Abstract
Levodopa therapy, as originally established by George Cotzias [2, 3], is the most powerful treatment for Parkinson’s disease (PD). Levodopa’s toxicity to neurons in vitro has raised concerns if it might hasten the progression of PD, although in vivo animal studies suggest it may be neuroprotective.
Objective
To discuss the results of the ELLDOPA trial that was carried out to determine if levodopa therapy influences the rate of progression of Parkinson’s disease (PD).
Design
ELLDOPA was a multicenter, parallel-group, double-blind, dosage-ranging, randomized, controlled clinical trial.
Setting
Academic movement disorders clinics at 38 sites in the United States and Canada.
Patients
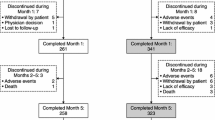
Three hundred and sixty-one patients with early PD of less than 2 years’ duration who did not require symptomatic therapy.
Interventions
Subjects were randomly assigned to one of four treatment groups: carbidopa/levodopa 12.5/50 mg t. i. d. (N=92), 25/100 mg t. i. d. (N=88), 50/200mg t. i. d. (N=91), or matching placebo (N=90). The dosage was gradually escalated over 9 weeks and then maintained until Week 40, at which time active treatment was withdrawn over 3 days. After 2 weeks without active treatment (Week 42), a final assessment of PD severity was obtained.
Outcome measures
The prespecified primary clinical outcome was the change in the total Unified Parkinson’s Disease Rating Scale (UPDRS) between baseline and Week 42, comparing the four treatment groups. The primary neuroimaging component of the study in a subgroup of 142 subjects was the percent change in striatal 123iodine 2-β-carboxymethoxy-3-β-(4-iodophe nyl)tropane (β-CIT) uptake between baseline and Week 40 visits. The neuroimaging substudy utilized single photon emission computed tomography (SPECT) of the dopamine transporter.
Results
All dosages of levodopa exerted clinical benefit compared to placebo on the UPDRS scores throughout the study, including 2 weeks after discontinuing levodopa. The UPDRS scores at Week 42 failed to reach the level encountered in the placebo group (change of 7.8±9.0, 1.9±6.0, 1.9±6.9, and –1.4±7.8, for placebo, 150 mg/day, 300 mg/ day, and 600 mg/day, respectively, p<0.0001). Nausea (p=0.001) and dyskinesias (p=0.0001) were more common in the levodopa groups, especially with the higher dosages. Freezing appeared around the same time, but was more common in the placebo (14 %) and 150 mg/day group (10 %). The percent decline of β-CIT uptake in the striatum was significantly more pronounced in the levodopa groups than the placebo group (–7.2%, –4%, –6%, and –1.4% in 600 mg/day, 300 mg/day, 150 mg/day, and placebo, respectively; p=0.035).
Conclusions
The clinical outcomes not only indicate that levodopa is effective in a dose-dependent manner in overcoming the signs and symptoms of PD, they also support the concept that the drug does not hasten the disease progression, but rather may slow down the rate of the disease. The clinical study failed to demonstrate any evidence of levodopa worsening early PD. However, the β-CIT SPECT substudy indicates the opposite effect, namely that levodopa causes a more rapid decline in the integrity of the dopamine transporter located in the nigrostriatal nerve terminals in the striatum. These contradictory findings warrant further investigation into the effect of levodopa on PD.
Other observations
The ELLDOPA study was the first levodopa dose-response study ever conducted. It showed that dose is a factor in the cause of producing motor complications of dyskinesias and wearing-off, and that these can develop as early as 5 to 6 months. On the other hand, freezing of gait could be delayed or its occurrence reduced by high dosage levodopa, compared to placebo or low-dose levodopa. Withdrawal of levodopa over a 3-day step-down can be safely carried out without inducing the neuroleptic-like syndrome. The UPDRS was shown to be a reliable linear marker for disease progression. The ELLDOPA study also called into question the interpretation of β-CIT SPECT in the presence of dopaminergic agents. Neuroimaging in ELLDOPA also showed that some people diagnosed with early PD do not have a dopaminergic deficit, calling into question how difficult the correct diagnosis may be in people with early symptoms of PD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fahn S, Przedborski S (2005) Parkinsonism. In: Rowland LP (ed) Merritt’s Neurology, 11th edn, Lippincott Williams and Wilkins, Philadelphia, pp 828–846
Riederer P, Wuketich S (1976) Time course of nigrostriatal degeneration in Parkinson’s disease. J Neural Transm 38:277–301
Hornykiewicz O (1982) Brain neuro-transmitter changes in Parkinson’s disease. In: Marsden CD, Fahn S (eds) Movement Disorders. Butterworth Scientific, London, pp 41–58
Cotzias GC, Van Woert MH, Schiffer LM (1967) Aromatic amino acids and modification of parkinsonism. N Engl J Med 276:374–379
Cotzias GC, Papavasiliou PS, Gellene R (1969) Modification of parkinsonism—chronic treatment with L-dopa. N Engl J Med 280:337–345
Miyasaki JM, Martin W, Suchowersky O, Weiner WJ, Lang AE (2002) Practice parameter: Initiation of treatment for Parkinson’s disease: An evidence-based review. Neurology 58:11–17
Fahn S (1989) Adverse effects of levodopa in Parkinson’s disease. In: Calne DB (ed) Drugs for the treatment of Parkinson’s Disease. Handbook of Experimental Pharmacology 88. Springer, Berlin, pp 385–409
Fahn S (1999) Parkinson disease, the effect of levodopa, and the ELLDOPA trial. Arch Neurol 56:529–535
Fahn S (1996) Is levodopa toxic? Neurology 47:S184–S195
Fahn S (1997) Levodopa-induced neurotoxicity: Does it represent a problem for the treatment of Parkinson’s disease? CNS Drugs 8:376–393
Agid Y (1998) Levodopa: is toxicity a myth? Neurology 50(4):858–863
Weiner WJ (1999) The initial treatment of Parkinson’s disease should begin with levodopa. Mov Disord 14(5):716–724
Factor SA (2000) The initial treatment of Parkinson’s disease. Mov Disord 15(2):360–361
Parkinson Study Group (2002) Dopamine transporter brain imaging to assess the effects of pramipexole vs levodopa on Parkinson disease progression. JAMA 287(13):1653–1661
Parkinson Study Group (2004) Pramipexole vs levodopa as initial treatment for Parkinson disease—A 4-year randomized controlled trial. Arch Neurol 61(7):1044–1053
Whone AL, Watts RL, Stoessl AJ, et al. (2003) Slower progression of Parkinson’s disease with ropinirole versus levodopa: The REAL-PET study. Ann Neurol 4(1):93–101
Rascol O, Brooks DJ, Korczyn AD, et al. (2000) A five-year study of the incidence of dyskinesia in patients with early Parkinson’s disease who were treated with ropinirole or levodopa. 056 Study Group. N Engl J Med 342(20):1484–1491
Morrish PK, Sawle GV, Brooks DJ (1996) An [F-18]dopa-PET and clinical study of the rate of progression in Parkinson’s disease. Brain 119:585–591
Marek K, Jennings D, Seibyl J (2002) Do dopamine agonists or levodopa modify Parkinson’s disease progression? Eur J Neurol 9(Suppl 3):15–22
Albin RL, Frey KA (2003) Initial agonist treatment of Parkinson disease: a critique. Neurology 60(3):390–394
Dauer W, Przedborski S (2003) Parkinson’s Disease: Mechanisms and Models. Neuron 39:889–909
Vila M, Przedborski S (2003) Targeting programmed cell death in neurodegenerative diseases. Nat Rev Neurosci 4(5):365–375
Fahn S, Sulzer D (2004) Neurodegeneration and neuroprotection in Parkinson disease. NeuroRx 1:139–154
Parkinson Study Group (2004) Levodopa and the progression of Parkinson’s disease. N Engl J Med 351(24):2498–2508
Parkinson Study Group (1993) Effects of tocopherol and deprenyl on the progression of disability in early parkinson’s disease. N Engl J Med 328:176–183
Parkinson Study Group (1996) Effect of lazabemide on the progression of disability in early Parkinson’s disease. Ann Neurol 40:99–107
Shults CW, Oakes D, Kieburtz K, Beal MF, Haas R, Plumb S, Juncos BL, Nutt J, Shoulson I, Carter J, Kompoliti K, Perlmutter JS, Reich S, Stern M, Watts RL, Kurlan R, Molho E, Harrison M, Lew M (2002) Effects of coenzyme Q(10) in early Parkinson disease—Evidence of slowing of the functional decline. Arch Neurol 59(10):1541–1550
Parkinson Study Group (2004) A controlled, randomized, delayed-start study of rasagiline in early Parkinson disease. Arch Neurol 61(4):561–566
de Jong GJ, Meerwaldt JD, Schmitz PIM (1987) Factors that influence the occurrence of response variations in Parkinson’s disease. Ann Neurol 22:4–7
Quinn N, Critchley P, Marsden CD (1987) Young onset Parkinson’s disease. Mov Disord 2:73–91
Blin J, Bonnet A-M, Agid Y (1988) Does levodopa aggravate Parkinson’s disease? Neurology 38:1410–1416
Roos RAC, Vredevoogd CB, van der Velde EA (1990) Response fluctuations in Parkinson’s disease. Neurology 40:1344–1346
Cedarbaum JM, Gandy SE, McDowell FH (1991) Early initiation of levodopa treatment does not promote the development of motor response fluctuations, dyskinesias, or dementia in Parkinson’s disease. Neurology 41:622–629
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Fahn, S., and the Parkinson Study Group. Does levodopa slow or hasten the rate of progression of Parkinson’s disease?. J Neurol 252 (Suppl 4), iv37–iv42 (2005). https://doi.org/10.1007/s00415-005-4008-5
Issue Date:
DOI: https://doi.org/10.1007/s00415-005-4008-5