Abstract
Changes in muscle activation and performance were studied in healthy men in response to 5 weeks of resistance training with or without “eccentric overload”. Subjects, assigned to either weight stack (grp WS; n = 8) or iso-inertial “eccentric overload” flywheel (grp FW; n = 9) knee extensor resistance training, completed 12 sessions of four sets of seven concentric–eccentric actions. Pre- and post-measurements comprised maximal voluntary contraction (MVC), rate of force development (RFD) and training mode-specific force. Root mean square electromyographic (EMGRMS) activity of mm. vastus lateralis and medialis was assessed during MVC and used to normalize EMGRMS for training mode-specific concentric (EMGCON) and eccentric (EMGECC) actions at 90°, 120° and 150° knee joint angles. Grp FW showed greater (p < 0.05) overall normalized angle-specific EMGECC of vastii muscles compared with grp WS. Grp FW showed near maximal normalized EMGCON both pre- and post-training. EMGCON for Grp WS was near maximal only post-training. While RFD was unchanged following training (p > 0.05), MVC and training-specific strength increased (p < 0.05) in both groups. We believe the higher EMGECC activity noted with FW exercise compared to standard weight lifting could be attributed to its unique iso-inertial loading features. Hence, the resulting greater mechanical stress may explain the robust muscle hypertrophy reported earlier in response to flywheel resistance training.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Skeletal muscle inherently possesses greater mechanical efficiency and ability to generate force in lengthening [eccentric (ECC)] than shortening [concentric (CON)] actions (Katz 1939; Komi and Buskirk 1972). Thus, the electromyographic (EMG) amplitude is less while lowering (ECC) than lifting (CON) a given weight (Moritani et al. 1987). In fact, the CON EMG amplitude may be more than twofold higher, inferring markedly less motor unit involvement in ECC than CON actions (Nardone et al. 1989). Such a response is paralleled by much less exercise-induced contrast shift of magnetic resonance images of muscle, following ECC actions (Adams et al. 1992). Thus, the metabolic demand is less when lowering (ECC) than lifting (CON) a given weight (Asmussen 1953; Dudley et al. 1991). EMG amplitude may also be lower in ECC than CON actions executed with maximal effort (Aagaard et al. 2000). Collectively, it appears that more load is placed upon each active muscle fiber in the ECC action. This may at least in part explain the greater hypertrophy reported following chronic resistance training comprising coupled ECC and CON actions or ECC actions compared with CON actions only (Hather et al. 1991; Higbie et al. 1996; Hortobagyi et al. 1996).
Given the greater mechanical efficiency of ECC actions during traditional weight training, no ECC action within a set will call for maximal activation. Additionally, and due to changes in biomechanical levers and muscle length, the ability to overcome the gravitational force of a weight is dictated by the “sticking point” occurring during the CON action. As a result, standard resistance exercise, employing a constant weight, calls for maximal activation only at the “sticking point” of the very last CON repetition resulting in failure to lift a particular weight. In contrast, the inertia of spinning flywheel(s) offers unrestrained resistance throughout the entire CON action and allows for brief episodes of ECC forces exceeding the CON forces, i.e., “ECC overload” (Norrbrand et al. 2008; Tesch et al. 2004). Thus, this exercise modality should evoke maximal muscle activation in the CON, and part of the ECC action, through each repetition of a set. Therefore, flywheel resistance exercise might elicit greater overall EMG amplitude than exercise using gravity-dependent weights. Because it is generally held that high mechanical loading, and in particular during ECC actions, is an essential stimulus to promote muscle hypertrophy (Hather et al. 1991; Higbie et al. 1996; Hortobagyi et al. 1996), features characteristic of flywheel exercise may also offer a more potent hypertrophic exercise stimulus than gravity-dependent weights. In fact, 3 and 5 weeks of flywheel knee extension training resulted in 4–5% (Seynnes et al. 2007) and 6–7% quadriceps hypertrophy (Norrbrand et al. 2008; Seynnes et al. 2007; Tesch et al. 2004). Traditional knee extension training programs, using weight stack, have failed to demonstrate comparable hypertrophy (Jones and Rutherford 1987; Narici et al. 1996; Ploutz et al. 1994).
Thus, muscle activation in the two loading modalities were compared before and after 5 weeks of seated open-chain knee extensor resistance exercise by controlling for, e.g., range of motion, time under tension per repetition and subject positioning.
The present experiments were set up with the assumption that the magnitude of muscle activation during resistance exercise correlates with the potential for skeletal muscle hypertrophy. Hence, this study aimed at comparing muscle activation, assessed by surface EMG, during flywheel and weight stack knee extension exercise. A second objective was to explore whether muscle activation would change following 5 weeks of flywheel or weight stack resistance training and, if so, compare neural training adaptations between exercise modes and in relation to potential changes in isometric and dynamic strength. Given the unique loading features of skeletal muscle prompted by flywheel inertia, we hypothesized that muscle activation during concentric and, more so, eccentric actions would be greater with resistance exercise using flywheel compared with standard weights. In view of readily induced neural adaptations with resistance training programs (Hakkinen et al. 1998; Moritani and deVries 1979), we further hypothesized that both exercise modes would induce marked increases in muscle strength.
Methods
General design
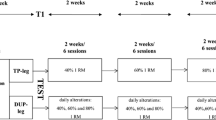
Seventeen healthy men performed 5 weeks of unilateral knee extensor training of the left limb. Subjects were assigned to 12 sessions of either flywheel (grp FW; n = 9) or standard weight stack (grp WS; n = 8) resistance exercise. Each session consisted of four sets of either seven maximal repetitions (grp FW) or seven RM (repetition maximum; grp WS) performed two to three times per week. Strength and root mean square EMG (EMGRMS) of the left limb were determined during maximal voluntary contraction (MVC) and training mode-specific coupled CON–ECC actions pre- and post-training. In addition, the rate of force development (RFD) was assessed. Verbal encouragement and feedback regarding exercise form were given during all training sessions and tests. Written informed consent was obtained from all subjects. The study protocol was approved by the regional ethics committee at the Karolinska Institutet, Stockholm, Sweden. Selected data from the study of grp WS have been reported earlier (Norrbrand et al. 2008).
Subjects
Seventeen healthy men with no or limited experience of lower limb resistance training, and no present or past reported knee pathology, volunteered for this study (Table 1). Eight men were assigned to resistance exercise using a knee extension weight stack (grp WS) apparatus, while nine men trained using a knee extension flywheel (grp FW) device. All subjects complied with the familiarization procedures of at least two sessions, the prescribed training program, and pre- and post-training tests.
Training equipment and resistance exercise protocol
The two devices used in the current study have been described in detail earlier (Norrbrand et al. 2008). Grp FW carried out unilateral training using a seated flywheel device (YoYo® Technology, Stockholm, Sweden (Tesch et al. 2004), equipped with a 4.2-kg flywheel with a moment inertia of 0.11 kg m2. While seated slightly reclined with back support (hip angle 90°), using restraint and grasping the handlebars, the trainee pushes against a crossbar mounted at the distal end of a pivoting moment arm, with the rotational axis aligned with the knee joint. From a starting position of about 80° knee angle (180° = full knee extension), flywheel rotation is initiated through the pull of a strap, anchored to the flywheel shaft and the distal part of the lever arm and looping around its curved cam. While pushing (CON) against the crossbar, the strap unwinds off the flywheel shaft thereby imparting force and energy into the flywheel. Once the pushing CON phase has been completed at about 165° knee angle, the strap rewinds by virtue of the kinetic energy of the flywheel and, thus, pulls the lever arm back. While attempting to resist the force produced by the pull of the rotating flywheel, which recoils the strap, the trainee executes an ECC action. After bringing the wheel to a stop at about 80° knee angle, a subsequent CON action is instantly initiated. During training, knee angle was measured using an electrogoniometer (Berg and Tesch 1994) fixed about the knee joint with a custom-built adjustable Velcro® strap system (Alfatex®, Deinze, Belgium). A miniature compression load cell (Model 276A, K-Toyo, Seoul, Korea) measured force through the pull of the strap. Force and knee joint angle were measured at a sampling rate of 100 Hz, using a Windows™-based data acquisition system (MuscleLab™, Ergotest, Langesund, Norway).
The seated knee extension weight stack machine (World Class®, Stockholm, Sweden), used by grp WS, is equipped with a cam system, which accommodates external torque through the range of motion. Similar to the flywheel device, it has a lever arm with its rotational axis aligned with the knee joint. While seated (90° hip angle; 80° knee angle) and grasping the handlebars, the trainee pushed against a perpendicular shin-padded, adjustable lever crossbar and executed CON actions to about 165° knee angle. The weight was then lowered in a controlled manner and brought to a stop before being raised again. A wire mounted onto the distal part of the lever arm and passing around the cam and two pulley wheels was attached to the weight stack. Weight plates of 5.0, 2.5 and 1.25 kg were used to set and adjust load. Load lifted and lowered, knee joint angle (see above) and vertical displacement of the weight, measured by means of a linear encoder, were recorded using the MuscleLab™ system (Norrbrand et al. 2008).
Training was performed two (week 1, 3 and 5) or three (week 2 and 4) times weekly. Each session consisted of (grp FW) or aimed at (grp WS) four sets of seven CON–ECC knee extensions using the left limb. Grp FW performed coupled actions with a repetition cycle of about 3 s, with CON and ECC actions each lasting about 1.5 s. Similarly, in grp WS, the repetition cycle was about 3 s; the CON and ECC actions were about 1 and 2 s, respectively. During familiarization, grp FW were asked to perform FW exercise with increasing effort, while training requested maximal effort in each muscle action. Subjects were requested to push with maximal effort through the entire CON action, resisting gently in the initial third of the ECC action, and then aim at bringing the wheel to a full stop at about 80° knee angle. While being familiarized, individual weights for grp WS were chosen to result in failure to lift and lower the weight with seven repetitions, i.e., seven RM, in a controlled manner. Subjects were requested to lift the weight to a target ~165° knee angle in about 1 s, and then lower the weight in a controlled manner in approximately 2 s. Training load was increased by 1.25 kg in the subsequent set if the subject could perform more than seven repetitions, or lowered by 1.25 kg if the subject failed to complete seven repetitions with good form. Thus, whereas repetitions/set was fixed at seven for grp FW, the number of repetitions/set over the 12 sessions averaged 7.2 ± 0.3 in grp WS. Sets were interspersed by 2-min rest periods. Exercise sessions were preceded by a 5 min warm-up consisting of three sets of seven actions with progressively increased effort.
Test protocol
Maximal voluntary contraction (MVC)
This test, used to assess non-training-specific maximal strength and activation, was performed in the FW device. Maximal voluntary isometric knee extensor force (maximal voluntary contraction; MVC) along with EMG (maximal EMG amplitude; see below) was determined with hip angle at about 90° and the lever arm fixed at 120° knee angle. At least two maximal isometric actions (2–3 s each), intervened by 1 min rest, were performed to assess MVC. If there was a difference in maximal force across the two trials of more than 5%, the subject was requested to perform additional attempt(s). The highest force, averaged over a 1-s window showing a steady force level, was considered MVC.
Rate of force development
To assess RFD, tests were performed in the FW device, with hip angle at about 90° and the lever arm fixed at 120° knee angle. While sustaining an isometric action at 20% of MVC, subjects were instructed to increase force as fast as possible to reach maximal force and maintain a steady force for about 2–3 s. At least two trials, intervened by 1 min rest, were performed to assess RFD. The force was analyzed at 0.1-s intervals within the first 0.5 s of each trial. The trial showing the highest force at 0.4 s was chosen for further analysis.
Dynamic coupled CON–ECC actions
To assess training mode-specific strength and EMG amplitude pre- and post-training, four sets of seven RM were performed in the WS (grp WS) and four sets of seven maximal actions in FW (grp FW) apparatuses. Sets were interspersed by 2-min rest periods. Both exercises were executed with a range of motion of 80–85°, starting the CON action at about 80° until about 165°. During pre- and post-tests, the range of motion of grp FW (pre: 81 ± 5°, post: 83 ± 3°) and grp WS (pre: 86 ± 6°, post: 82 ± 5°) showed a trend to interaction over time and group (p < 0.10), but no significant differences were shown (p > 0.05). Mean, peak and angle-specific force are shown in Tables 2 and 3. Because force measurements performed in the two devices were not compatible there were no comparisons across groups with regard to force.
Surface EMG recordings
Pre- and post-test in grps WS and FW
Multichannel surface EMG signals were acquired from mm. vastus lateralis (VL) and vastus medialis (VM) using two 8-electrode, 5-mm interelectrode distance adhesive arrays (ELSCH008; SPES Medica, Salerno, Italy). These detection systems allow recording of multichannel surface EMG signals during movement, with negligible movement artifacts (Pozzo et al. 2004). Before array placement, the skin was shaved, rubbed with abrasive paste and cleansed with a paper towel.
The arrays were placed between the innervation zone and the proximal or distal tendon regions, along the fiber direction. Innervation zones and tendon regions were identified at about 90° and 160° knee joint angles in isometric test actions with the use of a 16-silver bar electrode array (1-mm length, 5-mm diameter, 5-mm interelectrode distance; SA16/5, Ottino Bioelettronica, Torino, Italy). Surface EMG signals were sampled at 2,048 samples/s per channel and amplified with a multichannel amplifier (−3 dB bandwidth: 10–500 Hz) (Farina et al. 2004; Pozzo et al. 2004). Signals were converted to digital data by a 12-bit acquisition board (National Instruments DAQCard-6024E; Austin, TX), displayed in real-time, and recorded on a computer equipped with EMG acquisition software (Pozzo et al. 2004). To ensure similar positioning of the EMG electrodes during the pre- and post-tests, the electrode location and anatomical landmarks were marked and transferred to a plastic sheet.
Processing of EMG data from the acute FW and WS test, and pre- and post-tests
The central bipolar signal of the VM and VL array were used to compute EMGRMS values during MVC and dynamic actions. Maximal EMGRMS was computed from the 1-s window that was chosen to determine MVC. In dynamic actions, EMGRMS was assessed from the entire range of motion, and 150-ms windows at knee joint angles of 90°, 120° and 150° in the CON and ECC action, respectively. EMGRMS at 90°, 120° and 150° knee angles were further averaged over repetitions and sets. Given the inherent methodological problems with surface EMG recordings (Duchateau et al. 2006), EMGRMS was expressed relative to maximal EMG for each individual muscle, i.e., normalized angle-specific EMGCON and EMGECC.
Statistics
All comparisons were made separately for the CON and ECC action and the VL and VM muscles. Group comparisons were made using three-way (over group, time and angle: normalized angle-specific EMGCON and EMGECC, or over group, time and interval: RFD) and two-way (over group and time: MVC, MVC EMGRMS, and range of motion) repeated measures ANOVA (StatSoft, Tulsa, OK, USA). Comparisons of angle-specific force within groups were made using two-way (over time and angle) repeated measures ANOVA. When a difference between training groups or an interaction was found, planned comparisons were executed, and Bonferroni correction employed. Hence, any p value obtained was multiplied by the number of planned comparisons and considered significant if lower than 0.05. A Student’s t test was used for comparisons when applicable (mean and peak force over time within grp FW and grp WS). The significance level was set to 0.05. Values are expressed as mean ± standard deviation.
Results
MVC and maximal EMGRMS
MVC increased by (p < 0.05) 8.1% (pre: 575 ± 90; post: 622 ± 91 N) in grp FW and 4.8% (pre: 594 ± 113; post: 623 ± 103 N) in grp WS in response to training. Maximal EMGRMS of VL increased (p < 0.05) in grp FW, but not in grp WS (Fig. 1). VM showed no change.
Electromyographic amplitude (EMGRMS) of mm. vastus lateralis (VL, a) and medialis (VM, b) during maximal voluntary contraction (MVC) performed by the flywheel (grp FW) and weight stack (grp WS) training groups pre- and post-training. *An interaction over group and time, where grp FW showed an increase (‡), but not grp WS
Rate of force development
Force at 0.1–0.5 s, compared across groups and over time, showed a trend to increase (p < 0.10) from pre- to post-training (Fig. 2). There was no difference between groups.
Normalized angle-specific EMG
Grp FW showed greater (p < 0.05) normalized angle-specific EMGECC of both VL and VM compared with grp WS (Fig. 3). Specifically, grp FW showed greater (p < 0.05) normalized EMGECC of VL and VM at −90° pre- and post-training (Figs. 3, 4). In contrast, grp WS showed greater normalized EMGECC of VL and VM at −150° post-training (p < 0.05; Fig. 4). Normalized EMGECC showed no changes from pre- to post-test in either group (Fig. 4). Overall, normalized angle-specific EMGCON showed no group difference. Grp WS showed a trend (p < 0.10) to increased normalized EMGCON of VL at 120° following training (Fig. 4). Collectively, following training, normalized EMGCON of grp WS was not different from that of grp FW. However, grp FW showed greater normalized EMGECC than grp WS after training.
Normalized angle-specific electromyographic (EMG) amplitude of mm. vastus lateralis (VL, a) and medialis (VM, b) during concentric (CON, 90° to 150°) and eccentric (ECC, −150° to −90°) actions of unilateral knee extension exercise, performed by the flywheel (grp FW) and weight stack (grp WS) training groups before the training period (pre-test). *Greater overall normalized angle-specific EMG with FW compared with WS exercise (p < 0.05). ‡Greater normalized angle-specific EMG with FW compared with WS exercise (p < 0.05)
Normalized angle-specific electromyographic (EMG) amplitude of mm. vastus lateralis (VL) and medialis (VM) during concentric (CON, 90° to 150°) and eccentric (ECC, −150° to −90°) actions of unilateral knee extension exercise, performed by the flywheel (grp FW, a, b) and weight stack (grp WS, c, d) training group pre- and post-training. Differences between groups are not indicated in the figure (see “Results” and Fig. 3). †Trend to increase over time with training (p < 0.10)
Discussion
The main finding of the present study was the markedly greater EMG, and in particular EMGECC activity during flywheel (FW) compared with weight stack (WS) resistance exercise both before and after training. Hence, the greater muscle activation, and the resulting mechanical stress using FW, may serve to explain the very robust muscle hypertrophy reported earlier in response to 5 weeks of flywheel resistance training (Norrbrand et al. 2008; Seynnes et al. 2007; Tesch et al. 2004).
To ensure comparable conditions for the two training groups, a 5-week unilateral open-chain knee extension exercise protocol standardized with regard to frequency and number of training sessions, number of sets and repetitions and rest was executed. In this context, the necessity and challenge of controlling conditions when comparing the outcome of different exercise paradigms should be acknowledged (Toigo and Boutellier 2006). Thus, body positioning, range of motion and time under tension per repetition was, if not identical, very similar for the two loading paradigms. It can only be speculated that training, at a different action speed (Matheson et al. 2001; Pincivero et al. 2008), requesting more repetitions per set (Pincivero et al. 2006), using other exercise modes (Andersen et al. 2006; Wilk et al. 1996) or employing other strategies (Sahaly et al. 2003), would have impacted the outcome of the current study.
Prior to training, the highest normalized EMG during WS occurred near completion of the CON knee extension. This coincided with the “sticking point”, that inherently dictates the load that can be used in any exercise using weights. As a consequence, muscle activation during WS exercise is far from maximal through the major portion of the entire range of motion. Further, because the WS device uses constant resistance, only the last repetition of a set calls for maximal muscle activation at the point of failure to raise the weight. In contrast, the inertia of flywheel offers unrestrained resistance in each action, evoking maximal or near maximal activation from the onset and throughout a set, with force progressively decreasing with fatigue. The prescribed strategy used during FW training produced greater ECC than CON peak force (“ECC overload”). Thus, FW, but not WS resistance exercise, elicited maximal EMG throughout a large part of the CON action and maximal or near maximal EMG in the flexed position of the ECC action.
Interestingly, muscle activation was close to maximal already before training in grp FW. Grp WS showed a trend toward augmented EMGCON, where the normalized EMG of VL and VM were maximal or near maximal in the low range of motion (90° and 120°) after training. Accordingly, force during WS training increased mainly in the acceleration phase (90°–120°) of CON actions. Thus, following training, EMGCON was very similar across the two groups.
It is generally agreed that marked increases in MVC, as a result of resistance training, could occur through increased neural drive (Aagaard et al. 2002; Hakkinen and Komi 1983; Moritani and deVries 1979). The improved MVC (grp WS: 4.8%, grp FW: 8.1%) however appears to correlate with the associated hypertrophy (WS: 3.0%, FW: 6–7%) reported elsewhere (Norrbrand et al. 2008; Seynnes et al. 2007; Tesch et al. 2004), which contradicts the notion of increased neural drive. Further, the augmented maximal EMGRMS of VL in response to FW resistance training may be indicative of increased neural drive. Yet, a concomitant boost in EMGRMS of VM would have been expected (Aagaard et al. 2002; Moritani and deVries 1979). Instead, the EMGRMS of only VL in grp FW may have been potentiated by reduced amplitude cancellations (Duchateau et al. 2006) due to increased MU synchronization (Milner-Brown et al. 1975), and/or enlarged surface area of the hypertrophied muscle fibers beneath the pick-up area of the surface electrode (Higbie et al. 1996). Finally, increased neural drive, resulting from training, may also be reflected in greater RFD (Aagaard et al. 2002), but neither group showed significant changes following training. In view of this, and regardless of exercise mode, any increase in neural drive must have been negligible and thus probably did not contribute to gain of MVC or training-specific strength.
Mechanisms responsible for the increased mode-specific strength, most evident following WS training, were therefore more likely attributed to more orchestrated coordination of agonist and stabilizing muscles (Duchateau et al. 2006; Rutherford and Jones 1986), excelled timing of individual motor unit activation (Cracraft and Petajan 1977) and/or attenuated antagonist co-activation (Hakkinen et al. 1998). The modest transfer of training-specific strength to MVC, most apparent in grp WS, conforms with the general notion of such neural adaptation occurring during the first weeks of training (Moritani and deVries 1979; Rutherford and Jones 1986).
The present study was prompted by previous reports of manifest hypertrophy following FW knee extension training (Norrbrand et al. 2008; Seynnes et al. 2007; Tesch et al. 2004). The quadriceps hypertrophy following FW training amounted to 4–5% (Seynnes et al. 2007) and 6–7% (Norrbrand et al. 2008; Seynnes et al. 2007; Tesch et al. 2004) after 3 and 5 weeks, respectively. This rate of quadriceps hypertrophy has rarely been reported (Luthi et al. 1986), while the hypertrophic response of 3% in grp WS after 5 weeks of training (Norrbrand et al. 2008) is in accordance with several earlier reports (Jones and Rutherford 1987; Narici et al. 1996; Ploutz et al. 1994). It is generally held that both high mechanical loading and inclusion of ECC actions are essential stimuli to promote muscle hypertrophy (Hather et al. 1991; Higbie et al. 1996; Hortobagyi et al. 1996). Since skeletal muscle signaling and remodeling for hypertrophy appears to be initiated by mechanical myofibrillar disruption (Friden et al. 1983; Newham et al. 1983), reports of potentiated Z-line streaming or myofibrillar disruptions (Friden et al. 1983; Newham et al. 1983) and protein synthesis (Moore et al. 2005; Wong and Booth 1990a, b) resulting from exercise favoring acute ECC actions, confirm this notion. Hence, the greater (1) ECC than CON peak force and (2) normalized EMGECC in grp FW, compared with grp WS, is commensurate with the hypothesis that FW resistance exercise offered a more powerful stimulus for hypertrophy than WS resistance exercise.
In an effort to control for range of motion, there was a brief pause between the ECC and CON actions during WS training. In contrast, actions in the FW mode were coupled and involved stretch–shortening cycling, most likely facilitating potentiated CON muscle activation and force (Komi and Bosco 1978). Given the inherent features of the exercise system, the subsequent CON action prompts more mechanical ECC work and hence muscle loading. Because grp WS did not perform coupled actions, it cannot be excluded that stretch–shortening actions executed in the FW mode in part were responsible for the greater muscle activation shown here and the more substantial hypertrophy reported elsewhere (Norrbrand et al. 2008) in response to FW exercise.
While the goal of this study was to compare neural adaptations in previously untrained subjects following resistance training using either flywheel inertia or standard weights, it became evident that muscle activation produced by FW exercise was near maximal in major portions of both CON and ECC actions prior to training. In contrast, WS exercise evoked far from complete activation. Following training, the EMGCON was very similar between groups, while the normalized EMGECC remained much lower in grp WS. Thus, even after training, it appears that WS did not provide an optimal stimulus for hypertrophy. The current investigation may offer an explanation in support of previous notions of more substantial hypertrophy after flywheel than weight training. We suggest that the greater muscle activation, and particularly that during ECC muscle actions, is indicative of greater mechanical loading, resulting in more robust stimulus giving rise to signals promoting enhanced protein synthesis and eventually leading to greater muscle hypertrophy.
References
Aagaard P, Simonsen EB, Andersen JL, Magnusson SP, Halkjaer-Kristensen J, Dyhre-Poulsen P (2000) Neural inhibition during maximal eccentric and concentric quadriceps contraction: effects of resistance training. J Appl Physiol 89:2249–2257
Aagaard P, Simonsen EB, Andersen JL, Magnusson P, Dyhre-Poulsen P (2002) Increased rate of force development and neural drive of human skeletal muscle following resistance training. J Appl Physiol 93:1318–1326
Adams GR, Duvoisin MR, Dudley GA (1992) Magnetic resonance imaging and electromyography as indexes of muscle function. J Appl Physiol 73:1578–1583
Andersen LL, Magnusson SP, Nielsen M, Haleem J, Poulsen K, Aagaard P (2006) Neuromuscular activation in conventional therapeutic exercises and heavy resistance exercises: implications for rehabilitation. Phys Ther 86:683–697
Asmussen E (1953) Positive and negative muscular work. Acta Physiol Scand 28:364–382
Berg HE, Tesch A (1994) A gravity-independent ergometer to be used for resistance training in space. Aviat Space Environ Med 65:752–756
Cracraft JD, Petajan JH (1977) Effect of muscle training on the pattern of firing of single motor units. Am J Phys Med 56:183–194
Duchateau J, Semmler JG, Enoka RM (2006) Training adaptations in the behavior of human motor units. J Appl Physiol 101:1766–1775
Dudley GA, Tesch PA, Harris RT, Golden CL, Buchanan P (1991) Influence of eccentric actions on the metabolic cost of resistance exercise. Aviat Space Environ Med 62:678–682
Farina D, Pozzo M, Merlo E, Bottin A, Merletti R (2004) Assessment of average muscle fiber conduction velocity from surface EMG signals during fatiguing dynamic contractions. IEEE Trans Biomed Eng 51:1383–1393
Friden J, Sjostrom M, Ekblom B (1983) Myofibrillar damage following intense eccentric exercise in man. Int J Sports Med 4:170–176
Hakkinen K, Komi PV (1983) Electromyographic changes during strength training and detraining. Med Sci Sports Exerc 15:455–460
Hakkinen K, Kallinen M, Izquierdo M, Jokelainen K, Lassila H, Malkia E, Kraemer WJ, Newton RU, Alen M (1998) Changes in agonist–antagonist EMG, muscle CSA, and force during strength training in middle-aged and older people. J Appl Physiol 84:1341–1349
Hather BM, Tesch PA, Buchanan P, Dudley GA (1991) Influence of eccentric actions on skeletal muscle adaptations to resistance training. Acta Physiol Scand 143:177–185
Higbie EJ, Cureton KJ, Warren GL 3rd, Prior BM (1996) Effects of concentric and eccentric training on muscle strength, cross-sectional area, and neural activation. J Appl Physiol 81:2173–2181
Hortobagyi T, Hill JP, Houmard JA, Fraser DD, Lambert NJ, Israel RG (1996) Adaptive responses to muscle lengthening and shortening in humans. J Appl Physiol 80:765–772
Jones DA, Rutherford OM (1987) Human muscle strength training: the effects of three different regimens and the nature of the resultant changes. J Physiol (Lond) 391:1–11
Katz B (1939) The relation between force and speed in muscular contraction. J Physiol (Lond) 96:45–64
Komi PV, Bosco C (1978) Utilization of stored elastic energy in leg extensor muscles by men and women. Med Sci Sports 10:261–265
Komi PV, Buskirk ER (1972) Effect of eccentric and concentric muscle conditioning on tension and electrical activity of human muscle. Ergonomics 15:417–434
Luthi JM, Howald H, Claassen H, Rosler K, Vock P, Hoppeler H (1986) Structural changes in skeletal muscle tissue with heavy-resistance exercise. Int J Sports Med 7:123–127
Matheson JW, Kernozek TW, Fater DC, Davies GJ (2001) Electromyographic activity and applied load during seated quadriceps exercises. Med Sci Sports Exerc 33:1713–1725
Milner-Brown HS, Stein RB, Lee RG (1975) Synchronization of human motor units: possible roles of exercise and supraspinal reflexes. Electroencephalogr Clin Neurophysiol 38:245–254
Moore DR, Phillips SM, Babraj JA, Smith K, Rennie MJ (2005) Myofibrillar and collagen protein synthesis in human skeletal muscle in young men after maximal shortening and lengthening contractions. Am J Physiol Endocrinol Metab 288:E1153–E1159
Moritani T, deVries HA (1979) Neural factors versus hypertrophy in the time course of muscle strength gain. Am J Phys Med 58:115–130
Moritani T, Muramatsu S, Muro M (1987) Activity of motor units during concentric and eccentric contractions. Am J Phys Med 66:338–350
Nardone A, Romano C, Schieppati M (1989) Selective recruitment of high-threshold human motor units during voluntary isotonic lengthening of active muscles. J Physiol (Lond) 409:451–471
Narici MV, Hoppeler H, Kayser B, Landoni L, Claassen H, Gavardi C, Conti M, Cerretelli P (1996) Human quadriceps cross-sectional area, torque and neural activation during 6 months strength training. Acta Physiol Scand 157:175–186
Newham DJ, McPhail G, Mills KR, Edwards RH (1983) Ultrastructural changes after concentric and eccentric contractions of human muscle. J Neurol Sci 61:109–122
Norrbrand L, Fluckey JD, Pozzo M, Tesch PA (2008) Resistance training using eccentric overload induces early adaptations in skeletal muscle size. Eur J Appl Physiol 102:271–281
Pincivero DM, Gandhi V, Timmons MK, Coelho AJ (2006) Quadriceps femoris electromyogram during concentric, isometric and eccentric phases of fatiguing dynamic knee extensions. J Biomech 39:246–254
Pincivero DM, Coelho AJ, Campy RM (2008) Contraction mode shift in quadriceps femoris muscle activation during dynamic knee extensor exercise with increasing loads. J Biomech 41:3127–3132
Ploutz LL, Tesch PA, Biro RL, Dudley GA (1994) Effect of resistance training on muscle use during exercise. J Appl Physiol 76:1675–1681
Pozzo M, Merlo E, Farina D, Antonutto G, Merletti R, Di Prampero PE (2004) Muscle-fiber conduction velocity estimated from surface EMG signals during explosive dynamic contractions. Muscle Nerve 29:823–833
Rutherford OM, Jones DA (1986) The role of learning and coordination in strength training. Eur J Appl Physiol 55:100–105
Sahaly R, Vandewalle H, Driss T, Monod H (2003) Surface electromyograms of agonist and antagonist muscles during force development of maximal isometric exercises––effects of instruction. Eur J Appl Physiol 89:79–84
Seynnes OR, de Boer M, Narici MV (2007) Early skeletal muscle hypertrophy and architectural changes in response to high-intensity resistance training. J Appl Physiol 102:368–373
Tesch PA, Ekberg A, Lindquist DM, Trieschmann JT (2004) Muscle hypertrophy following 5-week resistance training using a non-gravity-dependent exercise system. Acta Physiol Scand 180:89–98
Toigo M, Boutellier U (2006) New fundamental resistance exercise determinants of molecular and cellular muscle adaptations. Eur J Appl Physiol 97:643–663
Wilk KE, Escamilla RF, Fleisig GS, Barrentine SW, Andrews JR, Boyd ML (1996) A comparison of tibiofemoral joint forces and electromyographic activity during open and closed kinetic chain exercises. Am J Sports Med 24:518–527
Wong TS, Booth FW (1990a) Protein metabolism in rat gastrocnemius muscle after stimulated chronic concentric exercise. J Appl Physiol 69:1709–1717
Wong TS, Booth FW (1990b) Protein metabolism in rat tibialis anterior muscle after stimulated chronic eccentric exercise. J Appl Physiol 69:1718–1724
Acknowledgments
We thank all the subjects who participated in this study. This study was funded by the Swedish Winter Sports Research Centre, Mid Sweden University, Östersund, the Swedish National Centre for Research in Sports (CIF), and the Swedish National Space Board (SNSB).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Arnold de Haan.
Rights and permissions
About this article
Cite this article
Norrbrand, L., Pozzo, M. & Tesch, P.A. Flywheel resistance training calls for greater eccentric muscle activation than weight training. Eur J Appl Physiol 110, 997–1005 (2010). https://doi.org/10.1007/s00421-010-1575-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-010-1575-7