premier 4000 blood gas analyzer (Instrumentation Laboratory, Bedford, MA, USA). Assayed time points included the two baseline visits, immediately prior to each TT, min 10, 20 and 30 during each TT, and min 5 and 15 following each TT. Due to technical difficulties with the analyzer, blood gas analytes were only obtained for 20 of the 28 subjects.
Serum and plasma blood analysis
Venous blood samples were collected at each of the two baseline visits, immediately prior to each TT, immediately following each TT, and 1 h following each TT. Following collection, serum separator and EDTA tubes were centrifuged at 3500g at 4 °C for 10 min and then aliquoted and immediately stored in a − 80 °C freezer for future batch-processing of serum thiobarbituric acid reactive substances (TBARS), serum erythropoietin (EPO), plasma cytokines, and plasma nitrate and nitrite (NOx). Serum TBARS were analyzed using a commercially available 96-well assay following the manufacturer’s procedures (Cayman, Ann Arbor, MI, USA), and resulted in an average intra-assay coefficient of variation (%CV) of 1.47%. Serum EPO was determined using a commercially available 96-well assay following the manufacturer’s procedures (Abcam, Cambridge, MA, USA), and resulted in an average intra-assay %CV of 4.7%. Plasma concentrations of IL-2, IL-4, IL-6, IL-8, IL-10, TNFα, GM-CSF and INFγ cytokines were determined using a commercially available 96-well multiplex assay following the manufacturer’s procedures (Bio-Rad, Hercules, CA, USA), and resulted in an average intra-assay %CV of ≤ 5.1% for all targets. Notably, IL-2, IL-4, GM-CSF and INFγ were not reported due to concentrations being below the sensitivity of the assay. For analysis of plasma NOx, samples were deproteinized in ice-cold ethanol, and assayed via ozone-based chemiluminescence using a NO analyzer (Sievers NOA 280i, Boulder, CO, USA). Briefly, samples were added to 0.1 M vanadium chloride in 1 M hydrochloric acid refluxing at 95 °C under nitrogen. A cooled photomultiplier tube housed in the analyzer detected respective reduction to NO and concentrations of NOx were determined by plotting signal (mV) against a calibration plot. Quantification of reticulocytes was performed using flow cytometry, whereby 5 µL of whole blood was mixed with 1 mL of BD Retic-Count (BD Biosciences, San Jose, CA, USA), triturated, gently vortexed and blocked from light. Flow cytometry was performed using a BD Accuri C6 (BD Biosciences, San Jose, CA, USA). Notably, serum and plasma analytes were only collected on 20 of the 28 participants due to one or more missed blood draws.
Flow-mediated dilation
FMD was used to determine endothelial reactivity in the right brachial artery with the use of a high-resolution ultrasound (Logiq S7 R2 Expert; General Electric, Fairfield, CT, USA) and a 3–12 MHz multi-frequency linear transducer. Specifically, the transducer was placed longitudinally 3–5 cm above the antecubital fossa and was either held manually or by use of a mechanical arm in a fixed position to measure brachial artery diameter and blood velocity. This was performed as previously described (Haun et al. 2016) via a duplex mode imaging (B-mode and Doppler), in which video was recorded with real-time analysis software (FMD Studio, Pisa, Italy). Furthermore, resting data was recorded for 1 min, and reactive hyperemia was produced by inflation of a blood pressure cuff placed 1–2 cm below the elbow for 5 min at 200 mmHg. Thereafter, the cuff was deflated and data were recorded for an additional 3 min. Vessel diameters were determined via automatic edge detection software (FMD Studio, Pisa, Italy) by measuring the distances between the walls of the intima. Additionally, blood velocity was determined by a defined region of interest via Doppler waveform. FMD is reported as relative (%FMD), normalized (nFMD) calculated as {100 × %FMD/[(4 × time averaged mean velocity)/vessel diameter] area under the curve − 1}. Blood flow was calculated from diameter and average blood velocity [Π × (diameter/2) 2 × time average mean velocity × 60]. Notably, FMD measures were only collected on a subset of 18 of the 28 participants.
Statistics
Statistical analyses were performed using SPSS for Windows version 21.0 (IBM, Seattle, WA, USA), and data herein are reported as mean ± standard deviation (SD) for all dependent variables. Prior to statistical analysis, the assumption of normality was tested using Shapiro–Wilk’s test for all dependent variables. Statistical analysis for average absolute power, relative average power and total distance over the 30-min TT was examined using one-tailed paired samples t tests in order to test our a priori hypothesis that BLN supplementation would improve performance. Familiarization compared to PLA was examined using a two-tailed paired samples t test to determine if a learning effect occurred. All other dependent variables were examined using two-tailed (condition by time) factorial repeated measures. If the assumption of heteroscedasticity for repeated measures was violated, a Greenhouse–Geisser correction factor was applied. Additionally, if significant interactions were found, data were further decomposed using one-way repeated measures ANOVA across time and paired samples t tests for between conditions. Significance for all null hypothesis testing was set at p ≤ 0.05, although p values “approaching significance” (0.05 < p < 0.10) were also discussed as meaningful given the exploratory nature of this analysis. Furthermore, 95% confidence intervals (CI) of the mean difference between conditions, and magnitude of the effect was calculated using Cohen d effect size with observed effects for cycling performance variables (i.e., average absolute power, average relative power and total distance between conditions), where 0.2, 0.5 and 0.8 were considered small, moderate and large effects.
Results
Effects of BLN supplementation on resting hemodynamics
Seven days of BLN supplementation had no effect on resting hemodynamic parameters. No significant differences were observed for baseline blood pressure readings for either diastolic (PLA vs. BLN, 74.60 ± 5.75 vs. 75.64 ± 7.73 mmHg) or systolic (PLA vs. BLN, 125.44 ± 7.21 vs. 125.76 ± 10.32 mmHg) pressures. Furthermore, no significant differences were observed for resting HR (PLA vs. BLN, 61.50 ± 10.84 bpm vs. 60.54 ± 12.12 bpm) or USG readings (PLA vs. BLN, 1.004 ± 0.021 vs. 1.005 ± 0.007).
Cycling performance
Due to technical difficulties, distance traveled over the 30-min TT was only collected on 24 of the 28 subjects. Notably, for the 18 subjects that underwent a familiarization (FAM) TT, average absolute power was not significantly different between the FAM and PLA conditions (225.1 ± 40.7 vs. 225.3 ± 35.8 W, p = 0.857), respectively.
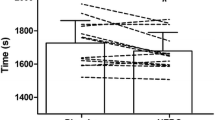
There was a statistically significant difference between conditions for average absolute power (p = 0.050) over the entire TT whereby BLN was greater than PLA [mean difference (95% CI), 6.3 W (− 1.0 to 13.6 W); Fig. 2a], however, this difference resulted in a small effect (d = 0.02). There were no statistically significant differences between conditions for average relative (bodyweight adjusted) power [0.08 W/kg (− 0.02 to 0.19); p = 0.058; Fig. 2b] or distance traveled over the 30-min TT [0.33 km (− 0.08 to 0.74); p = 0.064; Fig. 2c]. However, it is worth noting, based on our a priori alpha, that these aforementioned performance variables were trending towards a statistically significant difference, albeit the effect size analysis resulted in small effect sizes of d = 0.13 and 0.02, respectively.
Oxygen uptake and hemodynamics
There was no significant main effect of condition or condition by time interaction for relative VO2 over the TT. However, there was a significant main effect of time (p < 0.001; Fig. 3a). Additionally, there was a significant condition by time interaction for 5 min. average exercise efficiency (p = 0.045), with the simple effect of time being statistically significant for both PLA and BLN (p < 0.001) conditions. Post hoc analysis revealed a significant increase in average exercise efficiency (p = 0.029) in the BLN (7.63 ± 3.45 W/mL O2/kg/min) compared to PLA (6.23 ± 2.73 W/mL O2/kg/min) condition at the 30-min time point [1.33 W/mL O2/kg/min (− 0.01 to 2.66); Fig. 3b], which resulted in a moderate effect (d = 0.45). Furthermore, there were significant main effects of time for all exercise hemodynamic variables including HR (p < 0.001; Fig. 3c), SV (p < 0.001; Fig. 3d), total peripheral resistance (p < 0.001; Fig. 3e) and CO (p < 0.001). However, there were no significant condition or condition by time interactions for any of these variables.
Effects of BLN on VO2 and hemodynamic variables. a Relative VO2 at 5-min intervals during and at 5 and 15 min following the trial. b Power output relative to VO2 at 5-min intervals during the trial. c Heart rate over 5-min intervals during and at 5, 10 and 15 min following the trial. d Stroke volume over 5-min intervals during and at 5, 10 and 15 min following the trial. e Total peripheral resistance (TPR) over 5-min intervals during and at 5, 10 and 15 min following the trial. f Cardiac output over 5-min intervals during and at 5, 10 and 15 min following the trial. Data are presented as mean ± standard deviation. PLA placebo condition, BLN betalain condition. Symbols: asterisk indicates significant difference between condition at a given time point (p < 0.05)
Conduit artery reactivity and blood flow
There was a statistically significant condition by time interaction effect for brachial artery blood flow (p = 0.043; Fig. 4a). Post hoc analysis revealed no statistically significant differences between experimental conditions at any time point. However, blood flow in the BLN condition immediately post-exercise tended to be greater (p = 0.065) with the size of the effect being small to moderate (d = 0.32). Additionally, there was no statistically significant main effect of condition or condition by time interaction for FMD. However, there was a statistically significant main effect of time (p = 0.040; Fig. 4c). Finally, there were no statistically significant main effects or condition by time interaction for nFMD (Fig. 4d).
Effects of BLN on pre- and post-exercise blood flow and plasma NOx levels. a Brachial artery blood flow prior to the supplementation period (baseline), prior to the 30-min TT (PreCycle), and immediately following the 30-min TT (IP). b Plasma NOx levels at the PreCycle and IP time points. Data are presented as mean ± standard deviation. c, d Flow-mediated dilation (FMD) and normalized flow-mediated dilation (nFMD) at baseline, Precycle and IP. PLA placebo condition, BLN betalain condition
Blood analysis
There were no statistically significant condition, time, or condition by time effects for plasma NOx levels (Fig. 4b). There were statistically significant main effects of time for select serum and whole-blood variables including serum EPO (p < 0.001; Fig. 5b), whole-blood reticulocyte count (p < 0.001; Fig. 5c), whole-blood pH (p < 0.001; Fig. 5d), whole-blood glucose (p < 0.001; Fig. 5e), and whole-blood lactate (p < 0.001; Fig. 5f). However, there were no statistically significant effects of condition or condition by time interactions (p < 0.05) for any of the assayed markers.
Effects of BLN on select serum and whole-blood markers. a Serum TBARS levels prior to the supplementation period (baseline), prior to the 30-min TT (PreCycle), immediately following the 30-min TT (IP), and 1 h following the 30-min time trial (1 h Post). b Serum EPO levels at the baseline, PreCycle, IP, and 1 h Post time points. c Whole-blood reticulocyte counts at the baseline, PreCycle, IP and 1 h post time points. d–f Whole-blood pH, glucose, and lactate over 5-min intervals during and at 5 and 15 min following the trial. Data are presented as mean ± standard deviation. PLA placebo condition, BLN betalain condition
Plasma inflammatory markers
There were statistically significant main effects of time for serum IL-6 (p = 0.011; Fig. 6a), serum IL-8 (p = 0.002; Fig. 6b) serum IL-10 (p = 0.044; Fig. 6c) and serum TNF-α (p = 0.007; Fig. 6d). However, there were no statistically significant effects of condition or condition by time interactions (p < 0.05) for any of the assayed markers.
Effects of BLN on select plasma inflammatory markers. a Plasma IL-6 levels prior to the supplementation period (baseline), prior to the 30-min TT (PreCycle), immediately following the 30-min TT (IP), and 1 h following the 30-min time trial (1 h Post). b–d depict plasma IL-8, IL-10, and TNFα levels at the baseline, PreCycle, IP, and 1 h Post time points. Data are presented as mean ± standard deviation. PLA placebo condition, BLN betalain condition
Discussion
The objective of this study was to confirm the purported ergogenic effects of BLN supplementation as well as determine other meaningful physiological effects in trained cyclists. Our findings suggest that BLN supplementation, taken for 7 days prior to and immediately before a 30-min cycling TT, significantly improves power output and exercise efficiency. Additionally, BLN supplementation tended to modestly improve distance traveled and immediate post-exercise blood flow. However, BLN supplementation does not seem to alter serum NOx availability or relevant serum analytes of oxidative stress or inflammation.
Effects of BLN supplementation on exercise performance
Our findings of improved TT performance in trained competitive cyclists are in partial agreement with (Montenegro et al. 2017) who reported that BLN supplementation, using the same dosing strategy employed in the current study, improved 10-km TT running performance in a sample of competitive triathletes. However, these authors also reported that BLN supplementation failed to improve average power output during a 40-min sub-maximal cycling bout, which occurred immediately prior to the running TT. Similar findings were reported by Van Hoorebeke et al. (2016) who reported sub-chronic BLN supplementation resulted in improved 5-km TT performance in which trained running athletes improved their average speed and decreased their time to complete the TT. As in the Montenegro et al. (2017) study, running athletes in the Van Hoorebeke et al. (2016) study failed to improve sub-maximal running performance during a 30-min bout of treadmill exercise, which occurred immediately prior to the 5-km TT. Interestingly, Van Hoorebeke et al. (2016) did note a reduction in heart rate and change in lactate levels when exercise was performed at the same relative intensities under the BLN condition compared to PLA, suggestive of improved exercise efficiency.
Importantly, Montenegro et al. (2017) speculated that performance improvements could potentially be due to anti-nitrosative properties of BLN (Sakihama et al. 2012), which may lead to higher levels of nitric oxide and result in improved exercise blood flow. Our data partially supports the proposed hypothesis by Montenegro et al. (2017) given that we observed a modest improvement in post-exercise blood flow, but no increase in plasma NOx levels with BLN supplementation. In this regard, the modest improvement in post-exercise blood flow could be related to our findings of improved exercise performance and a potential mechanism by which BLN improves exercise performance. Furthermore, we observed that BLN supplementation did not elicit alterations in other markers related to potential improvements in performance (e.g., increased stroke volume, or reduced blood lactate or pH levels). Therefore, future research examining the efficacy of BLN supplementation should continue to explore how BLN increases exercise economy and blood flow during and following exercise.
BLN supplementation does not alter inflammatory markers
Past research has indicated that maximal cycling exercise increases serum levels of IL-6, TNFα, TBARS and erythrocytes in sedentary subjects (Steinberg et al. 2007). Furthermore, in elite cyclists following a 4-day cycling competition, increases in cytokine production were reported at each stage of a cycling race (Cordova Martinez et al. 2015). Brenner et al. (Brenner et al. 1999) reported that 2 h of cycling, compared to maximal intensity short duration and interval cycling, increased cytokine production (e.g., IL-6 and TNFα). Herein, we observed a time effect whereby serum IL-6 and IL-10 increases were relatively mild following cycling, albeit BLN supplementation did not abrogate these increases. Additionally, we did not observe exercise-induced increases in serum TBARS. Importantly, the average duration and distance traveled during the 30-min TT in the current study, when compared to other studies, was shorter by comparison and may explain the disparate findings. Moreover, given that trained cyclists were examined in the present study, it is possible that these participants were well adapted to the perhaps relatively less challenging stresses of the 30-min TT employed. Thus, studying the potential anti-inflammatory or anti-oxidant effects of BLN supplementation with more prolonged or intense exercise trials is warranted.
Effects of BLN supplementation on markers of hematopoiesis
Limited evidence suggests that BLN can potentially affect hematopoiesis and/or reduce hemolysis in vitro (Cho et al. 2017). We observed time effects regarding TT-induced increases in reticulocyte counts and serum EPO levels, although BLN supplementation did not augment these effects relative to the PLA condition. Indeed, our findings that exercise increased reticulocyte counts and serum EPO are in agreement with previous observations (Morici et al. 2005; Schwandt et al. 1991). However, BLN supplementation does not apparently affect these markers in vivo under the current study design.
Limitations
It should be noted that the use of TBARS as a marker of oxidative stress may not be the most relevant, and therefore, interpretation of the effects of BLN supplementation on oxidative stress in the current study should be read with caution. Furthermore, Garmin file corruption during the trial resulted in loss of distance traveled over the 30-min TT for four participants. Additionally, quantification of plasma betalains was not measured herein, and future investigations regarding absorption, transportation and quantification are warranted. Finally, the results of the current study are limited to trained cyclists, and thus, future investigations in recreationally trained populations are warranted.
Conclusion
We report that 7 days of sub-chronic supplementation of BLN prior to and immediately before a 30-min TT modestly improves power output, distance traveled, exercise efficiency, and blood flow without altering serum NOx bioavailability, serum markers of inflammation and oxidative stress, and/or hematopoiesis. These observed improvements in said performance parameters, although statistically small to moderate in magnitude and perhaps due to the fact that the tested group was highly trained, are in reasonable accord with previously published reports on BLN supplementation and further support the hypothesis that BLN may be of potential benefit to competitive athletes. In this regard, the observed changes in distance traveled (0.33 km (− 0.08 to 0.74); p = 0.064), although relatively small at first glance, could afford a practical advantage to competitive cyclists. For example, this change would represent approximately 100 additional meters traveled during a 5k TT which is of practical significance. It is interesting that BLN supplementation significantly improved exercise efficiency (W/ml/kg/min) during the last 5 min of the TT compared to PLA (p = 0.029, d = 0.45). Given that this is only the third study examining the potential ergogenic effects of BLN supplementation, we contend that more research is needed to examine the efficacy of this ingredient with more prolonged exercise as compared to “sprint” or “burst of energy” conditions, on a more diverse study population, or a larger population, and possibly with different dosing strategies.
Abbreviations
- ANOVA:
-
Analysis of variance
- BLN:
-
Betalain-rich beetroot concentrate
- CO:
-
Cardiac output
- EPO:
-
Erythropoietin
- FMD:
-
Flow-mediated dilation
- HR:
-
Heart rate
- NO:
-
Nitric oxide
- PLA:
-
Placebo
- RER:
-
Respiratory exchange ratio
- TBARS:
-
Thiobarbituric acid reactive substances
- TT:
-
Time trial
References
Albano C, Negro C, Tommasi N, Gerardi C, Mita G, Miceli A, De Bellis L, Blando F (2015) Betalains, phenols and antioxidant capacity in cactus pear [Opuntia ficus-indica (L.) Mill.] fruits from Apulia (South Italy) genotypes. Antioxidants (Basel) 4(2):269–280. https://doi.org/10.3390/antiox4020269
Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88(1):287–332. https://doi.org/10.1152/physrev.00015.2007
Bailey SJ, Winyard P, Vanhatalo A, Blackwell JR, Dimenna FJ, Wilkerson DP, Tarr J, Benjamin N, Jones AM (2009) Dietary nitrate supplementation reduces the O2 cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J Appl Physiol (Bethesda Md 1985) 107(4):1144–1155. https://doi.org/10.1152/japplphysiol.00722.2009
Barclay JK, Hansel M (1991) Free radicals may contribute to oxidative skeletal muscle fatigue. Can J Physiol Pharmacol 69(2):279–284
Brenner IK, Natale VM, Vasiliou P, Moldoveanu AI, Shek PN, Shephard RJ (1999) Impact of three different types of exercise on components of the inflammatory response. Eur J Appl Physiol Occup Physiol 80(5):452–460. https://doi.org/10.1007/s004210050617
Burt DG, Twist C (2011) The effects of exercise-induced muscle damage on cycling time-trial performance. J Strength Cond Res 25(8):2185–2192. https://doi.org/10.1519/JSC.0b013e3181e86148
Butera D, Tesoriere L, Di Gaudio F, Bongiorno A, Allegra M, Pintaudi AM, Kohen R, Livrea MA (2002) Antioxidant activities of sicilian prickly pear (Opuntia ficus-indica) fruit extracts and reducing properties of its betalains: betanin and indicaxanthin. J Agric Food Chem 50(23):6895–6901
Casa DJ, Armstrong LE, Hillman SK, Montain SJ, Reiff RV, Rich BS, Roberts WO, Stone JA (2000) National athletic trainers’ association position statement: fluid replacement for athletes. J Athl Train 35(2):212–224
Cermak NM, Gibala MJ, van Loon LJ (2012) Nitrate supplementation’s improvement of 10-km time-trial performance in trained cyclists. Int J Sport Nutr Exerc Metab 22(1):64–71
Cho J, Bing SJ, Kim A, Lee NH, Byeon SH, Kim GO, Jee Y (2017) Beetroot (Beta vulgaris) rescues mice from gamma-ray irradiation by accelerating hematopoiesis and curtailing immunosuppression. Pharm Biol 55(1):306–319. https://doi.org/10.1080/13880209.2016.1237976
Clifford T, Howatson G, West DJ, Stevenson EJ (2015) The potential benefits of red beetroot supplementation in health and disease. Nutrients 7(4):2801–2822. https://doi.org/10.3390/nu7042801
Clifford T, Constantinou CM, Keane KM, West DJ, Howatson G, Stevenson EJ (2017) The plasma bioavailability of nitrate and betanin from Beta vulgaris rubra in humans. Eur J Nutr 56(3):1245–1254. https://doi.org/10.1007/s00394-016-1173-5
Cordova Martinez A, Martorell Pons M, Sureda Gomila A, Tur Mari JA, Pons Biescas A (2015) Changes in circulating cytokines and markers of muscle damage in elite cyclists during a multi-stage competition. Clin Physiol Funct Imaging 35(5):351–358. https://doi.org/10.1111/cpf.12170
Criswell D, Powers S, Dodd S, Lawler J, Edwards W, Renshler K, Grinton S (1993) High intensity training-induced changes in skeletal muscle antioxidant enzyme activity. Med Sci Sports Exerc 25(10):1135–1140
Delgado-Vargas F, Jimenez AR, Paredes-Lopez O (2000) Natural pigments: carotenoids, anthocyanins, and betalains—characteristics, biosynthesis, processing, and stability. Crit Rev Food Sci Nutr 40(3):173–289. https://doi.org/10.1080/10408690091189257
Escribano J, Pedreño MA, García-Carmona F, Muñoz R (1998) Characterization of the antiradical activity of betalains from Beta vulgaris L. roots. Phytochem Anal 9(3):124–127
Gandia-Herrero F, Escribano J, Garcia-Carmona F (2009) The role of phenolic hydroxy groups in the free radical scavenging activity of betalains. J Nat Prod 72(6):1142–1146. https://doi.org/10.1021/np900131r
Gandia-Herrero F, Escribano J, Garcia-Carmona F (2010) Structural implications on color, fluorescence, and antiradical activity in betalains. Planta 232(2):449–460. https://doi.org/10.1007/s00425-010-1191-0
Haun CT, Kephart WC, Holland AM, Mobley CB, McCloskey AE, Shake JJ, Pascoe DD, Roberts MD, Martin JS (2016) Differential vascular reactivity responses acutely following ingestion of a nitrate rich red spinach extract. Eur J Appl Physiol 116(11–12):2267–2279. https://doi.org/10.1007/s00421-016-3478-8
Kanner J, Harel S, Granit R (2001) Betalains—a new class of dietary cationized antioxidants. J Agric Food Chem 49(11):5178–5185
Kephart WC, Wachs TD, Mac Thompson R, Brooks Mobley C, Fox CD, McDonald JR, Ferguson BS, Young KC, Nie B, Martin JS, Company JM, Pascoe DD, Arnold RD, Moon JR, Roberts MD (2016) Ten weeks of branched-chain amino acid supplementation improves select performance and immunological variables in trained cyclists. Amino Acids 48(3):779–789. https://doi.org/10.1007/s00726-015-2125-8
Kujala T, Loponen J, Pihlaja K (2001) Betalains and phenolics in red beetroot (Beta vulgaris) peel extracts: extraction and characterisation. Z Naturforsch C 56(5–6):343–348
Lansley KE, Winyard PG, Bailey SJ, Vanhatalo A, Wilkerson DP, Blackwell JR, Gilchrist M, Benjamin N, Jones AM (2011) Acute dietary nitrate supplementation improves cycling time trial performance. Med Sci Sports Exerc 43(6):1125–1131. https://doi.org/10.1249/MSS.0b013e31821597b4
Lee CH, Wettasinghe M, Bolling BW, Ji LL, Parkin KL (2005) Betalains, phase II enzyme-inducing components from red beetroot (Beta vulgaris L.) extracts. Nutr Cancer 53(1):91–103. https://doi.org/10.1207/s15327914nc5301_11
Martinez RM, Longhi-Balbinot DT, Zarpelon AC, Staurengo-Ferrari L, Baracat MM, Georgetti SR, Sassonia RC, Verri WA Jr, Casagrande R (2015) Anti-inflammatory activity of betalain-rich dye of Beta vulgaris: effect on edema, leukocyte recruitment, superoxide anion and cytokine production. Arch Pharm Res 38(4):494–504. https://doi.org/10.1007/s12272-014-0473-7
Montenegro CF, Kwong DA, Minow ZA, Davis BA, Lozada CF, Casazza GA (2017) Betalain-rich concentrate supplementation improves exercise performance and recovery in competitive triathletes. Appl Physiol Nutr Metab 42(2):166–172. https://doi.org/10.1139/apnm-2016-0452
Morici G, Zangla D, Santoro A, Pelosi E, Petrucci E, Gioia M, Bonanno A, Profita M, Bellia V, Testa U, Bonsignore MR (2005) Supramaximal exercise mobilizes hematopoietic progenitors and reticulocytes in athletes. Am J Physiol Regul Integr Comp Physiol 289(5):R1496–R1503. https://doi.org/10.1152/ajpregu.00338.2005
Mumford PW, Tribby AC, Poole CN, Dalbo VJ, Scanlan AT, Moon JR, Roberts MD, Young KC (2016) Effect of caffeine on golf performance and fatigue during a competitive tournament. Med Sci Sports Exerc 48(1):132–138. https://doi.org/10.1249/mss.0000000000000753
O’Neill CA, Stebbins CL, Bonigut S, Halliwell B, Longhurst JC (1996) Production of hydroxyl radicals in contracting skeletal muscle of cats. J Appl Physiol (Bethesda Md: 1985) 81(3):1197–1206. https://doi.org/10.1152/jappl.1996.81.3.1197
Pedersen BK (2000) Exercise and cytokines. Immunol Cell Biol 78(5):532–535
Pedersen BK, Steensberg A, Fischer C, Keller C, Ostrowski K, Schjerling P (2001) Exercise and cytokines with particular focus on muscle-derived IL-6. Exerc Immunol Rev 7:18–31
Pietrzkowski Z, Nemzer B, Spórna A, Stalica P, Tresher W, Keller R, Jimenez R, Michałowski T, Wybraniec S (2010) Influence of betalain-rich extract on reduction of discomfort associated with osteoarthritis. New Med 1:12–17
Powers SK, Criswell D, Lawler J, Ji LL, Martin D, Herb RA, Dudley G (1994) Influence of exercise and fiber type on antioxidant enzyme activity in rat skeletal muscle. Am J Physiol 266(2 Pt 2):R375–R380. https://doi.org/10.1152/ajpregu.1994.266.2.R375
Powers SK, DeRuisseau KC, Quindry J, Hamilton KL (2004) Dietary antioxidants and exercise. J Sports Sci 22(1):81–94. https://doi.org/10.1080/0264041031000140563
Reid MB, Haack KE, Franchek KM, Valberg PA, Kobzik L, West MS (1992) Reactive oxygen in skeletal muscle. I. Intracellular oxidant kinetics and fatigue in vitro. J Appl Physiol (Bethesda Md: 1985) 73(5):1797–1804. https://doi.org/10.1152/jappl.1992.73.5.1797
Sakihama Y, Maeda M, Hashimoto M, Tahara S, Hashidoko Y (2012) Beetroot betalain inhibits peroxynitrite-mediated tyrosine nitration and DNA strand cleavage. Free Radic Res 46(1):93–99. https://doi.org/10.3109/10715762.2011.641157
Schwandt HJ, Heyduck B, Gunga HC, Rocker L (1991) Influence of prolonged physical exercise on the erythropoietin concentration in blood. Eur J Appl Physiol Occup Physiol 63(6):463–466
Steinberg JG, Ba A, Bregeon F, Delliaux S, Jammes Y (2007) Cytokine and oxidative responses to maximal cycling exercise in sedentary subjects. Med Sci Sports Exer 39(6):964–968. https://doi.org/10.1097/mss.0b013e3180398f4b
Strack D, Vogt T, Schliemann W (2003) Recent advances in betalain research. Phytochemistry 62(3):247–269
Tesoriere L, Butera D, D’Arpa D, Di Gaudio F, Allegra M, Gentile C, Livrea MA (2003) Increased resistance to oxidation of betalain-enriched human low density lipoproteins. Free Radic Res 37(6):689–696
Tesoriere L, Allegra M, Butera D, Livrea MA (2004) Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: potential health effects of betalains in humans. Am J Clin Nutr 80(4):941–945. https://doi.org/10.1093/ajcn/80.4.941
Tesoriere L, Butera D, Allegra M, Fazzari M, Livrea MA (2005) Distribution of betalain pigments in red blood cells after consumption of cactus pear fruits and increased resistance of the cells to ex vivo induced oxidative hemolysis in humans. J Agric Food Chem 53 (4):1266–1270. https://doi.org/10.1021/jf048134&%23x002B;
Tesoriere L, Gentile C, Angileri F, Attanzio A, Tutone M, Allegra M, Livrea MA (2013) Trans-epithelial transport of the betalain pigments indicaxanthin and betanin across Caco-2 cell monolayers and influence of food matrix. Eur J Nutr 52(3):1077–1087. https://doi.org/10.1007/s00394-012-0414-5
Van Hoorebeke JS, Trias CO, Davis BA, Lozada CF, Casazza GA (2016) Betalain-rich concentrate supplementation improves exercise performance in competitive runners. Sports 4(3):40
Vidal PJ, Lopez-Nicolas JM, Gandia-Herrero F, Garcia-Carmona F (2014) Inactivation of lipoxygenase and cyclooxygenase by natural betalains and semi-synthetic analogues. Food Chem 154:246–254. https://doi.org/10.1016/j.foodchem.2014.01.014
Vincent HK, Powers SK, Demirel HA, Coombes JS, Naito H (1999) Exercise training protects against contraction-induced lipid peroxidation in the diaphragm. Eur J Appl Physiol Occup Physiol 79(3):268–273. https://doi.org/10.1007/s004210050505
Vincent HK, Powers SK, Stewart DJ, Demirel HA, Shanely RA, Naito H (2000) Short-term exercise training improves diaphragm antioxidant capacity and endurance. Eur J Appl Physiol 81(1–2):67–74. https://doi.org/10.1007/pl00013799
Yavari A, Javadi M, Mirmiran P, Bahadoran Z (2015) Exercise-induced oxidative stress and dietary antioxidants. Asian J Sports Med 6(1):e24898. https://doi.org/10.5812/asjsm.24898
Zielinska-Przyjemska M, Olejnik A, Dobrowolska-Zachwieja A, Grajek W (2009) In vitro effects of beetroot juice and chips on oxidative metabolism and apoptosis in neutrophils from obese individuals. Phytother Res 23(1):49–55. https://doi.org/10.1002/ptr.2535
Acknowledgements
This study was funded by a contract awarded to Auburn University (MDR) and the Edward Via College of Osteopathic Medicine—Auburn Campus (KCY and JSM) from FutureCeuticals (Momence, IL USA). Contract monies were used for participant compensation and assay-related costs.
Author information
Authors and Affiliations
Contributions
Conception and study design: MDR, KCY, JSM. Data collection: PWM, WCK, MAR, CTH, CBM, SCO, JCH, ANM, WCR, KCY. Data analysis: PMW, KCY, MDR, JSM. Manuscript preparation: PMW, MDR, KCY. All authors reviewed, edited and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have conflicts of interest to declare regarding the publication of these data.
Additional information
Communicated by Anni Vanhatalo.
Jeffrey S. Martin, Michael D. Roberts and Kaelin C. Young are co-principal investigators.
Rights and permissions
About this article
Cite this article
Mumford, P.W., Kephart, W.C., Romero, M.A. et al. Effect of 1-week betalain-rich beetroot concentrate supplementation on cycling performance and select physiological parameters. Eur J Appl Physiol 118, 2465–2476 (2018). https://doi.org/10.1007/s00421-018-3973-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-018-3973-1