Abstract
This study evaluated exercise modality [i.e. self-paced (SP) or fixed-intensity (FI) exercise] as a modulator of body temperature regulation under uncompensable heat stress. Eight well-trained male cyclists completed (work-matched) FI and SP cycling exercise bouts in a hot (40.6 ± 0.2°C) and dry (relative humidity 23 ± 3%) environment estimated to elicit 70% of \( \dot{V} \)O2max. Exercise intensity (i.e. power output) decreased over time in SP, which resulted in longer exercise duration (FI 20.3 ± 3.4 min, SP 23.2 ± 4.1 min). According to the heat strain index, the modification of exercise intensity in SP improved the compensability of the thermal environment which, relative to FI, was likely a result of the reductions in metabolic heat production (i.e. \( \dot{V} \)O2). Consequently, the rate of rise in core body temperature was higher in FI (0.108 ± 0.020°C/min) than in SP (0.082 ± 0.016°C/min). Interestingly, cardiac output, stroke volume, and heart rate during exercise were independent of exercise modality. However, core body temperature (FI 39.4 ± 0.3°C, SP 39.1 ± 0.4°C), blood lactate (FI 2.9 ± 0.8 mmol/L, SP 2.3 ± 0.7 mmol/L), perceived exertion (FI 18 ± 2, SP 16 ± 2), and physiological strain (FI 9.1 ± 0.9, SP 8.3 ± 1.1) were all higher in FI compared to SP at exhaustion/completion. These findings indicate that, when exercise is SP, behavioral modification of metabolic heat production improves the compensability of the thermal environment and reduces thermoregulatory strain. Therefore, under uncompensable heat stress, exercise modality modulates body temperature regulation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Body temperature is controlled by the regulation of autonomic and/or behavioral responses that modulate body heat exchange (Romanovsky 2007). According to the heat balance equation, when heat gain outweighs heat loss, body heat storage increases, elevating body temperatures. During fixed-intensity (FI) (constant power) exercise, metabolic heat production is constant, and therefore, heat loss is limited solely to autonomic responses. Consequently, core body temperature rises until heat balance is achieved as indicated by a ‘plateau’ in core temperature (Nielsen and Nielsen 1962; Saltin and Hermansen 1966).
During FI exercise when exposed to uncompensable heat stress, the attainment of heat balance is impossible (Cheung et al. 2000) with core temperature rising linearly as a function of the metabolic and ambient heat loads (Robinson 1963) until exhaustion occurs (Cheung 2007; Cheung and Sleivert 2004). This inability to continue exercising in a hot environment, i.e. heat exhaustion (Armstrong et al. 2007), is directly associated with the failure to achieve heat balance as heat exhaustion is accompanied by high core temperatures (Gonzalez-Alonso et al. 1999) and an increased challenge for the cardiovascular system to simultaneously meet the demands for both the working musculature and temperature regulation (Rowell 1974). Thus, thermal and cardiovascular regulatory processes have been included as important factors mediating the multi-system development of heat exhaustion (Armstrong et al. 2007; Cheung 2007; Cheung and Sleivert 2004; Gonzalez-Alonso et al. 2008; Nybo 2008).
In contrast, during self-paced (SP) exercise in the heat, the thermal compensability of the environment is likely improved by the exerciser by reducing exercise intensity and thus metabolic heat production (i.e. exercise work-rate; Tatterson et al. 2000; Tucker et al. 2004). This response has been suggested to occur primarily to control the rise in core body temperature (Schlader et al. 2010a), but as a consequence exercise performance is reduced relative to more moderate conditions (Tatterson et al. 2000; Tucker et al. 2004). Notably, under SP conditions potentially critical levels of physiological stress are anticipated (Marino 2004), and therefore, heat exhaustion is usually avoided (Brake and Bates 2002).
In order to facilitate heat loss, autonomic responses are likely maximized as exercise progresses in uncompensable heat. Hence, FI and SP exercise modalities (e.g. Tucker 2008) must modulate body temperature regulation differently, for example SP exercise seemingly utilizes behavior in order to improve the compensability of the thermal environment (Schlader et al. 2010a), while FI exercise continues until an apparent (multi-system) physiological ‘threshold’ is reached and exercise can no longer be sustained (Cheung 2007; Cheung and Sleivert 2004; Nybo 2008). Thus, the two exercise modalities evaluate different physiological processes, and accordingly, the theories underlying the observed decrements in exercise performance/duration (Galloway and Maughan 1997; Parkin et al. 1999; Tatterson et al. 2000; Tucker et al. 2004) have been built up in opposition (Cheung 2007; Tucker 2008). However, despite suggestions that future research should incorporate both modalities (Cheung 2007; Cheung and Flouris 2009), an established relationship between the physiological outcomes obtained from both FI and SP exercise remains elusive.
The purpose of this study was to evaluate exercise modality as a modulator of body temperature regulation, thus establishing a formal relationship between FI and SP exercise in the heat. This was achieved by first having our subjects complete a bout of FI exercise to voluntary exhaustion in the heat and, second, having them complete the same amount of work during the FI exercise, but where intensity was freely adjustable. It was hypothesized that the freedom to behaviorally thermoregulate during SP exercise would increase exercise duration and improve the compensability of the thermal environment. Additionally, given the expected decreases in exercise intensity and that heat exhaustion will be effectively avoided during SP exercise, we further hypothesized that the magnitude of change in the cardiovascular and thermoregulatory responses would be lower during and upon completion of SP exercise relative to FI exercise to voluntary exhaustion.
Methods
Experimental overview
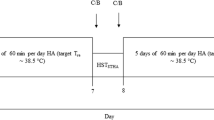
Eight well-trained male cyclists completed a FI and a SP cycling bout under uncompensable heat stress. During FI subjects rode until volitional exhaustion, while during SP the subjects completed the same amount of work as completed during FI, but exercise intensity (power output) was freely adjustable. All trials took place at the same time of day, and there were 7 days between each trial.
Subjects
Eight healthy and well-trained male cyclists agreed to participate in the study. The subject’s characteristics were (mean ± SD) age 34 ± 9 years, height 1.8 ± 0.1 m, weight 70.1 ± 6.2 kg, body surface area (BSA) 1.87 ± 0.11 m2, maximal oxygen uptake (\( \dot{V} \)O2max) 4.94 ± 0.41 l min−1, and percent body fat 10.6 ± 3.3%. For a complete description of the characteristics of these subjects please refer to Schlader et al. (2010b). Each subject was fully informed of the experimental procedures and possible risks before giving informed, written, consent. The protocol was approved by the University Human Ethics Committee and performed according to the Declaration of Helsinki. All subjects had participated in previous studies in our laboratory utilizing both FI and SP exercise protocols, and thus, they were fully familiarized with all experimental procedures.
Preliminary session
Approximately 1 week before the first experimental trial, subjects reported to the laboratory for anthropometric measurements, and submaximal and \( \dot{V} \)O2max testing. This session was conducted in a moderate environment (20.6 ± 2.0°C), with the exercise tests taking place on an electronically braked cycle ergometer (Lode Excalibur, Groningen, The Netherlands) set in the pedal rate independent mode. All subsequent exercise tests were completed on the same cycle ergometer. The tests first required the subjects to cycle for 6 min at each of four consecutive submaximal power outputs (100, 150, 200, and 250 W). Approximately 5 min after completion of the 250 W stage, during which time the subjects rested, an incremental exercise test (45 W min−1) to volitional exhaustion was completed. A linear relationship between the mean rate of oxygen consumption during the last 2 min of each submaximal stage and power output was determined. This equation was used to calculate a power output which would elicit 70% of \( \dot{V} \)O2max for each subject and was later used to construct the exercise protocols.
Experimental protocol
Approximately 7 days following the preliminary session, subjects arrived at the laboratory to complete their FI trial, which was followed 7 days later by their SP trial. All trials took place in a hot and dry environmental chamber: ambient temperature 40.6 ± 0.2°C, relative humidity 23 ± 3%. In order to control for the total amount of work completed, we were unable to randomize the two trials, and therefore, subjects were not blinded to which trial they completed. However, the subjects were completely unaware of the researcher hypotheses and the purpose of the study. Although all testing was completed during the southern hemisphere summer months of January and February, it was assumed that the subjects were not heat acclimatized as outdoor temperature averaged only 19 ± 4°C (FI 20 ± 5°C, SP 19 ± 6°C; P > 0.05).
All subjects arrived at the laboratory for both experimental trials having refrained from strenuous exercise, alcohol, caffeine, and tobacco for a period of 24 h. To minimize variations in pre-exercise muscle glycogen content and hydration status, subjects were required to complete a 48 h diet and activity log before FI and were instructed to follow the same diet and activities prior to SP. Further activity and diet standardization was achieved via a standardized training ride ~60 min in duration at a moderate intensity (74 ± 7% heart rate max) the day before each trial (24 h) and by the subjects consuming a standardized meal 3 h prior to the experimental trial. A euhydrated state was ensured by instructing the subjects to drink a pre-measured bolus of water (5 ml kg−1 of bodyweight) 2 h prior to each trial and was confirmed by nude bodyweight within 200 g, core temperature within 0.2°C, and resting heart rate within six beats of the values in the previous trials (Montain and Coyle 1992). Subjects were not allowed to drink during the trials and wore only cycling shorts and shoes.
Prior to arrival the subjects ingested a core body temperature pill, and upon arrival at the laboratory, a cannula was inserted into an antecubital vein and kept patent with saline. The subjects then voided, and nude bodyweight was measured. A heart rate monitor and skin thermistors were applied, and the subjects then entered the environmental chamber for completion of the experimental trial. Upon completion of exercise, the subjects were promptly removed from the chamber, and the heart rate monitor and skin thermistors were removed. Nude bodyweight was measured following towel drying, after which the cannula was removed. The duration of the procedures from the initial nude bodyweight to the final nude bodyweight was ~60 min.
Experimental procedures
Cycling exercise
A standard 5 min warm-up (at 150 W) in the environmental chamber was followed immediately by either FI or SP. In both trials, subjects were verbally encouraged at random intervals by the same researcher and were unaware of elapsed time, heart rate, or (in FI only) work completed. During SP, subjects were informed of the amount of work completed at intervals that corresponded with the taking of measurements (see below). Exercise continued until one of the following conditions occurred: (1) voluntary exhaustion, (2) core body temperature reached 40.0°C, (3) the subject displayed signs and/or symptoms of heat stroke (Armstrong et al. 2007), or (4) (in SP only) the required amount of work had been completed.
FI required subjects to cycle at a FI calculated to elicit 70% (70.8 ± 6.4%) of \( \dot{V} \)O2max until one of the previously mentioned conditions had been met. During FI, power output was independent of cycling cadence. SP required subjects to complete as quickly as possible the same amount of work as completed in FI. However, during SP, power output was proportionate to the cadence in a relationship derived from the submaximal and \( \dot{V} \)O2max tests.
Immediately prior to commencement, during, and upon completion of exercise, exercise time (SP only), heart rate (HR), mean skin temperature (T Sk), core body temperature (T C), thermal comfort, thermal sensation, and perceived exertion were recorded every 3 min during FI and at the same amount of work completed during SP (irrespective of time). Cardiovascular and metabolic responses were measured at the first recording interval (FI: at 3 min; SP: after 15 ± 4% of the required work had been completed) and immediately prior to exhaustion (FI)/completion (SP; 100% of the required work had been completed). Blood glucose and lactate were measured prior to exhaustion/completion, while percent changes in plasma volume and whole body sweat rate were determined pre- to post-exercise. In SP, power output was calculated from the amount of time it took to complete the required amount of work.
Measurements
The subjects’ height and weight were measured using a stadiometer (Seca, Bonn, Germany; accurate to 0.1 cm) and scale (Jadever, Taiwan; accurate to 0.01 kg), from which BSA was estimated (Dubois and Dubois 1916). Seven site skinfold thickness was determined using a Harpenden Skinfold Caliper (Baty International, West Sussex, UK) at the chest, axilla, triceps, subscapula, abdomen, suprailliac, and thigh, and subsequently, percent body fat (Siri 1961) was estimated from body density (Jackson and Pollock 1978).
The HR was monitored using a Polar heart rate monitor (Polar Vantage XL, Polar Electro). Cardiac output (\( \dot{Q} \)) was estimated via the CO2 rebreathing method described by DeFares (1958), and used previously in our laboratory (Schlader et al. 2010b). End-tidal CO2 (PETCO2) data were measured (O2/CO2 Gas Analyzer, ADInstruments, Australia), acquired (PowerLab, ADInstruments, Australia), and displayed (Chart5, ADInstruments, Australia) continuously throughout the entire procedure. Differences between PETCO2 and venous and arterial PCO2 were corrected as demonstrated previously by Paterson and Cunningham (1976) and Jones et al. (1979). The CO2 content difference was calculated according to McHardy (1967). Stroke volume (SV) was calculated from the Fick equation. Blood pressure was measured in duplicate manually using a stethoscope and a sphygmomanometer over the right brachial artery. All blood pressure measurements were made by the same experienced operator. Mean arterial pressure (MAP, in mmHg) was estimated by auscultation of the systolic (SBP) and diastolic blood pressure (DBP) according to the following equation: \( {\text{MAP}} = {\text{DBP}} + 1/3({\text{SBP}} - {\text{DBP}}). \)
Expired gases were collected for 1 min via standard Douglas bags. The expired gases were analyzed for CO2 and O2 concentrations (AEI Technologies, USA) and volume (dry gas meter, Harvard, UK), and values converted to STPD. Rates of O2 uptake (\( \dot{V} \)O2), CO2 elimination (\( \dot{V} \)CO2), respiratory exchange ratio (RER), and expiratory minute ventilation (\( \dot{V} \)E) were calculated.
According to previously reported guidelines (Byrne and Lim 2007), subjects ingested a calibrated telemetric core body temperature sensor (CorTemp™, Palmetto FL, USA; accurate to 0.05°C) 6 h prior to their experimental trial, which served as an indication of T C. Four calibrated surface thermistors (Grant Instruments Ltd., Cambridgeshire, UK; accurate to 0.02°C) were secured in place with Transpore Surgical Tape (3 M Healthcare, St. Paul, Minnesota, USA) to the chest, thigh, leg, and arm on the right side of the body for determination of T Sk (Ramanathan 1964). In the present study, increased thermoregulatory strain is any combination of an increased rate of rise of T C or higher T C at exhaustion/completion.
The modified physiological strain index (PSI) was used as an indicator of the combined thermoregulatory (T C) and cardiovascular (HR) strain (Moran et al. 1998) and has been used previously in comparing the physiological outcomes from FI and SP exercise (Schlader et al. 2010a). PSI was calculated as follows: PSI = 5(T Ct − T Co)/(40.0 − T Co) + 5(HRt − HRo)/(HRmax − HRo), where T Co resting core temperature (°C); T Ct core temperature at exhaustion (FI) or completion (SP, °C); HRo resting heart rate (bpm); HRt heart rate at exhaustion (FI) or completion (SP; bpm); HRmax max heart rate measured during the \( \dot{V} \)O2max test. The PSI was modified as shown previously (Schlader et al. 2010a).
The heat strain index (HSI) served as an estimate of the thermal compensability of the environment, where an HSI > 1.0 indicates uncompensable heat stress, and an HSI < 1.0 indicates compensable heat stress (Cheung et al. 2000). The HSI was calculated as the ratio of the required evaporative cooling for heat balance (E req, in W/m2) and the maximal evaporative capacity of the environment (E max, in W/m2) (Cheung et al. 2000). E req was calculated in the following manner (Cheung et al. 2000): E req = M − W ± (C + R) ± (C res − E res), where M is metabolic heat production (in W/m2), calculated as follows (Kenney 1998): \( M = {{(352(0.23{\text{RER}} + 0.77)\dot{V}{\text{O}}_{2} )} \mathord{\left/ {\vphantom {{(352(0.23{\text{RER}} + 0.77)\dot{V}{\text{O}}_{2} )} {\text{BSA}}}} \right. \kern-\nulldelimiterspace} {\text{BSA}}}. \) W is heat transfer from the generation of external work (in W/m2). C + R is heat transfer from convection (C, in W/m2) and radiation (R, in W/m2), calculated as the sum of: C = h c(T Sk − T A) (Kerslake 1972) and R = 4.7(T A − T Sk) (Kenney 1998), where h c is the convective heat transfer coefficient (in W/m2/°C) (Kerslake 1972) and T A is the ambient temperature (in °C). C res + E res is the respiratory conductive (C res) and evaporative (E res) heat transfer and was calculated as follows (Kenney 1998): C res + E res = [0.0012M (34 − T A)] + [0.0023M(44 − P A)], where P A is ambient vapor pressure (in kPa). E max was calculated according to the following equation (Kerslake 1972): E max = LR × h c × (P Sk − P A), where LR is the Lewis relation (16.5°C/kPa), and P Sk is the saturated vapor pressure at the skin (in kPa). Additionally, evaporative heat loss (E, in W/m2) was estimated according to the following equation (Kerslake 1972): \( E = (P_{\text{Sk}} - P_{\text{A}} ) \times \sqrt v \times 124, \) where v is air velocity (0.5 m/s).
Ratings of perceived exertion (RPE) were measured on the 15-grade Borg rating of perceived exertion scale (from 6 to 20; Borg 1970) and was administered according to the recommendations set forth by Noble and Robertson (1996). Thermal comfort and thermal sensation were determined on modified four [from 1 (comfortable) to 4 (very uncomfortable)] and seven [from 1 (cold) to 7 (hot)] point scales (Gagge et al. 1967).
After correcting for respiratory and metabolic water losses (Mitchell et al. 1972), changes in nude bodyweight were used to estimate whole body sweat rate.
Whole blood lactate and glucose were assayed using a YSI lactate analyzer (Yellow Springs, OH, USA) and an Accu-Check Advantage glucose meter (Mannheim, Germany). Whole blood hemoglobin and hematocrit were measured via an automated analyzer (ABL800 FLEX, Radiometer, Denmark). Percentage changes in plasma volume were calculated according to previously described methods (Dill and Costill 1974).
Statistical analysis
Each subject completed a different amount of work, and therefore, it was not possible to statistically compare ‘raw’ power output responses over time and between trials. Accordingly, for each subject during each trial, power outputs during SP and FI were standardized and compared by analyzing the log transformed relationship between time (dependent variable) and the cumulative work completed (independent variable) for each subject during each trial. Both time and cumulative work completed were log transformed, and these data were fitted with a significant (P < 0.001; R 2 = 0.99 ± 0.00) linear regression model (y = y0 + ax). For all subjects, the mean y-intercept (Y0) and slopes (a) in the two conditions were compared via paired t tests. This analysis transformed the changes in power output during SP linear allowing for statistical comparison with FI. These data were analyzed using SigmaPlot statistical software (v. 10.0, Systat Software Inc., Chicago, IL, USA).
Mean differences between trials were compared via paired t tests. To identify any changes in the cardiovascular, metabolic, and heat exchange variables over time during exercise, a two-way (trial × time) repeated measures analysis of variance (ANOVA) was also conducted. These data were assessed for approximation to a normal distribution and sphericity, and no corrections were necessary. These data were analyzed using SPSS statistical software (V. 17, Chicago, IL, USA). A priori statistical significance was set at P < 0.05. All data are reported as mean ± SD unless noted otherwise.
Notably, given the large effect size (>0.9) of exercise modality on our primary independent variables (i.e. power output and time), an N = 8 was deemed sufficient to identify differences between trials and over time via both repeated measures ANOVA and paired t tests (Tran 1997). Furthermore, there was a large discrepancy in the amount of work completed during FI and therefore SP (range 232–312 kJ). Accordingly, the 3 min fixed measurement period corresponded to a higher or lower percentage of work completed per subject, with each subject ultimately having a different number of data points depending upon their time to exhaustion in FI. Consequently, it was not possible to analyze or display mean data during the exercise bout other than at the start (i.e. at 15 ± 4% of the required work completed) and at completion/exhaustion (i.e. at 100% of the required work completed). Figure 2 demonstrates the typical observed responses of selected variables during both trials.
Results
Exercise duration and power output
Seven (of eight) subjects exercised until voluntary exhaustion in FI, while exercise was stopped for one subject upon the attainment of a T C of 40.0°C. In SP, all subjects successfully completed the required amount of work. In both trials, subjects completed 281 ± 32 kJ of work; however, SP took ~12% longer (P < 0.001) than FI (Table 1). Accordingly, mean power output was higher (P < 0.001) in FI (234 ± 25 W) than in SP (207 ± 23 W; Fig. 1). Relative to FI, power output slowed (P < 0.05) over time in SP (Table 2).
Temperature regulation and heat exchange
Pre-exercise T C was similar (P > 0.05) in FI (37.2 ± 0.2°C) and SP (37.2 ± 0.2°C). The rate of rise in T C during exercise was higher (P < 0.01) in FI (0.108 ± 0.020°C/min) than in SP (0.082 ± 0.016°C/min), with T C being higher (P < 0.05) at exhaustion in FI (Table 2). A typical T C response during both trials is presented in Fig. 2. Average T Sk was similar (P > 0.05) in both FI (37.4 ± 0.3°C) and SP (37.4 ± 0.3°C). The compensability of the thermal environment and body heat exchange are presented in Table 3 and Fig. 3.
Modes of heat exchange (mean ± SE) during (at 15% of total work completed) and upon exhaustion/completion (100% of total work completed) of fixed-intensity (FI) and self-paced (SP) exercises. M metabolic heat production, W heat transfer from the generation of external work, E heat transfer from evaporation, C + R the sum of heat transfer from convection (C) and radiation (R), C res + E res the sum of respiratory conductive (C res) and evaporative (E res) heat transfer. *Significantly different than SP (P < 0.05); †Significantly different than 15% (P < 0.05)
Cardiovascular responses and physiological strain
Pre-exercise HR was similar (P > 0.05) in FI (71 ± 8 bpm) and SP (69 ± 12 bpm). MAP, \( \dot{Q} \), SV, and HR data during exercise are presented in Fig. 4. PSI was higher (P < 0.01) at the end of SP (Table 1). Typical responses during each trial are presented in Fig. 2.
Mean arterial pressure (MAP), cardiac output, stroke volume, and heart rate (mean ± SE) during (at 15% of total work completed) and upon completion (100% of total work completed) of fixed-intensity (FI) and self-paced (SP) exercises. *Significantly different than SP (P < 0.01); †significantly different than 15% (P < 0.05)
Metabolic and fluid balance measures
\( \dot{V} \)O2 was similar (P > 0.05) upon completion of 15% of the required work (FI 3.50 ± 0.45 L/min, SP 3.28 ± 0.47 L/min), but was higher (P < 0.05) in FI upon exhaustion/completion (FI 3.39 ± 0.50 L/min: SP 3.11 ± 0.41 L/min). Blood glucose and lactate at exhaustion/completion are presented in Table 1. Percent changes in plasma volume (FI −8.9 ± 6.9%, SP −6.6 ± 4.4%) and whole body sweat rates (FI 13.5 ± 1.3 g/min; SP 12.7 ± 2.7 g/min) were similar (P > 0.05) between the two trials.
Perceptual measures
The RPE was higher (P < 0.05) upon exhaustion/completion in FI (Table 1). In both conditions, completion/exhaustion was unanimously (P > 0.05) regarded as very uncomfortable (4.0 ± 0.0) and hot (7.0 ± 0.0).
Discussion
This study is the first to formally evaluate exercise modality as a modulator of body temperature regulation in the heat. This was achieved by directly comparing the thermoregulatory, cardiovascular, and heat exchange responses during work-matched FI and SP exercise in uncompensable heat. In support of our hypotheses, we found that voluntary reductions in power output during SP exercise improved thermal compensability and extended exercise duration. Additionally, thermoregulatory strain was higher during and upon completion of FI exercise. Surprisingly, however, we did not observe differences in cardiovascular responses between exercise modalities.
Relative to FI exercise, in the current study, we found that when exercise was SP power output was gradually reduced with exercise duration (Fig. 1). This was demonstrated by the log transformed relationship between time and cumulative work completed. This analysis showed that the power outputs were similar upon exercise commencement (i.e. the mean y-intercepts were similar), yet became reduced over time during the SP trial (i.e. the mean slope was lower than that during the FI trial; Table 2). Ultimately, this reduction in power output resulted in a longer exercise duration in the SP trial (Table 1). Previously, exercise intensity has generally been found not to differ between SP and FI trials undertaken in a moderate thermal environment (Billat et al. 2001, 2006; Lander et al. 2009). In fact, there is some evidence that exercise performance, and therefore intensity, is actually improved (faster) when exercise is SP compared to that during FI exercise (Garcin et al. 2008). This discrepancy is likely due to the uncompensable heat stress in which our trials were conducted.
Generally, in moderate thermal conditions, FI exercise has previously been found to be more physiologically stressful than SP exercise despite a similar exercise intensity (Billat et al. 2006; Lander et al. 2009). These studies indicated that a similar exercise intensity elicited significant reductions in mean oxygen uptake and heart rate (Billat et al. 2006), core body temperature (Lander et al. 2009), neuromuscular activity (Lander et al. 2009), and post-exercise blood lactate concentrations (Billat et al. 2006; Lander et al. 2009). These findings are not unanimous however (Binkley et al. 2002). That the current study found an increased rate of rise in core temperature during exercise and higher core temperatures, blood lactate concentrations, and perception of effort upon exhaustion/completion (Table 1) supports the contention that SP exercise is less physiologically demanding than FI exercise.
The current study has demonstrated that modification of body heat exchange (Fig. 3) and the corresponding improvement in thermal compensability (Table 3) successfully allowed the same exercise task to be completed with less thermoregulatory strain (Table 1; Fig. 2). Given that evaporative heat loss was similar and that conductive and radiative heat gain changed similarly between trials (Fig. 3), the compensability improvement was likely a direct result of the observed reductions in metabolic heat production (Fig. 3). Differences in respiratory conductive and evaporative heat transfer were observed upon completion/exhaustion (Fig. 3). However, these are almost certainly driven by the observed differences in oxygen uptake and therefore metabolic heat production. It is important to note that while the compensability of the environment was improved at the end of the SP trial, it was still considered uncompensable (Table 3). This observation is likely due to the methodology utilized. For instance, the subjects were instructed to successfully complete the given amount of work as quickly as possible; therefore, it is likely they compromised heat gain for exercise performance, thus making true thermal compensability impossible during the relatively short period of exercise.
It is notable that heat balance was not attained (i.e. M − W ≠ C + R + E + C res + E res) in either the SP or FI trials. It has been suggested previously that, when given the freedom to self-pace, subjects decrease their exercise intensity in order to ultimately reduce the rate of body heat storage to zero (Tucker et al. 2006). Given the reduced rate of rise in core temperature and, upon completion/exhaustion, a lower core temperature (Table 1) and rate of metabolic heat production (Fig. 3), the present study certainly supports that the rate of heat storage is reduced when SP exercise is undertaken in the heat. Nevertheless, given that it was not the intent of this study to identify the physiological signals initiating reductions in intensity during SP exercise, the current study does not allow for further speculation.
Unexpectedly, we found no differences between exercise modalities for the cardiovascular responses measured during the initial stages of exercise or upon completion/exhaustion (Fig. 4). Nevertheless, given that reductions in power output (Fig. 1; Table 2), metabolic heat production (Fig. 3), and oxygen uptake were observed during SP exercise, it would be naïve to conclude that there were not slight differences in oxygen delivery. Therefore, it is highly likely that our CO2 rebreathing methodology (Defares 1958) used to estimate cardiac output may not have been sensitive enough to observe these differences. Besides this, the observed increase in heart rate and decreases in stroke volume and cardiac output (Fig. 4) are typical characteristics of prolonged exercise in the heat associated with marked dehydration and hyperthermia (Gonzalez-Alonso et al. 1995, 2000).
This study supports exercise modality as a modulator of body temperature regulation. Compared to that during FI exercise, during SP exercise behavioral reductions in metabolic heat production (Fig. 3) apparently reduced the rate of rise in core temperature, allowing the same amount of work to be completed but with less thermoregulatory strain (Table 1). These findings support the hypothesis of a thermoregulatory continuum (Schlader et al. 2010a). For example, in the present paradigm, behavior was preferentially elicited (i.e. SP exercise); however, during instances where behavior is restricted (i.e. FI exercise), when the signaling inputs to behave are ignored, or when behavior is inadequate, heat exhaustion ensues. It is only upon the failure of both behavior and exhaustion that temperature regulation may be lost, homeostasis compromised, and exertional heat stroke becomes imminent (Schlader et al. 2010a). This is supported by the high incidence of exertional heat stroke when high motivation and external factors (e.g. a drill sergeant, coach, fellow runners, etc.) dictate exercise intensity (Armstrong et al. 2007; Epstein et al. 1999; Shibolet et al. 1976) and a lower incidence when people exercise alone or self-pace (Brake and Bates 2002).
Notably, this behavioral arrangement is similarly reflected by that of the thermoregulatory system at rest (cf. Cheung and Flouris 2009). At rest, behavior is preferentially (Schlader et al. 2010c) initiated by changes in skin temperature, thereby preventing changes in core temperature (Schlader et al. 2009). It is only when behavior is restricted or insufficient that core temperature rises and energy/water consuming autonomic thermoregulatory responses are elicited (Romanovsky 2007). In the circumstance that both behavioral and autonomic thermoregulatory responses are inadequate, core temperature will rise/fall uncontrollably, and body temperature regulation may be lost.
Given that we were unable to work-match and randomize our experimental trials, we must acknowledge that our non-randomized experimental design may have impacted our results. We attempted to minimize these effects by using well familiarized subjects and by blinding them to our hypotheses and the purpose of the study. Ultimately, our non-randomized repeated measures experimental design was deemed the sole manner in which we could test our hypotheses, and therefore, any potential order effect was deemed acceptable. Additionally, given that intestinal temperature is slower to react to changes in body temperature than other indices of core temperature, e.g. esophageal temperature (Byrne and Lim 2007), we acknowledge that our index of core temperature may have underestimated the actual changes. Furthermore, we also acknowledge that our methods of estimating body heat exchange are less sensitive than those of calorimetry; notably, these facilities are rare. We suggest that future investigations in this area account for these limitations. However, we feel the data presented add significantly to the current literature.
Conclusions
The data presented indicate that under uncompensable heat stress exercise modality is a modulator of body temperature regulation. Compared to FI exercise, we have shown that, when given the opportunity to self-pace, reductions in exercise intensity decrease metabolic heat production and thus improve the compensability of the thermal environment. Consequently, when the same exercise task was completed when exercise intensity is either fixed or freely adjustable, exercise was accompanied by lower levels of thermoregulatory strain when exercise was SP. In conclusion, this study presents experimental evidence of a formal relationship between the physiological outcomes observed during and upon completion/exhaustion of SP and FI exercise in the heat.
References
Armstrong LE, Casa DJ, Millard-Stafford M, Moran DS, Pyne SW, Roberts WO (2007) American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med Sci Sports Exerc 39:556–572
Billat VL, Slawinski J, Danel M, Koralsztein JP (2001) Effect of free versus constant pace on performance and oxygen kinetics in running. Med Sci Sports Exerc 33:2082–2088
Billat VL, Wesfreid E, Kapfer C, Koralsztein JP, Meyer Y (2006) Nonlinear dynamics of heart rate and oxygen uptake in exhaustive 10,000 m runs: influence of constant vs freely paced. J Physiol Sci 56:103–111
Binkley HM, Beckett J, Casa DJ, Kleiner DM, Plummer PE (2002) National Athletic Trainers’ Association position statement: exertional heat illnesses. J Athl Train 37:329–343
Borg GA (1970) Perceived exertion as an indicator of somatic stress. Scand J Rehabil Med 2:92–98
Brake DJ, Bates GP (2002) Deep body core temperatures in industrial workers under thermal stress. J Occup Environ Med 44:125–135
Byrne C, Lim CL (2007) The ingestible telemetric body core temperature sensor: a review of validity and exercise applications. Br J Sports Med 41:126–133
Cheung SS (2007) Hyperthermia and voluntary exhaustion: integrating models and future challenges. Appl Physiol Nutr Metab 32:808–817
Cheung SS, Flouris AD (2009) Maximal oxygen uptake regulation as a behavioral mechanism. J Appl Physiol 106:345
Cheung SS, Sleivert GG (2004) Multiple triggers for hyperthermic fatigue and exhaustion. Exerc Sport Sci Rev 32:100–106
Cheung SS, McLellan TM, Tenaglia S (2000) The thermophysiology of uncompensable heat stress. Physiological manipulations and individual characteristics. Sports Med 29:329–359
Defares JG (1958) Determination of PvCO2 from the exponential CO2 rise during rebreathing. J Appl Physiol 13:159–164
Dill DB, Costill DL (1974) Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol 37:247–248
Dubois D, Dubois EF (1916) A formula to estimate approximate surface area if height and weight be known. Arch Intern Med 17:863–871
Epstein Y, Moran DS, Shapiro Y, Sohar E, Shemer J (1999) Exertional heat stroke: a case series. Med Sci Sports Exerc 31:224–228
Gagge AP, Stolwijk JA, Hardy JD (1967) Comfort and thermal sensations and associated physiological responses at various ambient temperatures. Environ Res 1:1–20
Galloway SD, Maughan RJ (1997) Effects of ambient temperature on the capacity to perform prolonged cycle exercise in man. Med Sci Sports Exerc 29:1240–1249
Garcin M, Danel M, Billat V (2008) Perceptual responses in free vs constant pace exercise. Int J Sports Med 29:453–459
Gonzalez-Alonso J, Morarodriguez R, Below PR, Coyle EF (1995) Dehydration reduces cardiac-output and increases systemic and cutaneous vascular-resistance during exercise. J Appl Physiol 79:1487–1496
Gonzalez-Alonso J, Teller C, Andersen SL, Jensen FB, Hyldig T, Nielsen B (1999) Influence of body temperature on the development of fatigue during prolonged exercise in the heat. J Appl Physiol 86:1032–1039
Gonzalez-Alonso J, Mora-Rodriguez R, Coyle EF (2000) Stroke volume during exercise: interaction of environment and hydration. Am J Physiol Heart Circ Physiol 278:H321–H330
Gonzalez-Alonso J, Crandall CG, Johnson JA (2008) The cardiovascular challenge of exercising in the heat. J Physiol 586:45–53
Jackson AS, Pollock ML (1978) Generalized equations for predicting body density of men. Br J Nutr 40:497–504
Jones NL, Robertson DG, Kane JW (1979) Difference between end-tidal and arterial PCO2 in exercise. J Appl Physiol 47:954–960
Kenney WL (1998) Heat flux and storage in hot environments. Int J Sports Med 19(Suppl 2):S92–S95
Kerslake DM (1972) The stress of hot environments. Cambridge University Press, London
Lander PJ, Butterly RJ, Edwards AM (2009) Self-paced exercise is less physically challenging than enforced constant pace exercise of the same intensity: influence of complex central metabolic control. Br J Sports Med 43:789–795
Marino FE (2004) Anticipatory regulation and avoidance of catastrophe during exercise-induced hyperthermia. Comp Biochem Physiol B Biochem Mol Biol 139:561–569
Mchardy GJR (1967) Relationship between differences in pressure and content of carbon dioxide in arterial and venous blood. Clin Sci 32:299–309
Mitchell JW, Nadel ER, Stolwijk JA (1972) Respiratory weight losses during exercise. J Appl Physiol 32:474–476
Montain SJ, Coyle EF (1992) Influence of graded dehydration on hyperthermia and cardiovascular drift during exercise. J Appl Physiol 73:1340–1350
Moran DS, Shitzer A, Pandolf KB (1998) A physiological strain index to evaluate heat stress. Am J Physiol 275:R129–R134
Nielsen B, Nielsen M (1962) Body temperature during work at different environmental temperatures. Acta Physiol Scand 56:120–129
Noble BJ, Robertson RJ (1996) The Borg scale: development, administration, and experimental use. In: Noble BJ, Robertson RJ (eds) Perceived exertion. Human Kinetics, Champaign, pp 59–92
Nybo L (2008) Hyperthermia and fatigue. J Appl Physiol 104:871–878
Parkin JM, Carey MF, Zhao S, Febbraio MA (1999) Effect of ambient temperature on human skeletal muscle metabolism during fatiguing submaximal exercise. J Appl Physiol 86:902–908
Paterson DH, Cunningham DA (1976) Comparison of methods to calculate cardiac-output using CO2 rebreathing method. Eur J Appl Physiol 35:223–230
Ramanathan NL (1964) A new weighting system for mean surface temperature of the human body. J Appl Physiol 19:531–533
Robinson S (1963) Temperature regulation in exercise. Pediatrics 32:S691–S702
Romanovsky AA (2007) Thermoregulation: some concepts have changed. Functional architecture of the thermoregulatory system. Am J Physiol Regul Integr Comp Physiol 292:R37–R46
Rowell LB (1974) Human cardiovascular adjustments to exercise and thermal-stress. Physiol Rev 54:75–159
Saltin B, Hermansen L (1966) Esophageal, rectal, and muscle temperature during exercise. J Appl Physiol 21:1757–1762
Schlader ZJ, Prange HD, Mickleborough TD, Stager JM (2009) Characteristics of the control of human thermoregulatory behavior. Physiol Behav 98:557–562
Schlader ZJ, Stannard SR, Mundel T (2010a) Exercise and heat stress: performance, fatigue and exhaustion—a hot topic. Br J Sports Med. doi:10.1136/bjsm.2009.063024
Schlader ZJ, Mundel T, Barnes MJ, Hodges LD (2010b) Peak cardiac power output in healthy, trained males. Clin Physiol Funct Imaging 30:480–484
Schlader ZJ, Stannard SR, Mundel T (2010c) Human thermoregulatory behavior during rest and exercise—a prospective review. Physiol Behav 99:269–275
Shibolet S, Lancaster MC, Danon Y (1976) Heat stroke: a review. Aviat Space Environ Med 47:280–301
Siri WE (1961) Body composition from fluid spaces and density: analysis of methods. In: Brozek J, Henschel A (eds) Techniques for measuring body composition. National Academy of Sciences, National Research Council, Washington, DC, pp 223–243
Tatterson AJ, Hahn AG, Martin DT, Febbraio MA (2000) Effects of heat stress on physiological responses and exercise performance in elite cyclists. J Sci Med Sport 3:186–193
Tran ZV (1997) Estimating sample size in repeated-measures analysis of variance. Meas Phys Educ Exerc Sci 1:89–102
Tucker R (2008) Thermoregulation, fatigue and exercise modality. Med Sport Sci 53:26–38
Tucker R, Rauch L, Harley YX, Noakes TD (2004) Impaired exercise performance in the heat is associated with an anticipatory reduction in skeletal muscle recruitment. Pflugers Arch 448:422–430
Tucker R, Marle T, Lambert EV, Noakes TD (2006) The rate of heat storage mediates an anticipatory reduction in exercise intensity during cycling at a fixed rating of perceived exertion. J Physiol 574:905–915
Acknowledgments
The authors wish to express their gratitude to the subjects who participated in the experiments. This study was funded by a Massey University Postgraduate Research Support Grant. Zachary J. Schlader is funded by a New Zealand International Doctoral Research Scholarship (Education New Zealand).
Conflict of interest
There are no conflicts of interest to report.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by George Havenith.
Rights and permissions
About this article
Cite this article
Schlader, Z.J., Raman, A., Morton, R.H. et al. Exercise modality modulates body temperature regulation during exercise in uncompensable heat stress. Eur J Appl Physiol 111, 757–766 (2011). https://doi.org/10.1007/s00421-010-1692-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-010-1692-3