Abstract
Purpose
A previous study observed a joint passive torque increase above baseline ~30 min after a high-intensity stretching. This study examined the effect of a high-intensity stretching on ankle dorsiflexion passive torque, medial gastrocnemius (MG) shear modulus, and plantar flexors maximal voluntary isometric force (MVIC).
Method
Participants (n = 11, age 27.2 ± 6.5 years, height 172.0 ± 10.0 cm, weight 69.5 ± 10.4 kg) underwent two stretching sessions with plantar flexors isometric contractions performed: (1) 5 min before, 1 min after, and every 10 min after stretching (MVC session); (2) 5 min before, and 60 min after the stretching (no-MVC session).
Results
In both sessions, no changes were observed for MG shear modulus (p > 0.109). In the no-MVC session, passive torque decreased 1 min after stretching (−7.5 ± 8.4 %, p = 0.015), but increased above baseline 30 min after stretching (+6.3 ± 9.3 %, p = 0.049). In the MVC session, passive torque decreased at 1 min (−10.1 ± 6.3 %, p < 0.001), 10 min (−6.3 ± 8.2 %, p = 0.03), 20 min (−8.0 ± 9.2 %, p = 0.017), and 60 min (−9.2 ± 12.4 %, p = 0.034) after the stretching, whereas the MVIC decreased at 1 min (−5.0 ± 9.3 %, p = 0.04) and 10 min (−6.7 ± 8.7 %, p = 0.02) after stretching.
Conclusion
The ankle passive torque increase 30 min following the stretch was not due to the MG shear modulus response; consequently, response may be due to changes in surrounding connective tissue mechanical properties.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stretching protocols are commonly designed to increase the maximal range of motion (ROM) of a joint and decrease the passive muscle tension or stiffness, in both clinical and sports contexts. Although several studies have reported acute decreases in passive joint torque after different stretching protocols (Magnusson et al. 1996; McNair et al. 2001; Freitas et al. 2014), it has been shown that passive torque returns to baseline values within 10 to 60 min after stretching (Magnusson et al. 1996; McHugh and Cosgrave 2010; Mizuno et al. 2013). While most of these studies have used static stretching protocols with rest between repetitions (Magnusson et al. 1996; Mizuno et al. 2013), few studies have focused on protocols based on non-resting between repetitions (NRI) intending to induce a higher stretch intensity, and consequently a greater gain in maximal ROM (Freitas et al. 2015b, 2016b). This stretching method is similar to proprioceptive neuromuscular facilitation (PNF) contract–relax stretching except it does not include muscle contractions, and has been recently shown to induce a greater stretching intensity compared to conventional resting interval static stretching (Freitas et al. 2015b, 2016a). However, we recently observed a significant joint passive torque increase above the baseline condition at 30 and 60 min after high-intensity knee flexors stretching induced by an NRI protocol (Freitas et al. 2016a). This finding is perplexing concerning the aims of using high-intensity stretching protocols (e.g., NRI) and requires further clarification. Thus, a detailed characterization of the muscle mechanical response after an intense stretch is crucial for both fundamental and applied purposes. The present study was designed to explore three main key points.
First, the passive joint torque represents the passive resistance to stretch given by the whole muscle–articular complex, including muscular and non-muscular structures (Riemann et al. 2001; Nordez et al. 2006, 2008). Recently, localized muscle mechanical properties have been characterized using Supersonic Shear Imaging elastography (Maïsetti et al. 2012; Freitas et al. 2015a). The shear modulus measured is linearly correlated to the muscle Young’s modulus (Eby et al. 2013), and can be used as an estimation of changes in passive muscle tension (Maïsetti et al. 2012; Koo et al. 2013). Two previous studies showed that static stretching decreases the muscle shear modulus (Freitas et al. 2015a; Taniguchi et al. 2015). This technique could, therefore, be very relevant to analyze the effects of intense stretching on localized muscle properties.
Second, most changes in passive torque occur at the beginning of the knee extension range of motion (Freitas et al. 2016b), indicating that the shape of the torque–angle relationship and the dissipation coefficient could also be altered (Nordez et al. 2010). Thus, the assessment of the muscle load–unload stretching response could be useful in describing the acute effects of an intensive stretching on muscle dissipative mechanical properties.
Third, an increase in the passive muscle tension and dissipation coefficient after an exercise stimulus is often associated with muscle damage (Whitehead et al. 2001a, 2003a; Lacourpaille et al. 2014). For instance, Larcoupaille (2014) observed a muscle passive shear modulus (i.e., stiffness) and dissipation coefficient increase 1 h after eccentric muscle contractions. Since a previous study has reported changes in muscle damage markers as a consequence of a static stretching intervention (Smith et al. 1993), an intense stretching protocol may cause (small) muscle damage due to high tissue deformation. In that case, the maximal isometric muscle force should be decreased at 1 h after the stretching protocol (McHugh et al. 1999), considering that the strength loss induced by stretching is recovered in less than 1 h (Mizuno et al. 2014). Consequently, to better understand the acute effects of intense stretching, maximal voluntary isometric contractions (MVIC) tests have to be performed in addition to passive torque and shear modulus measurements.
The present study was designed to determine the acute effects (within 1 h) of high-intensity ankle plantar flexors stretching on: (1) ankle dorsiflexion passive torque and medial gastrocnemius (MG) shear modulus; (2) MG and ankle dissipative properties; (3) plantar flexors maximal isometric force. An NRI stretching protocol was used to impose passive high-intensity stretching on the ankle plantar flexors. We hypothesized that (1) the MG shear modulus would increase due to high stretching-induced muscle damage, and (2) the MVIC would decrease at 1 h after stretch indicating the occurrence of muscle damage.
Materials and methods
Participants
Eleven participants (physically active; men, n = 5; women, n = 6; age 27.2 ± 6.5 years; height 172.0 ± 10.0 cm; weight 69.5 ± 10.4 kg) volunteered for this study, and consent was obtained from each participant. All participants were familiarized with the experimental setup. The local ethics committee approved the study (#2013/01), and all the procedures were in accordance with the Declaration of Helsinki.
Variables and equipment
Passive torque–angle
An isokinetic dynamometer (Biodex 3 medical, Shirley, NY, USA) was used to impose maximal passive ankle dorsiflexion (i.e., ankle plantar flexors stretching), and to measure ankle angle and torque during the stretching and MVIC tests. The lateral malleolus was aligned with the axis of the Biodex dynamometer. The foot was perpendicularly positioned with the leg and corresponded to 0°. Mechanical data were recorded at a sampling rate of 1000 Hz (MP36, BIOPAC, Goleta, California, USA). A good inter-day reliability of ankle dorsiflexion passive torque–angle assessment was previously reported (Freitas et al. 2015a).
Medial gastrocnemius shear modulus
An Aixplorer ultrasound scanner (version 7.0; Supersonic Imagine, Aix-en-Provence, France) coupled with a linear transducer array (4–15 MHz. Super Linear 15-4, Vermon, Tours, France) was used in shear wave elastography mode (musculo-skeletal preset) to assess the medial gastrocnemius (MG) shear modulus. The ultrasound probe was positioned over the MG mid-belly (i.e., 50 % of the muscle length) by an experienced examiner (i.e., more than 100 h). The transducer was placed perpendicularly to the skin and according to the orientation of the muscle fascicles (Blazevich 2006). Assuming a linear elastic behavior, the muscle shear modulus (μ) was calculated as follows (1):
where ρ is the density of soft tissues (1000 kg m3) and Vs is the shear wave speed. The maps of the shear modulus were collected at 1 Hz with a spatial resolution of 1 × 1 mm. A good intra-day (Maïsetti et al. 2012) and inter-day (Freitas et al. 2015a) reliability of MG shear modulus has been previously reported.
Protocol
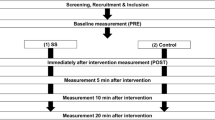
Participants performed three sessions on different days with at least 1 week of rest in between (Fig. 1a). In the familiarization session, participants performed some static stretching trials (with submaximal intensities) in the experimental setup. In the following two sessions, an intense NRI stretching protocol that targeted the ankle plantar flexors muscles was performed (Fig. 1b) (Freitas et al. 2015b). During one of the sessions (i.e., MVC session), the ankle plantar flexors MVIC was assessed 5 min before and 1, 10, 20, 30, 40, 50, and 60 min after the stretching protocol. In the other session (i.e., no-MVC session), the MVIC was assessed only 5 min before and 60 min after the stretching protocol. The MVIC assessment consisted of three trials of 3-s maximal voluntary plantar flexors contractions, with 15-s rest between repetitions. The ankle was in a neutral position (i.e., leg positioned perpendicularly to the foot, 0°). The experimental (i.e., MVC and no-MVC) sessions were randomized.
Each session started with the MVIC testing. Then, after 5-min rest, four ankle plantar-to-dorsiflexion cycles were performed with an angular velocity of 5°/s for conditioning purposes, followed by an additional fifth cycle performed at 2°/s for pre-assessment purpose (Nordez et al. 2010). The cycles were performed between 40° of ankle plantar flexion and 20° of ankle dorsiflexion. For testing the post-stretching effects, an ankle cycle (2°/s) was performed 1, 10, 20, 30, 40, 50, and 60 min after each stretching protocol. In the MVC session, the cycle was always performed before the MVIC trials. The ankle was positioned at 20° of plantar flexion during the resting time between assessments, to maintain the MG in a slack position (Hug et al. 2013).
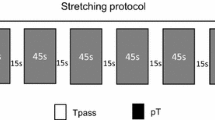
The NRI stretching protocol consisted of performing the maximal number of passive stretching repetitions without rest between repetitions (Freitas et al. 2016b). In all repetitions, the stretching maneuvers were performed with an angular velocity of 2°/s until participants reached end ROM (point of discomfort) (Fig. 1b). At the end ROM of each repetition, the joint was immobilized for 90 s in the stretching position so joint relaxation could occur. At the end of relaxation, the participants were asked if they could stretch further. When participants could not stretch further, the NRI protocol was finalized thus allowing (due to stretch tolerance variability) the maximal number of NRI repetitions (and consequently the total stretching duration) could differ between participants. This procedure was used to ensure that the highest stretch intensity was achieved during the stretching intervention. The same number of NRI repetitions and the ankle angles performed in the first session were reproduced in the second session. Thus, the stretching intensity and duration were, therefore, the same across sessions.
Data analysis
All data processing procedures were performed using Matlab (Matlab, Mathworks, Natick, MA, US). MG shear modulus recordings were exported from software (Version 7.0, Supersonic Imagine, Aix en Provence, France) in .mp4 video format and sequenced in .jpeg image files. Briefly, image processing converted the colored map into shear modulus values. For each image, the average value of shear modulus was calculated over a region of interest corresponding to the largest muscular region of MG. The ankle passive torque–angle and shear modulus values were determined for every 2° of ankle angle through the range of motion used.
For both passive torque–angle and shear modulus–angle data, the areas under the loading (E) and unloading (ER) curves, the hysteresis area (ED = E − ER), and the normalized hysteresis area (DC = ED/E) were calculated as described by Nordez et al. (2010).
The MVIC value for each test was determined as the highest torque value across the three trials performed. The value of each trial was calculated as the average obtained in 1-s window at the peak torque value.
Statistical analysis
All data were analyzed using IBM SPSS Statistics 19.0 (IBM Corporation, New York, USA). The data were first normalized to baseline (pre) values (mean ± SD). Normal distribution was confirmed using Shapiro–Wilk test. Both shear modulus and passive torque values at the angles corresponding to the 80 % of the ankle range of motion (i.e., 12° of dorsiflexion) were determined for comparisons between the different testing moments. Such angle was used to prevent muscle activity due to high muscle lengthening. To determine if participants were in the same condition at the beginning of the stretching protocol, a t test was used to compare the MG shear modulus, the passive torque, and the MVIC of the pre-stretching condition between the two sessions. A two-way ANOVA [protocol (MVC, no-MVC) × time (pre, 1, 10, 20, 30, 40, 50, 60 min)] was performed for comparisons of ankle passive torque, passive torque-DC, MG shear modulus, and MG shear modulus-DC values. Two-way repeated measures ANOVA [protocol (MVC, no-MVC) × time (pre, 60 min)] was performed for MVIC, and one-way repeated measures ANOVA was performed for MVIC for all observations in the MVC session. When an interaction for protocols × time was observed, a one-way repeated measures followed by contrasts (pre vs. post stretching time testing) was performed for time moments, and paired t tests were performed to compare protocols for each testing time. A one-way repeated measure followed by contrasts was performed for MVIC in the MVC session. Statistical significance was set at p < 0.05.
Results
No differences were found for passive torque (p = 0.29), MG shear modulus (p = 0.21), and MVIC (p = 0.11) between the pre-condition of the two stretching sessions. Although a different number of stretching repetitions and total stretch duration were performed among participants [2NRI (stretch duration = 180 s): n = 2; 3NRI (270 s): n = 3; 4NRI (360 s): n = 2; 5NRI (450 s): n = 4], the same number of stretching repetitions and stretch duration was performed in both sessions. A typical example of ankle passive torque and MG shear modulus response to stretching in both sessions before and 20 min after the NRI stretching can be observed in Fig. 2.
Passive torque Significant effects of protocol × time (p = 0.017), time (p < 0.001) and protocol (p = 0.027) were observed for passive torque (Fig. 3a). For the no-MVC session, the passive torque decreased 1 min after stretching (−7.5 ± 8.4 %, p = 0.015), then increased above baseline at 30 min (+6.3 ± 9.3 %, p = 0.049) and recovered to baseline at 40 min (p = 0.23). In the MVC session, passive torque decreased at 1 min (−10.1 ± 6.3 %, p < 0.001), 10 min (−6.3 ± 8.2 %, p = 0.03), 20 min (−8.0 ± 9.2 %, p = 0.017), and 60 min (−9.2 ± 12.4 %, p = 0.034) after the stretching. Passive torque was significantly different between sessions at 20 min (+7.5 ± 13.9 % vs. −8.0 ± 82 %, p = 0.013) and 30 min (+6.3 ± 9.3 % vs. −5.6 ± 13.2 %, p = 0.017). A significant effect for protocol × time (p = 0.019) and time (p = 0.001) on passive torque-DC was observed (Fig. 3b). Passive torque-DC increased at 20 min (+59.0 ± 81.7 %, p = 0.024) in the no-MVC session, but not at 30 min (+31.5 ± 56.7 %, p = 0.06).
Measurements before and 1, 10, 20, 30, 40, 50, and 60 min after the stretching protocol in both MVC and no-MVC sessions: a ankle passive torque at 12° of dorsiflexion; b passive torque-DC; c MG shear modulus at 12° of dorsiflexion; d maximal plantar flexors isometric contraction. All values are normalized to the baseline condition. *Statistically different from the baseline condition (p < 0.05), #statistically different between sessions (p < 0.05)
MG shear modulus The MG shear modulus is depicted for both stretching sessions in Fig. 3c. No significant effects were observed in the MG shear modulus (p > 0.464), and MG shear modulus-DC (p > 0.109).
Maximal muscle isometric force A significant effect was observed for time (p = 0.022) in the MVIC for the MVC session only (Fig. 3d), whereas a decrease was observed at 1 min (−5.0 ± 9.3 %, p = 0.04) and 10 min (−6.7 ± 8.7 %, p = 0.02). No significant effect was observed for time (p = 0.48) and protocols (p = 0.47) in both stretching sessions.
Discussion
This study investigated the effects of a high-intensity plantar flexors stretching protocol on ankle passive torque, MG shear modulus, and plantar flexors MVIC. It was observed that the ankle passive torque decreased immediately after the intense stretching used for a given common ankle angle, and increased above the baseline 30 min following the stretching. However, contrary to our hypothesis, no significant changes in MG shear modulus were found. In addition, while a significant (but transient) decrease in MVIC was found after stretching, a different time course was observed compared to the changes in passive torque (Fig. 3), invalidating the damage hypothesis to explain such change.
The MG has been reported as the most tensioned plantar flexor muscle during dorsiflexion (Edama et al. 2015, Hirata et al. 2016); thus, it is assumed to be an important contributor to dorsiflexion passive torque during plantar flexors stretching when the knee is fully extended. The MG shear modulus is reported to decrease immediately after 10 min (i.e., constant angle) at submaximal (80 %) ROM (Freitas et al. 2015a), and 5 min (i.e., constant angle) at maximal ROM (Taniguchi et al. 2015) of static stretching, and recovers to baseline values within 20 min (Taniguchi et al. 2015). In the present study, no changes were observed after stretching on the MG shear modulus and on MG dissipative properties, despite the significant decrease found in ankle passive torque. However, it should be noted that the stretching intensity used in the present study was much greater than those performed previously (Taniguchi et al. 2015). Thus, it is possible that the muscle shear modulus response to static stretching depends on both stretching intensity (i.e., degree of tissues length/tension) and stretching method (e.g., with rest or no-rest between repetitions). Considering that the muscle shear modulus reflects the changes in passive muscle tension (Maïsetti et al. 2012; Koo et al. 2013), this result also indicates that alterations in joint passive torque do not reflect changes in a localized muscle passive tension. A similar conclusion was previously obtained for submaximal stretching intensities (Freitas et al. 2015a). Therefore, future studies should explore the tissues linked to changes in passive torque after various stretching protocols with various intensities.
As previously expected (Freitas et al. 2016a), an increase in ankle passive torque 30 min was found after the NRI protocol. This mechanical response was accompanied by an increase in the ankle passive torque–angle dissipation coefficient. However, we unexpectedly observed a passive torque decrease when plantar flexors MVIC were performed each 10 min after stretching, suppressing the acute increase in passive torque. Previous studies have showed that muscle contractions change the joint passive torque, and these are likely to be dependent on the type of muscle contraction (Magnusson et al. 1995; Whitehead et al. 2001; Klee et al. 2002; Whitehead et al. 2003; Kay et al. 2015a). For instance, Kay and Blazevich (2009) (Kay et al. 2015) reported an ankle dorsiflexion passive torque decrease after concentric (Kay and Blazevich 2009) and isometric (Kay et al. 2015) plantar flexors contraction. In addition, muscle and tendon passive stiffness also decreased (Kay and Blazevich 2009; Kay et al. 2015). Thus, we speculate that isometric and concentric muscle contractions performed after the NRI stretching protocol may counteract the joint mechanical effects through changes in connective tissues (e.g., tendon and fascias) crossing the joint.
The hypothesis that a high-intensity stretch could cause muscle damage and underlie the joint passive torque increase is not supported by the results of this study. Four reasons justify this conclusion. First, no change on muscle shear modulus was observed when passive torque was increased above baseline, whereas it is expected to increase when the muscle is damaged (Lacourpaille et al. 2014). Second, MVIC of plantar flexors decreased after the stretching, according to reported values in the literature (Kay and Blazevich 2012; Simic et al. 2013), but recovered within 20 min. Previous literature reports that strength loss induced by stretching is transient (less than 1 h) (Mizuno et al. 2014), whereas such recovery does not occur after muscle damage. Thus, we conclude that the effects of NRI on MVIC are due to stretching-induced strength loss (e.g., shift in muscle active force–length curve). If muscle damage occurred due to the stretching intervention, the time course of change (i.e., decrease) in MVIC should remain for a longer time period. Third, the ankle passive torque only increased at 20 min (p = 0.06) and 30 min after stretching, and it was suppressed when muscle MVICs were performed. Four, the MG shear modulus-DC did not change after stretching, whereas a change was observed in the passive torque-DC. Thus, we assume that the passive torque increase above baseline after the NRI stretching (i.e., no-MVC session) was not due to muscle damage.
Considering the no change in MG shear modulus, and the passive torque increase at 30 min after NRI stretching (Freitas et al. 2016a), our results suggest that the mechanism underlying the passive torque response may be: (i) not represented by all plantar flexors muscles components, and/or (ii) related to a non-muscular tissue. For instance, the muscle contractions are thought to stress the surrounding connective tissue (i.e., intra- and extra-muscular); these structures (e.g., tendon) are, therefore, deformed during muscle contractions, and thus their stiffness decreases with no changes in muscle stiffness (Kay and Blazevich 2009; Kay et al. 2015). In addition, because the passive torque-DC also increased with the increase of passive torque at 20–30 min post-stretching, it is reasonable to assume that ankle viscosity was also affected. Interestingly, a previous study observed an increase in fascia stiffness accompanied by a change in fascia water content after an intense stretch (Schleip et al. 2012). In the present study, we did not measure the connective tissue passive tension, and thus we cannot confirm that a change in tension occurred 20–30 min after NRI stretching. Future studies may want to confirm this hypothesis by, for instance, determining the tendon stiffness at 20–30 min after a high-intensity stretching.
It should be noted that NRI stretching is very similar to conventional PNF contract–relax stretching. The only difference is that NRI does not include isometric muscle contractions. During PNF stretching, the joint is moved until the end ROM, and after a relaxation period, muscles being stretched produce an isometric contraction and subsequently they are stretched [with no-rest (i.e., unload) period] to greater ROM. During NRI, in the end relaxation, the limb is moved to a new ROM. Thus, despite NRI not being a popular concept among professionals, it may be performed with high incidence.
Four main limitations should be considered in the present study. First, previous stretching studies that assessed ankle dorsiflexion passive torque have simultaneously measured the leg muscles surface electromyographic (EMG) activation, to ensure a passive condition expressed by low (<1 % of MVIC) EMG activation (Gajdosik 2006). In addition, a low-joint angular velocity (<2º/s) is often used to impose muscle–tendon stretching (Kay and Blazevich 2009; Hug et al. 2013; Freitas et al. 2015a). However, a greater muscular activation is often observed when the ankle is dorsiflexed close to the maximal range of motion (i.e., ankle angle >80 % of maximal ROM; or >25º of dorsiflexion (Gajdosik 2006; Freitas et al. 2015b)). In the present study, we have not quantified the muscle activation due to data collection problems. Although, we have carefully inspected the passive angle–torque curves to detect if a muscle contraction would occur during the stretching. In addition, since the plantar flexors were stretched only until 20º of dorsiflexion, it can be assumed that the EMG activity was not relevant and the cycles were performed in a passive condition. However, we recognize this assumption as the main study limitation. Second, the number of plantar flexion isometric contractions could have induced some fatigue in the MVC sessions (i.e., a total number of 24 isometric contractions), since the repetitions were separated by ~30 s and the contractions bouts by 10 min, although the maximal force decrease was not different within 1 min and 60 min after stretching in the MVC session. Thus, we assume that no fatigue occurred and, consequently, the total number of isometric contractions did not affect the study conclusions. Third, repeated bout effect could potentially affect the mechanical responses after stretch in the second session, although the repeated bout effect only alters the magnitude of muscle damage effects and not the type of response. In addition, experimental sessions were randomized across participants, and thus potential bias induced by repeated bout effect was minimized. Finally, although MVIC is reported to change when muscle damage occurs, this marker may not be sensitive enough to detect (potentially small) muscle damage. In addition, since the joint active torque–angle relationship has been reported to shift to the right after stretching (Cramer et al. 2007), the transient MVIC decrements could be explained by this shift, and not due to muscle damage.
Conclusions
Intense stretching did not appear to induce plantar flexors damage and the MG shear modulus response to a high-intensity plantar flexors stretching did not follow the changes in ankle passive torque. Thus, the response of other agonistic muscles or non-muscular structures (e.g., fascia) might explain the joint passive torque response. A practical application of this study is that resistance to stretching (i.e., joint passive torque) can be reduced after high-intensity stretching when adding isometric contractions to the intense stretching, and with the maximal muscle force production restored within 20 min. Future studies should investigate the load characteristics (i.e., stretching and muscle contractions) that acutely and chronically decrease the resistance to stretching.
Abbreviations
- DC:
-
Dissipation coefficient
- E:
-
Area under the load curve
- ED:
-
Energy dissipated (i.e., hysteresis area)
- ER:
-
Area under the unload curve
- MVC:
-
Muscular voluntary contraction session
- MG:
-
Medial gastrocnemius
- MVIC:
-
Muscular voluntary isometric contraction
- No-MVC:
-
No muscular voluntary contraction session
- NRI:
-
Non-rest interval
- ROM:
-
Range of motion
- PNF:
-
Proprioceptive neuromuscular facilitation stretching
References
Blazevich AJ (2006) Effects of physical training and detraining, immobilisation, growth and aging on human fascicle geometry. Sports Med 36:1003–1017
Cramer JT, Beck TW, Housh TJ et al (2007) Acute effects of static stretching on characteristics of the isokinetic angle–torque relationship, surface electromyography, and mechanomyography. J Sports Sci 25:687–698
Eby SF, Pengfei S, Shigao C et al (2013) Validation of shear wave elastography in skeletal muscle. J Biomech 46:2381–2387. doi:10.1016/j.jbiomech.2013.07.033
Edama M, Onishi H, Kumaki K et al (2015) Effective and selective stretching of the medial head of the gastrocnemius. Scand J Med Sci Sports 25:242–250. doi:10.1111/sms.12203
Freitas SR, Vilarinho D, Vaz JR et al (2014) Responses to static stretching are dependent on stretch intensity and duration. Clin Physiol Funct Imaging 35:478–484. doi:10.1111/cpf.12186
Freitas SR, Andrade RJ, Lilian L et al (2015a) Muscle and joint responses during and after static stretching performed at different intensities. Eur J Appl Physiol 115:1263–1272. doi:10.1007/s00421-015-3104-1
Freitas SR, Vaz JR, Gomes L et al (2015b) A new tool to assess the perception of stretching intensity. J Strength Cond Res 29:2666–2678. doi:10.1519/JSC.0000000000000926
Freitas SR, Vaz JR, Andrade RJ, Bruno P, Mil-Homens P (2016a) Stretching effects: high-intensity & moderate-duration vs. Low-intensity & long-duration. Int J Sports Med 37:239–244. doi:10.1055/s-0035-1548946
Freitas SR, Vaz JR, Bruno PM et al (2016b) Are rest intervals between stretching repetitions effective to acutely increase range of motion? Int J Sports Physiol Perform 10:191–197. doi:10.1123/ijspp.2014-0192
Gajdosik RL (2006) Influence of a low-level contractile response from the soleus, gastrocnemius and tibialis anterior muscles on viscoelastic stress-relaxation of aged human calf muscle-tendon units. Eur J Appl Physiol 96:379–388. doi:10.1007/s00421-005-0091-7
Hirata K, Miyamoto-Mikami E, Kanehisa H, Miyamoto N (2016) Muscle-specific acute changes in passive stiffness of human triceps surae after stretching. Eur J Appl Physiol 116:911–918. doi:10.1007/s00421-016-3349-3
Hug F, Lacourpaille L, Maïsetti O, Nordez A (2013) Slack length of gastrocnemius medialis and Achilles tendon occurs at different ankle angles. J Biomech 46:2534–2538. doi:10.1016/j.jbiomech.2013.07.015
Kay AD, Blazevich AJ (2009) Isometric contractions reduce plantar flexor moment, Achilles tendon stiffness, and neuromuscular activity but remove the subsequent effects of stretch. J Appl Physiol 107:1181–1189. doi:10.1152/japplphysiol.00281.2009
Kay AD, Blazevich AJ (2012) Effect of acute static stretch on maximal muscle performance: a systematic review. Med Sci Sports Exerc 44:154–164. doi:10.1249/MSS.0b013e318225cb27
Kay AD, Husbands-Beasley J, Blazevich AJ (2015) Effects of Contract-Relax, Static Stretch, and Isometric Contractions on Muscle-Tendon Mechanics. Med Sci Sports Exerc 47:2181–2190. doi:10.1249/MSS.0000000000000632
Klee A, Andreas K, Klaus W (2002) Stretch and contraction specific changes in passive torque in human M. Rectus Femoris. Eur J Sport Sci 2:1–10. doi:10.1080/17461391.2002.10142576
Koo TK, Guo J-Y, Cohen JH, Parker KJ (2013) Relationship between shear elastic modulus and passive muscle force: an ex vivo study. J Biomech 46:2053–2059. doi:10.1016/j.jbiomech.2013.05.016
Lacourpaille L, Nordez A, Hug F et al (2014) Time-course effect of exercise-induced muscle damage on localized muscle mechanical properties assessed using elastography. Acta Physiol (Oxf) 211:135–146. doi:10.1111/apha.12272
Magnusson SP, Simonsen EB, Aagaard P et al (1995) Contraction specific changes in passive torque in human skeletal muscle. Acta Physiol Scand 155:377–386. doi:10.1111/j.1748-1716.1995.tb09987.x
Magnusson SP, Simonsen EB, Aagaard P, Kjaer M (1996) Biomechanical responses to repeated stretches in human hamstring muscle in vivo. Am J Sports Med 24:622–628
Maïsetti O, Hug F, Bouillard K, Nordez A (2012) Characterization of passive elastic properties of the human medial gastrocnemius muscle belly using supersonic shear imaging. J Biomech 45:978–984. doi:10.1016/j.jbiomech.2012.01.009
McHugh MP, Cosgrave CH (2010) To stretch or not to stretch: the role of stretching in injury prevention and performance. Scand J Med Sci Sports 20:169–181. doi:10.1111/j.1600-0838.2009.01058.x
McHugh MP, Connolly DA, Eston RG et al (1999) The role of passive muscle stiffness in symptoms of exercise-induced muscle damage. Am J Sports Med 27:594–599
McNair PJ, Dombroski EW, Hewson DJ, Stanley SN (2001) Stretching at the ankle joint: viscoelastic responses to holds and continuous passive motion. Med Sci Sports Exerc 33:354–358
Mizuno T, Matsumoto M, Umemura Y (2013) Decrements in stiffness are restored within 10 min. Int J Sports Med 34:484–490. doi:10.1055/s-0032-1327655
Mizuno T, Matsumoto M, Umemura Y (2014) Stretching-induced deficit of maximal isometric torque is restored within 10 minutes. J Strength Cond Res 28:147–153. doi:10.1519/JSC.0b013e3182964220
Nordez A, Cornu C, McNair P (2006) Acute effects of static stretching on passive stiffness of the hamstring muscles calculated using different mathematical models. Clin Biomech 21:755–760. doi:10.1016/j.clinbiomech.2006.03.005
Nordez A, Casari P, Cornu C (2008) Effects of stretching velocity on passive resistance developed by the knee musculo-articular complex: contributions of frictional and viscoelastic behaviours. Eur J Appl Physiol 103:243–250. doi:10.1007/s00421-008-0695-9
Nordez A, McNair PJ, Casari P, Cornu C (2010) Static and cyclic stretching: their different effects on the passive torque-angle curve. J Sci Med Sport 13:156–160. doi:10.1016/j.jsams.2009.02.003
Riemann BL, DeMont RG, Ryu K, Lephart SM (2001) The effects of sex, joint angle, and the gastrocnemius muscle on passive ankle joint complex stiffness. J Athl Train 36:369–375
Schleip R, Duerselen L, Vleeming A et al (2012) Strain hardening of fascia: static stretching of dense fibrous connective tissues can induce a temporary stiffness increase accompanied by enhanced matrix hydration. J Bodyw Mov Ther 16:94–100. doi:10.1016/j.jbmt.2011.09.003
Simic L, Sarabon N, Markovic G (2013) Does pre-exercise static stretching inhibit maximal muscular performance? A meta-analytical review. Scand J Med Sci Sports 23:131–148. doi:10.1111/j.1600-0838.2012.01444.x
Smith LL, Brunetz MH, Chenier TC et al (1993) The effects of static and ballistic stretching on delayed onset muscle soreness and creatine kinase. Res Q Exerc Sport 64:103–107. doi:10.1080/02701367.1993.10608784
Taniguchi K, Shinohara M, Nozaki S, Katayose M (2015) Acute decrease in the stiffness of resting muscle belly due to static stretching. Scand J Med Sci Sports 25:32–40. doi:10.1111/sms.12146
Whitehead NP, Weerakkody NS, Gregory JE et al (2001) Changes in passive tension of muscle in humans and animals after eccentric exercise. J Physiol 533:593–604
Whitehead NP, Morgan DL, Gregory JE, Proske U (2003) Rises in whole muscle passive tension of mammalian muscle after eccentric contractions at different lengths. J Appl Physiol 95:1224–1234. doi:10.1152/japplphysiol.00163.2003
Acknowledgments
The authors declare no conflict of interest, and are grateful for the support of the Portuguese Scientific Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Olivier Seynnes.
Rights and permissions
About this article
Cite this article
Freitas, S.R., Andrade, R.J., Nordez, A. et al. Acute muscle and joint mechanical responses following a high-intensity stretching protocol. Eur J Appl Physiol 116, 1519–1526 (2016). https://doi.org/10.1007/s00421-016-3410-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-016-3410-2