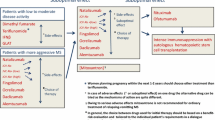
Abstract
Immunotherapy for multiple sclerosis (MS) has developed extremely successfully during the past decade and a number of new strategies were developed for the treatment of the disease. Examples include therapeutic strategies targeting leukocyte differentiation molecules, costimulatory molecules, anti-adhesion molecules, chemotaxis, novel immunomodulators, autologous stem cell transplantation, anti-infectious therapies and strategies for neuroprotection, neurorepair and remyelination. Here we describe examples of monoclonal antibodies, a novel immunosuppressant and interesting neuroprotective strategies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bar-Or A, Calabresi PA, Arnlod D,Markowitz C, Shafer S, Kasper LH,Waubant E, Gazda S, Fox RJ, PanzaraM, Sarkar N, Agarwal S, Smith CH(2008) Rituximab in relapsing-remittingmultiple sclerosis: a 72-week,open-label, phase I trial. Ann Neurol63:395–400
Beutler E (1992) Cladribine (2-chlorodeoxyadenosine).Lancet 340:952–956
Beutler E, Sipe JC, Romine JS, KoziolJA, McMillan R, Zyroff J (1996) Thetreatment of chronic progressivemultiple sclerosis with cladribine. ProcNatl Acad Sci USA 93:1716–1720
Bielekova B, Catalfamo M, Reichert-Scrivner S, Packer A, Cerna M, WaldmannTA, McFarland H, Henkart PA,Martin R (2006) RegulatoryCD56(bright) natural killer cells mediateimmunomodulatory effects ofIL-2Ralpha-targeted therapy (daclizumab)in multiple sclerosis. Proc NatlAcad Sci USA 103:5941–5946
Bielekova B, Richert N, Howard T,Blevins G, Markovic-Plese S, McCartinJ, Frank JA, Wurfel J, Ohayon J, WaldmannTA, McFarland HF, Martin R(2004) Humanized anti-CD25 (daclizumab)inhibits disease activity inmultiple sclerosis patients failing torespond to interferon beta. Proc NatlAcad Sci USA 101:8705–8708
Brinkmann V, Davis MD, Heise CE,Albert R, Cottens S, Hof R, Bruns C,Prieschl E, Baumruker T, Hiestand P,Foster CA, Zollinger M, Lynch KR(2002) The immune modulatorFTY720 targets sphingosine 1-phosphatereceptors. J Biol Chem 277:21453–21457
Chiba K (2005) FTY720, a new class ofimmunomodulator, inhibits lymphocyteegress from secondary lymphoidtissues and thymus by agonistic activityat sphingosine 1-phosphate receptors.Pharmacol Ther 108:308–319
Coles A, Group TCS (2007) Efficacy ofAlemtuzumab in Treatment-NaiveRelapsing-Remitting Multiple Sclerosis:Analysis after Two Years of StudyCAMMS223. Neurology 68(Suppl 1):A100
Coles AJ, Wing M, Smith S, Coraddu F,Greer S, Taylor C, Weetman A, Hale G,Chatterjee VK, Waldmann H, CompstonA (1999) Pulsed monoclonal antibodytreatment and autoimmune thyroiddisease in multiple sclerosis.Lancet 354:1691–1695
Coles AJ, Wing MG, Molyneux P,Paolillo A, Davie CM, Hale G, Miller D,Waldmann H, Compston A (1999)Monoclonal antibody treatment exposesthree mechanisms underlyingthe clinical course of multiple sclerosis.Ann Neurol 46:296–304
Filippi M, Rovaris M, Iannucci G,Mennea S, Sormani MP, Comi G (2000)Whole brain volume changes in patientswith progressive MS treated withcladribine. Neurology 55:1714–1718
Filippi M, Rovaris M, Rice GP, SormaniMP, Iannucci G, Giacomotti L, Comi G(2000) The effect of cladribine on T(1)’black hole’ changes in progressive MS.J Neurol Sci 176:42–44
Fujino M, Funeshima N, Kitazawa Y,Kimura H, Amemiya H, Suzuki S, LiXK (2003) Amelioration of experimentalautoimmune encephalomyelitis inLewis rats by FTY720 treatment.J Pharmacol Exp Ther 305:70–77
Hauser SL, Waubant E, Arnold DL,Vollmer T, Antel J, Fox RJ, Bar-Or A,Panzara M, Sarkar N, Agarwal S,Langer-Gould A, Smith CH (2008) B-celldepletion with rituximab in relapsing-remitting multiple sclerosis.N Engl J Med 358:676–688
Hohlfeld R, Wekerle H (2004) Autoimmuneconcepts of multiple sclerosisas a basis for selective immunotherapy:from pipe dreams to (therapeutic)pipelines. Proc Natl Acad Sci USA 101:14599–14606
Kappos L, Radu EW, Antel J (2005)Promising results with a novel oralimmunomodulator-FTY720-in relapsingmultiple sclerosis. Mult Scler 11:S13
Kleinschnitz C, Meuth SG, Kieseier BC,Wiendl H (2007) Immunotherapeuticapproaches in MS: update onpathophysiology and emerging agentsor strategies 2006. Endocr MetabImmune Disord Drug Targets 7:35–63
McFarland HF (2008) The B cell – oldplayer, new position on the team.N Engl J Med 358:664–665
Rice GP, Filippi M, Comi G (2000)Cladribine and progressive MS: clinicaland MRI outcomes of a multicentercontrolled trial. Cladribine MRI StudyGroup. Neurology 54:1145–1155
Romine JS, Sipe JC, Koziol JA, Zyroff J,Beutler E (1999) A double-blind, placebo-controlled, randomized trial ofcladribine in relapsing-remittingmultiple sclerosis. Proc Association ofAmerican Physician 111:35–44
Rose JW, Burns JB, Bjorklund J, Klein J,Watt HE, Carlson NG (2007) Daclizumabphase II trial in relapsing andremitting multiple sclerosis: MRI andclinical results. Neurology 69:785–789
Rose JW, Watt HE, White AT, CarlsonNG (2004) Treatment of multiple sclerosiswith an anti-interleukin-2 receptormonoclonal antibody. Ann Neurol56:864–867
Sakaguchi S (2005) Naturally arisingFoxp3-expressing CD25+CD4+ regulatoryT cells in immunological toleranceto self and non-self. Nat Immunol6:345–352
Sipe JC (2005) Cladribine for multiplesclerosis: review and current status.Expert Rev Neurother 5:721–727
Sipe JC, Romine JS, Koziol J, Zyroff J,McMillan R, Beutler E (1997) Cladribineimproves relapsing-remitting MS: adouble blind placebo controlled study.Neurology 48:A340
Sipe JC, Romine JS, Koziol JA,McMillan R, Zyroff J, Beutler E (1994)Cladribine in treatment of chronicprogressive multiple sclerosis [seecomments]. Lancet 344:9–13
Webb M, Tham CS, Lin FF, Lariosa-Willingham K, Yu N, Hale J, Mandala S,Chun J, Rao TS (2004) Sphingosine1-phosphate receptor agonists attenuaterelapsing-remitting experimentalautoimmune encephalitis in SJL mice.J Neuroimmunol 153:108–121
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest Sven G. Meuth received honoraria for lecturing and travel expenses for attending meetings from Bayer Healthcare/Bayer Vital and Merck Serono and serves as a consultant for Merck Serono. Heinz Wiendl has received honoraria for lecturing, travel expenses for attending meetings and financial support for research from Bayer Health Care, Biogen Idec, Merck Serono, Novartis, Sanofi Aventis, and TEVA. Christoph Kleinschnitz received honoraria for lecturing and travel expenses for attending meetings from Biogen Idec/Elan, Schering, Serono, Bayer and Sanofi Aventis.
Rights and permissions
About this article
Cite this article
Meuth, S.G., Kleinschnitz, C. & Wiendl, H. Recent clinical trials and future therapies. J Neurol 255 (Suppl 6), 93–96 (2008). https://doi.org/10.1007/s00415-008-6017-7
Issue Date:
DOI: https://doi.org/10.1007/s00415-008-6017-7