Abstract
Purpose
Symptomatic retinal arterial macroaneurysms (RAM) are primarily investigated by fundus fluorescein angiography after presenting with visual disturbance. The natural history includes spontaneous regression and occasionally occlusion of the arteriole distal to the aneurysm. RAM may be managed conservatively. Interventional treatment options include focal argon laser photocoagulation, Nd:YAG laser hyaloidotomy, and pars plana vitrectomy. The purpose of this study was to elicit the rates of distal vessel occlusion and aneurysm thrombosis in RAM at presentation, and their relevance to the treatment of RAM. Furthermore, visual outcomes were examined.
Methods
Retrospective review of cases of RAM presenting to a tertiary ophthalmology care centre was accomplished in a university teaching hospital. The angiographic features, treatment indications, and visual outcomes in patients with RAM were recorded. Angiographic features noted were distal vessel patency and aneurysm thrombosis at presentation.
Results
Ten patients with RAM were identified. Ninety percent had an angiographically patent distal arteriole, with 40 % showing spontaneous thrombosis of the aneurysm sac at presentation. Patients presenting with a spontaneously thrombosed RAM were managed conservatively, those with flow within the aneurysm wall were treated with focal laser, and those with subhyaloid haemorrhage underwent Nd:YAG laser hyaloidotomy. LogMAR visual acuity improved from 0.3 (±0) at presentation to 0.15 (±0.1) in the conservative group, and from 0.78 (±0.23) to 0.24 (±0.18) in those who underwent one intervention. One patient lost vision after multiple RAM.
Conclusion
Thrombosis within the aneurysm wall is an important feature in deciding to treat RAM, and selective use of interventions improves vision in affected patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Retinal arterial macroaneurysms (RAM) are an acquired abnormality of the retinal vasculature. They comprise a dilatation or outpouching of a major retinal arteriole. The term retinal arterial macroaneurysm was first described in 1973 to distinguish these aneurysms from those found in retinal veins and capillaries [1].
RAM are an isolated phenomenon in two thirds of cases, and occur in conjunction with retinal venous obstruction in one third [2]. Ten percent of cases are bilateral [1]. Risk factors for the development of a RAM include female sex, retinal vein occlusion, and hypertension [2, 3]. RAM are rare, with a reported prevalence of 1 in 4500 people [4]. They are usually asymptomatic until acute haemorrhage and/or exudation affect the macula [1]. RAM have been subdivided into two subgroups- haemorrhagic and exudative [5]. Haemorrhagic RAM are associated with systemic hypertension while exudative RAM are more frequently associated with retinal vein occlusions. RAM are most commonly found superotemporally (53 %), followed by the inferotemporally (38 %), inferonasally (5 %), and superonasally (4 %) [5].
The most common clinical features of symptomatic RAM are retinal haemorrhage (81 %) and exudate (70 %) [2]. RAM-associated retinal haemorrhage may be preretinal, intraretinal, or subretinal [3]. Acute multilevel haemorrhage surrounding a white or yellow spot should arouse suspicion for RAM. Less commonly, vitreous haemorrhage (30 %) is observed [2].
RAM have a characteristic saccular or fusiform hyperfluorescence on fundus fluorescein angiogram (FFA) [6]. Fusiform RAM are described as filling rapidly in early phases of the angiogram, with saccular RAM showing minimal early filling [7]. The latter fill in middle to late phases, with heterogenous hyperfluorescence possibly due to intraluminal clot. Data on the rate of aneurysm thrombosis and the patency of distal vasculature at presentation is sparse. To date, it has not been determined if this is of clinical significance. One study of 44 RAM showed that at least 48 % demonstrated fine capillary changes, including capillary closure, both adjacent and distal to the RAM [7]. Spectral domain optical coherence tomography (OCT) has been used to further delineate the morphologic characteristics of RAM [8]. Hypo-reflectivity was noted within the aneurysm in all the cases examined at presentation, likely representing patent blood flow. This was in contrast with the hyper-reflectivity found on thrombosis of the aneurysm after photocoagulation. It is unclear if these RAM were exclusively fusiform- given that is the more common subtype associated with a patent lumen, without thrombosis at presentation.
Symptomatic RAM associated with haemorrhage may resolve spontaneously with improvement in visual acuity, particularly if the bleeding is in the inner retinal layers [9]. The natural history of RAM is described as frequent spontaneous regression and improvement in visual acuity [2, 10]. However, identifying those which are more likely to regress spontaneously is a challenge. Therefore, a number of factors must be considered before deciding to treat symptomatic RAM- the deficit in visual acuity, location of haemorrhage (the retinal layer(s) involved), and involvement of macula. This study also proposes the inclusion of specific angiographic features of the RAM in the decision to treat. Treatment options include argon laser photocoagulation of the RAM [11–13], Nd:YAG laser hyaloidotomy for subhyaloid haemorrhage, [14, 15] and pars plana vitrectomy (PPV) for vitreous haemorrhage [16]. Studies are emerging advocating the use of intravitreal anti-VEGF (vascular endothelial growth factor) in selected cases of RAM, e.g., foveal involvement [17, 18].
Common findings observed on resolution include kinking of the vessel at the site of the RAM, arteriolar constriction proximal to the RAM, arterial sheathing distal to the RAM and arteriolar occlusion distal to the RAM [12, 19]. Higher rates of vessel occlusion occur after direct aneurysmal laser (16 %) as opposed to perianeurysmal laser exclusively (0 %) [2]. Spontaneous occlusion of the distal arteriole occurs in 4–25 % [2, 20, 21].
Risk factors for poorer visual outcome include foveal exudation or subfoveal haemorrhage [22, 23]. No long term visual benefit has been shown where no treatment was compared to laser photocoagulation [10]. Visual prognosis of submacular haemorrhage due to ruptured RAM is poor, but that of macular oedema, preretinal haemorrhage, or vitreous haemorrhage is relatively good [24].
The aims of this study are to describe the angiographic features, treatment indications, and visual outcomes in a case series of patients with RAM. We propose the inclusion of angiographic patency of RAM in the decision to treat RAM.
Methods
A retrospective analysis of all patients with a diagnosis of RAM presenting to the ophthalmology department of a tertiary referral hospital from 2003 to 2013 was carried out. Medical records were reviewed and the following details recorded: age, sex, past medical and ophthalmic history, presenting complaint, Snellen best corrected visual acuity (BCVA) at presentation, affected eye and retinal quadrant, characteristics of haemorrhage and retinal layers affected, FFA appearance, intervention, and final BCVA. Snellen VA has been converted to the LogMAR equivalent for ease of analysis. Results are expressed as the mean ± standard deviation. Statistical significance is determined as P < 0.05, as calculated by the paired Student’s t-test.
Results
A total of ten patients were identified. The mean age at presentation was 71.1 years (±9 years). Ninety percent were female, 60 % had a previous diagnosis of hypertension, and 80 % of the RAM were in the left eye. Nine patients had one RAM, and one patient developed five RAM over 5 years. The patient with multiple RAM underwent systemic investigations, which were normal (including assessment for Von Hippel Lindau). Nine patients (90 %) presented with blurred vision, and the other RAM was noted during diabetic retinopathy screening. Most were superotemporal to the disc (90 %), with a minority located inferonasally (10 %). The average distance of the RAM from the optic nerve was 2.9 disc diameters (+/− 1.2). All patients were investigated with FFA. OCT was not available in this ophthalmology department throughout the duration of the study, and as a result is not included in the analysis.
Angiographic findings
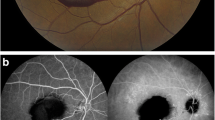
FFA was available for review in all cases. Macular oedema was evident in 50 % of patients. Nine patients had retinal haemorrhage, categorised as preretinal (40 %), intraretinal (80 %), and subretinal (60 %). There was trilaminar haemorrhage in 30 % of cases (Fig. 1a). All RAM were saccular. Forty percent had flow in the aneurysm wall while 60 % had thrombosed at presentation. Thrombosis within the RAM wall bore no correlation to the presence or absence of macular oedema. There was patent arterial flow in the vessel distal to the RAM in 90 % at presentation (Fig. 1b and c). The only patient who had an occluded artery distal to the RAM had an exudative type RAM as well as a thrombosed RAM at presentation. They were managed conservatively with hypertension control. Regarding RAM subtype, 10 % of cases were purely exudative, and 90 % haemorrhagic with or without macular exudation.
Angiographic findings in patients affected by RAM at presentation, and follow-up. a FFA at presentation demonstrating trilaminar haemorrhage. b FFA 1 month after Nd:YAG hyaloidotomy demonstrating resolution of the premacular haemorrhage, thrombosis of the aneurysm wall, and a patent distal arteriole. c FFA at presentation demonstrating flow within the aneurysm wall of a superotemporal RAM, and a patent distal arteriole. d FFA 1 year after focal argon laser demonstrating thrombosis of the aneurysm and a patent distal arteriole. There is some additional intraretinal haemorrhage inferior to the macula, and arteriovenous nipping, related to hypertensive retinopathy. e FFA at presentation demonstrating a RAM superotemporal to the fovea with hyperfluorscence in a saccular aneurysm wall and patent distal arteriole. f FFA 8 months after focal laser demonstrating thrombosis of the aneurysm wall and a patent distal arteriole. There is a second RAM superior to the original RAM
Interventions
Sixty percent of patients underwent an interventional procedure- two underwent posterior laser hyaloidotomy (Fig. 1a and b), and four underwent focal peri-aneurysm laser (Fig. 1c and d). One patient who had peri-aneurysmal laser subsequently required vitrectomy. She developed multiple sequential RAM and non-resolving vitreous haemorrhage (Fig. 1e and f). The indications for intervening were premacular subhyaloid haemorrhage and/or patent flow within the aneurysm wall. Macular oedema was not an absolute indication for intervention; hypertension control alone was sufficient to resolve macula oedema without other intervention in one case. As mentioned above, not all patients with macular oedema had non-thrombosed RAM.
Visual acuity
BCVA in the group managed conservatively (n = 4) was 0.3 (±0) at presentation, improving to 0.15 (±0.1) at final follow-up (p = 0.06). In the group who had one intervention (n = 5), BCVA was 0.78 (±0.23) at presentation improving significantly to 0.24 (±0.18) at final measurement (p = 0.003). When the treatment groups are subdivided, patients who underwent hyaloidotomy alone improved from 0.75 (±0.25) to 0.10 (±0.10). Those who had photocoagulation alone improved from 0.80 (±0.2) to 0.5 (±0.3). The patient with multiple RAM underwent five procedures, and BCVA was 0.0 at presentation, and 1.7 at follow-up. The mean gain in visual acuity was 0.15 (±0.1) in those without intervention, and 0.54 (±0.27) in those who had one intervention. The patient who had >1 intervention lost vision (+1.7) due to macular scarring.
Discussion
RAM are rare, precluding large studies on management and outcomes. We identified ten affected patients managed in the ophthalmology unit of a tertiary referral hospital in Ireland. Consistent with other studies [2, 3], the majority of patients in this series were female (90 %), and had a history of hypertension (60 %). The majority of RAM were haemorrhagic (90 %). This conforms with previous studies linking the association between hypertension and haemorrhagic RAM [7]. All cases were unilateral, differing from reported bilaterality in 10 % [1]. The majority of RAM were in the left eye. There is no predominant laterality reported in RAM, and this cannot be explained. Ninety percent presented with blurred vision, consistent with other studies [7]. Conclusions drawn from this study are limited to symptomatic RAM. The assumption made is that asymptomatic RAM are likely under-represented in studies, as they go undiagnosed in many cases unless ophthalmic examination or retinal imaging is performed for another reason.
The majority of RAM were superotemporal (90 %). The average distance of the haemorrhagic RAM in the series from the optic disc was 2.9 disc diameters- this contrasts with previous reports of an average distance of 1.5 disc diameters [7]. Fifty percent had macular oedema compared to 30 % [2] and 77 % [22] in other studies. BCVA improved from 0.3 to 0.14 (±0.1) in those managed conservatively (p = 0.06), and from 0.78 (±0.23) to 0.24 (±0.18) in those with one intervention (p = 0.003). The interventional group had a worse mean presenting BCVA and final BCVA, but achieved a higher mean gain in vision. This is in contrast with previous groups who did not find a difference in final BCVA after intervention [25]. Sixty percent of patients had an interventional procedure, all of whom had haemorrhagic RAM subtype. The indication to intervene was premacular subhyaloid haemorrhage, and/or patent flow within the aneurysm wall (non-thrombosed RAM lumen). Non-thrombosed RAM represented 40 % of the cases presented. Patent flow within the aneurysm wall represents the possibility of further leakage whether as fresh haemorrhage, or peri-aneursymal retinal oedema. This is delineated by the fact that aneurysm thrombosis can be observed after photocoagulation: it is a desired endpoint of treatment [26]. The rate of RAM thrombosis at presentation is unreported in any of the published series, and to date is an overlooked aspect of the FFA. It is interesting to note that macular oedema was not exclusive to non-thrombosed RAM. Arteriolar occlusion is reported to occur in between 4 and 25 % of RAM spontaneously, and in an additional 16 % of cases following RAM laser photocoagulation [2, 21]. This compares with the series presented, which had a spontaneously occluded distal arteriole in 10 %.
In our experience visual acuity in patients presenting with RAM can be improved with assessment and control of systemic risk factors, and by utilising the angiographic features on presentation to choose the most appropriate intervention. Sixty percent of RAM presented were thrombosed at presentation, representing a higher likelihood of recovery without intervention. Perianeurysm argon laser to angiographically patent RAM can improve visual acuity, while Nd:YAG laser posterior laser hyaloidotomy should be considered when premacular subhyaloid haemorrhage is present.
References
Robertson DM (1973) Macroaneurysms of the retinal arteries. Trans Am Acad Ophthalmol Otolaryngol 77(1):OP55–OP67
Panton RW, Goldberg MF, Farber MD (1990) Retinal arterial macroaneurysms: risk factors and natural history. Br J Ophthalmol 74(10):595–600
Moosavi RA, Fong KC, Chopdar A (2006) Retinal artery macroaneurysms: clinical and fluorescein angiographic features in 34 patients. Eye (Lond) 20(9):1011–1020. doi:10.1038/sj.eye.6702068
Xu L, Wang Y, Jonas JB (2007) Frequency of retinal macroaneurysms in adult Chinese: the Beijing eye study. Br J Ophthalmol 91(6):840–841. doi:10.1136/bjo.2006.107342
Tezel T, Gunalp I, Tezel G (1994) Morphometrical analysis of retinal arterial macroaneurysms. Doc Ophthalmol 88(2):113–125
Pitkanen L, Tommila P, Kaarniranta K, Jaaskelainen JE, Kinnunen K (2014) Retinal arterial macroaneurysms. Acta Ophthalmol 92(2):101–104. doi:10.1111/aos.12210
Lavin MJ, Marsh RJ, Peart S, Rehman A (1987) Retinal arterial macroaneurysms: a retrospective study of 40 patients. Br J Ophthalmol 71(11):817–825
Lee EK, Woo SJ, Ahn J, Park KH (2011) Morphologic characteristics of retinal arterial macroaneurysm and its regression pattern on spectral-domain optical coherence tomography. Retina 31(10):2095–2101. doi:10.1097/IAE.0b013e3182111711
Tsujikawa A, Sakamoto A, Ota M, Oh H, Miyamoto K, Kita M, Yoshimura N (2009) Retinal structural changes associated with retinal arterial macroaneurysm examined with optical coherence tomography. Retina 29(6):782–792. doi:10.1097/IAE.0b013e3181a2f26a
Brown DM, Sobol WM, Folk JC, Weingeist TA (1994) Retinal arteriolar macroaneurysms: long-term visual outcome. Br J Ophthalmol 78(7):534–538
Hudomel J, Imre G (1973) Photocoagulation treatment of solitary aneurysm near the macula lutea. Report of a case. Acta Ophthalmol (Copenh) 51(5):633–638
Abdel-Khalek MN, Richardson J (1986) Retinal macroaneurysm: natural history and guidelines for treatment. Br J Ophthalmol 70(1):2–11
Psinakis A, Kokolakis S, Theodossiadis PG, Koutsandrea C (1989) Pulsatile arterial macroaneurysm: management with argon laser photocoagulation. J Fr Ophtalmol 12(10):673–676
Dahreddine M, Eldirani H, Mutsinzi E, Hirsch A (2011) Retinal arterial macroaneurysm complicated by premacular hemorrhage: treatment by YAG laser disruption. J Fr Ophtalmol 34(2):131.e1–135.e5. doi:10.1016/j.jfo.2010.09.018
Zghal-Mokni I, Nacef L, Yazidi B, Malek I, Bouguila H, Ayed S (2007) Clinical and progressive features of macular hemorrhage secondary to retinal artery macroaneurysms. J Fr Ophtalmol 30(2):150–154
Nakamura H, Hayakawa K, Sawaguchi S, Gaja T, Nagamine N, Medoruma K (2008) Visual outcome after vitreous, sub-internal limiting membrane, and/or submacular hemorrhage removal associated with ruptured retinal arterial macroaneurysms. Graefes Arch Clin Exp Ophthalmol 246(5):661–669. doi:10.1007/s00417-007-0724-0
Pichi F, Morara M, Torrazza C, Manzi G, Alkabes M, Balducci N, Vitale L, Lembo A, Ciardella AP, Nucci P (2013) Intravitreal bevacizumab for macular complications from retinal arterial macroaneurysms. Am J Ophthalmol 155(2):287.e1–294.e1. doi:10.1016/j.ajo.2012.07.029
Cahuzac A, Scemama C, Mauget-Faysse M, Sahel JA, Wolff B (2016) Retinal arterial macroaneurysms: clinical, angiographic, and tomographic description and therapeutic management of a series of 14 cases. Eur J Ophthalmol 26(1):36–43. doi:10.5301/ejo.5000641
Holland PM (1984) Evolution of a retinal arterial macroaneurysm. Ann Ophthalmol 16(12):1167–1170
Fineman MS Ho A (2012) Color atlas and synopsis of clinical ophthalmology, Wills eye institute: retina, 2nd edn. Lippincott Williams and Wilkins
Cleary PE, Kohner EM, Hamilton AM, Bird AC (1975) Retinal macroaneurysms. Br J Ophthalmol 59(7):355–361
Yang CS, Tsai DC, Lee FL, Hsu WM (2005) Retinal arterial macroaneurysms: risk factors of poor visual outcome. Ophthalmologica 219(6):366–372. doi:10.1159/000088380
Palestine AG, Robertson DM, Goldstein BG (1982) Macroaneurysms of the retinal arteries. Am J Ophthalmol 93(2):164–171
Saito K, Iijima H (1997) Visual prognosis and macular pathology in eyes with retinal macroaneurysms. Nihon Ganka Gakkai Zasshi 101(2):148–151
Koinzer S, Heckmann J, Tode J, Roider J (2015) Long-term, therapy-related visual outcome of 49 cases with retinal arterial macroaneurysm: a case series and literature review. Br J Ophthalmol 99(10):1345–1353. doi:10.1136/bjophthalmol-2014-305884
Lee KY, Tomidokoro A, Sakata R, Konno S, Mayama C, Saito H, Hayashi K, Iwase A, Araie M (2010) Cross-sectional anatomic configurations of peripapillary atrophy evaluated with spectral domain-optical coherence tomography. Invest Ophthalmol Vis Sci 51(2):666–671. doi:10.1167/iovs.09-3663
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Informed consent
For this type of study (retrospective review) formal consent is not required.
Rights and permissions
About this article
Cite this article
Hughes, E.L., Dooley, I.J., Kennelly, K.P. et al. Angiographic features and disease outcomes of symptomatic retinal arterial macroaneurysms. Graefes Arch Clin Exp Ophthalmol 254, 2203–2207 (2016). https://doi.org/10.1007/s00417-016-3388-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3388-9