Abstract
Many reviews conclude that metabolites play an important role with respect to muscle hypertrophy during resistance exercise, but their actual physiologic contribution remains unknown. Some have suggested that metabolites may work independently of muscle contraction, while others have suggested that metabolites may play a secondary role in their ability to augment muscle activation via inducing fatigue. Interestingly, the studies used as support for an anabolic role of metabolites use protocols that are not actually designed to test the importance of metabolites independent of muscle contraction. While there is some evidence in vitro that metabolites may induce muscle hypertrophy, the only study attempting to answer this question in humans found no added benefit of pooling metabolites within the muscle post-exercise. As load-induced muscle hypertrophy is thought to work via mechanotransduction (as opposed to being metabolically driven), it seems likely that metabolites simply augment muscle activation and cause the mechanotransduction cascade in a larger proportion of muscle fibers, thereby producing greater muscle growth. A sufficient time under tension also appears necessary, as measurable muscle growth is not observed after repeated maximal testing. Based on current evidence, it is our opinion that metabolites produced during resistance exercise do not have anabolic properties per se, but may be anabolic in their ability to augment muscle activation. Future studies are needed to compare protocols which produce similar levels of muscle activation, but differ in the magnitude of metabolites produced, or duration in which the exercised muscles are exposed to metabolites.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is generally recommended for individuals to perform resistance training at least twice per week as part of a comprehensive exercise program (American College of Sports Medicine 2009). Chronic resistance training increases muscle mass which may improve self-image (Hausenblas and Fallon 2006) and be of clinical importance for attenuating the risk of chronic disease (Wolfe 2006). As such, a greater understanding of the mechanisms governing muscle growth is necessary to ensure proper exercise programming and potential therapeutic alternatives to resistance exercise. Current physical activity guidelines recommend that individuals looking to increase muscle mass should exercise with a load corresponding to at least 70% of an individual’s one-repetition maximum (1RM) strength (American College of Sports Medicine 2009). This statement, however, does not account for the large body of scientific evidence demonstrating similar increases in muscle size when comparing lower (30–50% 1RM) and higher (75–90% 1RM) loads, provided the exercises are performed until volitional failure (Mitchell et al. 2012; Ogasawara et al. 2013; Morton et al. 2016). Furthermore, blood flow-restricted exercise has also been shown to promote similar increases in muscle size to that of low load training to volitional failure (Fahs et al. 2015; Farup et al. 2015) and traditional high load training (Laurentino et al. 2012). The notion that all of these protocols produce similar muscle hypertrophy has led many (Schoenfeld 2013; Ozaki et al. 2015, 2016) to hypothesize that these different exercise modalities may be working through alternative mechanisms (i.e., metabolites) to induce muscle growth.
Therefore, the purpose of this review is to discuss the current evidence surrounding the role that metabolite production during exercise (e.g., lactate, inorganic phosphate) has in regulating exercise-induced skeletal muscle hypertrophy. Specifically, we are interested in answering the question as to whether a heightened production and/or prolonged duration of metabolites (in addition to that which is typically present during resistance exercise) has an additive effect on muscle growth. Notably, it is our opinion that increasing the duration and/or production of metabolites during resistance exercise does not have anabolic properties per se, but rather promotes muscle hypertrophy indirectly via inducing muscle fatigue and augmenting muscle activation. The act of repeated muscle contraction appears to be of primary importance to fatigue the muscle and stress a larger proportion of motor units. While there are an overwhelming number of potential mechanisms that may contribute to skeletal muscle hypertrophy during (e.g., blood flow, cytokines) or independent (e.g., amino acids, testosterone supplementation) of resistance exercise, this review focuses solely on the contributions of metabolites to muscle hypertrophy.
How does traditional resistance exercise induce skeletal muscle hypertrophy?
While the purpose of this review is not to provide an in-depth review of the molecular mechanisms governing contraction-induced muscle hypertrophy (for a review see Marcotte et al. 2015), a brief overview of the proposed molecular mechanism will provide the reader with the necessary information to detail why, in our opinion, current evidence would suggest that metabolites may not have direct anabolic properties. It is currently thought that muscle growth via resistance exercise occurs through a process of mechanotransduction, in which the mechanical stress of muscle contraction is converted into a chemical signal. This is thought to work through the binding of individual focal adhesion proteins such as focal adhesion kinase (FAK), which collectively form focal adhesions attached to integrin receptors (Goldmann 2012). These focal adhesions are thought to mediate the mechanotransduction process, by allowing for the tension placed on the muscle cell to be transmitted from the extracellular matrix, to the inside of the cell and to the cell nucleus (Goldmann 2012). This chemical signal then increases muscle mass through activation of the mechanistic target of rapamycin complex 1 (mTORC1), but this complex is not solely activated through resistance exercise, as other stimuli such as growth factors and amino acids can also increase mTORC1 activation (Marcotte et al. 2015). The specific process by which resistance exercise activates the mTORC1 complex is not entirely understood, but ultimately involves the removal of tuberous sclerosis complex 2 (TSC2) which then allows for activation of the mTORC1 (Jacobs et al. 2017). While resistance exercise may also assist in elevating protein synthesis by translocating the mTORC1 complex to the lysosome where it can be activated (Jacobs et al. 2017), this process is also facilitated by the consumption of amino acids (Bar-Peled and Sabatini 2014). Therefore, the combined effect of removing the mTORC1 inhibitor and relocating mTORC1 to the lysosome produces robust muscle hypertrophy (West and Baar 2013).
Can metabolites induce skeletal muscle hypertrophy through an alternate mechanism?
It has been suggested that muscle hypertrophy occurring during blood flow restriction may not work via mechanotransduction given its low mechanical stress (Pearson and Hussain 2015). Alternatively, intramuscular (Suga et al. 2009, 2010, 2012) and whole blood (Takarada et al. 2000a, b; Takano et al. 2005) metabolite measurements detail large accumulations during blood flow restriction protocols, but the specific measurement techniques are not the focus of this review. The idea that blood flow restriction exercise activates the mitogen activated protein kinase (MAPK) pathway (Fry et al. 2010), which may be independent of the exercise load (Dentel et al. 2005), has led to the hypothesis that a metabolic sensor (as opposed to a mechanical sensor) may regulate the MAPK pathway (Dentel et al. 2005) providing a potential mechanism for muscle growth that is not load dependent. The metabolites which are commonly hypothesized to have anabolic effects are those which have been shown to be altered in response to blood flow-restricted exercise protocols [e.g., increased lactate (Takarada et al. 2000a, b), increased inorganic phosphate (Suga et al. 2012)], under the assumption that it is the increase in metabolite production that augments muscle growth. This would seem to be a plausible explanation given that applying blood flow restriction during low load resistance exercise increases muscle size compared to that of a repetition matched control group in the absence of blood flow restriction (Nielsen et al. 2012). In this review, we will briefly discuss the current evidence supporting the anabolic potential of a few metabolites which have been suggested by some to augment muscle growth during resistance exercise. The purpose of this section is not to overview the anabolic potential of each metabolite which is produced during resistance exercise, but rather to briefly discuss the aforementioned metabolites commonly stated to have anabolic effects. It is important to understand that during resistance exercise, there are numerous metabolites being produced that could be playing a role. Although this is not an exhaustive review of every potential metabolite present following resistance exercise, any metabolite produced would still be accounted for within the chronic training studies.
Of the metabolites produced during resistance exercise, lactate appears to have the most support as a potential anabolic molecule for muscle hypertrophy (Nalbandian and Takeda 2016). Support for lactate having anabolic properties comes from cell culture studies where it has been shown to induce myogenesis (Willkomm et al. 2014; Oishi et al. 2015) and increase phosphorylation of p70S6K (a downstream target of mTORC1) (Oishi et al. 2015), providing some evidence that metabolites may induce muscle hypertrophy in vitro. There is, however, limited evidence in vivo with only one study illustrating that 30 min of treadmill running for 4 weeks produced greater increases (relative to an exercising control group) in the muscle weight of rats after oral lactate (100 mg/kg) and caffeine (36 mg/kg) consumption (Oishi et al. 2015). The authors speculated that lactate may be anabolic via alterations in proteins expressed in the exercised rat muscles, which included increased myogenin (which regulates differentiation of satellite cells), decreased myostatin (a negative regulator of muscle growth), and increased follistatin (an inhibitor of myostatin). The authors also acknowledged that the caffeine supplementation may have contributed to the anabolic stimulus through increased intracellular calcium, but the lactate and caffeine compounds were not analyzed independently in the in vivo study. It is also possible that lactate may work to increase muscle size via indirectly increasing testosterone production as was shown in rat cells bathed in lactate (10 mmol for 1 h) (Lin et al. 2001); however, as mentioned in a recent review (Nalbandian and Takeda 2016), there is no evidence to support these findings in vivo. Even if lactate were to increase testosterone in humans, acute elevations in endogenously produced testosterone which result from resistance exercise have been shown to have no added benefit for increasing muscle size (West et al. 2010). Inorganic phosphate has received some support as a potential anabolic molecule, but these adaptations are related to bone tissue as opposed to skeletal muscle (Beck 2003; Spina et al. 2013). Finally, reactive oxygen species have garnered some support for having anabolic effects in vitro via upregulating insulin-like growth factor 1 (IGF-1), but this effect appears to occur in a dose-dependent fashion with higher levels of reactive oxygen species having a potentially detrimental effect on IGF-1 signaling (Barbieri and Sestili 2012).
While a number of reviews have been published centered on the importance of metabolic accumulation for inducing muscle growth in humans (Schoenfeld 2013; Ozaki et al. 2015, 2016), there are currently no studies (in humans) which provide direct evidence that metabolites produced during resistance exercise facilitate muscle growth greater than that of muscle contraction itself. All the current studies used as support for the anabolic role of metabolites simply detail that metabolites are associated with muscle growth. In other words, these studies are not designed to assess the role of metabolites because it is not just the metabolic environment that is being altered, but, rather, some other training variable is manipulated which subsequently results in a greater accumulation of metabolites. This is a very important concept because the level of local muscle fatigue (i.e., reaching or approaching volitional failure) appears to be the most important factor with respect to muscle growth (Mitchell et al. 2012; Morton et al. 2016) and this is likely related to the level of muscle activation. In other words, metabolites are present because the increased level of effort equates to more energy being utilized and more metabolites being produced, and thus represent more a consequence of contraction rather than a cause of muscle growth. Here are just a few examples of study designs used to support the role of metabolites for inducing muscle hypertrophy: (1) hypoxic environments (Nishimura et al. 2010); (2) blood flow-restricted exercise (Takarada et al. 2000b); (3) eccentric vs. concentric contractions (Smith and Rutherford 1995); (4) short vs. long muscle contractions (Schott et al. 1995; Tanimoto et al. 2008); (5) short vs. long rest periods provided between multiple sets of exercise [(Kraemer et al. 1987; Villanueva et al. 2015); and (6) rest periods that are provided within sets (i.e., breaking up a 10RM into two sets of 5 repetitions] (Goto et al. 2005). Two of these studies are even titled “The role of metabolites in strength training” (Schott et al. 1995; Smith and Rutherford 1995) despite not directly testing the role of metabolites in strength training. In fact, none of these aforementioned studies assess the effects of metabolites independent of muscle contraction, which makes it difficult to assess whether the greater muscle growth is due to the act of muscle contraction itself, or the additional metabolites that are produced. Importantly, and as suggested previously (Moritani et al. 1992), metabolites may be anabolic in their ability to augment muscle activation and this idea has received further support (Loenneke et al. 2011) after the role of exercise-induced hormonal elevations (also thought to potentially work independently of muscle contraction) were shown to be of little importance for muscle growth (West et al. 2010). Although fatigue is likely multifactorial and cannot be solely attributed to metabolites, fatigue has been shown to stimulate the recruitment of additional higher threshold motor units (Bigland-Ritchie et al. 1986).
Metabolites may induce muscle growth indirectly via motor unit recruitment
Motor unit recruitment assessed via electromyography
Inorganic phosphate and hydrogen ion accumulation may contribute to muscle hypertrophy by inhibiting cross-bridge cycling and inducing muscle fatigue (Debold 2012). Electromyography (EMG) amplitude is often used as an indirect measure of motor unit recruitment based on the assumption that more motor units firing at a given time will produce more action potentials and thus a higher EMG amplitude. Blood flow restriction has been shown to augment EMG amplitude over unrestricted repetition and load matched protocols (Yasuda et al. 2009; Lauver et al. 2017), while both protocols performed to volitional failure result in similar EMG amplitudes (Wernbom et al. 2009). Since low load protocols that are repetition matched produce greater muscle growth when blood flow restriction is applied (Laurentino et al. 2012), but exercising to volitional failure negates these differences (Fahs et al. 2015; Farup et al. 2015), it would appear that the primary benefit of applying blood flow restriction during resistance exercise may be related to greater muscle fatigue (Loenneke et al. 2012a). This is supported by the fact that the application of blood flow restriction does not augment muscle growth occurring during high load resistance exercise (Laurentino et al. 2008) and this is likely because muscle activation cannot be appreciably augmented (Takarada et al. 2000b; Dankel et al. 2017a), since high load exercise is already very fatiguing. If metabolites could induce muscle growth independent of the traditional mechanotransduction cascade, high load resistance exercise would likely benefit from the application of blood flow restriction, but this is not the case (Laurentino et al. 2008). Therefore, it seems more likely that it is the recruitment of higher threshold motor units, and thus the activation of a large proportion of muscle fibers that is of primary importance for muscle growth. While metabolites would be produced during both high and low load resistance exercise, their contribution to motor unit recruitment would be particularly evident during low load resistance exercise given that higher loads will recruit more motor units at the onset of exercise to produce the higher force that is necessary (Fig. 1). It is our opinion that metabolites are not mandatory for muscle growth, but provide one mechanism through which additional motor units can be recruited as a result of local muscle fatigue.
While metabolites are produced during both high load and low load resistance exercise, they appear to be particularly beneficial during low load exercise in their ability to assist with fatiguing lower threshold motor units. This then causes the recruitment of additional motor units (and thus muscle fibers) that were not active at the start of exercise. Notably, it is our hypothesis that metabolites do not provide an alternative mechanism for inducing muscle hypertrophy during resistance exercise, but are rather an alternative means to increase muscle activation. It also appears as though each muscle fiber must be activated enough times within a given duration to allow for the mechanical stimulus to produce a sufficient chemical signal within the cell to initiate muscle hypertrophy
The argument can be made, however, that blood flow restriction exercise does not activate as many higher threshold motor units as traditional high load training (see Fig. 6 in Manini and Clark 2009). We believe this is more likely an artifact of the load being lifted rather than a true discrepancy in motor unit activation, and may detail a potential limitation with using surface EMG amplitude to directly infer motor unit recruitment. It has been shown that during fatiguing contractions, some motor units may not only reduce their firing frequencies, but shut off completely to allow for recovery from fatigue (Person 1974). As a sufficient force is still being produced to maintain the appropriate force output to continue exercising, these fatigued motor units will pass their force production duties onto other motor units (termed substitution). This demonstrates that the control of motor units during fatiguing contractions may work independently of the common drive theory (De Luca et al. 1982; De Luca and Erim 1994) as each motor unit can have their own control properties within the motor neuron pool. This individualized control would allow for the recovery of motor units during prolonged fatiguing contractions (Westgaard and De Luca 1999). Notably, this has been shown to occur during isometric contractions of the knee extensors producing force up to 50% of MVC, and is more pronounced during dynamic contractions when changes in movement patterns are present (Person 1974).
The occurrence of motor unit substitution (Person 1974) details a limitation with inferring muscle activation from EMG amplitude when comparing protocols using different loads. Such studies can be potentially misleading, as exercising with a heavier load does not allow for motor unit substitution since a higher force output must be maintained. Therefore, low load exercise to volitional failure is likely activating the same proportion of motor units (and thus muscle fibers) to that of high load training to volitional failure, although this would not necessarily be evident based on the results of EMG amplitude (Fig. 2). In other words, the EMG amplitude at a given time point would be lower during low load resistance exercise, even if the same number of motor units have been stimulated, and this is because some motor units have been substituted and subsequently turned off when the EMG amplitude is recorded. It would make conceptual sense that both high load and low load training to volitional failure would produce similar levels of muscle activation given the muscle is incapable of producing the force necessary to complete another repetition.
Fibers that are depicted in white would not hypertrophy as they have not been activated to undergo the mechanotransduction cascade. Fibers depicted in black are currently activated and assisting in force production and will undergo the mechanostransduction cascade to induce muscle growth. The black striped design depicts a muscle fiber that has been activated, but has not contracted a sufficient number of times to induce the hypertrophy stimulus. Fibers depicted in gray have already received the mechanotransduction stimulus and will hypertrophy but have been selectively turned off to allow for recovery. If these protocols were assessed using electromyography (EMG) amplitude, the level of muscle activation would be proportional to the number of solid black and black striped muscle fibers. Notably, this explains why EMG amplitude often favors the high load condition even though a similar number of muscle fibers received the stimulus for muscle growth
Motor unit recruitment assessed via phosphorus-31 magnetic resonance spectroscopy
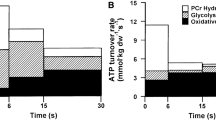
An alternative, non-invasive method used to estimate muscle activation involves a method called phosphorus-31 magnetic resonance spectroscopy (Park et al. 1987; Vandenborne et al. 1991). This method is used to assess the recruitment of different muscle fibers based on the depletion of ATP, reduction in pH and total phosphate, and increase in inorganic phosphate, all of which will occur more rapidly in type II muscle fibers during exercise. Importantly, this method of assessing muscle fiber contribution during exercise is less sensitive to changes in exercise intensity and is more related to involvement of muscle fibers based on energy metabolism (Vandenborne et al. 1991). One research group has used this method to conduct several studies comparing motor unit activation between low load blood flow restriction exercise and high load exercise, while assessing motor unit recruitment based on the level of inorganic phosphate splitting (Suga et al. 2009, 2010, 2012). When individuals performed 60 repetitions of plantar flexion exercise at 20% 1RM with and without blood flow restriction (Suga et al. 2010), it was shown that only those exercising with blood flow restriction recruited higher threshold motor units. Another study by the same research group illustrated that blood flow restriction exercise did not increase the activation of type II muscle fibers to the extent of high load exercise (Suga et al. 2009). Since both the blood flow restriction (20% 1RM) and high load condition (65% 1RM) completed the same number of repetitions (60 repetitions), the hypertrophic potential was likely different as a much greater number of repetitions can be completed at 20% 1RM with blood flow restriction (51 repetitions) as compared to 50% 1RM without blood flow restriction (25 repetitions) (Barcelos et al. 2015). Therefore, it is likely that the blood flow restriction protocol was not very fatiguing, and as such fewer metabolites were produced, and the lack of motor unit recruitment actually does not provide much information in this case because lower motor unit recruitment would be expected. A follow-up study from this research group illustrated that multi-set protocols comparing blood flow restriction to high load training yielded very similar responses with respect to metabolite production and recruitment of higher threshold motor units (Suga et al. 2012). Since this is more representative of the majority of blood flow restriction protocols that are employed (Loenneke et al. 2012b; Dankel et al. 2016b), it seems likely that both indeed result in similar levels of motor unit recruitment/muscle activation. This method is also not without limitation and future studies may wish to use immunohistochemistry techniques to examine muscle biopsy samples taken post-exercise to confirm that this method of assessing muscle activation is valid.
What types of studies are needed to determine the role of metabolites in muscle hypertrophy?
Using the electrical stimulation model
It has been proposed that the role of metabolites can be tested by comparing electrical stimulation with and without blood flow restriction provided there are no differences in motor unit recruitment (Meyer 2006), as any differences in muscle hypertrophy could not be attributed to differences in muscle activation. Although electrical stimulation studies have demonstrated a greater effect when applying blood flow restriction (Natsume et al. 2015; Gorgey et al. 2016), it is unknown whether differences in muscle activation were present as this was not measured and may be difficult to assess given the artifact produced by the pulse of the electrical stimulus (Collins 2007). Evidence for a role of the central nervous system during electrical stimulation exists, in that anesthetic nerve blockage (which removes the influence of the central nervous system) results in a significant torque reduction during electrical stimulation (Collins 2007). This involvement of the central nervous system details that sensory fibers (group III and IV afferents) may provide feedback to the central nervous system to alter motor command (Laurin et al. 2015) and the involvement of the central nervous system is apparent regardless of whether the electrical stimulation is applied to the nerve or muscle belly (Bergquist et al. 2012), but motor unit activity during electrical stimulation does not follow the orderly recruitment pattern commonly observed with voluntary exercise (Collins 2007). It is possible that electrical stimulation can be used to test the role of metabolites, provided maximal electrical stimulation is used in which no more motor units can possibly be recruited. Therefore, differences in muscle growth could not be attributed to differences in motor unit recruitment, although this would likely be an uncomfortable protocol to complete.
What other models can be tested?
To answer this specific question, researchers should avoid simply looking at the level of metabolites produced during different protocols, and instead manipulate (via study design) a control and experimental group that receive the same stimulus but differ in metabolite production. Our laboratory has recently employed a study in which we compared one set of exercise to volitional failure with another condition, performing the exactly same protocol followed by 3 min of post-exercise blood flow restriction, which was done to pool metabolites within the muscle (Dankel et al. 2016a). Therefore, any difference between conditions could be attributed to the intervention (i.e., the 3 min post-exercise blood flow restriction period). Not only was this strategy ineffective for augmenting muscle growth, but it appeared to be detrimental, specifically in the female population. Being that the metabolites were likely to be pooled within the muscle post-exercise (this was indirectly assessed via depressed torque), the metabolites did not have an opportunity to work synergistically with muscle contraction occurring with additional sets of exercises, and thus this would refute the hypothesis that metabolites may induce muscle hypertrophy independent of muscle contraction. Of course, we cannot rule out the possibility that restricting blood flow to pool metabolites may have had a detrimental sex-specific effect on muscle growth, possibly through reduced nutrient delivery (Timmerman et al. 2010). It is also possible that the reduced ATP synthesis and accumulation of AMP, ADP and oxidative stress from restricting blood flow in the absence of exercise (Goldfarb et al. 2008; Garten et al. 2015) may have activated the AMP-activated protein kinase (AMPK) pathway, potentially limiting the mTORC1 activity and contributing to protein degradation (Atherton et al. 2015). It remains unclear, however, why this would produce sex-specific differences in muscle hypertrophy. Future studies wishing to assess the specific role of metabolites should attempt to alter the metabolic environment within the exercised muscle while holding constant as many variables as possible. Additionally, studies should attempt to assess the independent effects of metabolites to answer the specific question: Can metabolites augment muscle growth through an independent mechanism not reliant on inducing muscle fatigue?
Why would low load exercise work any differently?
Given the previously stated idea that muscle growth is thought to work via mechanotransduction (i.e., induced by muscle contraction), it may seem counterintuitive that exercising with a lower load would change the mechanism causing muscle growth. Regardless, it has been stated that “…it is questionable whether such mechanotransduction processes would contribute to blood flow restriction (BFR) resistance training-induced hypertrophy given its low mechanical stress nature.” (Pearson and Hussain 2015). The idea that the load or intensity of muscle contraction during low load resistance exercise is insufficient to induce the mechanotransduction cascade would seemingly go against how motor units are presently thought to contribute to force. To illustrate, the twitch force of a given motor unit does not differ based on the magnitude of force (e.g., % maximal isometric contraction) that is being produced (LeFever and De Luca 1982); thus, each firing of a motor neuron sends a nerve impulse that always causes the innervated muscle fiber to produce the maximal force possible. The maximal amount of force that is produced by an individual muscle fiber may differ based on fatigue or the Treppe effect (slight increase in tension with repeated twitches), but it will still be contracting at its maximal capability. In other words, a human can alter their voluntary force output through the number of motor units recruited, but an individual motor unit cannot.
The overall stimulus to each muscle fiber must then be related to the number of times it is stimulated, given that each stimulus always produces 100% of its possible contractile force at a given time. Therefore, it is not necessarily the level of contractile force produced by the muscle fibers that is of importance, but, rather, the proportion of the muscle (i.e., the percentage of muscle fibers) that is required to produce force, given that overall muscle hypertrophy will be proportional to the number of muscle fibers stimulated (Marcotte et al. 2015). As such, any protocol producing a high muscle activation and sufficient number of contractions within each muscle fiber would likely result in the activation of mTORC1 in a similar proportion of muscle fibers, thereby producing a similar hypertrophic stimulus. The idea that blood flow-restricted exercise is also increasing muscle size through activation of mTORC1 has been shown previously (Gundermann et al. 2014), which would support the notion that they may be working through the same contraction-dependent activation of mTORC1. Furthermore, a study comparing 12 weeks of knee extension exercises found that both low load (30% 1RM), blood flow restriction exercise and high load (6–10 RM) exercise resulted in similar acute and chronic changes in the expression of 29 genes related to muscle function, with no significant differences found between conditions for any of the genes measured (Ellefsen et al. 2015). These studies support our hypothesis that these protocols may be working via the same mechanism, and it would be hard to rationalize the existence of a point along the loading spectrum (i.e., 0–100% 1RM) at which the hypertrophic stimulus from resistance exercise would switch from being mechanically driven to becoming metabolically driven.
Is muscle activation the sole driver of muscle hypertrophy?
Although it appears that a muscle fiber must be activated during resistance exercise to receive the mechanotransduction stimulus, activation of the muscle fiber per se is not sufficient to induce muscle growth. For example, we have previously conducted a study in which individuals performed maximal voluntary isometric and isotonic (1RM) testing every day for 21 straight days and this was insufficient to induce muscle growth (Dankel et al. 2017b). The maximal nature of these tests would suggest that all of the motor units that could be voluntarily activated were indeed activated; however, this did not produce muscle growth. It should be mentioned that these individuals performed exactly the same protocol, in addition to three sets of exercises on the contralateral arm and this was sufficient in inducing muscle growth. Collectively, these findings would suggest that not simply activation, but also a sufficient number of motor unit firings within a reasonable duration (i.e., a sufficient time under tension) may be necessary to trigger this mechanotransduction cascade in each muscle fiber (Fig. 2). This may be related to an insufficient amplitude and/or duration of intracellular calcium, as short duration/high amplitude calcium signals activate the calcium–calmodulin protein kinase II (CaMKII) pathway (Chin 2005). Intracellular calcium (Ito et al. 2013) and the synthesis of phosphatidic acid (You et al. 2014) may also be limiting the magnitude of muscle growth present during such a short duration of muscle contraction, as both have been postulated to play an important role upstream of the mTORC1 pathway. It is likely that whatever is causing the lack of muscle hypertrophy is related to a reduced activation of the mTORC1 pathway, as blocking this pathway completely abolishes compensatory hypertrophy in rats (Bodine et al. 2001) and largely reduces the muscle protein synthesis response to resistance exercise in humans (Gundermann et al. 2014).
The stimulus necessary to induce muscle growth does not appear to be difficult to achieve, as even three 10 s isometric contractions per day was shown to induce measurable muscle hypertrophy. It is our hypothesis that the same number of active motor units and the same number of firings within each motor unit (i.e., the same percentage of muscle fibers contracting the same number of times) would be insufficient to induce muscle growth, if these motor unit firings were spread throughout the day. In other words, would thirty 1 s maximal isometric contractions dispersed evenly throughout the day produce the same results as the three 10 s isometric contractions used by Ikai and Fukunaga (1970)? If not, then it appears that it is not strictly the proportion of activated motor units and the number of motor unit firings (as these would be very similar between conditions), but rather a sufficient number of motor unit firings within a given duration may be necessary to induce a sufficient chemical change capable of inducing muscle growth (Fig. 1). This may be related to accumulations of phosphatidic acid which has been shown to be of critical importance for the mechanical activation of mTORC1 (Hornberger et al. 2006; O’Neil et al. 2009). The combination of the percentage of muscle fibers stimulated, and the duration in which each is stimulated, would appear to be the most important factors governing contraction-induced muscle hypertrophy, as our laboratory has shown that even maximally flexing the elbow flexors through the full range of motion with no external load produced similar muscle hypertrophy to that of high load resistance exercise (Counts et al. 2016). Although the longer duration in which the muscle fibers are placed under tension would produce more metabolites given the greater energy utilization, it is our opinion that metabolites are simply permissive for muscle growth rather than being obligatory.
Conclusion
It is commonly stated that the accumulation of metabolites during resistance exercise is one of the mechanisms responsible for the hypertrophic stimulus. The idea that low load resistance training and low load blood flow-restricted resistance training produce similar increases in muscle size to that of high load resistance training is commonly used as evidence for this statement. It is currently unknown whether metabolites produced during resistance exercise have anabolic properties that are independent of muscle contraction, or whether they are anabolic through their ability to augment muscle activation by inducing muscle fatigue. Based on the available evidence, it is our opinion that the anabolic role of exercise-induced metabolites lies in their ability to assist with augmenting muscle activation. To test this hypothesis, future studies should compare two identical protocols that differ only based on the presence, magnitude, or duration of metabolites, with careful caution to not allow metabolites to alter the level of fatigue produced between protocols.
Abbreviations
- 1RM:
-
One-repetition maximum
- AMPK:
-
AMP-activated protein kinase
- CAMKII:
-
Calcium–calmodulin protein kinase II
- EMG:
-
Electromyography
- FAK:
-
Focal adhesion kinase
- MAPK:
-
Mitogen-activated protein kinase
- mTORC1:
-
Mechanistic target of rapamycin complex 1
- TSC2:
-
Tuberous sclerosis complex 2
References
American College of Sports Medicine (2009) American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med Sci Sports Exerc 41:687–708. doi:10.1249/MSS.0b013e3181915670
Atherton PJ, Phillips BE, Wilkinson DJ (2015) Exercise and regulation of protein metabolism. Prog Mol Biol Transl Sci 135:75–98. doi:10.1016/bs.pmbts.2015.06.015
Barbieri E, Sestili P (2012) Reactive oxygen species in skeletal muscle signaling. J Signal Transduct 2012:982794
Barcelos LC, Nunes PRP, de Souza LRMF et al (2015) Low-load resistance training promotes muscular adaptation regardless of vascular occlusion, load, or volume. Eur J Appl Physiol 115:1559–1568. doi:10.1007/s00421-015-3141-9
Bar-Peled L, Sabatini DM (2014) Regulation of mTORC1 by amino acids. Trends Cell Biol 24:400–406. doi:10.1016/j.tcb.2014.03.003
Beck GR (2003) Inorganic phosphate as a signaling molecule in osteoblast differentiation. J Cell Biochem 90:234–243. doi:10.1002/jcb.10622
Bergquist AJ, Wiest MJ, Collins DF (2012) Motor unit recruitment when neuromuscular electrical stimulation is applied over a nerve trunk compared with a muscle belly: quadriceps femoris. J Appl Physiol Bethesda Md 1985 113:78–89. doi:10.1152/japplphysiol.00074.2011
Bigland-Ritchie B, Cafarelli E, Vøllestad NK (1986) Fatigue of submaximal static contractions. Acta Physiol Scand Suppl 556:137–148
Bodine SC, Stitt TN, Gonzalez M et al (2001) Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat Cell Biol 3:1014–1019. doi:10.1038/ncb1101-1014
Chin ER (2005) Role of Ca2+/calmodulin-dependent kinases in skeletal muscle plasticity. J Appl Physiol Bethesda Md 1985 99:414–423. doi:10.1152/japplphysiol.00015.2005
Collins DF (2007) Central contributions to contractions evoked by tetanic neuromuscular electrical stimulation. Exerc Sport Sci Rev 35:102–109. doi:10.1097/jes.0b013e3180a0321b
Counts BR, Buckner SL, Dankel SJ et al (2016) The acute and chronic effects of “NO LOAD” resistance training. Physiol Behav 164:345–352. doi:10.1016/j.physbeh.2016.06.024
Dankel SJ, Buckner SL, Jessee MB et al (2016a) Post-exercise blood flow restriction attenuates muscle hypertrophy. Eur J Appl Physiol 116:1955–1963. doi:10.1007/s00421-016-3447-2
Dankel SJ, Jessee MB, Abe T, Loenneke JP (2016b) The effects of blood flow restriction on upper-body musculature located distal and proximal to applied pressure. Sports Med Auckl NZ 46:23–33. doi:10.1007/s40279-015-0407-7
Dankel SJ, Buckner SL, Jessee MB et al (2017a) Can blood flow restriction augment muscle activation during high-load training? Clin Physiol Funct Imaging. doi:10.1111/cpf.12414
Dankel SJ, Counts BR, Barnett BE et al (2017b) Muscle adaptations following 21 consecutive days of strength test familiarization compared with traditional training. Muscle Nerve. doi:10.1002/mus.25488
De Luca CJ, Erim Z (1994) Common drive of motor units in regulation of muscle force. Trends Neurosci 17:299–305
De Luca CJ, LeFever RS, McCue MP, Xenakis AP (1982) Control scheme governing concurrently active human motor units during voluntary contractions. J Physiol 329:129–142
Debold EP (2012) Recent insights into the molecular basis of muscular fatigue. Med Sci Sports Exerc 44:1440–1452. doi:10.1249/MSS.0b013e31824cfd26
Dentel JN, Blanchard SG, Ankrapp DP et al (2005) Inhibition of cross-bridge formation has no effect on contraction-associated phosphorylation of p38 MAPK in mouse skeletal muscle. Am J Physiol Cell Physiol 288:C824–C830. doi:10.1152/ajpcell.00500.2004
Ellefsen S, Hammarström D, Strand TA et al (2015) Blood flow-restricted strength training displays high functional and biological efficacy in women: a within-subject comparison with high-load strength training. Am J Physiol Regul Integr Comp Physiol 309:R767–R779. doi:10.1152/ajpregu.00497.2014
Fahs CA, Loenneke JP, Thiebaud RS et al (2015) Muscular adaptations to fatiguing exercise with and without blood flow restriction. Clin Physiol Funct Imaging 35:167–176. doi:10.1111/cpf.12141
Farup J, de Paoli F, Bjerg K et al (2015) Blood flow restricted and traditional resistance training performed to fatigue produce equal muscle hypertrophy. Scand J Med Sci Sports 25:754–763. doi:10.1111/sms.12396
Fry CS, Glynn EL, Drummond MJ et al (2010) Blood flow restriction exercise stimulates mTORC1 signaling and muscle protein synthesis in older men. J Appl Physiol Bethesda Md 1985 108:1199–1209. doi:10.1152/japplphysiol.01266.2009
Garten RS, Goldfarb A, Crabb B, Waller J (2015) The impact of partial vascular occlusion on oxidative stress markers during resistance exercise. Int J Sports Med 36:542–549. doi:10.1055/s-0034-1396827
Goldfarb AH, Garten RS, Chee PDM et al (2008) Resistance exercise effects on blood glutathione status and plasma protein carbonyls: influence of partial vascular occlusion. Eur J Appl Physiol 104:813–819. doi:10.1007/s00421-008-0836-1
Goldmann WH (2012) Mechanotransduction and focal adhesions. Cell Biol Int 36:649–652. doi:10.1042/CBI20120184
Gorgey AS, Timmons MK, Dolbow DR et al (2016) Electrical stimulation and blood flow restriction increase wrist extensor cross-sectional area and flow meditated dilatation following spinal cord injury. Eur J Appl Physiol 116:1231–1244. doi:10.1007/s00421-016-3385-z
Goto K, Ishii N, Kizuka T, Takamatsu K (2005) The impact of metabolic stress on hormonal responses and muscular adaptations. Med Sci Sports Exerc 37:955–963
Gundermann DM, Walker DK, Reidy PT et al (2014) Activation of mTORC1 signaling and protein synthesis in human muscle following blood flow restriction exercise is inhibited by rapamycin. Am J Physiol Endocrinol Metab 306:E1198–1204. doi:10.1152/ajpendo.00600.2013
Hausenblas HA, Fallon EA (2006) Exercise and body image: a meta-analysis. Psychol Health 21:33–47. doi:10.1080/14768320500105270
Hornberger TA, Chu WK, Mak YW et al (2006) The role of phospholipase D and phosphatidic acid in the mechanical activation of mTOR signaling in skeletal muscle. Proc Natl Acad Sci USA 103:4741–4746. doi:10.1073/pnas.0600678103
Ikai M, Fukunaga T (1970) A study on training effect on strength per unit cross-sectional area of muscle by means of ultrasonic measurement. Int Z Für Angew Physiol Einschl Arbeitsphysiol 28:173–180. doi:10.1007/BF00696025
Ito N, Ruegg UT, Kudo A et al (2013) Activation of calcium signaling through Trpv1 by nNOS and peroxynitrite as a key trigger of skeletal muscle hypertrophy. Nat Med 19:101–106. doi:10.1038/nm.3019
Jacobs BL, McNally RM, Kim KJ et al (2017) Identification of mechanically regulated phosphorylation sites on tuberin (TSC2) that control mechanistic target of rapamycin (mTOR) signaling. J Biol Chem. doi:10.1074/jbc.M117.777805
Kraemer WJ, Noble BJ, Clark MJ, Culver BW (1987) Physiologic responses to heavy-resistance exercise with very short rest periods. Int J Sports Med 8:247–252. doi:10.1055/s-2008-1025663
Laurentino G, Ugrinowitsch C, Aihara AY et al (2008) Effects of strength training and vascular occlusion. Int J Sports Med 29:664–667. doi:10.1055/s-2007-989405
Laurentino GC, Ugrinowitsch C, Roschel H et al (2012) Strength training with blood flow restriction diminishes myostatin gene expression. Med Sci Sports Exerc 44:406–412. doi:10.1249/MSS.0b013e318233b4bc
Laurin J, Pertici V, Dousset E et al (2015) Group III and IV muscle afferents: role on central motor drive and clinical implications. Neuroscience 290:543–551. doi:10.1016/j.neuroscience.2015.01.065
Lauver JD, Cayot TE, Rotarius T, Scheuermann BW (2017) The effect of eccentric exercise with blood flow restriction on neuromuscular activation, microvascular oxygenation, and the repeated bout effect. Eur J Appl Physiol. doi:10.1007/s00421-017-3589-x
LeFever RS, De Luca CJ (1982) A procedure for decomposing the myoelectric signal into its constituent action potentials–part I: technique, theory, and implementation. IEEE Trans Biomed Eng 29:149–157
Lin H, Wang SW, Wang RY, Wang PS (2001) Stimulatory effect of lactate on testosterone production by rat leydig cells. J Cell Biochem 83:147–154
Loenneke JP, Fahs CA, Wilson JM, Bemben MG (2011) Blood flow restriction: the metabolite/volume threshold theory. Med Hypotheses 77:748–752. doi:10.1016/j.mehy.2011.07.029
Loenneke JP, Balapur A, Thrower AD et al (2012a) Blood flow restriction reduces time to muscular failure. Eur J Sport Sci 12:238–243. doi:10.1080/17461391.2010.551420
Loenneke JP, Wilson JM, Marín PJ et al (2012b) Low intensity blood flow restriction training: a meta-analysis. Eur J Appl Physiol 112:1849–1859. doi:10.1007/s00421-011-2167-x
Manini TM, Clark BC (2009) Blood flow restricted exercise and skeletal muscle health. Exerc Sport Sci Rev 37:78–85. doi:10.1097/JES.0b013e31819c2e5c
Marcotte GR, West DWD, Baar K (2015) The molecular basis for load-induced skeletal muscle hypertrophy. Calcif Tissue Int 96:196–210. doi:10.1007/s00223-014-9925-9
Meyer RA (2006) Does blood flow restriction enhance hypertrophic signaling in skeletal muscle? J Appl Physiol Bethesda Md 1985 100:1443–1444. doi:10.1152/japplphysiol.01636.2005
Mitchell CJ, Churchward-Venne TA, West DWD et al (2012) Resistance exercise load does not determine training-mediated hypertrophic gains in young men. J Appl Physiol 113:71. doi:10.1152/japplphysiol.00307.2012
Moritani T, Sherman WM, Shibata M et al (1992) Oxygen availability and motor unit activity in humans. Eur J Appl Physiol 64:552–556
Morton RW, Oikawa SY, Wavell CG et al (2016) Neither load nor systemic hormones determine resistance training-mediated hypertrophy or strength gains in resistance-trained young men. J Appl Physiol Bethesda Md 1985 121:129–138. doi:10.1152/japplphysiol.00154.2016
Nalbandian M, Takeda M (2016) Lactate as a signaling molecule that regulates exercise-induced adaptations. Biology 5:38
Natsume T, Ozaki H, Saito AI et al (2015) Effects of electrostimulation with blood flow restriction on muscle size and strength. Med Sci Sports Exerc 47:2621–2627. doi:10.1249/MSS.0000000000000722
Nielsen JL, Aagaard P, Bech RD et al (2012) Proliferation of myogenic stem cells in human skeletal muscle in response to low-load resistance training with blood flow restriction. J Physiol 590:4351–4361. doi:10.1113/jphysiol.2012.237008
Nishimura A, Sugita M, Kato K et al (2010) Hypoxia increases muscle hypertrophy induced by resistance training. Int J Sports Physiol Perform 5:497–508
O’Neil TK, Duffy LR, Frey JW, Hornberger TA (2009) The role of phosphoinositide 3-kinase and phosphatidic acid in the regulation of mammalian target of rapamycin following eccentric contractions. J Physiol 587:3691–3701. doi:10.1113/jphysiol.2009.173609
Ogasawara R, Loenneke JP, Thiebaud RS, Abe T (2013) Low-load bench press training to fatigue results in muscle hypertrophy similar to high-load bench press training. Int J Clin Med 4:114. doi:10.4236/ijcm.2013.42022
Oishi Y, Tsukamoto H, Yokokawa T et al (2015) Mixed lactate and caffeine compound increases satellite cell activity and anabolic signals for muscle hypertrophy. J Appl Physiol Bethesda Md 1985 118:742–749. doi:10.1152/japplphysiol.00054.2014
Ozaki H, Abe T, Mikesky AE et al (2015) Physiological stimuli necessary for muscle hypertrophy. J Phys Fit Sports Med 4:43–51
Ozaki H, Loenneke JP, Buckner SL, Abe T (2016) Muscle growth across a variety of exercise modalities and intensities: contributions of mechanical and metabolic stimuli. Med Hypotheses 88:22–26. doi:10.1016/j.mehy.2015.12.026
Park JH, Brown RL, Park CR et al (1987) Functional pools of oxidative and glycolytic fibers in human muscle observed by 31P magnetic resonance spectroscopy during exercise. Proc Natl Acad Sci USA 84:8976–8980
Pearson SJ, Hussain SR (2015) A review on the mechanisms of blood-flow restriction resistance training-induced muscle hypertrophy. Sports Med Auckl NZ 45:187–200. doi:10.1007/s40279-014-0264-9
Person RS (1974) Rhythmic activity of a group of human motoneurones during voluntary contraction of a muscle. Electroencephalogr Clin Neurophysiol 36:585–595. doi:10.1016/0013-4694(74)90225-9
Schoenfeld BJ (2013) Potential mechanisms for a role of metabolic stress in hypertrophic adaptations to resistance training. Sports Med 43:179–194. doi:10.1007/s40279-013-0017-1
Schott J, McCully K, Rutherford OM (1995) The role of metabolites in strength training. II. Short versus long isometric contractions. Eur J Appl Physiol 71:337–341
Smith RC, Rutherford OM (1995) The role of metabolites in strength training. I. A comparison of eccentric and concentric contractions. Eur J Appl Physiol 71:332–336
Spina A, Sorvillo L, Esposito A et al (2013) Inorganic phosphate as a signaling molecule: a potential strategy in osteosarcoma treatment. Curr Pharm Des 19:5394–5403
Suga T, Okita K, Morita N et al (2009) Intramuscular metabolism during low-intensity resistance exercise with blood flow restriction. J Appl Physiol Bethesda Md 1985 106:1119–1124. doi:10.1152/japplphysiol.90368.2008
Suga T, Okita K, Morita N et al (2010) Dose effect on intramuscular metabolic stress during low-intensity resistance exercise with blood flow restriction. J Appl Physiol Bethesda Md 1985 108:1563–1567. doi:10.1152/japplphysiol.00504.2009
Suga T, Okita K, Takada S et al (2012) Effect of multiple set on intramuscular metabolic stress during low-intensity resistance exercise with blood flow restriction. Eur J Appl Physiol 112:3915–3920. doi:10.1007/s00421-012-2377-x
Takano H, Morita T, Iida H et al (2005) Hemodynamic and hormonal responses to a short-term low-intensity resistance exercise with the reduction of muscle blood flow. Eur J Appl Physiol 95:65–73. doi:10.1007/s00421-005-1389-1
Takarada Y, Nakamura Y, Aruga S et al (2000a) Rapid increase in plasma growth hormone after low-intensity resistance exercise with vascular occlusion. J Appl Physiol Bethesda Md 1985 88:61–65
Takarada Y, Takazawa H, Sato Y et al (2000b) Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. J Appl Physiol 88:2097–2106
Tanimoto M, Sanada K, Yamamoto K et al (2008) Effects of whole-body low-intensity resistance training with slow movement and tonic force generation on muscular size and strength in young men. J Strength Cond Res 22:1926–1938. doi:10.1519/JSC.0b013e318185f2b0
Timmerman KL, Lee JL, Dreyer HC et al (2010) Insulin stimulates human skeletal muscle protein synthesis via an indirect mechanism involving endothelial-dependent vasodilation and mammalian target of rapamycin complex 1 signaling. J Clin Endocrinol Metab 95:3848–3857. doi:10.1210/jc.2009-2696
Vandenborne K, McCully K, Kakihira H et al (1991) Metabolic heterogeneity in human calf muscle during maximal exercise. Proc Natl Acad Sci 88:5714–5718. doi:10.1073/pnas.88.13.5714
Villanueva MG, Lane CJ, Schroeder ET (2015) Short rest interval lengths between sets optimally enhance body composition and performance with 8 weeks of strength resistance training in older men. Eur J Appl Physiol 115:295–308. doi:10.1007/s00421-014-3014-7
Wernbom M, Järrebring R, Andreasson MA, Augustsson J (2009) Acute effects of blood flow restriction on muscle activity and endurance during fatiguing dynamic knee extensions at low load. J Strength Cond Res 23:2389–2395. doi:10.1519/JSC.0b013e3181bc1c2a
West DWD, Baar K (2013) May the Force move you: TSC-ing the mechanical activation of mTOR. J Physiol 591:4369–4370. doi:10.1113/jphysiol.2013.260216
West DWD, Burd NA, Tang JE et al (2010) Elevations in ostensibly anabolic hormones with resistance exercise enhance neither training-induced muscle hypertrophy nor strength of the elbow flexors. J Appl Physiol Bethesda Md 1985 108:60–67. doi:10.1152/japplphysiol.01147.2009
Westgaard RH, de Luca CJ (1999) Motor unit substitution in long-duration contractions of the human trapezius muscle. J Neurophysiol 82:501–504
Willkomm L, Schubert S, Jung R et al (2014) Lactate regulates myogenesis in C2C12 myoblasts in vitro. Stem Cell Res 12:742–753. doi:10.1016/j.scr.2014.03.004
Wolfe RR (2006) The underappreciated role of muscle in health and disease. Am J Clin Nutr 84:475–482
Yasuda T, Brechue WF, Fujita T et al (2009) Muscle activation during low-intensity muscle contractions with restricted blood flow. J Sports Sci 27:479–489. doi:10.1080/02640410802626567
You J-S, Lincoln HC, Kim C-R et al (2014) The role of diacylglycerol kinase ζ and phosphatidic acid in the mechanical activation of mammalian target of rapamycin (mTOR) signaling and skeletal muscle hypertrophy. J Biol Chem 289:1551–1563. doi:10.1074/jbc.M113.531392
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
None.
Additional information
Communicated by Michael Lindinger.
Rights and permissions
About this article
Cite this article
Dankel, S.J., Mattocks, K.T., Jessee, M.B. et al. Do metabolites that are produced during resistance exercise enhance muscle hypertrophy?. Eur J Appl Physiol 117, 2125–2135 (2017). https://doi.org/10.1007/s00421-017-3690-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-017-3690-1