Abstract
Background
Precise IOL calculation in post-refractive surgery patients is still a challenge for the cataract surgeon. The purpose of this study is to test whether adding Orbscan II values into the double-K method improves IOL calculation in this group of patients.
Methods
A prospective study with 43 eyes previously submitted to refractive surgery that underwent cataract extraction. IOL calculation was performed with double-K method. Post-K value was derived from Orbscan total-mean power map. The average corneal curvature of the general population (43.8D) was used as the pre-K value. Refraction results 30 days after surgery were compared with refraction that would be obtained if we used: (1) post-K values from keratometry, (2) post-K values from topography, and (3) pre-K values from Orbscan total-mean power. Anterior chamber depth measures obtained with the IOL Master and Orbscan II were compared.
Results
Mean postoperative spherical equivalent (SE) was −0.25 ± 1.10 D in eyes submitted to radial keratotomy , –1.04 ± 1.42 D in eyes previously submitted to myopic Lasik, and +0.05 ± 1.76 D in those submitted to hyperopic surgeries. Had we inputted post-K values derived from keratometer and from topography, we would have obtained significantly higher postoperative refractive errors in eyes previously submitted to myopic refractive surgery (p < 0.05). Refractions using pre-K derived from the central 8 mm Orbscan instead of 43.8 D were similar in all studied groups (p > 0.05). Anterior chamber depth measured with IOL Master or Orbscan were similar.
Conclusions
Orbscan measurements used as the post-K values into the double-K method provide a precise IOL calculation, especially in post myopic refractive surgery patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One of the great challenges in performing cataract surgery in post-refractive surgery patients is obtaining an accurate refractive outcome, especially in previously myopic patients. Intraocular lens (IOL) calculation is less reliable in these cases, generally inducing hyperopic errors after cataract surgery [1–3]. The two main sources of biometric errors are the IOL formulas and the inaccuracy of post-refractive surgery central corneal curvature measurements [4].
First and second generation formulas were developed for normal corneas where a constant relationship exists between anterior and posterior curvatures. In non-operated corneas, this ratio is approximately 82%; that is, posterior curvature radio is 1.2 mm less than the anterior, which leads to a corneal refractive index of 1.3375. After refractive surgery, this relationship is not constant anymore, which leads to a change in corneal refractive index [4–6]. Furthermore, these formulas consider the corneal curvature to calculate the effective lens position, and flat corneal measurements erroneously estimate the effective lens position [4].
In 2003, Aramberri described a method that considers a double keratometry, where original pre-refractive surgery cornea curvature (pre-K) is used to obtain the effective IOL position, and the post-refractive corneal curvature (post-K) for the vergence formula. This method has considerably improved IOL calculation after refractive surgery [7].
The other important source of error is the improper central corneal curvature measurement after refractive surgery. Keratometers and Placido ring topographers usually generate a measure that is steeper than the real central corneal curvature, because they calculate an average of the inner reflected ring on the cornea, not reflecting the real central corneal curvature [5, 6]. Adjusted keratometry, contact lens and clinical history methods are other alternatives, but also with poor predictability [1–3, 8, 9].
Corneal tomography with Orbscan II (Bausch & Lomb, USA), Pentacam (Oculus, Germany) or Galilei (Ziemer Group, Switzerland), on the other hand, have been shown to be more precise for evaluating central corneal curvature after refractive surgery, because they analyze corneal elevation, including the central area [4–6, 9–13]. Furthermore, the total corneal power assessed by these equipments includes data from the corneal thickness, from both corneal surfaces instead of only from the anterior surface, and physiologic refraction indices instead of a fictitious keratometric refractive index [9, 12, 13].
Finally, another problem is that many times we don’t have the corneal curvature before refractive surgery (pre-K) to input into the double-K method. An alternative is to use the general population mean keratometry (43.8D), which is usually precise enough for this method [14].
We present herein the refractive results of 43 eyes submitted to cataract surgery which had previously been submitted to refractive surgery, in which we used the double-K method, where the post-K value was obtained from the Orbscan II and 43.8D considered as pre-K value.
Methods
A prospective study was performed in 43 eyes of 30 patients previously submitted to radial keratotomy (26 eyes), myopic Lasik (eight eyes), and hyperopic Lasik or PRK (nine eyes) who underwent cataract surgery. Table 1 provides detailed preoperative information of each case. None of the patients had precise information about their corneal measurements before the refractive surgery.
IRB/Ethics Committee decided approval was not required for this study, but an informed consent was obtained for participants.
Patients were submitted to a tomography of the anterior segment using Orbscan II (Bausch Lomb, Germany) to obtain the data to insert in the Aramberri double-K method for IOL calculation [7]. Pre-K value used in the double-K method was 43.8 D, as the average value for the general population [14]. Post-K values were assessed from Orbscan II average of total-mean power from the central 2 mm for eyes submitted to myopic correction, and from the central 4 mm for eyes submitted to hyperopic surgery [4, 5, 9–12].
Axial length and anterior chamber depth (ACD) were obtained through partial coherence interferometry (IOL Master®, Carl Zeiss Meditec, Germany). ACD obtained by Orbscan II was compared to the one obtained from IOL Master.
All surgeries were performed without complications with peribulbar anesthesia and phacoemulsification with a self-sealing temporal clear corneal 2.75 mm incision, implanting all IOLs in the bag. The chosen IOL to be implanted was the one that best matched emmetropia. The formula used in the study was SRK/T for eyes longer than 22.51 mm and Hoffer-Q for eyes shorter than 22.50 mm.
IOLs implanted were 20 Tecnis ZA9003 (AMO, USA), six Acrysof Natural SN60AT (Alcon, USA), four multifocal Tecnis ZM900 (AMO, USA), three C-Flex 970-C (Rayner, England), five Sensar AR-40e (AMO, USA), two multifocal ReZoom (AMO, USA), two Acrysof toric (Alcon, USA), and one multifocal M-Flex 630F (Rayner, England).
Postoperative spherical equivalent (SE) 30 days after surgery was compared with the refraction that would be obtained if we used: (1) post-K values derived from keratometry performed with the Tomey auto-keratometer (Tomey, USA), (2) post-K values derived from topography performed with the Tomey TMS-3 topographer (Tomey, USA), and (3) pre-K average values derived from the central 8 mm area from Orbscan II [7].
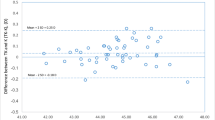
Statistical analysis was performed with SPSS version 17.0. Means were compared with the Student´s paired-samples t-test, at 5% significance. Bland and Altman technique was performed to evaluate the agreement between our results and the other methods.
Results
Mean SE after cataract surgery in eyes previously submitted to radial keratotomy was −0.25 D ± 1.10 D (range −2.12 D to +1.70D). In patients previously submitted to myopic Lasik, mean postoperative SE was −1.04 D ±1.42 D (range −3.10 D to +0.65 D). In patients previously submitted to hyperopia surgery, mean postoperative SE was +0.05 D ± 1.76 D (range −2.25D to +2.40 D) (Table 2). Refraction outcomes of each patient after cataract surgery are listed in Table 1.
Simulation of the postoperative refraction using post-K values obtained from keratometer and topography, as well as pre-K values obtained from Orbscan II, is shown in Tables 2 and 4. Table 3 shows the limits of agreement (CI 95 %) among our refraction results and the others that were simulated (Table 4).
Results obtained using the average value from the central 2 mm area measured by Orbscan II as the post-K value in the double-K method were statistically better than the ones we would have expected using the post-K values derived from keratometry (p < 0.05), in all eyes that had undergone myopic and hyperopic refractive surgery.
Using post-K values from topography, we would also obtain worst refractive results in eyes previously submitted to myopic refractive surgery (p < 0.05). In eyes previously submitted to hyperopic correction, refractive results were also better, but there was no statistically significant difference (p > 0.05).
If we had used pre-K values derived from Orbscan (instead of the average corneal curvature of 43.8D), postoperative SE would be similar in all groups studied.
IOL calculations using ACD measurements obtained either from the Orbscan II or from the IOL Master were similar in all groups (p > 0.05).
Discussion
One of the great challenges in calculating IOL power after refractive surgery is to precisely determine central corneal curvature. Both keratometry and topography based on reflection of Placido rings are not accurate, usually underestimate the actual central curvature of the anterior surface of the cornea and the total corneal power. As a consequence, hyperopia is common after cataract surgery in these patients, especially in eyes previously submitted to myopia correction [1–3, 7, 8, 15].
Corneal tomography can be used to scan the entire corneal surface, including its geometric center, which provides real measurements of the corneal central curvature. The calculation of the central corneal power after refractive surgery (post-K) by a quantitative area topography using Orbscan and Pentacam has been shown to provide better biometric results [4–6, 9–13, 16, 17].
Our results also showed a very precise IOL calculation when using central corneal power derived from Orbscan II as the post-K value and 43.8D as pre-K value into the double-K method, especially in eyes previously submitted to myopic correction. This approach provided a significantly better refraction outcome than that obtained using post-K values from either keratometry or corneal topography.
In the eyes previously submitted to hyperopic correction, we also found significant better SE results when compared to post-K from keratometry. When comparing with post-K values derived from topography results were also better, although without significance, which could be explained by the small number of patients in this group.
On the other hand, had we used pre-K value derived from the 8 mm area from Orbscan (instead of 43.8D), we would probably get similar refractive outcomes in patients previously submitted to myopic or hyperopic refractive surgery.
As for ACD measurement, we found no difference in the IOL calculation using the values from IOL Master or from Orbscan II. Our results revealed a good level of agreement between these two optical devices, just like the results reported by Reddy and coauthors [18]. Therefore, we believe that either one can provide reliable values.
The application of the double-K method [7], which considers the effective position of the IOL, produced refractive results that are closer to emmetropia, and which are in accordance with those described by other authors [2, 3, 8, 15, 19].
In recent years, many formulas have been described by several authors always trying to improve IOL power calculation after refractive surgery, such as Borasio [6], Shammas [15, 20], Haigis [21], and Masket [22]. They all showed good results, but their series did not include such a large number of patients submitted to radial keratotomy, and both myopic and hyperopic laser procedures.
Conclusion
Our study showed that the post-K value derived from the Orbscan II is a very good alternative to be inputted into the double K method to improve IOL power calculation in cataract patients previously submitted to radial keratotomy, myopic Lasik, and hyperopic laser surgeries. Compared to post-K values derived from keratometry and topography, this method seems to produce better refractive outcomes, especially in patients previously submitted to myopic correction.
References
Koch DD, Liu JF, Hyde LL, Rock RL, Emery JM (1989) Refractive complications of cataract surgery after radial keratotomy. Am J Ophthalmol 108:676–682
Gimbel HV, Sun R (2001) Accuracy and predictability of intraocular lens power calculation after laser in situ keratomileusis. J Cataract Refract Surg 27:571–576
Hoffer KJ (2009) Intraocular lens power calculation after previous laser refractive surgery. J Cataract Refract Surg 35:759–765
Ho JD, Liou SW, Tsai RJF, Tsai CY (2008) Estimation of effective lens position using a rotating Scheimpflug camera. J Cataract Refract Surg 34:2119–2127
Qazi MA, Cua IY, Roberts CJ, Pepose JS (2007) Determining corneal power using Orbscan II videokeratography for intraocular lens calculation after excimer laser surgery for myopia. J Cataract Refract Surg 33:21–30
Borasio E, Stevens J, Smith GT (2006) Estimation of true corneal power after keratorefractive surgery in eyes requiring cataract surgery: BESSt formula. J Cataract Refract Surg 32:2004–2014
Aramberri J (2003) Intraocular lens power calculation after corneal refractive surgery: double-K method. J Cataract Refract Surg 29:2063–2068
Feiz V, Mannis MJ, Garcia-Ferrer F, Kandavel G, Darlington JK, Kim E, Caspar J, Wang W (2001) Intraocular lens power calculation after laser in situ keratomileusis for myopia and hyperopia: a standardized approach. Cornea 20:792–797
Sónego-Krone S, López-Moreno G, Beaujon-Balbi OV, Arce CG, Schor P, Campos M (2004) A direct method to measure the power of the central cornea after myopic laser in situ keratomileusis. Arch Ophthalmol 122:159–166
Maidana EJ, Alzamora JB, Arce CG, Schor P, Campos M (2005) Método para determinar cuál era el poder central de la córnea antes de la cirugía refractiva de miopía. Presented at: XXV Pan American Congress of Ophthalmology, March 18-21,Santiago, Chile
Gelender H (2006) Orbscan II-assisted intraocular lens power calculation for cataract surgery following myopic laser in situ keratomileusis. Trans Am Ophthalmol Soc 104:402–413
Arce CG, Soriano ES, Weisenthal RW, Hamilton SM, Rocha KM, Alzamora JB, Maidana EJ, Vadrevu VL, Himmel K, Schor P, Campos M (2009) Calculation of intraocular lens power using Orbscan II quantitative area topography after corneal refractive surgery. J Refract Surg 25:1061–1074
Savini G, Barboni P, Profazio V (2008) Corneal power measurements with the Pentacam Scheimpflug camera after myopic excimer laser surgery. J Cataract Refract Surg 34:809–813
Packer M, Brown LK, Hoffman RS, Fine IH (2004) Intraocular lens power calculation after incisional and thermal keratorefractive surgery. J Cataract Refract Surg 30:1430–1434
Shammas HJ, Shammas MC, Garabet A, Kim JH, Shammas A, LaBree L (2003) Correcting the corneal power measurements for intraocular lens power calculations after myopic laser in situ keratomileusis. Am J Ophthalmol 136:426–432
Garg A (2005) Optical biometry with IOL Master. In: Garg A, Hoyos JE, Dementiev D (eds) Mastering techniques of IOL power calculations. Jaypee Brothers, New Delhi, pp 51–54
Cairns G, McGhee CNJ (2005) Orbscan computerized topography: attributes, applications, and limitations. J Cataract Refract Surg 31:205–220
Reddy AR, Pande MV, Finn P, El-Gogary H (2004) Comparative estimation of anterior chamber depth by ultrasonography, Orbscan II, and IOL Master. J Cataract Refract Surg 30:1268–1271
Wang L, Booth MA, Koch DD (2004) Comparison of intraocular lens power calculation methods in eyes that have undergone LASIK. Ophthalmology 111:1825–1831
Shammas HJ, Shammas MC (2007) No-history method of intraocular lens power calculation for cataract surgery after myopic laser in situ keratomileusis. J Cataract Refract Surg 33:31–36
Haigis W (2008) Intraocular lens calculation after refractive surgery for myopia: Haigis-L formula. J Cataract Refract Surg 34:1658–1663
Masket S, Masket SE (2006) Simple regression formula for intraocular lens power adjustment in eyes requiring cataract surgery after excimer laser photoablation. J Cataract Refract Surg 32:430–434
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors do not have any financial interest.
Rights and permissions
About this article
Cite this article
Kwitko, S., Marinho, D.R., Rymer, S. et al. Orbscan II and double-K method for IOL calculation after refractive surgery. Graefes Arch Clin Exp Ophthalmol 250, 1029–1034 (2012). https://doi.org/10.1007/s00417-012-1974-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-012-1974-z