Abstract
Background
Huntington disease is a fatal inherited neurodegenerative disease. Because the end result of Huntington disease is death due to Huntington disease-related causes, there is a need for better understanding and caring for individuals at their end of life.
Aim
The purpose of this study was to develop a new measure to evaluate end of life planning.
Design
We conducted qualitative focus groups, solicited expert input, and completed a literature review to develop a 16-item measure to evaluate important aspects of end of life planning for Huntington disease. Item response theory and differential item functioning analyses were utilized to examine the psychometric properties of items; exploratory factor analysis was used to establish meaningful subscales.
Participants
Participants included 508 individuals with pre-manifest or manifest Huntington disease.
Results
Item response theory supported the retention of all 16 items on the huntington disease quality of life (“HDQLIFE”) end of life planning measure. Exploratory factor analysis supported a four-factor structure: legal planning, financial planning, preferences for hospice care, and preferences for conditions (locations, surroundings, etc.) at the time of death. Although a handful of items exhibited some evidence of differential item functioning, these items were retained due to their relevant clinical content. The final 16-item scale includes an overall total score and four subscale scores that reflect the different end of life planning constructs.
Conclusions
The 16-item HDQLIFE end of life planning measure demonstrates adequate psychometric properties; it may be a useful tool for clinicians to clarify patients’ preferences about end of life care.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Individuals with an expansion mutation in the huntingtin gene will eventually develop clinically significant symptoms of Huntington disease (HD), with average age of onset around 40 years. HD causes insidious and progressive motor, behavior and cognitive decline. As the typical course ranges from 15 to 20 years after diagnosis until death due to HD-related causes, it is important for individuals with HD and their families to plan for end of life care, and for physicians to understand late-stage HD well enough to provide effective counseling to these families [1]. Unfortunately, few studies have focused on end of life (EOL) in HD. Talking about EOL and death and dying can be uncomfortable for the patient, the family, and the clinician; [2] research suggests that individuals with HD often do not discuss their EOL thoughts or wishes with physicians [3]. Many physicians neglect to initiate and even avoid these discussions, despite data suggesting that they recognize the importance of discussing EOL options with all patients, particularly those with a terminal disease [4, 5]. Furthermore, hospice care is underutilized in individuals with HD and those that are engaged in these programs are enrolled longer than other clinical groups and tend to die in a long-term care facility rather than at home (this is regardless of an emphasis for hospice care to enable patients to die at home) [6]. Improving the dialogue between patients and physicians about EOL should be an essential component for caring treatment of these patients [3]. To this end, a brief assessment tool with patient-reported EOL preferences and planning may provide an avenue for physicians, social workers, nursing and palliative care staff, and patients to initiate these difficult conversations.
Position papers and guidelines for clinical care at the EOL constitute the bulk of the published literature on EOL in HD [5, 7,8,9,10]. Papers that address the quality of EOL care focus on the important issue of family-reported satisfaction [11], but do not consider the patient’s perspective. A few studies have gathered patient data about EOL concerns but only as a secondary focus rather than a primary aim [3, 12,13,14]. These few studies provide evidence that EOL issues contribute to health-related quality of life [12, 13]. Not surprisingly, the few studies that focused solely on EOL in HD reported that preferences for EOL care and planning varied widely. Some individuals had clear preferences about EOL and took steps to articulate these preferences in the form of advance directives and euthanasia. Others had only general ideas, particularly about circumstances to avoid, but may not have made these desires known to others. Finally, some may be unable to articulate clear preferences [3, 14].
Further, while concerns have been raised about advanced care planning and associated increases in anxiety [15], this has not been addressed in research. Specifically, there is research in the general aging population [16] as well as in patients with cancer [17, 18] to suggest that advance care planning may reduce family stress, anxiety, and depression. Furthermore, a detailed systematic review of advance care planning across clinical populations reported that advance care planning either did not change or improved quality of life (i.e., no evidence suggested that advance care planning diminished HRQOL) [19]. Therefore, articulating end of life preferences likely has a positive downstream effect on the health-related quality of life (HRQOL) for individuals with HD and their families.
Thus, the purpose of this study was to develop a new measure to evaluate EOL preferences for individuals with HD. The state-of-the-science approach to the development and validation of this measure will ultimately improve EOL care for individuals with HD.
Methods
Individuals with pre-manifest (i.e., those with the CAG expansion for HD but no clinical diagnosis) or manifest HD were invited to participate in this study. Individuals were at least 18 years of age, able to read and comprehend English, capable of providing informed consent and had either a positive test for the CAG expansion for HD or a clinical diagnosis of HD. Participants were recruited through HD treatment centers (University of Michigan, University of Iowa, University of California-Los Angeles, Indiana University, Johns Hopkins University, Rutgers University, Struthers Parkinson’s Center, and Washington University in St. Louis), the Predict-HD study which encompasses a well-characterized, pre-manifest HD sample (Cleveland Clinic, Indiana University, Johns Hopkins University, University of California-San Francisco, University of Iowa, and Washington University) [20,21,22], electronic medical record review [23], community outreach (HD support groups and HD specialized nursing homes in Phoenix, AZ; Tucson, AZ; Denver, CO; Jacksonville, FL; Des Moines, IA; Louisville, KY; Lansing, MI, Robbinsdale, MN; Lakewood, NJ; Plainfield, NJ; New York City, NY; Dallas, TX; Seattle, WA), the National Research Roster for Huntington Disease, and articles/advertisements in HD-specific newsletters and websites. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards (University of Michigan Medical School Institutional Review Board, HUM00055669, approved 02/01/2012; Cleveland Clinic Institutional Review Board, IRB 13-460, approved 04/26,/2017; Indiana University Institutional Review Board [IRB-01], Protocol 1208009383, approved 09/07/2012; Johns Hopkins Medicine Institutional Review Board, Study NA_00079341, approved 12/13/2012; University of Medicine and Dentistry of New Jersey, subsumed by Rutgers University, Institutional Review Board, Study ID Pro2012002196, approved 04/04/2013; Park Nicollet Institutional Review Board, Study 04334-13-A, approved 11/15/2013; University of California San Francisco Institutional Review Board, IRB 13-10880 Reference 065701, approved 09/04/2013; University of California Los Angeles Institutional Review Board, IRB 12-000743, approved 06/12/2012; University of Iowa Institutional Review Board, IRB ID 201301724, approved 01/17/2013; and Washington University St. Louis Institutional Review Board, IRB ID 201206052, approved 08/14/2012). Detailed study protocol information is reported elsewhere [24]. Briefly, participants completed both an in-person clinical assessment and a series of self-report measures using an online data capture platform (i.e., assessmentcenter.net). The EOL measures that are the focus of the current report are described below.
HDQLIFE end of life planning
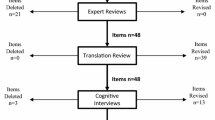
The HDQLIFE EOL Planning measure is comprised of 16 items developed from focus group data, expert input, and a comprehensive literature review (see “Appendix”) [25]. Items were refined through an iterative process using expert review, translatability review, and cognitive interviews with individuals with HD following established methodological standards for new measurement development. Items also underwent a literacy review using the Lexile framework [26] to ensure that the items did not exceed a fifth grade reading level. This measure was completed as part of the online self-report assessment battery.
Participant characterization
Participants were characterized as either pre-manifest or manifest HD using data collected through the in-person clinical assessment. Specifically, pre-manifest HD participants had a positive test for the CAG expansion that causes HD (i.e., CAG > 35), but no clinical diagnosis (which was determined using the final question on the Total Motor Scale from the Unified Huntington Disease Rating Scale [UHDRS; 27] which asks the clinician to indicate whether or not the participant has motor manifestations that are unequivocal signs of manifest HD; if the rater did not feel with at least 99% confidence that the participant had manifest HD, the participant was classified as pre-manifest). Manifest HD participants had a clinical diagnosis of HD (confirmed by medical record review, and a 99% or greater confidence rating score by a clinician on the final question on the Total Motor Scale from the UHDRS [27]). Manifest participants were classified as either early- or late HD using the 5-item clinician-administered Total Functional Capacity (TFC) Scale [28] from the Unified Huntington Disease Rating Scale (UHDRS) [27]. The TFC evaluates day-to-day functioning for occupation, finances, domestic chores, activities of daily living, and care level. Scores range from 0 to 13, with higher scores indicating better functioning. Participants with an HD diagnosis and TFC sum scores of 7–13 were classified as early-stage HD (stages I and II) and those with an HD diagnosis and TFC sum scores of 0–6 were classified as late HD (stages II, IV, and V) [29]. TFC data was missing for two participants who could not be characterized.
Analysis approach
Item response theory (IRT) analyses
The 16 items from HDQLIFE EOL planning were evaluated using Samejima’s graded response model (GRM) [30] in IRTPRO 2.1 [31]. In this two-parameter logistic IRT model, item responses were used to estimate the “measure” (the person’s score on the latent trait). The two parameters are item location on the latent trait and item slope, which indicates how well the item discriminates (distinguishes) between person differences across the latent trait [32]. Items demonstrating good model fit were retained, while items displaying significant misfit (S-X2, p < 0.01) were candidates for exclusion.
Differential item functioning (DIF) was used to evaluate stability of measurement properties between sub-groups using IRT-scaled score-based ordinal logistic regression; [33] DIF was evaluated on sex, age (≤ 40 vs. > 40; ≤ 50 vs. > 50 years), and education (high school graduate or less vs. greater than high school). Items with DIF (non-negligible DIF criterion: R 2 > 0.02 and p < 0.01) were discussed by the study team and were candidates for exclusion.
Reliability and construct validity
Exploratory factor analysis (EFA) with a promax rotation was used to determine the number of factors within the item pool according to Eigenvalues (> 1) and the number of factors before the break in the scree plot. A promax rotation was used to determine items and their associated factor (criterion > 0.4) and inter-factor correlations. Each unidimensional set of items (determined by EFA) was then subjected to confirmatory factor analysis (CFA) to assess model fit using the second randomly generated dataset. CFA used the following criteria: (1) Comparative Fit Index (CFI) > 0.90, (2) root mean square error of approximation (RMSEA) < 0.1 [34,35,36,37], and (3) residual correlations < 0.15 [38,39,40]. An iterative process including clinical input was taken into account to finalize item exclusion/inclusion [38,39,40]. EFA and CFA were conducted using MPLUS 6.11 [41]. In addition, marginal reliabilities (i.e., IRT-based estimates of reliability) were examined to determine acceptable reliability of the measure (> 0.80).
Scoring
The IRT-scaled scores (theta) were converted into a standardized T score (mean 50, SD 10, referenced to the HD population represented by the current sample). Descriptive data for the different HD groups are provided, and one-way analysis of variance was used to determine if there were group differences for any of the subscale scores or the total score for this new measure.
Results
A large sample (n = 508) of individuals with pre-manifest (n = 197) or manifest HD (n = 195 early—[(41% stage I and 59% stage II) and n = 114 late-stage HD (73% stage III, 24% stage IV, and 3% stage V)] completed the 16 items. Detailed demographic data are reported in Table 1.
IRT analyses
IRT parameter estimates from the GRM indicated slopes ranged from 0.60 to 4.77 and thresholds ranged from − 2.80 to 7.91 (see Table 2); 11 of the 16 items examined using S-X2 model fit statistics had misfit statistics (p < 0.05). Information was good for scaled scores between − 1.5 and 1.0; see Fig. 1 for the scale information function. There was no DIF for age. Two items had elevated R 2 (i.e., > 0.02) and DIF (p < 0.01) for education (the items for Life Insurance and Finances) and four items had elevated R 2 (i.e., > 0.02) and DIF (p < 0.01) for gender (location of death preference, conversations about death and dying, funeral arrangements and preference about death). These items were discussed by the team as potential candidates for exclusion. Given that the team recognized that the content of the items was relevant and clinically meaningful, and that items were not consistently biased (i.e., for age, gender, and education), the team decided to retain these items in the final version of the measure. Thus, all 16 items were retained for inclusion in this scale.
HDQLIFE end of life planning and preparation short form. In general, we want total information to be > 9.0 and standard error to be < 0.33 (this provides a reliability of 0.9). This figure shows excellent total information and standard error for end of life planning and preparation short form scale scores between − 1.5 and 1.0
Reliability and construct validity
EFA indicated that the data could be explained by four factors (Table 3). The first factor included three items that generally represented legal planning for EOL; the second factor included three items concerning preferences for EOL care; the third factor included five items that generally represented preferences about death and dying; and the fourth factor included four items that generally represented financial planning. Information was good (i.e., marginal reliability = 0.86), for the 16-item scale. Marginal reliability for the individual factors was adequate to very good (0.85 for legal planning; 0.61 for preferences for care; 0.78 for preferences about death and dying; and 0.66 for financial planning).
Scoring
Table 4 provides a conversion table for the raw summed score to a scaled score and a conversion table for the four subscales. Lower scores indicate the presence of less planning and preparation on issues of EOL. The three HD groups did not differ on any of the EOL planning scores (all p > 0.25; Table 5).
Discussion
This study presents the development of a new patient-reported outcome measure to evaluate EOL planning and preparation in HD, the HDQLIFE EOL planning measure. This measure is a part of the HDQLIFE measurement system [24], which includes several HD-specific HRQOL measures [42, 43], as well as two HD-specific patient-reported outcomes of EOL concerns (concern with death and dying; meaning and purpose) [44]. This new measure provides a patient-reported assessment to foster end of life planning in this population and includes both a total score and four meaningful subscale scores, including legal planning, preferences for care, preferences for death and dying, and financial planning. The total and subdomain scores all use a T metric, with a mean of 50 and standard deviation of 10; higher scores indicate more preparation and planning. As such, a low total score, and/or low scores on any single subdomain (i.e., scores ≤ 40 or those individuals that have engaged in less planning and preparation than 68.27% of people with HD) might be used as either the starting point for a general discussion about EOL wishes (in the case of low total score) or for more targeted discussion about legal planning, preferences for care, preferences for death and dying, and/or financial planning.
This overall reliability of the HDQLIFE planning measure was very good, and subdomain scores ranged from adequate to very good. The reliability of Preferences for Care and Financial Planning subdomains were slightly lower than recommended for a new measure [45, 46], so additional consideration when using these scales is appropriate. Regardless, construct validity of these four factors was supported by EFA. There is evidence that individuals with HD often do not discuss their EOL thoughts or wishes with physicians.2 Thus, this study provides preliminary support for the reliability and validity of this new measure that can be used to foster productive and meaningful discussions about this sensitive topic in conjunction with established clinical guidelines of EOL care [2].
While this new measure provides a brief assessment of EOL planning and preparation, this study does have several limitations. First, reliability and validity data are preliminary; a more thorough evaluation of the psychometric properties of this measurement tool, and further analysis of the clinical utility of this tool should be examined to foster discussions about EOL planning. In addition, while item concepts are broad, legal issues related to end of life planning vary by state (within the United States), as well as by country; more work is needed to understand how these differences may impact the clinical utility of this tool. Furthermore, establishing generalizability of findings to a broader HD sample requires more work. Specifically, a large portion of study participants were recruited from specialized HD clinical centers, whereas most persons with HD do not attend such centers. Similarly, a large portion of participants were recruited through the PREDICT-HD study, which targets persons who have elected to undergo gene testing, something that only a small percentage of individuals at risk for HD typically do [47]. Furthermore, while our study examined individuals across the HD disease spectrum, the reliability of self-report data, especially in individuals in the later stages of the disease, has not been assessed. As such, future work that examines the accuracy of responses (e.g., data confirmation by other family members) for self-report responses in this population is warranted. Additional work is needed to determine when, and how frequently this measure might be used to optimize treatment discussions and clarify end of life planning preferences for these individuals and to better understand how patient-reported planning and preparation influences family-reported satisfaction regarding quality of care at the end of life.
Regardless of these limitations, this is the first HD-specific EOL planning and preparation measure. This brief measure demonstrates adequate psychometric properties and has the potential to improve clinical care at the end of life by providing an easy starting point for patient and clinician discussions about EOL care. Furthermore, although this measure was designed specifically for HD, it may also have clinical utility in other neurodegenerative conditions. As such, this provides an important step in the evaluation of patient preferences and planning in relationship to EOL care.
References
Nance MA, Sanders G (1996) Characteristics of individuals with Huntington disease in long-term care. Mov Disord 11(5):542–548
Kirwin JL, Edwards RA (2013) Helping patients articulate end-of-life wishes: a target for interprofessional participation. Ann Palliat Med 2(2):95–97
Booij SJ et al (2014) Thinking about the end of life: a common issue for patients with Huntington’s disease. J Neurol 261(11):2184–2191
Booij SJ et al (2013) A plea for end-of-life discussions with patients suffering from Huntington’s disease: the role of the physician. J Med Ethics 39(10):621–624
Booij SJ et al (2014) Perhaps the subject of the questionnaire was too sensitive: Do we expect too much too soon? Wishes for the end of life in Huntington’s Disease—the perspective of European physicians. J Huntingtons Dis 3(3):229–232
Johnson MO, Frank S, Mendlik M, Casarett D (2017) Utilization of hospice services in a population of patients with Huntington’s Disease. J Pain Symptom Manage. https://doi.org/10.1016/j.jpainsymman.2017.09.004
Dellefield ME, Ferrini R (2011) Promoting excellence in end-of-life care: lessons learned from a cohort of nursing home residents with advanced Huntington disease. J Neurosci Nurs 43(4):186–192
Dubinsky R et al (2004) Lifting the veil of Huntington’s disease: Recommendations to the field from the Huntington’s disease Peer Workgroup. Roberrt Wood Johnson Foundation, Princeton
Klager J et al (2008) Huntington’s disease: a caring approach to the end of life. Care Manag J 9(2):75–81
Mullahy CM, Jensen DK (2005) End-of-life care: a special calling for case managers. Case Manag 16(1):40–42
Wachterman MW et al (2016) Quality of end-of-life care provided to patients with different serious illnesses. JAMA Intern Med 176(8):1095–1102
Carlozzi NE, Tulsky DS (2012) Identification of health-related quality of life (HRQOL) issues relevant to individuals with Huntington disease. J Health Psychol
Ho AK, Hocaoglu MB (2009) The impact of huntington’s on quality of life Huntington’s on quality of life: a survey across disease stages using the HDQ-D1. Clin Genet. https://doi.org/10.1111/j.1399-0004.2009.01225.x
Booij SJ et al (2013) Euthanasia and physician-assisted suicide in Huntington’s disease in The Netherlands. Int Psychogeriatr 25(2):339–340
Perkins HS (2007) Controlling death: the false promise of advance directives. Ann Intern Med 147(1):51–57
Detering KM et al (2010) The impact of advance care planning on end of life care in elderly patients: randomised controlled trial. BMJ 340:c1345
Voogt E et al (2005) Attitudes of patients with incurable cancer toward medical treatment in the last phase of life. J Clin Oncol 23(9):2012–2019
Lyon ME et al (2014) A longitudinal, randomized, controlled trial of advance care planning for teens with cancer: anxiety, depression, quality of life, advance directives, spirituality. J Adolesc Health 54(6):710–717
Brinkman-Stoppelenburg A, Rietjens JA, van der Heide A (2014) The effects of advance care planning on end-of-life care: a systematic review. Palliat Med 28(8):1000–1025
Paulsen JS et al (2006) Preparing for preventive clinical trials: the predict-HD study. Arch Neurol 63(6):883–890
Paulsen JS et al (2008) Detection of Huntington’s disease decades before diagnosis: the predict-HD study. J Neurol Neurosurg Psychiatry 79(8):874–880
Paulsen JS et al (2014) Clinical and biomarker changes in premanifest Huntington disease show trial feasibility: a decade of the PREDICT-HD study. Front Aging Neurosci 6:78
Hanauer DA et al (2015) Supporting information retrieval from electronic health records: a report of University of Michigan’s nine-year experience in developing and using the Electronic Medical Record Search Engine (EMERSE). J Biomed Inform 55:290–300
Carlozzi NE et al (2016) HDQLIFE: development and assessment of health-related quality of life in Huntington disease (HD). Qual Life Res 25(10):2441–2455
Carlozzi NE, Tulsky DS (2013) Identification of health-related quality of life (HRQOL) issues relevant to individuals with Huntington disease. J Health Psychol 18(2):212–225
MetaMetrics (1995) The LEXILE framework for reading. MetaMetrics Inc, Durham
Huntington Study Group (1996) Unified Huntington’s Disease Rating Scale: reliability and consistency. Mov Disord 11(2):136–142
Shoulson I, Fahn S (1979) Huntington disease—clinical care and evaluation. Neurology 29(1):1–3
Marder K et al (2000) Rate of functional decline in Huntington’s disease. Huntington Study Group. Neurology 54(2):452–458
Samejima F, van der Liden WJ, Hambleton R (1996) The graded response model. In: van der Liden WJ (ed) Handbook of modern item response theory. Springer, NY, pp 85–100
Cai L, Thissen D, du Toit SHC (2011) IRTPRO for Windows [Computer software]. Scientific Software International, Lincolnwood
van der Linden WJ, Hambleton RK (1997) Handbook of modern item response theory. Springer, New York
Crane PK et al (2006) Differential item functioning analysis with ordinal logistic regression techniques. DIFdetect and difwithpar. Med Care 44(11 Suppl 3):S115–S123
Kline RB (2005) Principles and practice of structural equation modeling, 2nd edn. Guilford Press, New York
Bentler PM (1990) Comparative fit indexes in structural models. Psychol Bull 107(2):238–246
Hu LT, Bentler PM (1999) Cutoff criteria for fit indexes in covariance structure analysis: conventional criteria versus new alternatives. Struct Equ Model Multidiscip J 6(1):1–55
Hatcher L (1994) A step-by-step approach to using SAS for factor analysis and structural equation modeling. SAS Institute Inc, Cary
McDonald RP (1999) Test theory: a unified treatment. Lawrence Erlbaum Associates Inc, Mahwah
Reise SP, Morizot J, Hays RD (2007) The role of the bifactor model in resolving dimensionality issues in health outcomes measures. Qual Life Res 16(Suppl 1):19–31
Cook KF, Kallen MA, Amtmann D (2009) Having a fit: impact of number of items and distribution of data on traditional criteria for assessing IRT’s unidimensionality assumption. Qual Life Res 18(4):447–460
Muthén LK, Muthén BO (2011) Mplus User’s Guide. Muthén & Muthén, Los Angeles
Carlozzi NE et al (2016) HDQLIFE: the development of two new computer adaptive tests for use in Huntington disease, Speech Difficulties, and Swallowing Difficulties. Qual Life Res 25(10):2417–2424
Carlozzi NE et al (2016) The development of a new computer adaptive test to evaluate chorea in Huntington disease: HDQLIFE Chorea. Qual Life Res 25(10):2429–2439
Carlozzi NE et al (2016) New measures to capture end of life concerns in Huntington disease: Meaning and Purpose and Concern with Death and Dying from HDQLIFE (a patient-reported outcomes measurement system). Qual Life Res 25(10):2403–2415
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Academic Press, New York
DeVellis R (2017) Scale development: theory and applications. In: Bickman L, Rog DJ (eds) Applied social research methods series, 4th edn. Sage, Los Angeles
Tibben A (2007) Predictive testing for Huntington’s disease. Brain Res Bull 72(2–3):165–171
Zhang Y et al (2011) Indexing disease progression at study entry with individuals at-risk for Huntington disease. Am J Med Genet Part B Neuropsychiatr Genet 156B(7):751–763
Acknowledgments
Work on this manuscript was supported by the National Institutes of Health (NIH), National Institute of Neurological Disorders and Stroke (R01NS077946) and the National Center for Advancing Translational Sciences (UL1TR000433). In addition, a portion of this study sample was collected in conjunction with the Predict-HD study. The Predict-HD data were supported by the NIH, National Institute of Neurological Disorders and Stroke (R01NS040068), the NIH, Center for Inherited Disease Research (provided support for sample phenotyping), and the CHDI Foundation (award to the University of Iowa). We thank the University of Iowa, the Investigators and Coordinators of this study, the study participants, the National Research Roster for Huntington Disease Patients and Families, the Huntington Study Group, and the Huntington Disease Society of America. We acknowledge the assistance of Jeffrey D. Long, Hans J. Johnson, Jeremy H. Bockholt, Roland Zschiegner, and Jane S. Paulsen. We also acknowledge Roger Albin, Kelvin Chou, and Henry Paulsen for the assistance with participant recruitment. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
HDQLIFE Site Investigators and Coordinators: Noelle Carlozzi, Praveen Dayalu, Stephen Schilling, Amy Austin, Matthew Canter, Siera Goodnight, Jennifer Miner, Nicholas Migliore (University of Michigan, Ann Arbor, MI); Jane Paulsen, Nancy Downing, Isabella DeSoriano, Courtney Shadrick, Amanda Miller (University of Iowa, Iowa City, IA); Kimberly Quaid, Melissa Wesson (Indiana University, Indianapolis, IN); Christopher Ross, Gregory Churchill, Mary Jane Ong (Johns Hopkins University, Baltimore, MD); Susan Perlman, Brian Clemente, Aaron Fisher, Gloria Obialisi, Michael Rosco (University of California Los Angeles, Los Angeles, CA); Michael McCormack, Humberto Marin, Allison Dicke (Rutgers University, Piscataway, NJ); Joel S. Perlmutter, Stacey Barton, Shineeka Smith (Washington University, St. Louis, MO); Martha Nance, Pat Ede (Struthers Parkinson’s Center); Stephen Rao, Anwar Ahmed, Michael Lengen, Lyla Mourany, Christine Reece, (Cleveland Clinic Foundation, Cleveland, OH); Michael Geschwind, Joseph Winer (University of California-San Francisco, San Francisco, CA), David Cella, Richard Gershon, Elizabeth Hahn, Jin-Shei Lai (Northwestern University, Chicago, IL).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
Carlozzi, N. E. currently has research Grants from the NIH; she is also supported by Grant funding from the NIH and CHDI. She provides patient-reported outcome measurement selection and application consultation for Teva Pharmaceuticals. She declares no conflicts of interest. Hahn, E. A. currently has research Grants from the NIH; she is also supported by Grant funding from the NIH and PCORI, and by research contracts from Merck and EMMES; she declares no conflicts of interest. Frank, S. receives salary support from the Huntington Study Group for a study sponsored by Auspex Pharmaceuticals. There is no conflict of interest. Perlmutter, J. S. currently has funding from the NIH, HDSA, CHDI, and APDA. He has received honoraria from the University of Rochester, American Academy of Neurology, Movement Disorders Society, Toronto Western Hospital, St. Luke’s Hospital in St Louis, Emory University, Penn State, Alberta innovates, Indiana Neurological Society, Parkinson Disease Foundation, Columbia University, St. Louis University, Harvard University and the University of Michigan; he declares no conflicts of interest. Downing, N. R. declares no conflicts of interest. McCormack, M. K. currently has Grants from the NJ Department of Health; he declares no conflicts of interest. Barton, S. K. is supported by grant funding from the Huntington Disease Society of America, CHDI Foundation and the NIH. She declares no conflicts of interest. Nance, M. A. declares no conflicts of interest. Schilling, S. G. has a research Grant from NSF. He also is supported by Grant funding from NIH. He declares no conflicts of interest.
Appendix
Appendix
HDQLIFE end of life planning |
|---|
1. Advance directive |
0 = I have not thought about getting an advance directive |
1 = I have thought about getting an advance directive |
2 = I have taken steps to obtain an advance directive |
3 = I have an advance directive |
2. Health care power of attorney |
0 = I have not thought to getting a health care power of attorney |
1 = I thought about getting a health care power of attorney |
2 = I have taken steps to identify a health care power of attorney |
3 = I have a health care power of attorney |
3. Nursing home care |
0 = I have not thought about living in a nursing home |
1 = I have thought about the type of nursing home I would like to go to |
2 = I have taken steps to arrange nursing home care |
3 = I have established nursing home care |
4. Location of death preference |
0 = I have not thought about where I would like to die (i.e., at home, in the hospital) |
1 = I have thought about where I would like to die (i.e., at home, in the hospital) |
2 = I have taken steps to arrange where I would like to die (i.e., at home, in the hospital) |
3 = I have identified a location where I would like to die (i.e., at home, in the hospital) |
5. Conversations about death and dying |
0 = I have not thought about starting a conversation about death with my friends, family or members in the community (e.g., church/synagogue) |
1 = I have thought about starting a conversation about death with my friends, family or members in the community (e.g., church/synagogue) |
2 = I have taken steps to start a conversation about death with my friends, family or members in the community (e.g., church/synagogue) |
3 = I have has a conversation about death with my friends, family or members in the community (e.g., church/synagogue) |
6. Living will |
0 = I not thought about getting a living will |
1 = I have thought about getting a living will |
2 = I have taken steps to prepare a living will |
3 = I have a living will |
7. Life insurance |
0 = I have not thought about getting life insurance |
1 = I have thought about getting life insurance |
2 = I have taken steps to get life insurance |
3 = I have life insurance |
8. Palliative care (management of pain, symptoms and stress) |
0 = I have not thought about palliative care |
1 = I have thought about palliative care |
2 = I have taken steps to arrange palliative care |
3 = I am receiving palliative care |
9. Child care planning |
Not applicable |
0 = I have not thought about the care of my children if I become unable to care for them |
1 = I have thought about the care of my children if I become unable to care for them |
2 = I have taken steps to arrange care for my children if I become unable to care for them |
3 = My children are receiving care from others because I am no longer able to care for them |
10. Finances |
0 = I not thought about the necessary financial resources for my long-term care |
1 = I have thought about the necessary financial resources for my long-term care |
2 = I have taken steps to arrange for the necessary financial resources for my long-term care |
3 = I have the necessary financial resources for my long-term care |
11. Estate planning |
0 = I have not thought about what will happen to my estate after my death |
1 = I have thought about what will happen to my estate after my death |
2 = I have made plans for my estate after my death |
12. Support to make decisions |
0 = I have not thought about who will help me make decisions (e.g., financial, health/medical) if I am unable to |
1 = I have thought about who will help me make decisions (e.g., financial, health/medical) if I am unable to |
2 = I have arranged for someone to help me make decisions (e.g., financial, health/medical) if I am unable to |
13. Hospice care |
0 = I have not thought about hospice care |
1 = I have thought about hospice care |
2 = I have taken steps to arrange hospice care |
3 = I am receiving hospice care |
14. Rescusitation preference |
0 = I have not thought about my preference for resuscitation if I stop breathing |
1 = I have thought about my desires for resuscitation if I stop breathing |
2 = I have made my desires about resuscitation clear to others if I stop breathing |
15. Funeral arrangements |
0 = I have not thought about what others should do with my body after I die (e.g., burial, cremation) |
1 = I have thought about what others should do with my body after I die (e.g., burial, cremation) |
2 = I have made arrangements about what others should do with my body after I die (e.g., burial, cremation) |
16. Preference about death |
0 = I have not thought about how I would like to die |
1 = I have thought about how I would like to die |
2 = I have told others of my preferences regarding how I would like to die |
Rights and permissions
About this article
Cite this article
Carlozzi, N.E., Hahn, E.A., Frank, S.A. et al. A new measure for end of life planning, preparation, and preferences in Huntington disease: HDQLIFE end of life planning. J Neurol 265, 98–107 (2018). https://doi.org/10.1007/s00415-017-8677-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8677-7